Intermolecular Attractions and the Properties of Liquids and













- Slides: 36

Intermolecular Attractions and the Properties of Liquids and Solids

Chapter 12 Intermolecular Forces • Important differences between gases, solids, and liquids: – Gases • Expand to fill their container – Liquids • Retain volume, but not shape – Solids • Retain volume and shape 2 At room temperature, some are solid, others are liquid, others are gaseous. Why?

• Physical Properties of Gases, Liquids and Solids determined by – How tightly molecules are packed together – Strength of attractions between molecules

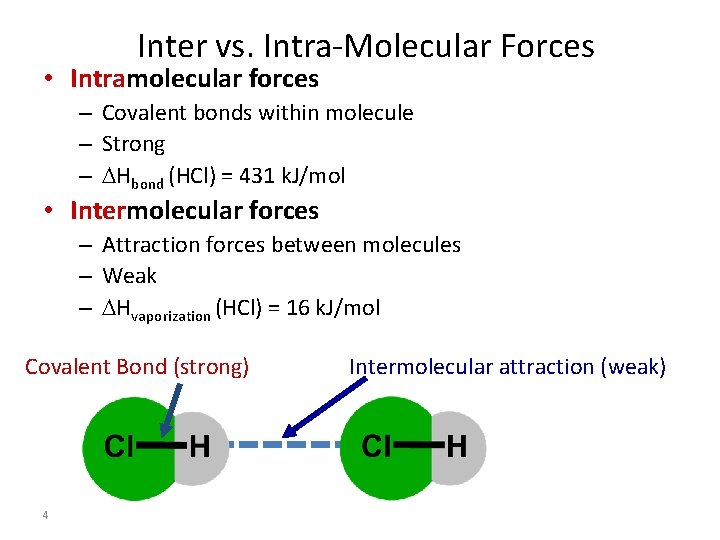
Inter vs. Intra-Molecular Forces • Intramolecular forces – Covalent bonds within molecule – Strong – Hbond (HCl) = 431 k. J/mol • Intermolecular forces – Attraction forces between molecules – Weak – Hvaporization (HCl) = 16 k. J/mol Covalent Bond (strong) 4 Intermolecular attraction (weak)

• When substance melts or boils – Intermolecular forces are broken – Not covalent bonds • • Responsible for existence of condensed states of matter Responsible for bulk properties of matter – Boiling Points and Melting Points

Electronegativity Review Electronegativity: Measure of attractive force that one atom in a covalent bond has for electrons of the bond 6

Bond Dipoles • Two atoms with different electronegativity values share electrons unequally • Electron density is uneven – Higher charge concentration around more electronegative atom • Bond dipoles – Indicated with delta (δ) notation – Indicates partial charge has arisen 7

Three Important Types of Intermolecular Forces 1. Dipole-dipole forces – Hydrogen bonds 2. London dispersion forces 3. Ion-dipole forces – Ion-induced dipole forces 8

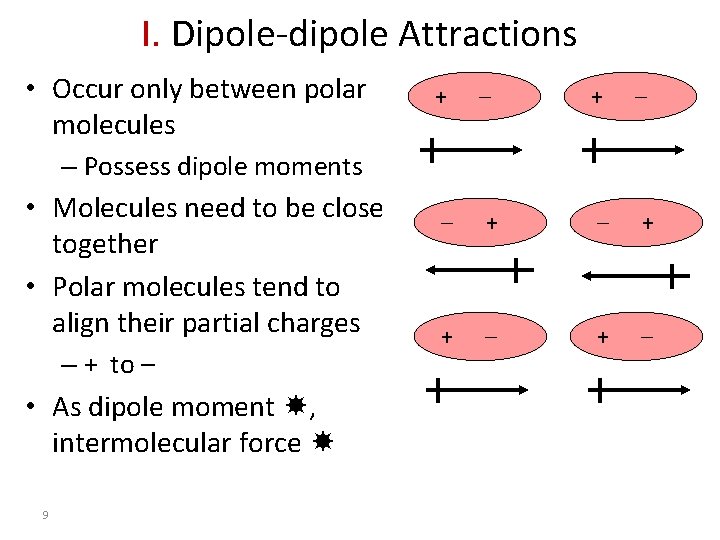
I. Dipole-dipole Attractions • Occur only between polar molecules + + + + – Possess dipole moments • Molecules need to be close together • Polar molecules tend to align their partial charges – + to – • As dipole moment , intermolecular force 9

