Interchangeability of followon products Sndor KerpelFronius M D

Interchangeability of follow-on products Sándor Kerpel-Fronius, M. D. , D. Sc. Semmelweis University Department of Pharmacology and Pharmacotherapy Budapest, Hungary Email: kerpel-fronius. sandor @med. semmelweis-uni. hu KERPEL-FRONIUS S. 1

Interchangeability Science and politics Money Pharmaceutical Industry Health care providers Patient care Efficacy & safety of drug therapy Social justice Equal access to high quality, affordable medicines Follow-on medicines Reference pricing KERPEL-FRONIUS S. 2

Interchangeability and Substitution Different working definitions https: //www. bio. org/sites/default/files/Sept%2010%20 -%20 Compa ny%20 Presentation%20 -%2014 h 00%20 -%20 Abb. Vie. pdf; v Interchangeability: Health or Regulatory Authority Designation (US FDA) BPCI Act. Biologics Price Competition and Innovation Act of 2009. Federal Register 2010; H. R. 3590 -686 -702 Ø Expected to produce the same clinical result as the reference product in any given patient; Ø Repeated switching between biosimilar and reference product presents no greater safety or efficacy risk than continued use of the reference product v Substitution: Pharmacist Action v. When a pharmacist substitutes a certain prescribed product by another equivalent product Ø Automatic substitution: when substitution is done without the prescribing physician’s involvement (Interchangeable) v Medical Switching: Treating Physician Decision Ø When a prescribing physician changes medication, usually because of KERPEL-FRONIUS S. 3 efficacy or safety issue(s)
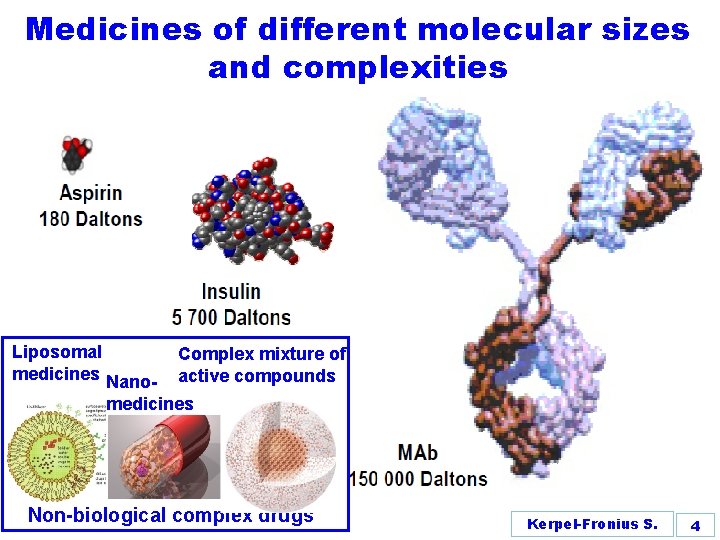
Medicines of different molecular sizes and complexities Liposomal medicines Nano- Complex mixture of active compounds medicines Non-biological complex drugs Kerpel-Fronius S. 4
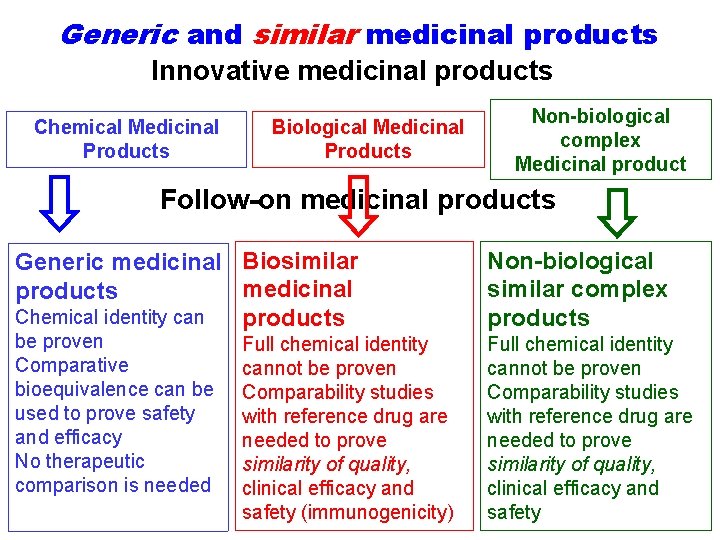
Generic and similar medicinal products Innovative medicinal products Chemical Medicinal Products Biological Medicinal Products Non-biological complex Medicinal product Follow-on medicinal products Generic medicinal Biosimilar medicinal products Chemical identity can products be proven Comparative bioequivalence can be used to prove safety and efficacy No therapeutic comparison is needed Full chemical identity cannot be proven Comparability studies with reference drug are needed to prove similarity of quality, clinical efficacy and safety (immunogenicity) Non-biological similar complex products Full chemical identity cannot be proven Comparability studies with reference drug are needed to prove similarity of quality, clinical efficacy and safety
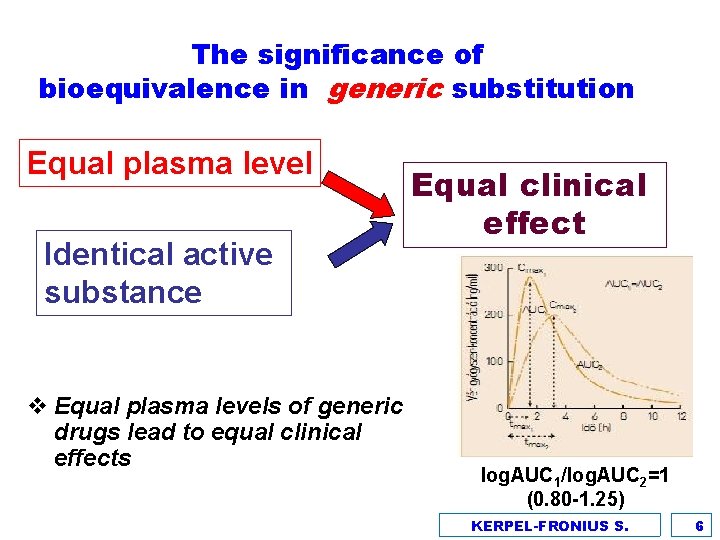
The significance of bioequivalence in generic substitution Equal plasma level Identical active substance v Equal plasma levels of generic drugs lead to equal clinical effects Equal clinical effect log. AUC 1/log. AUC 2=1 (0. 80 -1. 25) KERPEL-FRONIUS S. 6

Causes of possible differences between generic drugs Same active substance Different excipients Same active substance v Same active substance but different inactive vehicles v In the same active substance different amounts and/or chemically different impurities v Different quality and/or stability of the medicinal product, etc. v These differences usually lead to negligible medical consequences only KERPEL-FRONIUS S: 7
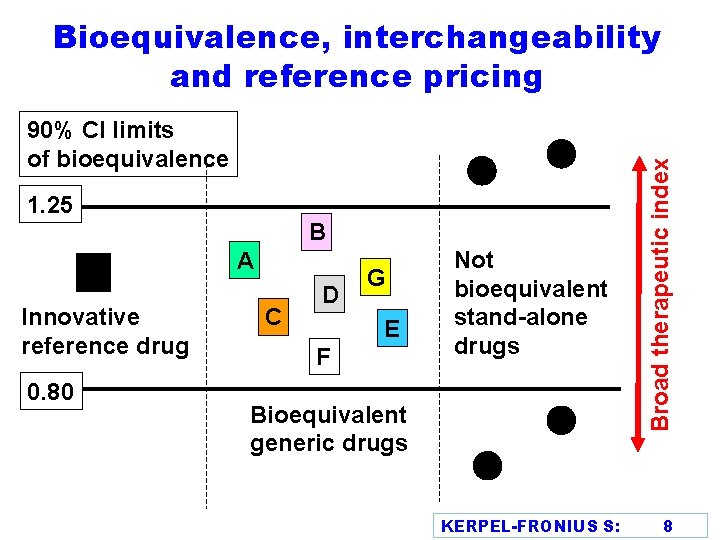
90% CI limits of bioequivalence 1. 25 B A Innovative reference drug 0. 80 C D G E F Not bioequivalent stand-alone drugs Bioequivalent generic drugs KERPEL-FRONIUS S: Broad therapeutic index Bioequivalence, interchangeability and reference pricing 8

