Interatomic bonding 1 Bonding forces of atoms All

Interatomic bonding 1

Bonding forces of atoms All forces playing role in bonding are electrostatic Coulomb forces. Nuclei attract electrons, but nuclei repulse each other as well as electrons do. So, bonding is rather complicated, it is a many-body system, exact solution can be found by the Schrödinger equation only. 2
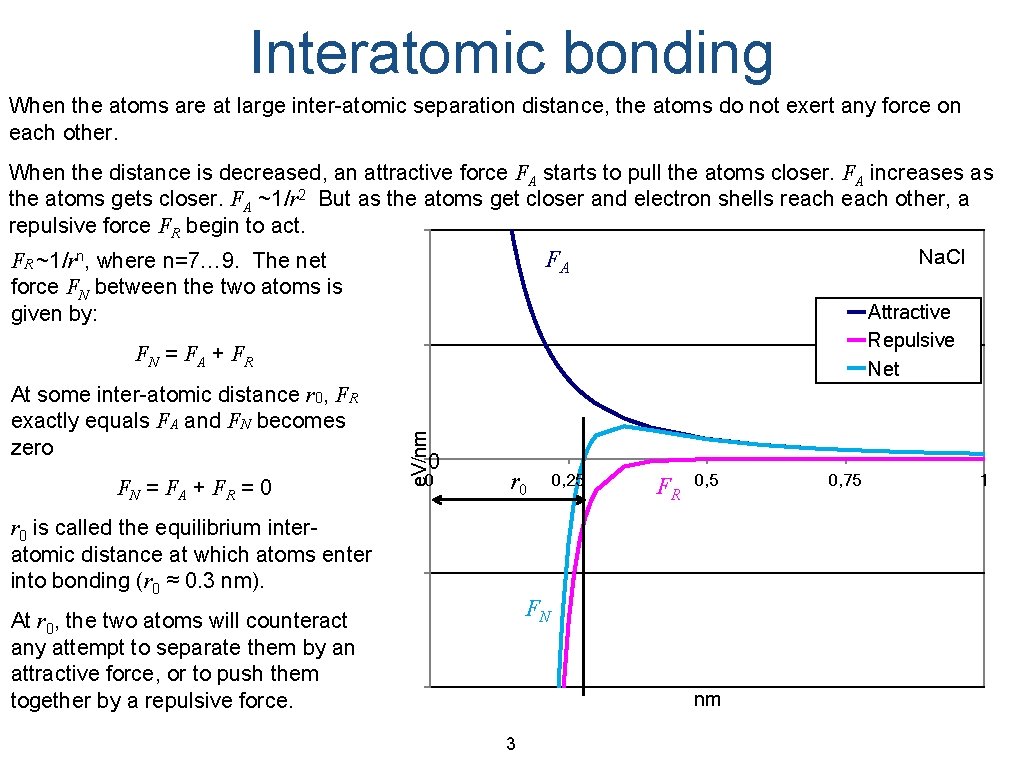
Interatomic bonding When the atoms are at large inter-atomic separation distance, the atoms do not exert any force on each other. When the distance is decreased, an attractive force FA starts to pull the atoms closer. FA increases as the atoms gets closer. FA ~1/r 2 But as the atoms get closer and electron shells reach other, a repulsive force FR begin to act. Na. Cl FA FR ~1/rn, where n=7… 9. The net force FN between the two atoms is Attractive given by: Repulsive Net At some inter-atomic distance r 0, FR exactly equals FA and FN becomes zero FN = FA + FR = 0 e. V/nm FN = FA + FR 0 0 r 0 is called the equilibrium interatomic distance at which atoms enter into bonding (r 0 ≈ 0. 3 nm). 0, 25 FR 0, 5 FN At r 0, the two atoms will counteract any attempt to separate them by an attractive force, or to push them together by a repulsive force. nm 3 0, 75 1
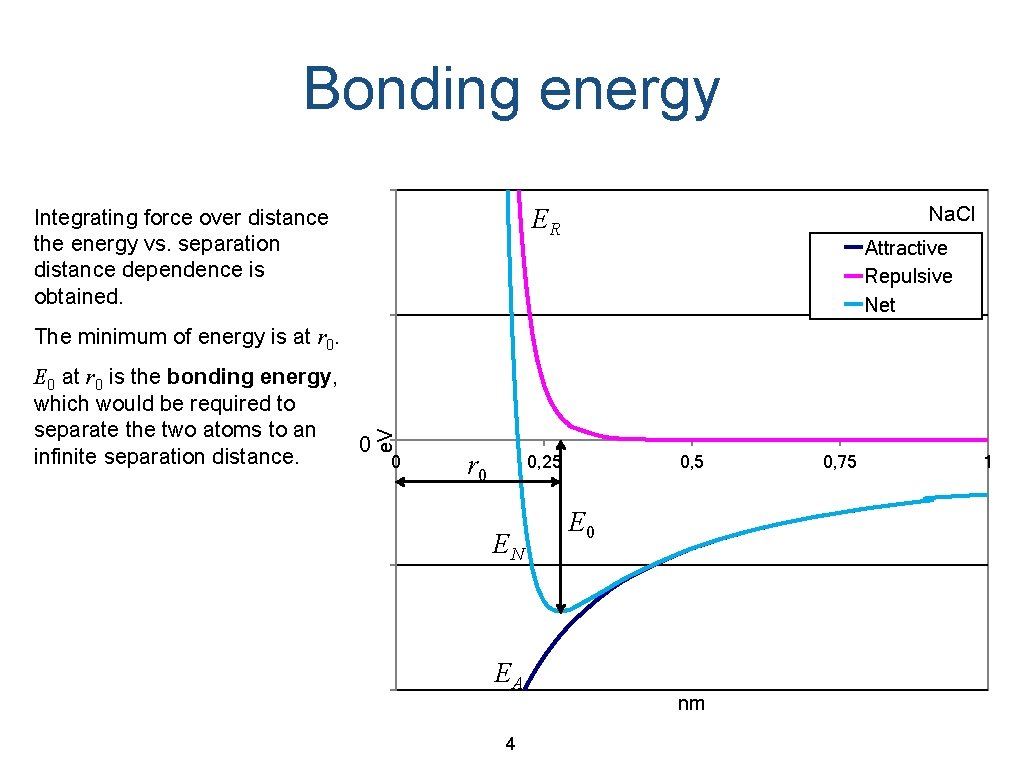