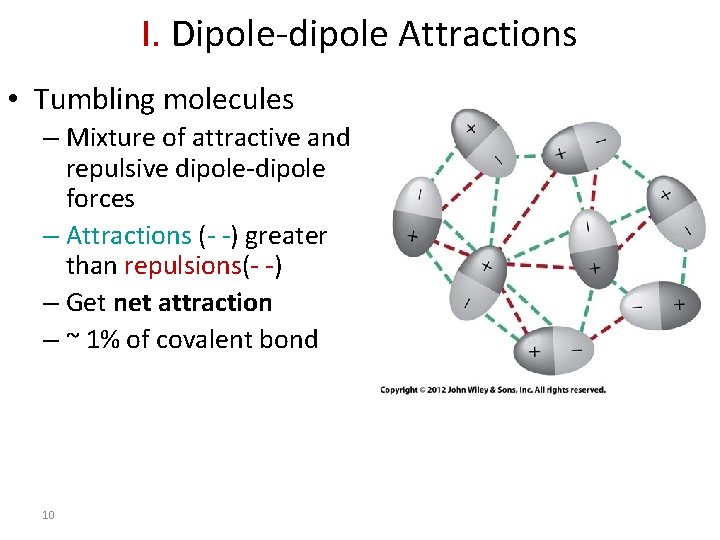
I. Dipole-dipole Attractions • Tumbling molecules – Mixture of attractive and repulsive dipole-dipole forces – Attractions (- -) greater than repulsions(- -) – Get net attraction – ~ 1% of covalent bond 10

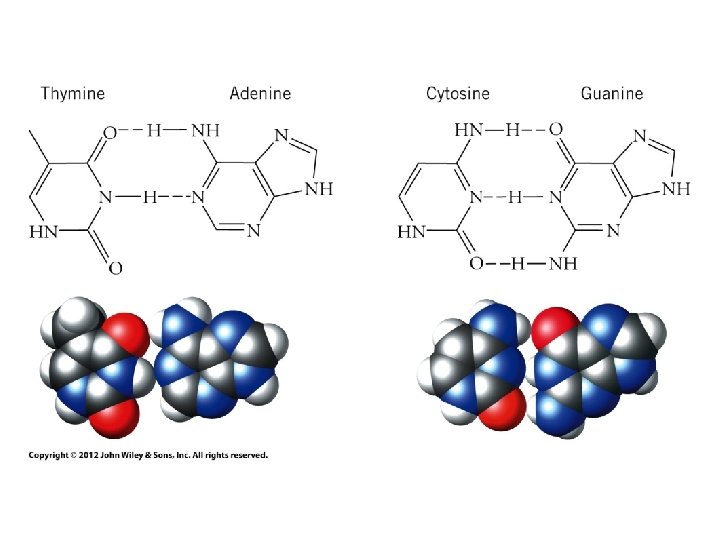
Hydrogen Bonds • Special type of Dipole-Dipole Interaction – Very strong dipole-dipole attraction – ~40 k. J/mol • Occurs between H and highly electronegative atom (O, N, or F) – H—F, H—O, and H—N bonds very polar • Positive end of one can get very close to negative end of another 11