Narrow therapeutic index drugs v Medicnes with steep dose-activity curves v They have less than a 2 -fold difference in median lethal dose and median effective dose values v They have less than a 2 -fold difference in the minimum toxic concentrations and minimum effective concentrations in the blood v Safe and effective use of the drug products requires careful titration and patient monitoring Minimum (median) toxic plasma concentration Minimum (median) therapeutic plasma concentration <2 KERPEL-FRONIUS S: 9
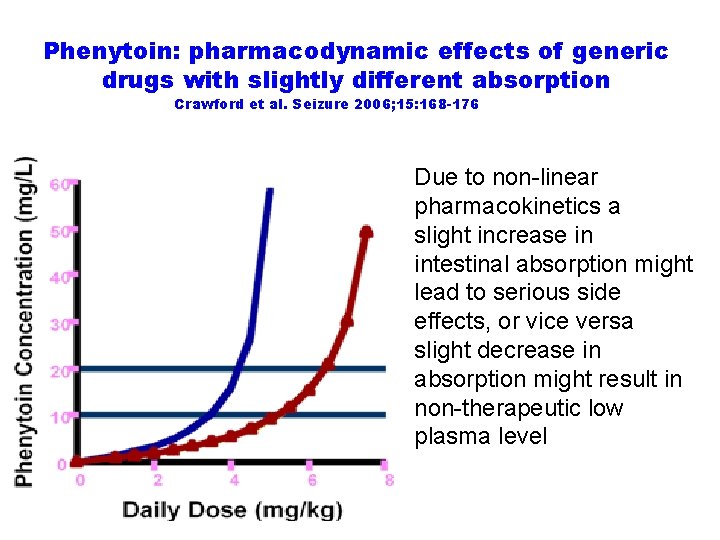
Phenytoin: pharmacodynamic effects of generic drugs with slightly different absorption Crawford et al. Seizure 2006; 15: 168 -176 Due to non-linear pharmacokinetics a slight increase in intestinal absorption might lead to serious side effects, or vice versa slight decrease in absorption might result in non-therapeutic low plasma level

Drugs with a narrow therapeutic index v Recommended but generally not accepted CI limits for drugs with narrow therapeutic window (95% CI (0. 9 -1. 11) Ø Aminophylline/Theophylline Ø Lithium Ø Thyroxine Ø Warfarin Ø Antiepileptics (apart from benzodiazepines) Ø Immunosuppressive agents Ø Cytotoxic agents Ø Antiarrhythmics Ø Tricyclic antidepressive drugs v Substitution should be done under tight medical control (retitration of the dose, if necessary with therapeutic drug monitoring, etc. ) KERPEL-FRONIUS S: 10

Biosimilar application (FDA) Sherman RE, Associate Director for Medical Policy Center of Drug Evaluation and Research Biologics Price Competition and Innovation (BPCI) Act Biosimilarity means v biological product is highly similar to the reference product notwithstanding minor differences in clinically inactive components v there are no clinically meaningful differences between the biological product and the reference product in terms of the safety, purity, and potency of the product. Specifications of biosimilar applications v biosimilarity to a reference product v same mechanism(s) of action for the proposed condition(s) of use–only to the extent known for the reference product v condition(s) of use proposed in labelling have been previously approved for the reference product v same route of administration, dosage form and strength as the reference product
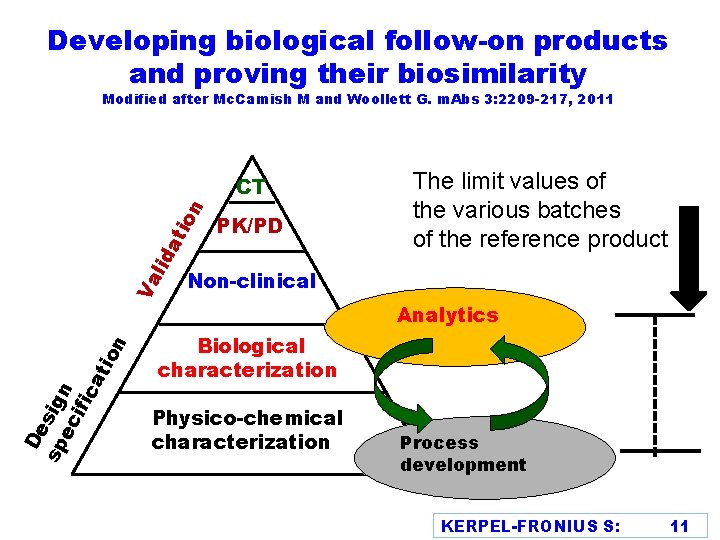
Developing biological follow-on products and proving their biosimilarity Modified after Mc. Camish M and Woollett G. m. Abs 3: 2209 -217, 2011 De sp sign ec ifi ca tio n Va lid at ion CT PK/PD The limit values of the various batches of the reference product Non-clinical Analytics Biological characterization Physico-chemical characterization Process development KERPEL-FRONIUS S: 11

Similar biological medicinal products Clinical advantages and problems Proven identical and/or similar properties: • Identical amino acid composition • Similar pharmacokinetic properties • Similar pharmacologic effects Dissimilar properties: Biosimilar medicines • Post-translational modifications • Immunogenic properties • The clinical significance of the differences are uncertain KERPEL-FRONIUS S. 14