Bonding energy Na. Cl ER Integrating force over distance the energy vs. separation distance dependence is obtained. Attractive Repulsive Net The minimum of energy is at r 0. e. V E 0 at r 0 is the bonding energy, which would be required to separate the two atoms to an 0 infinite separation distance. 0 r 0 0, 25 EN EA 4 0, 5 E 0 nm 0, 75 1

Types of bonding Primary bonding Atoms with incomplete outer shells strive to reach the noble gas by sharing or transferring electrons among each other for maximal stability - primary bonding. Strong (100 -1000 k. J/mol or 1 -10 e. V/atom) • ionic bonding • covalent bonding • metallic bonding Secondary bonding No electron transferred or shared Weak (< 100 k. J/mol or < 1 e. V/atom) • van der Waals (dipole) bonding • hydrogen bonding 5

Ionic Bonding Callister: Fundamentals of Material Science and Engineering, 4 th Edition, Fig. 2 -6 Ionic Bonding is typical for elements that are situated at the horizontal extremities of the periodic table - large difference in electronegativity. Atoms from the left (metals) give up their valence electrons to the (non-metallic) atoms from the right that get one or a few electrons to acquire stable or noble gas electron configuration. As a result of this transfer mutual ionization occurs: atom that gives up electron(s) becomes positively charged ion (cation), atom that accepts electron(s) becomes negatively charged ion (anion). Ions are attracted by strong Coulomb interaction. (Oppositely