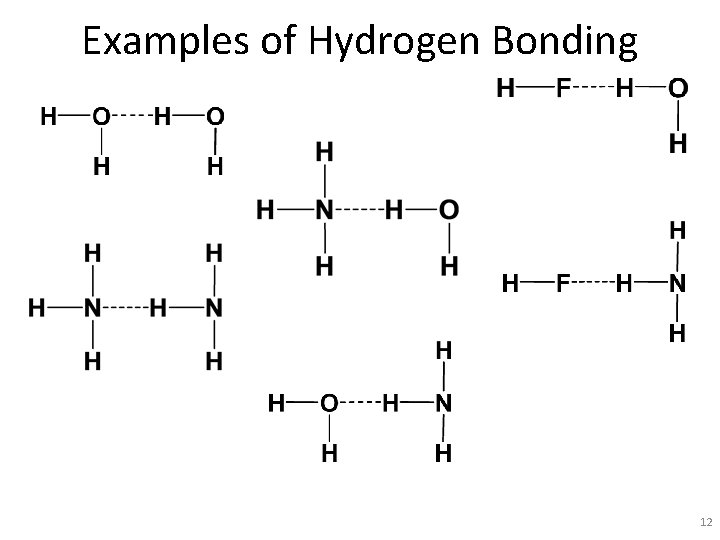
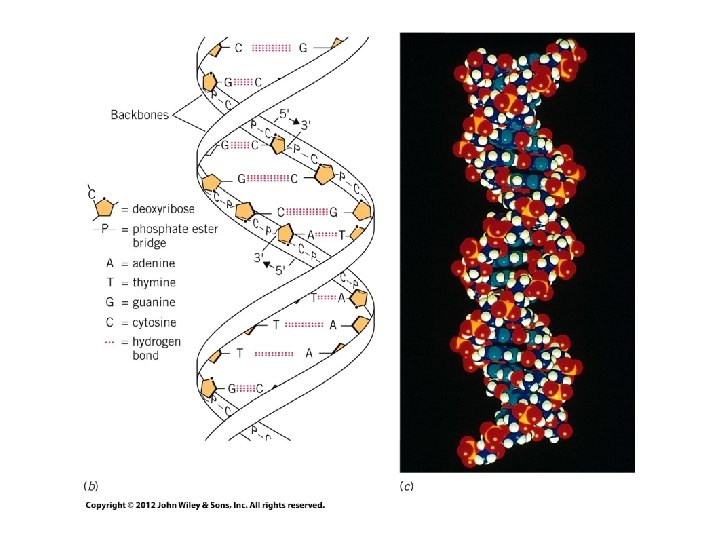
Examples of Hydrogen Bonding 12

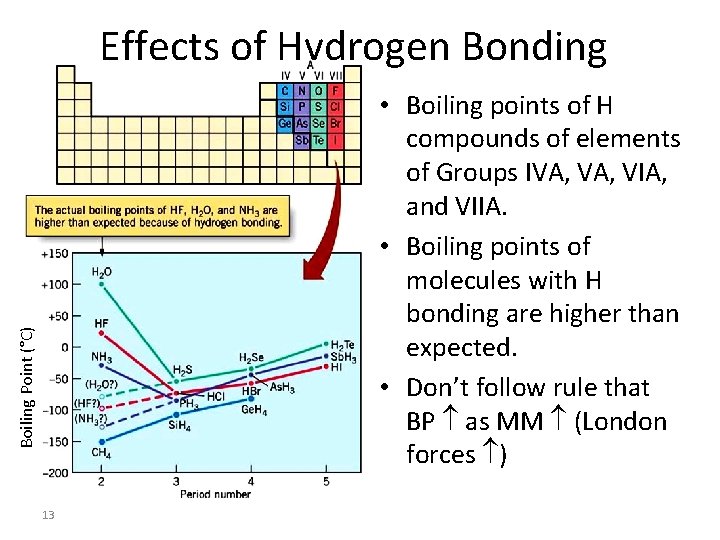
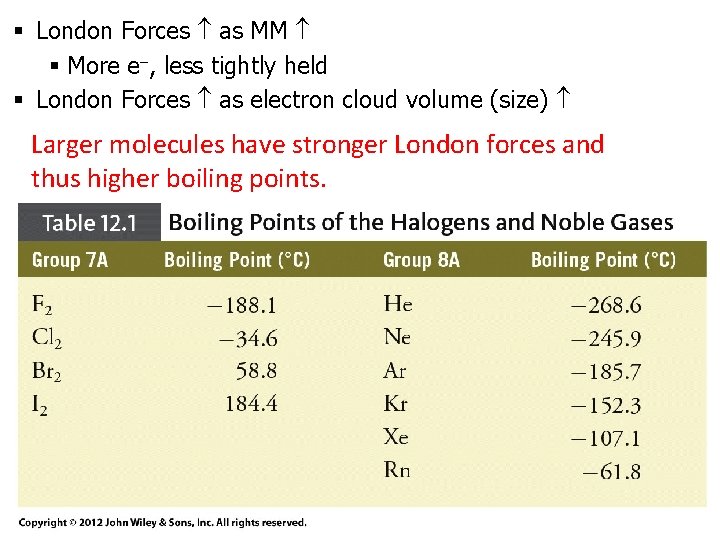
Effects of Hydrogen Bonding Boiling Point (°C) • Boiling points of H compounds of elements of Groups IVA, VIA, and VIIA. • Boiling points of molecules with H bonding are higher than expected. • Don’t follow rule that BP as MM (London forces ) 13