Steps for proving biosimilarity of follow-on m. Abs EMA guideline draft (EMA/CHMP/BMWP/403543/2010 Similarity v Identical amino acid composition v Similarity of physical-chemical measurements v Non-clinical comparative experiments Ø In vitro biological experiments Ø In vivo efficacy evaluation in animal models if feasible, comparative safety evaluation v Human comparative studies Ø Clinical pharmacodynamic studies Ø Clinical pharmacokinetic studies Ø Evaluation of dose: pharmacodynamic relationships Ø Therapeutic trials Identity KERPEL-FRONIUS S.
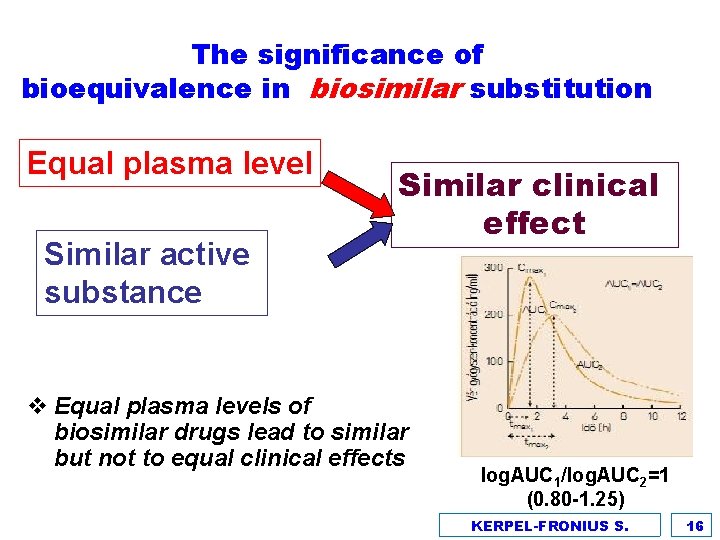
The significance of bioequivalence in biosimilar substitution Equal plasma level Similar active substance Similar clinical effect v Equal plasma levels of biosimilar drugs lead to similar but not to equal clinical effects log. AUC 1/log. AUC 2=1 (0. 80 -1. 25) KERPEL-FRONIUS S. 16
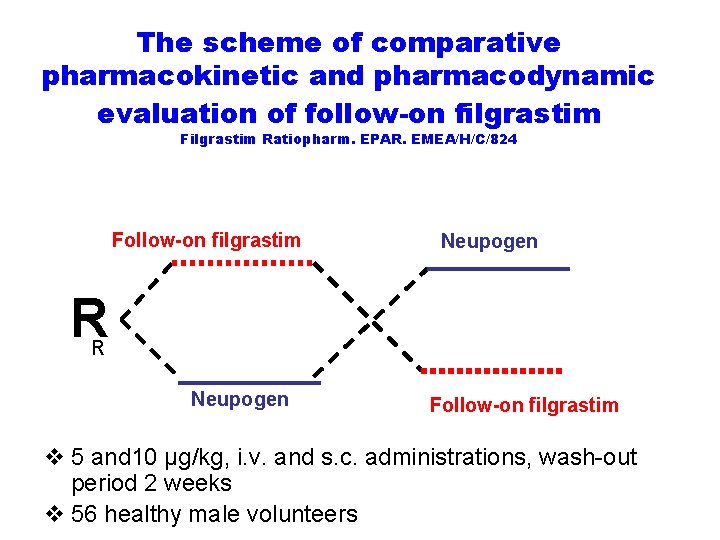
The scheme of comparative pharmacokinetic and pharmacodynamic evaluation of follow-on filgrastim Filgrastim Ratiopharm. EPAR. EMEA/H/C/824 Follow-on filgrastim Neupogen R R Neupogen Follow-on filgrastim v 5 and 10 μg/kg, i. v. and s. c. administrations, wash-out period 2 weeks v 56 healthy male volunteers
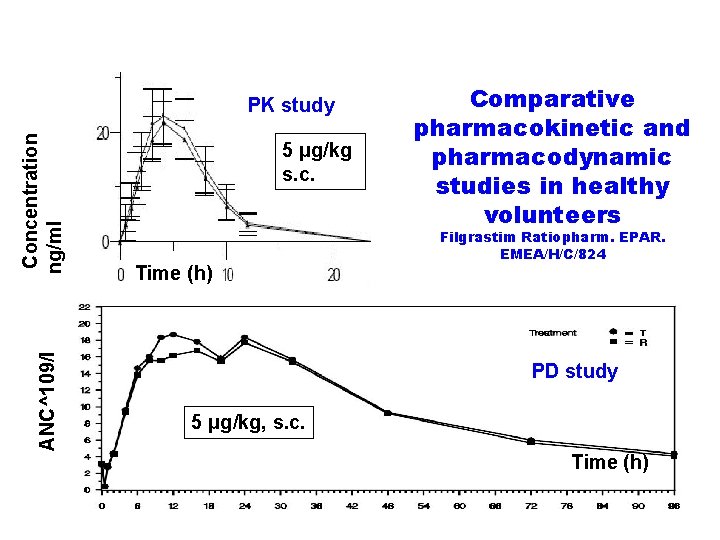
ANC^109/l Concentration ng/ml PK study 5 μg/kg s. c. Time (h) Comparative pharmacokinetic and pharmacodynamic studies in healthy volunteers Filgrastim Ratiopharm. EPAR. EMEA/H/C/824 PD study 5 μg/kg, s. c. Time (h)
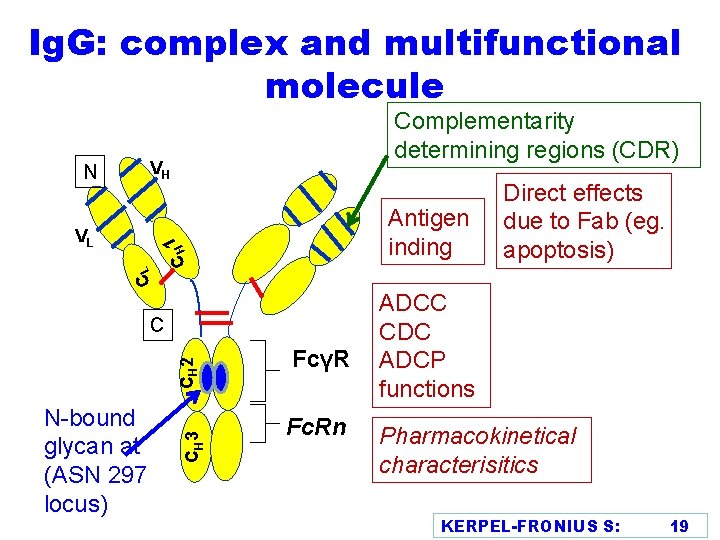
Ig. G: complex and multifunctional molecule Complementarity determining regions (CDR) VH CL 1 CH N VL Antigen inding N-bound glycan at (ASN 297 locus) CH 3 CH 2 C FcγR Fc. Rn Direct effects due to Fab (eg. apoptosis) ADCC CDC ADCP functions Pharmacokinetical characterisitics KERPEL-FRONIUS S: 19
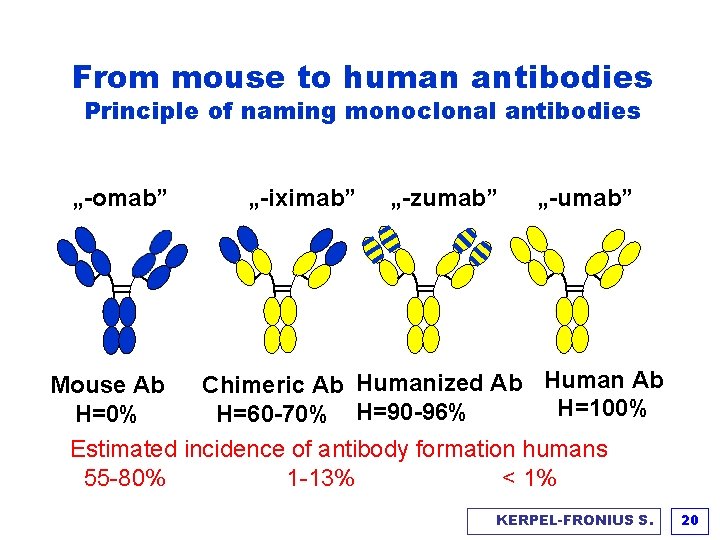
From mouse to human antibodies Principle of naming monoclonal antibodies „-omab” „-iximab” „-zumab” „-umab” Chimeric Ab Humanized Ab Human Ab Mouse Ab H=100% H=60 -70% H=90 -96% H=0% Estimated incidence of antibody formation humans 55 -80% 1 -13% < 1% KERPEL-FRONIUS S. 20
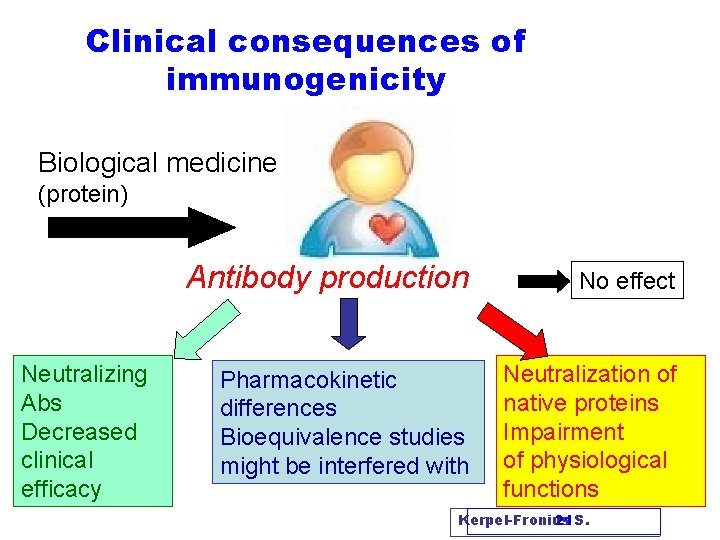
Clinical consequences of immunogenicity Biological medicine (protein) Neutralizing Abs Decreased clinical efficacy Antibody production No effect Pharmacokinetic differences Bioequivalence studies might be interfered with Neutralization of native proteins Impairment of physiological functions Kerpel-Fronius 21 S.