charged atoms attract each other. ) An ionic bond is non-directional (ions may be attracted to another in any direction). Callister: Fundamentals of Material Science and Engineering, 4 th Edition, Fig. 2 -7 As electrons are bonded to nuclei, ionic materials are good electrical and thermal insulators. Ceramics are mostly ionic. 6

Ionic bonding Example: rock salt (Na. Cl) Na has 11 electrons, 1 more than needed for a full outer shell (Neon) Cl has 17 electron, 1 less than needed for a full outer shell (Argon) Electron transfer reduces the energy of the system of atoms, that is, electron transfer is energetically favorable. Note relative sizes of ions: Na shrinks and Cl expands. 7
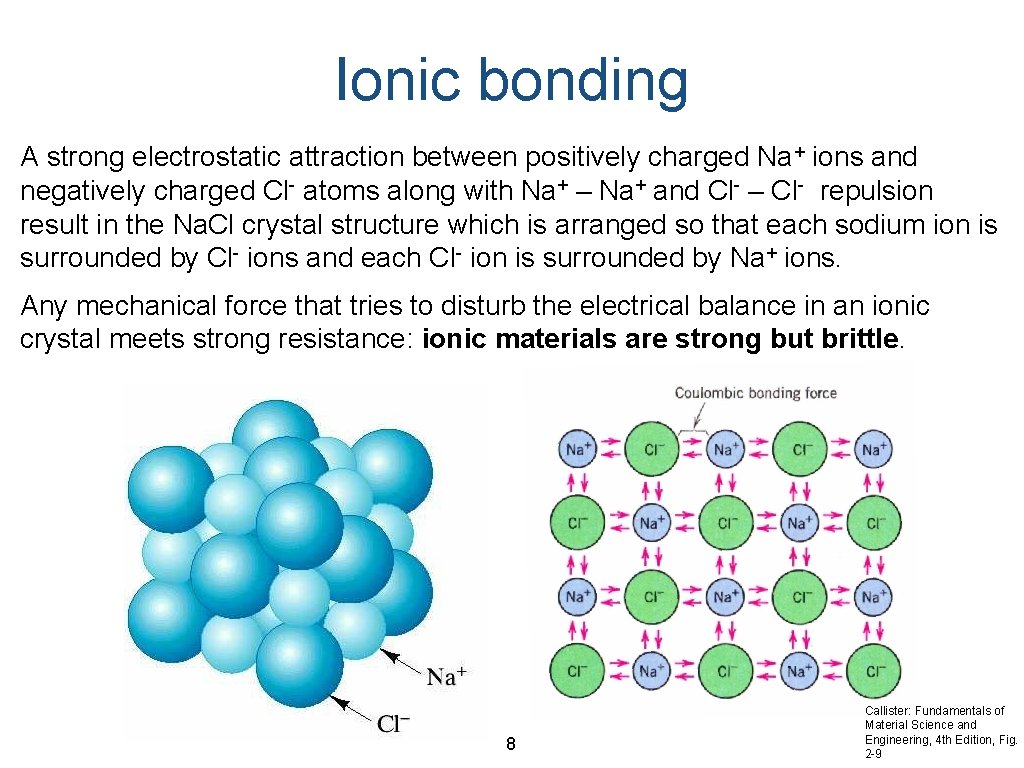
Ionic bonding A strong electrostatic attraction between positively charged Na+ ions and negatively charged Cl- atoms along with Na+ – Na+ and Cl- – Cl- repulsion result in the Na. Cl crystal structure which is arranged so that each sodium ion is surrounded by Cl- ions and each Cl- ion is surrounded by Na+ ions. Any mechanical force that tries to disturb the electrical balance in an ionic crystal meets strong resistance: ionic materials are strong but brittle. 8 Callister: Fundamentals of Material Science and Engineering, 4 th Edition, Fig. 2 -9