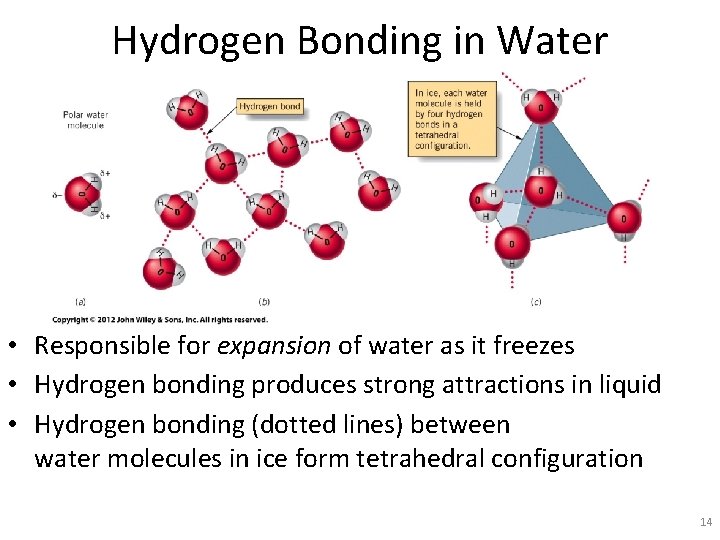
Hydrogen Bonding in Water • Responsible for expansion of water as it freezes • Hydrogen bonding produces strong attractions in liquid • Hydrogen bonding (dotted lines) between water molecules in ice form tetrahedral configuration 14

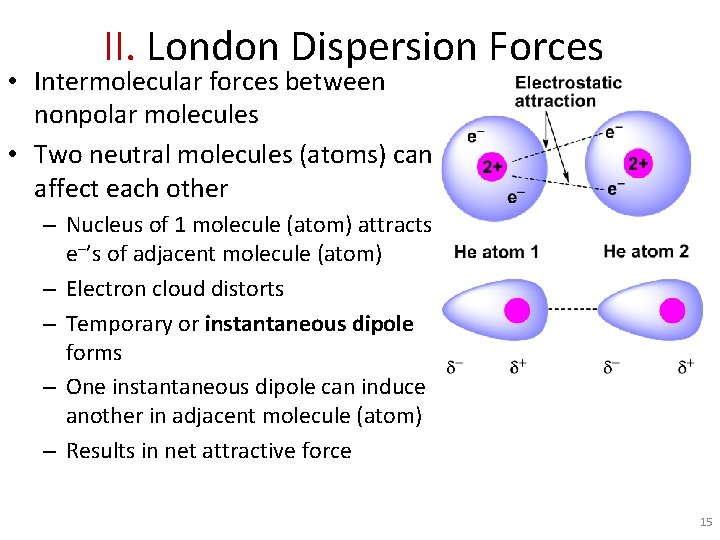
II. London Dispersion Forces • Intermolecular forces between nonpolar molecules • Two neutral molecules (atoms) can affect each other – Nucleus of 1 molecule (atom) attracts e ’s of adjacent molecule (atom) – Electron cloud distorts – Temporary or instantaneous dipole forms – One instantaneous dipole can induce another in adjacent molecule (atom) – Results in net attractive force 15

London Dispersion Forces Instantaneous dipole-induced dipole attractions • – London Dispersion Forces – London forces – Dispersion forces • Decrease as 1/d 6 (d = distance between molecules) • Effect enhanced with increased particle mass • Operate between all molecules – Neutral or net charged – Nonpolar or polar 16

§ London Forces as MM § More e , less tightly held § London Forces as electron cloud volume (size) Larger molecules have stronger London forces and thus higher boiling points.