Antigenecity - immunogenicity v Antigenicity: is the ability to combine with the final products of the humoral and/or cell mediated immune response. It is not activating the immune response rather it combines with the final products of the immune response. v Immunogenicity: is antigenicity in an inflammatory milieu resulting in a successful humoral response. v All immunogens are antigenic but all antigens are not immunogenic KERPEL-FRONIUS S. 22

The clinical importance of Anti-drug antibodies (ADAs) & Immune Complexes (IC) Krishna M & and Nadler SG, Front. Immunol. 7: 21, 2016 doi: 10. 3389/fimmu. 2016. 00021 v The formation and detection of Anti-Drug Antibodies (ADAs) can be usually equated with the measure of immunogenicity v Most adverse effects are a consequence of formation of immune complexes (ICs) between the ADA and therapeutic proteins Ø pharmacological abrogation Ø impact on therapeutic exposure Ø hypersensitivity reactions v The level of IC, kinetics of interaction, size, polyclonal diversity, distribution, and Fc-mediated effects determine the type and severity of ADRs KERPEL-FRONIUS S. 23
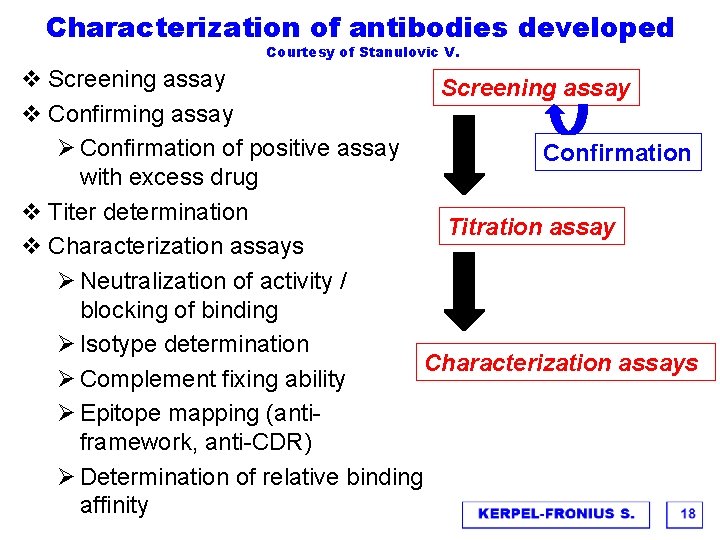
Characterization of antibodies developed Courtesy of Stanulovic V. v Screening assay v Confirming assay Ø Confirmation of positive assay Confirmation with excess drug v Titer determination Titration assay v Characterization assays Ø Neutralization of activity / blocking of binding Ø Isotype determination Characterization assays Ø Complement fixing ability Ø Epitope mapping (antiframework, anti-CDR) Ø Determination of relative binding affinity
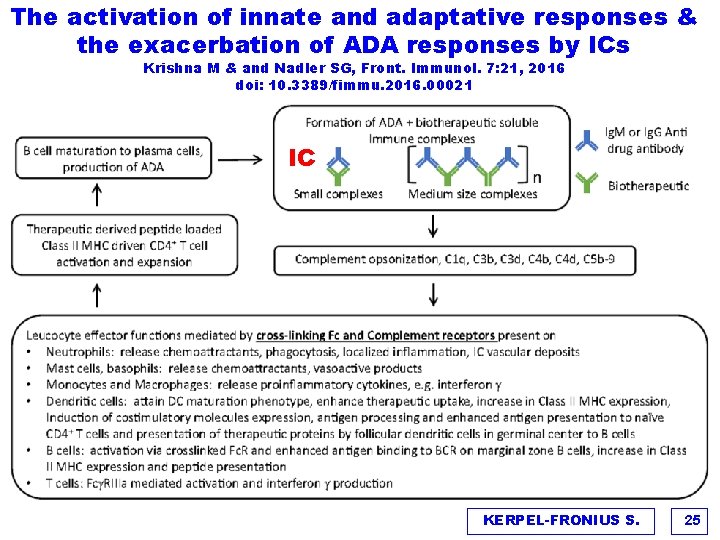
The activation of innate and adaptative responses & the exacerbation of ADA responses by ICs Krishna M & and Nadler SG, Front. Immunol. 7: 21, 2016 doi: 10. 3389/fimmu. 2016. 00021 IC KERPEL-FRONIUS S. 25
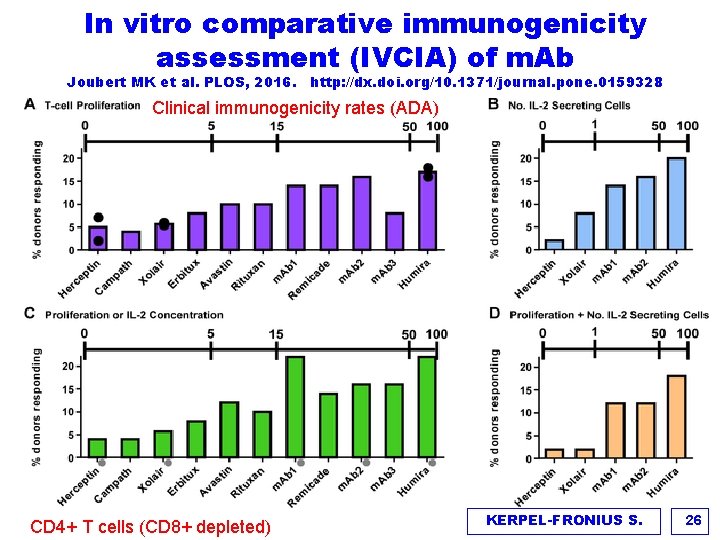
In vitro comparative immunogenicity assessment (IVCIA) of m. Ab Joubert MK et al. PLOS, 2016. http: //dx. doi. org/10. 1371/journal. pone. 0159328 Clinical immunogenicity rates (ADA) CD 4+ T cells (CD 8+ depleted) KERPEL-FRONIUS S. 26
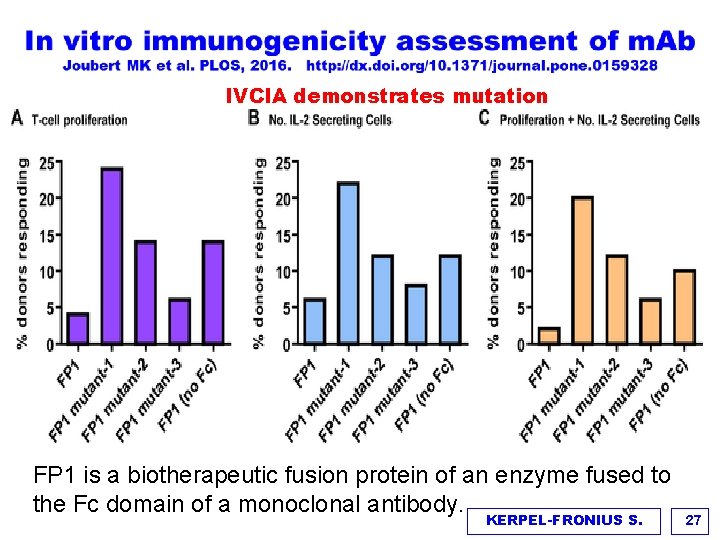
IVCIA demonstrates mutation FP 1 is a biotherapeutic fusion protein of an enzyme fused to the Fc domain of a monoclonal antibody. KERPEL-FRONIUS S. 27
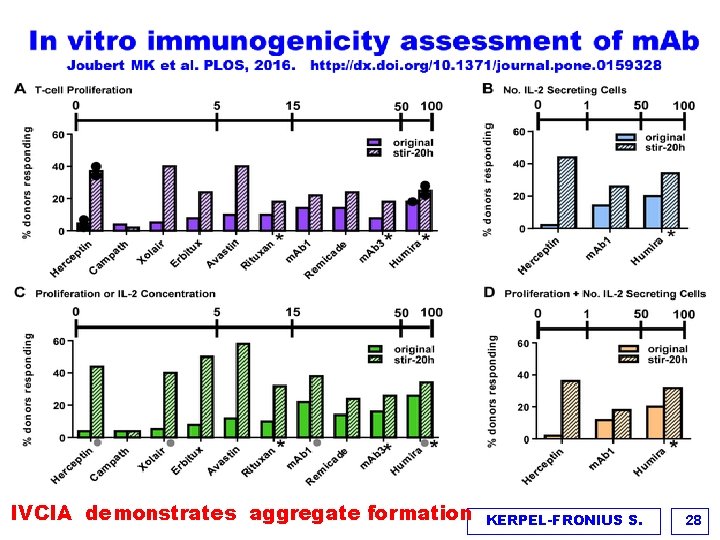
IVCIA demonstrates aggregate formation KERPEL-FRONIUS S. 28
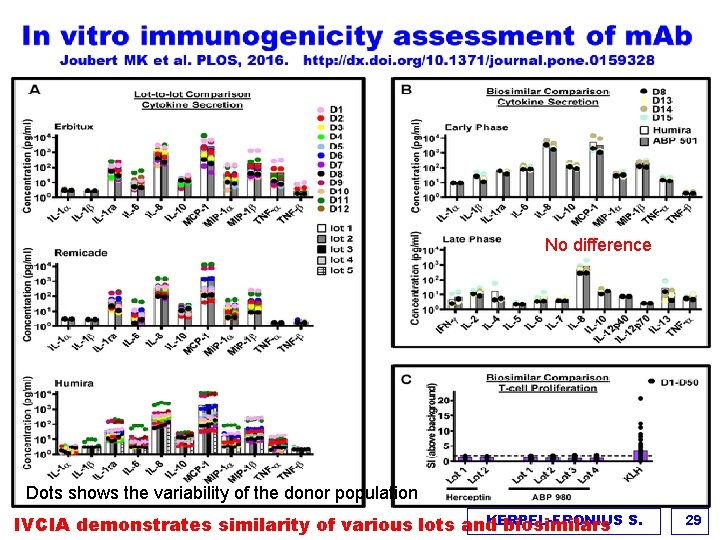
No difference Dots shows the variability of the donor population KERPEL-FRONIUS IVCIA demonstrates similarity of various lots and biosimilars S. 29

Extrapolation of immunogenicity accross indications Shomron Ben-Horin et al. Expert Review of Gastroenterology & Hepatology 9 (2015) v Antibody conformational assay can demonstrate crossimmunogenicity equivalence Ø Antibodies were raised against several different peptides derived from infliximab. Ø Recognition of 14 batches of reference (Remicade) versus biosimilar infliximab (Remsima) demonstrated similar profiles of exposed epitopes on the two molecules Ø The two molecules have very similar higher order immuno -dominant structures v Immunogenicity is similar in RA and IBD indicating a broadly similar rate across these two indications, supporting the validity of extrapolating immunogenicity data accross indications KERPEL-FRONIUS S. 30
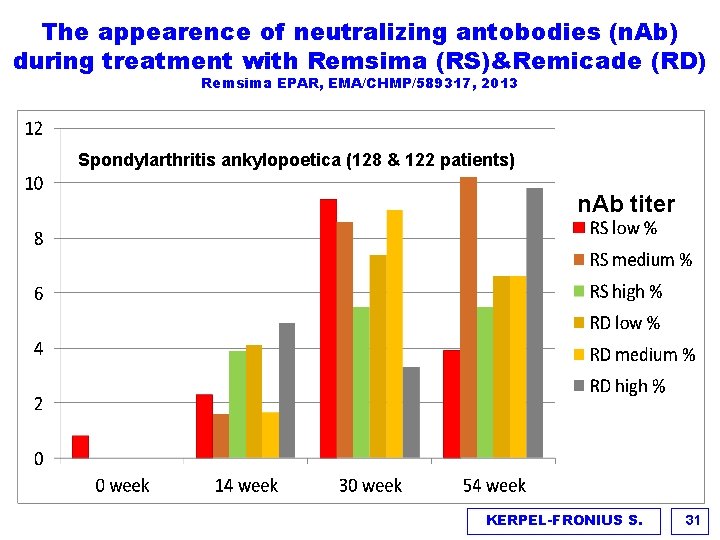
The appearence of neutralizing antobodies (n. Ab) during treatment with Remsima (RS)&Remicade (RD) Remsima EPAR, EMA/CHMP/589317, 2013 Spondylarthritis ankylopoetica (128 & 122 patients) n. Ab titer KERPEL-FRONIUS S. 31
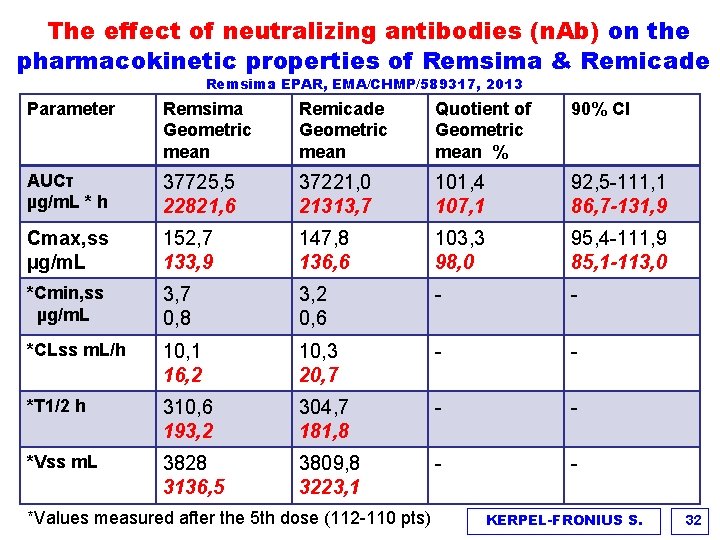
The effect of neutralizing antibodies (n. Ab) on the pharmacokinetic properties of Remsima & Remicade Remsima EPAR, EMA/CHMP/589317, 2013 Parameter Remsima Geometric mean Remicade Geometric mean Quotient of Geometric mean % 90% CI AUCτ µg/m. L * h 37725, 5 22821, 6 37221, 0 21313, 7 101, 4 107, 1 92, 5 -111, 1 86, 7 -131, 9 Cmax, ss µg/m. L 152, 7 133, 9 147, 8 136, 6 103, 3 98, 0 95, 4 -111, 9 85, 1 -113, 0 *Cmin, ss µg/m. L 3, 7 0, 8 3, 2 0, 6 - - *CLss m. L/h 10, 1 16, 2 10, 3 20, 7 - - *T 1/2 h 310, 6 193, 2 304, 7 181, 8 - - *Vss m. L 3828 3136, 5 3809, 8 3223, 1 - - *Values measured after the 5 th dose (112 -110 pts) KERPEL-FRONIUS S. 32

Interchangeability and Substitution Different working definitions https: //www. bio. org/sites/default/files/Sept%2010%20 -%20 Compa ny%20 Presentation%20 -%2014 h 00%20 -%20 Abb. Vie. pdf; v Interchangeability: Health or Regulatory Authority Designation (US FDA) BPCI Act. Biologics Price Competition and Innovation Act of 2009. Federal Register 2010; H. R. 3590 -686 -702 Ø Expected to produce the same clinical result as the reference product in any given patient; Ø Repeated switching between biosimilar and reference product presents no greater safety or efficacy risk than continued use of the reference product v Substitution: Pharmacist Action Ø When a pharmacist substitutes a certain prescribed product by another equivalent product Ø Automatic substitution: when substitution is done without the prescribing physician’s involvement (Interchangeable) v Medical Switching (substitution) : Treating Physician Decision Ø When a prescribing physician changes medication, usually because of KERPEL-FRONIUS S. 33 efficacy or safety issue(s)

FDA Biosimilar Interchangeability Guidance lhttps: //www. fda. gov/downloads/Drugs/Guidance. Compliance. Regulatory. Information /Guidances/UCM 537135. pdf v The requirements will vary based on the properties of the product submitted Ø Relevant factors including product complexity and product-specific immunogenicity risk Ø Low structural complexity with meaningful fingerprint-like analytical similarity to the reference product and has a low incidence of serious adverse events related to immunogenicity. Appropriately designed switching study might be enough for proving interchangeability. Ø High structural complexity, no meaningful fingerprint-like analytical similarity and exhibits serious adverse events related to immunogenicity. Switching study and postmarketing data for the product as a licensed biosimilar might support a demonstration of interchangeability KERPEL-FRONIUS S. 34

FDA Biosimilar Interchangeability Guidance lhttps: //www. fda. gov/downloads/Drugs/Guidance. Compliance. Regulatory. Information /Guidances/UCM 537135. pdf v Switching studies should demonstrate whether alternating between a biosimilar and its reference product two or more times impacts the safety or efficacy of the treatment course. v If the product is only intended to be administered once than switching study might not be needed and requested v "If an apparent difference in immune response or adverse events is noticed between the switching and non -switching arms of the study…it would raise concerns as to whether the proposed interchangeable product is interchangeable, regardless of whether the proposed interchangeable product or the reference product or the switching of the two products actually caused the event, " FDA KERPEL-FRONIUS S. 35
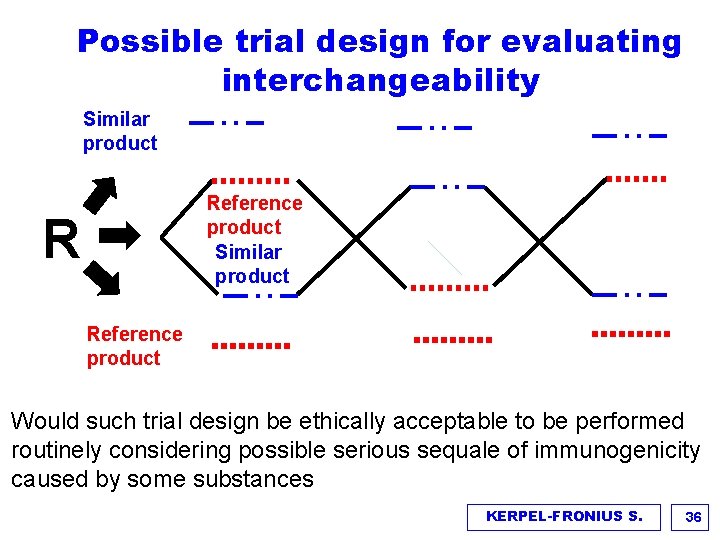
Possible trial design for evaluating interchangeability Similar product Reference product Similar product R Reference product Would such trial design be ethically acceptable to be performed routinely considering possible serious sequale of immunogenicity caused by some substances KERPEL-FRONIUS S. 36