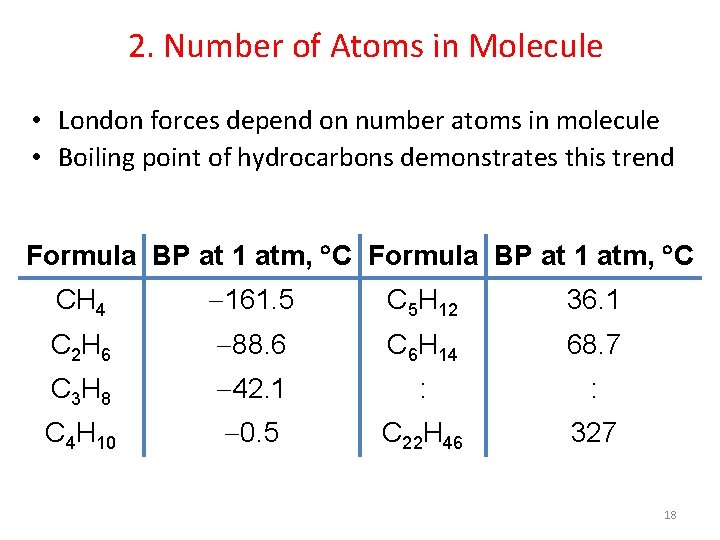
2. Number of Atoms in Molecule • London forces depend on number atoms in molecule • Boiling point of hydrocarbons demonstrates this trend Formula BP at 1 atm, C CH 4 161. 5 C 5 H 12 36. 1 C 2 H 6 88. 6 C 6 H 14 68. 7 C 3 H 8 42. 1 : : C 4 H 10 0. 5 C 22 H 46 327 18

III. Ion-dipole Attractions • Attractions between ion and charged end of polar molecules – Attractions can be quite strong as ions have full charges (a) Negative ends of water dipoles surround cation (b) Positive ends of water dipoles surround anion 19

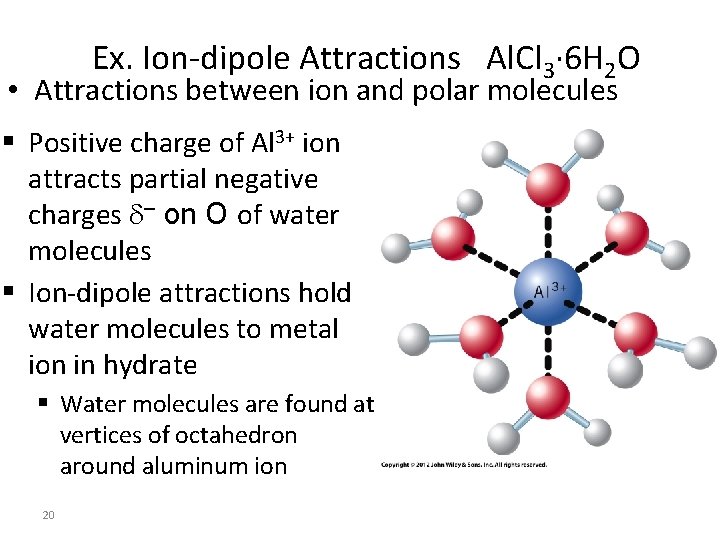
Ex. Ion-dipole Attractions Al. Cl 3· 6 H 2 O • Attractions between ion and polar molecules § Positive charge of Al 3+ ion attracts partial negative charges – on O of water molecules § Ion-dipole attractions hold water molecules to metal ion in hydrate § Water molecules are found at vertices of octahedron around aluminum ion 20

Using Intermolecular Forces • Often can predict physical properties (like BP and MP) by comparing strengths of intermolecular attractions Strongest – Ion-Dipole – Hydrogen Bonding – Dipole-Dipole Weakest – London Dispersion Forces • Larger, longer, heavier molecules have stronger IMFs • Smaller, more compact, lighter molecules have weaker IMFs 21

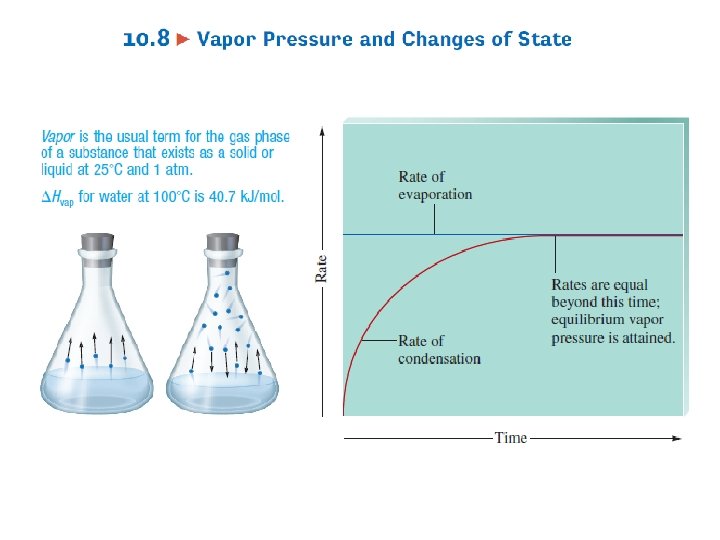
Phase Changes • Changes of physical state – Deal with motion of molecules • As temperature changes – Matter will undergo phase changes • Liquid Gas – Evaporation – As heat H 2 O, forms steam or water vapor – Requires energy or source of heat to occur 22