Abb. Vie Submits Citizen Petition on Interchangeable Biosimilars http: //www. biologicsblog. com/blog/abbvie-submits-citizen-petition-oninterchangeable-biosimilars/ Abb. Vie recommendations for the Safety Standards for Determining Interchangeability v FDA needs to ensure that biological products listed as substitutable are, in fact, interchangeable for all the indications and conditions of use of the reference product. v In the case of a biological product administered more than once to a patient, the risk in terms of safety or diminished efficacy of alternating or switching between use of the biological product and the reference product is not greater than the risk of using the reference product without such alternation or switch. v Abb. Vie suggest that any FDA guidance on interchangeability should address the real-world possibility that different interchangeable biological products corresponding to a single reference product will be substituted and switched over the course of a single patient’s treatment. KERPEL-FRONIUS S. 37

90% CI limits of bioequivalence 1. 25 Bioequivalent biosimilar drugs B A Innovative reference drug 0. 80 C D G E F Not bioequivalent stand-alone drugs Broad therapeutic index Interchangeability, the pharmacological significance of bioequivalence, biosimilarity and immunogenicity According to FDA, switching studies should be designed to determine whether alternating between a biosimilar and its reference product two or more times impacts the safety or efficacy of the treatment course. What will happen in real life having many biosimilars of the same KERPEL-FRONIUS S: 8 reference product? ?

Changing strategy for comapring biological medicinal agents v Presently in vivo comparative clinical studies are necessary and are required for proving biosimilarity in humans v Improved physico-chemical methods, in vitro efficacy, antigenecity and human cell based immunogenicity assays are rapidly developed for proving biological equivalence v It is expected that the improving specificities of in vitro tests predicting biosimilarity including similar immunogenicity might diminish the need for extensive clinical comparison of efficacy and safety v The relative significance of comparative clinical trials to determine clinical efficacy, safety and immunogenicity of biological medicinal agents is expected to be decreased in the future KERPEL-FRONIUS S. 39
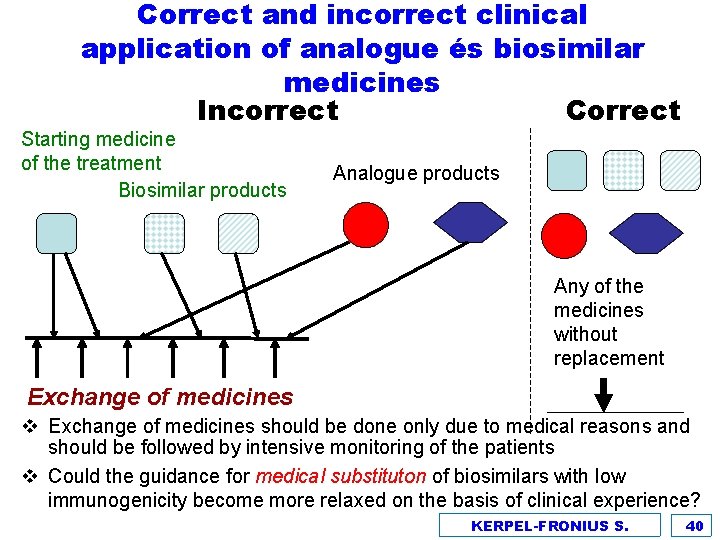
Correct and incorrect clinical application of analogue és biosimilar medicines Incorrect Correct Starting medicine of the treatment Biosimilar products Analogue products Any of the medicines without replacement Exchange of medicines v Exchange of medicines should be done only due to medical reasons and should be followed by intensive monitoring of the patients v Could the guidance for medical substituton of biosimilars with low immunogenicity become more relaxed on the basis of clinical experience? KERPEL-FRONIUS S. 40

Non-biological complex drugs (NBCD) Crommelin DJA: European Journal of Pharmaceutical Sciences 76: 10– 17, 2015 Properties v Consist of a multitude of closely related structures v The entire complex is the active pharmaceutical ingredient v The properties cannot be fully characterized by physicochemical analysis v The well-controlled, robust manufacturing process is fundamental to reproduce the product Diversity of products, different mechanisms of action Glatiramoids (mixture of peptides, copaxone), liposomes, nanomedicines, iron-carbohydrate-complexes, albuminecytostatic complexes, dry powder inhalers, ocular/intravenous emulsions, dermal patches, etc. Differences in safety: severity and frequency of side effects KERPEL-FRONIUS S. 41

Biosimilar development of enoxaparin sodium EPAR: Inhixa, Thorinane EMA/536977/2016 KERPEL-FRONIUS S. 42
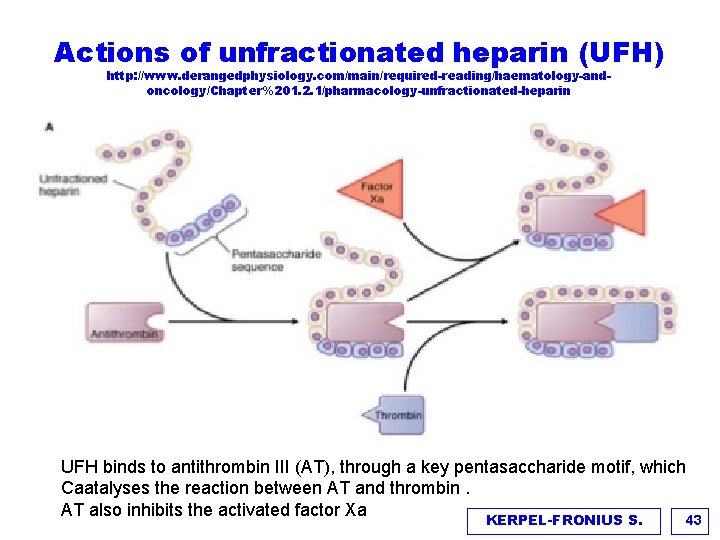
Actions of unfractionated heparin (UFH) http: //www. derangedphysiology. com/main/required-reading/haematology-andoncology/Chapter%201. 2. 1/pharmacology-unfractionated-heparin UFH binds to antithrombin III (AT), through a key pentasaccharide motif, which Caatalyses the reaction between AT and thrombin. AT also inhibits the activated factor Xa KERPEL-FRONIUS S. 43
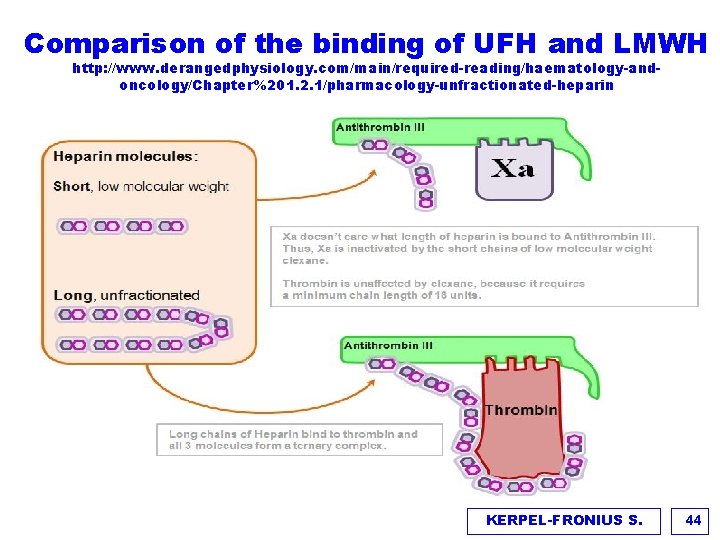
Comparison of the binding of UFH and LMWH http: //www. derangedphysiology. com/main/required-reading/haematology-andoncology/Chapter%201. 2. 1/pharmacology-unfractionated-heparin KERPEL-FRONIUS S. 44
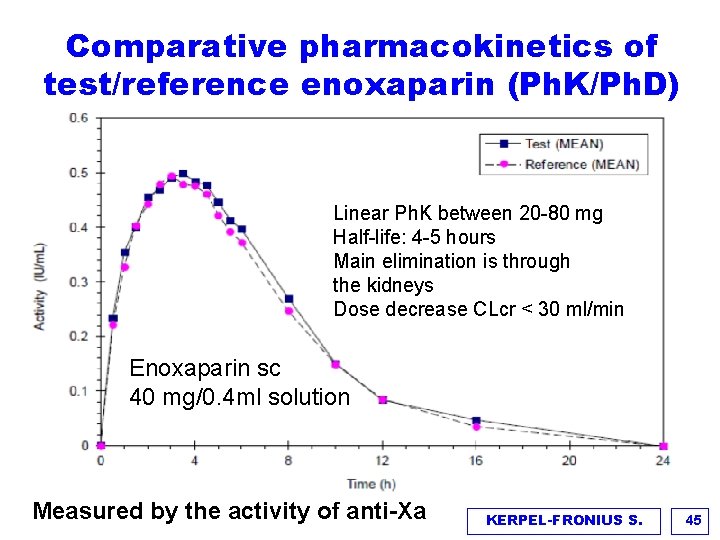
Comparative pharmacokinetics of test/reference enoxaparin (Ph. K/Ph. D) Linear Ph. K between 20 -80 mg Half-life: 4 -5 hours Main elimination is through the kidneys Dose decrease CLcr < 30 ml/min Enoxaparin sc 40 mg/0. 4 ml solution Measured by the activity of anti-Xa KERPEL-FRONIUS S. 45
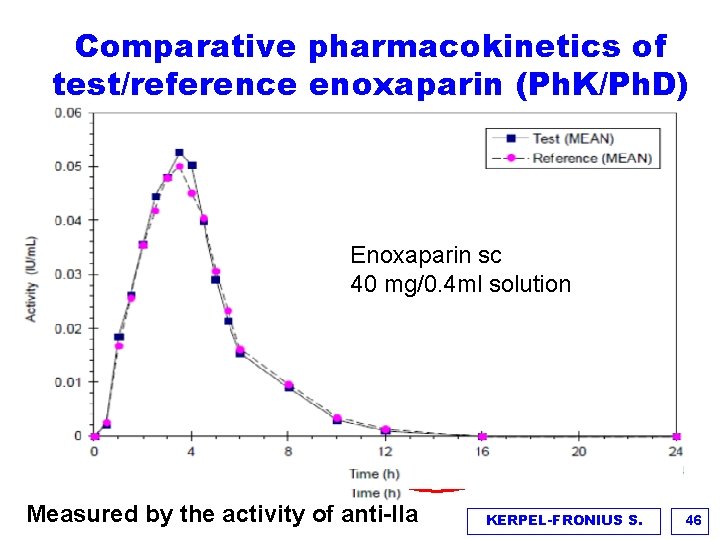
Comparative pharmacokinetics of test/reference enoxaparin (Ph. K/Ph. D) Enoxaparin sc 40 mg/0. 4 ml solution Inaccurate measurements Measured by the activity of anti-IIa KERPEL-FRONIUS S. 46
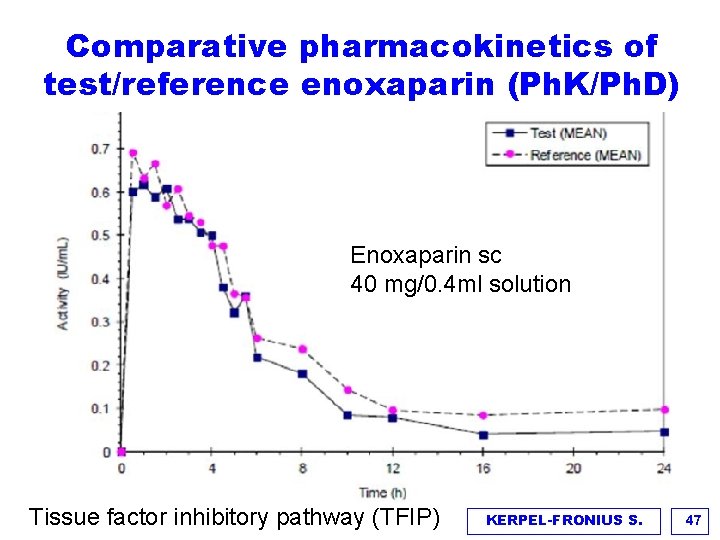
Comparative pharmacokinetics of test/reference enoxaparin (Ph. K/Ph. D) Enoxaparin sc 40 mg/0. 4 ml solution Tissue factor inhibitory pathway (TFIP) KERPEL-FRONIUS S. 47
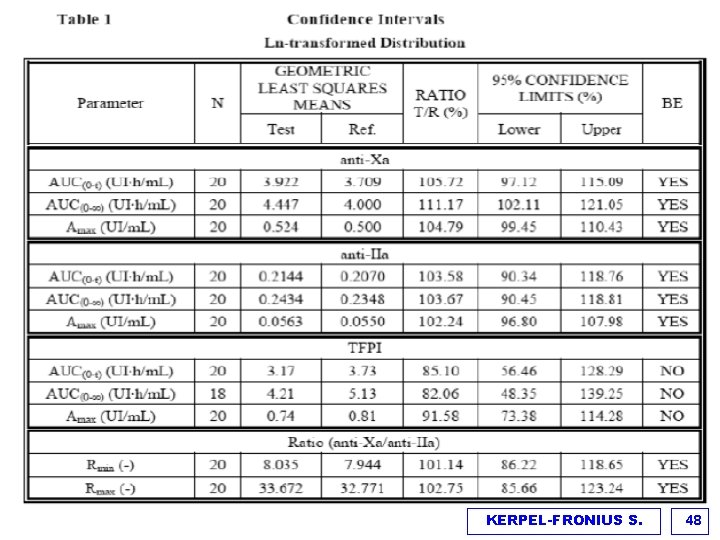
KERPEL-FRONIUS S. 48
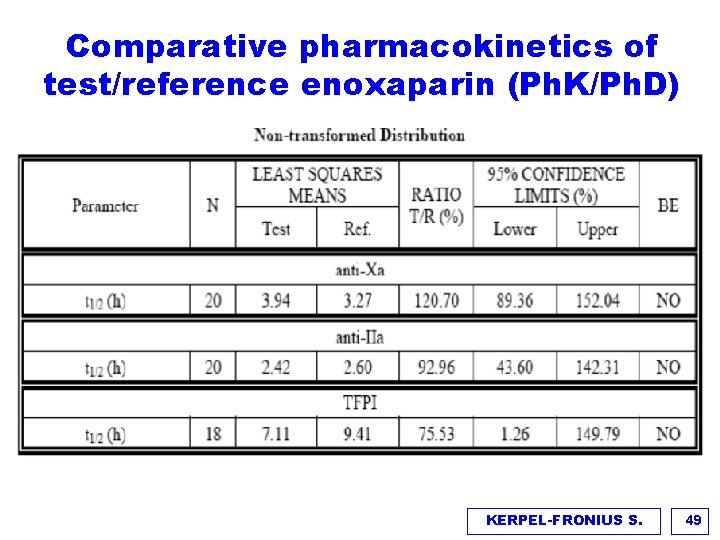
Comparative pharmacokinetics of test/reference enoxaparin (Ph. K/Ph. D) KERPEL-FRONIUS S. 49

Comparative pharmacokinetics of test/reference enoxaparin (Ph. K/Ph. D) Outcomes and endpoints v Primary endpoints: AUC (0 -t) and Amax of anti-Xa and anti-Iia activity v Equivalence criteria: 90% (95%) confidence intervals for ln transformed T/R ratios have to lie between 0. 80 and 1. 25% v Secondary PK/PD parameters: TFIP, Rmin, Rmax, tmin, tmax of anti-Xa/anti-II activity ratio Results v t 1/2 did not fulfill biosimilarity criteria, however the differences of t 1/2 between T&R enoxaparins for Anti-Xa and Anti-IIa were 40 min and 11 min which were not considered clinically meaningful. (Variability was due to inaccuracy of measuring the points at the terminal elimination phase) v Only 20% of the polysaccharide mixture inhibits Xa and Iia. The molecular mass distribution is similar in T & R enoxaparin v TFPI: indirect parameter representing endothelial function, endogenous TFPI and circadian rhythm makes measurements difficult and less reliable KERPEL-FRONIUS S. 50