Phase Changes • Solid Gas – Sublimation – Ice cubes in freezer, leave in long enough disappear – Endothermic • Gas Liquid – Cooling or Condensation – Dew is H 2 O vapor condensing onto cooler ground – Exothermic 23

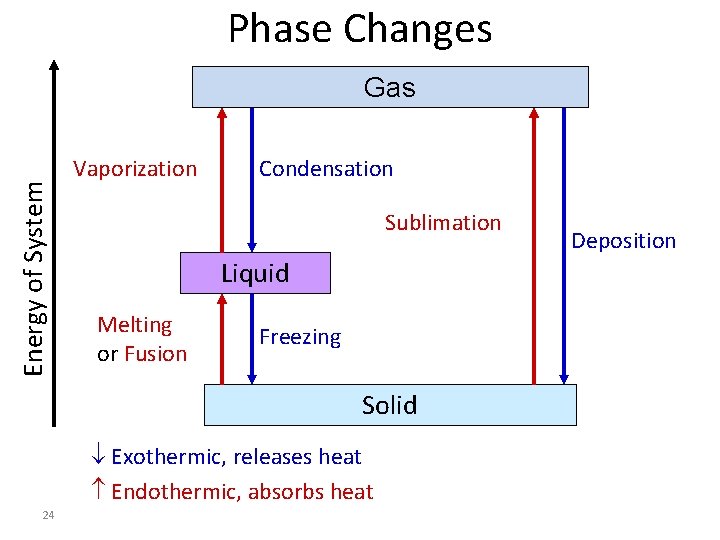
Phase Changes Energy of System Gas Vaporization Condensation Sublimation Liquid Melting or Fusion Freezing Solid Exothermic, releases heat Endothermic, absorbs heat 24 Deposition



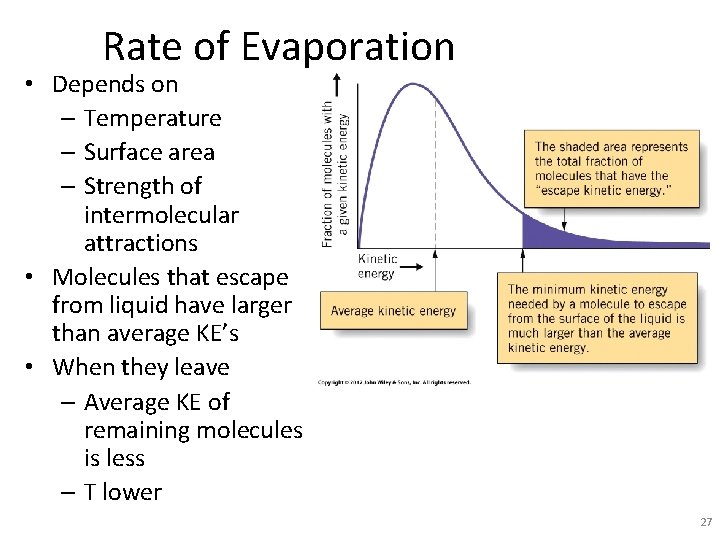
Rate of Evaporation • Depends on – Temperature – Surface area – Strength of intermolecular attractions • Molecules that escape from liquid have larger than average KE’s • When they leave – Average KE of remaining molecules is less – T lower 27

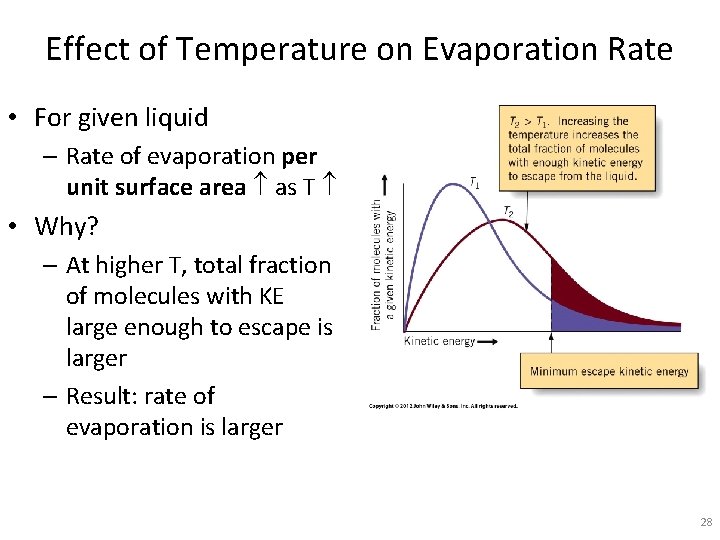
Effect of Temperature on Evaporation Rate • For given liquid – Rate of evaporation per unit surface area as T • Why? – At higher T, total fraction of molecules with KE large enough to escape is larger – Result: rate of evaporation is larger 28

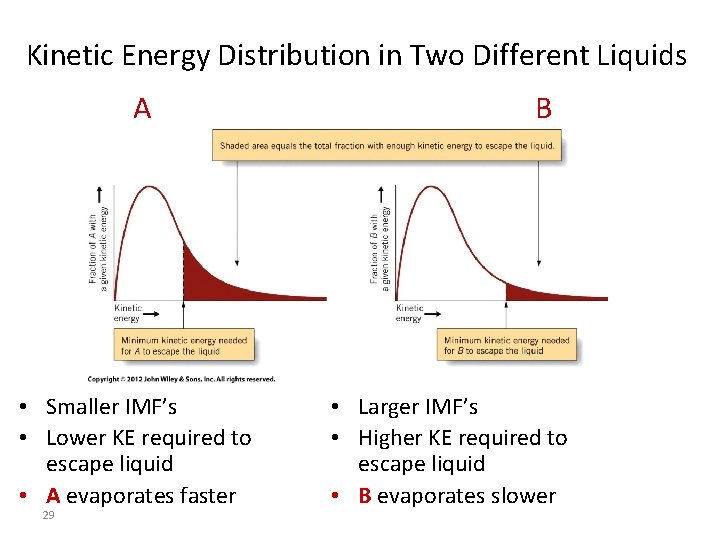
Kinetic Energy Distribution in Two Different Liquids A • Smaller IMF’s • Lower KE required to escape liquid • A evaporates faster 29 B • Larger IMF’s • Higher KE required to escape liquid • B evaporates slower

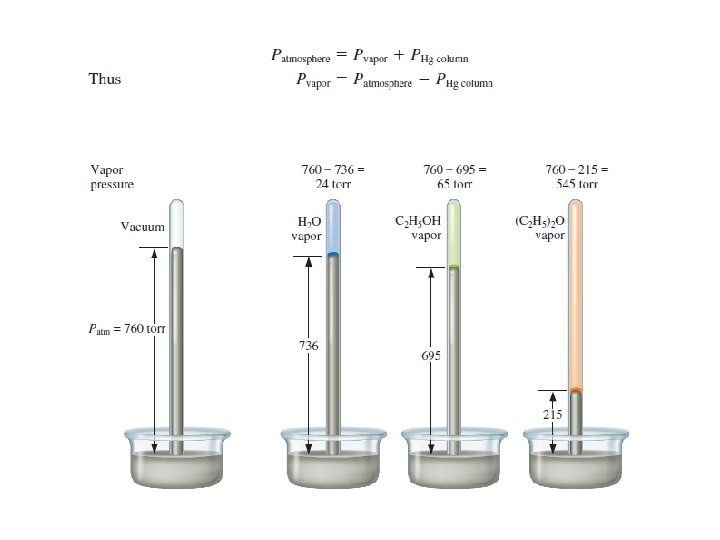
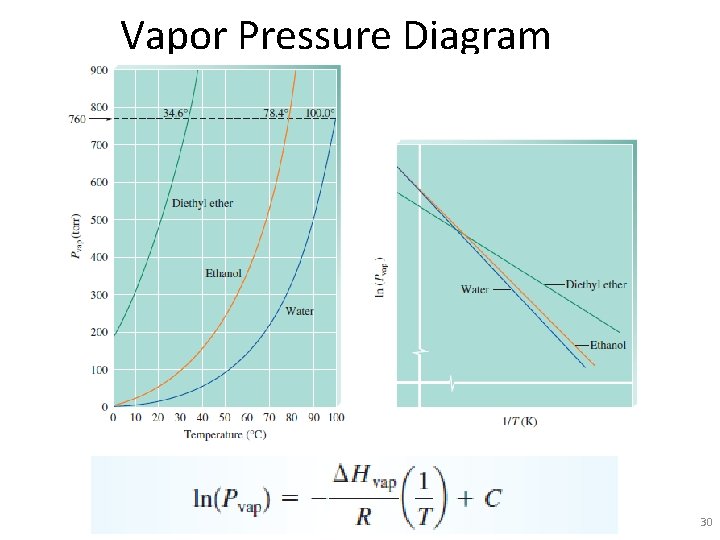
Vapor Pressure Diagram 30