In vitro immunogenicity testing of T & R enoxaparins Immunogenicity strategy v Prevalence of antibody formation is 1 -10% v Risk of heparin induced thrombopenia and thrombosis (HITT) 0. 2%. Clinical immunogenicity testing is not practical v Immunogenicity was tested by comparing in vitro models for human immunity Ø in vitro formation of PF 4 -enoxaparin (platelet factor 4) complexes using T & R enoxaparin Ø In-vitro PBMC model: PF-4 + enoxaparin, CD 83 marker of APC maturation was used as indicator KERPEL-FRONIUS S. 51

In vitro immunogenicity testing of T & R enoxaparins Immunogenicity strategy v Serotonin release from platelet rich samples by adding PF 4 -enoxaprin complexes v TFPI release from HUVEC after T & R enoxaparin stimultion The tests showed high variabilities and did not exhibit suitable level of product-specific discriminatory capacity, but reassuringly did not reveal differences v The stuctures of PF 4 -enoxaprin complexes did not show differences v Binding of T & R to immobilized PF 4 on plasmon showed comparable bindings by surface plasmon resonance KERPEL-FRONIUS S. 52

In vitro immunogenicity testing of T & R enoxaparins Immunogenicity strategy v Serotonin release from platelet rich samples by adding PF 4 -enoxaprin complexes v TFPI release from HUVEC after T & R enoxaparin stimultion The tests showed high variabilities and did not exhibit suitable level of product-specific discriminatory capacity, but reassuringly did not reveal differences v The stuctures of PF 4 -enoxaprin complexes did not show differences v Binding of T & R to immobilized PF 4 on plasmon showed comparable bindings by surface plasmon resonance KERPEL-FRONIUS S. 53
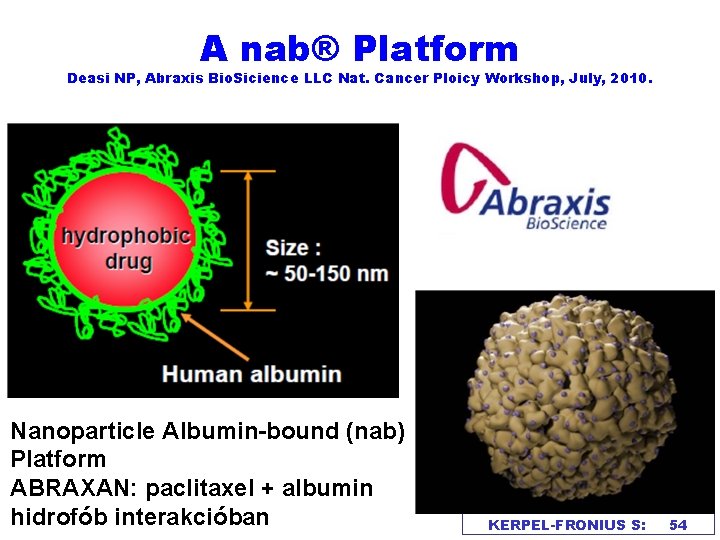
A nab® Platform Deasi NP, Abraxis Bio. Sicience LLC Nat. Cancer Ploicy Workshop, July, 2010. Nanoparticle Albumin-bound (nab) Platform ABRAXAN: paclitaxel + albumin hidrofób interakcióban KERPEL-FRONIUS S: 54
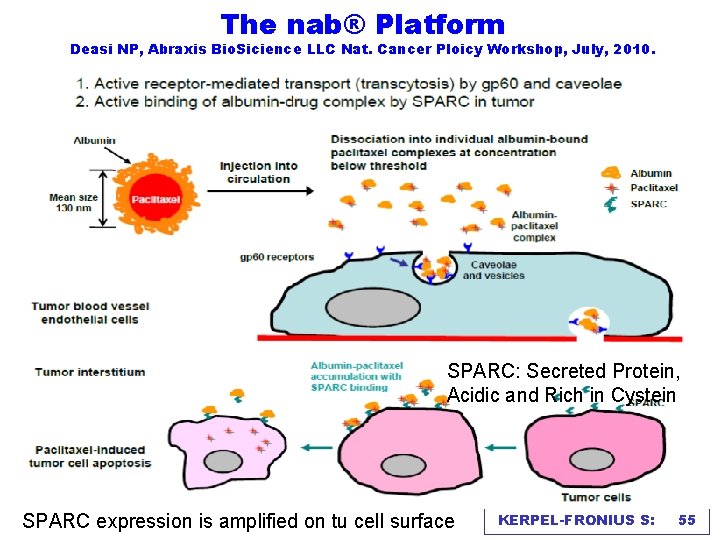
The nab® Platform Deasi NP, Abraxis Bio. Sicience LLC Nat. Cancer Ploicy Workshop, July, 2010. SPARC: Secreted Protein, Acidic and Rich in Cystein SPARC expression is amplified on tu cell surface KERPEL-FRONIUS S: 55
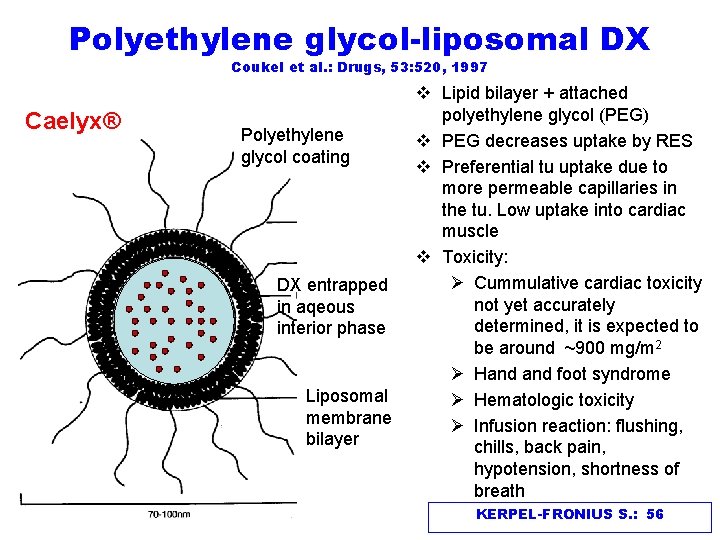
Polyethylene glycol-liposomal DX Coukel et al. : Drugs, 53: 520, 1997 Caelyx® Polyethylene glycol coating DX entrapped in aqeous interior phase Liposomal membrane bilayer v Lipid bilayer + attached polyethylene glycol (PEG) v PEG decreases uptake by RES v Preferential tu uptake due to more permeable capillaries in the tu. Low uptake into cardiac muscle v Toxicity: Ø Cummulative cardiac toxicity not yet accurately determined, it is expected to be around ~900 mg/m 2 Ø Hand foot syndrome Ø Hematologic toxicity Ø Infusion reaction: flushing, chills, back pain, hypotension, shortness of breath KERPEL-FRONIUS S. : 56

Interchangeability of non-biological complex drugs Nanoparticle albumine-bound platform (nab) v. The recommendations for interchangeability might be expected to be made case by case or group by group. v. Beside proving equivalent therapeutic effect, the evaluation of interchangeability will most probably depend on the expected variable medical risk caused by the repeated substitution of the different complex medicinal agents KERPEL-FRONIUS S. 57

Interchangeability of follow-on medicinal products CI limits of Innovative and followthe bioon medicinal availability products ratio Innovative and generic drugs with narrow therapeutic index Innovative biological and biosimilar medicinal products Innovative complex and non-biological similar medicinal products 90% Professionals performing Method of interchange inter-change of medicinal products Pharmacist Simple interor treating change physician Production of antibodies following interchange No 95% Dose adjustment needed Treating physician No 90% Only if medically indicated Treating physician Frequent according to molecule 90% Pharmacist Variable or physician according to Might occur according to safety risk category KERPEL-FRONIUS S. 58

The application of biological medicinal products in the EU Marketing authorization Efficacy, safety, quality Risk management plan The use of biological medicinal products not registered in the EU is not permitted and additionally might be dangerous „Drug fortress European Union”
- Slides: 59