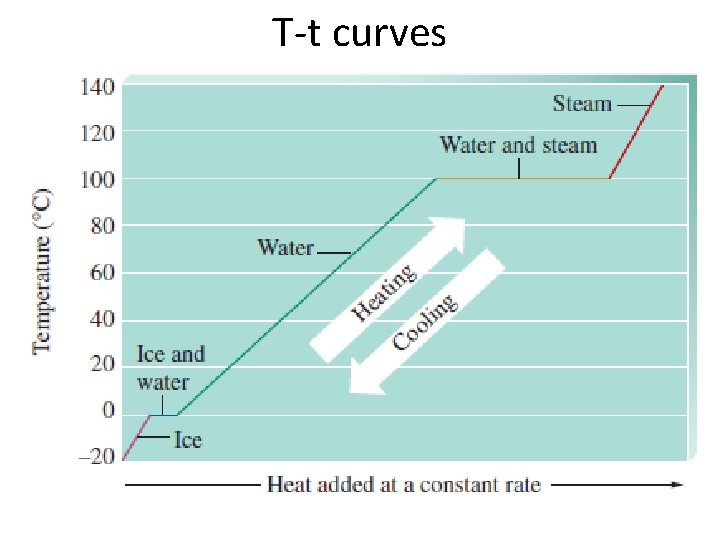
T-t curves

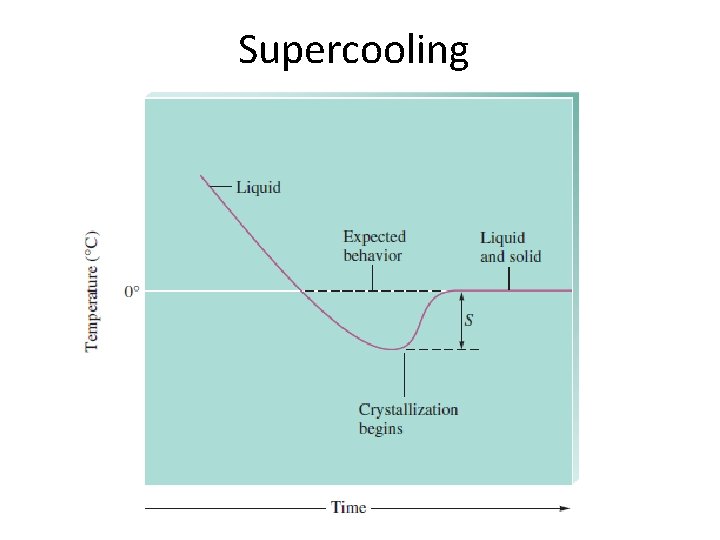
Supercooling

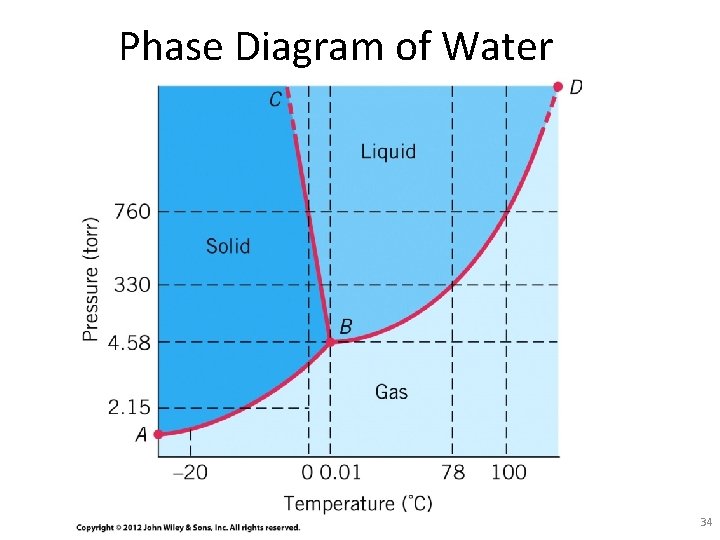
Phase Diagram of Water 34