Metallic bonding Metallic atoms have at most 3 valence electrons. In the course of bonding valence electrons are detached from atoms, and spread in an 'electron sea' that 'glues' the positive ions together. They belong to the metal as a whole. The remaining non valence electrons and the atomic nuclei form ion cores, which are positively charged. A metallic bond is non-directional (bonds form in any direction), atoms pack closely. The bonds do not break when atoms are rearranged – metals can experience a significant degree of plastic deformation, are ductile as compared to ionic materials. Examples of typical metallic bonding: Cu, Al, Au, Ag. Transition metals (Fe, Ni, etc. ) form mixed bonds that are comprising of metallic bonds and covalent bonds involving their 3 d-electrons. As a result the transition metals are more brittle. Metallic bonds may be strong or weak with energies ranging from 68 k. J/mole (0. 7 e. V/atom) to 850 k. J/mole (8. 8 e. V/atom). Metals are good conductors of electricity and heat (as a consequence of their free electrons). 9 Callister: Fundamentals of Material Science and Engineering, 4 th Edition, Fig 2 -11
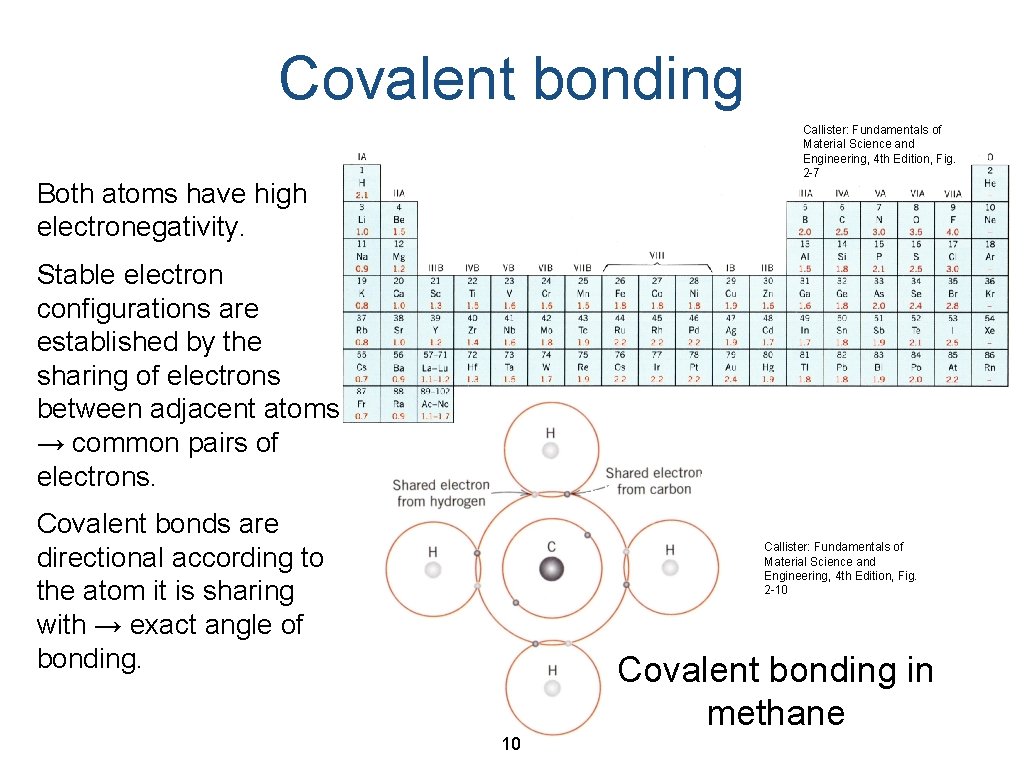
Covalent bonding Callister: Fundamentals of Material Science and Engineering, 4 th Edition, Fig. 2 -7 Both atoms have high electronegativity. Stable electron configurations are established by the sharing of electrons between adjacent atoms → common pairs of electrons. Covalent bonds are directional according to the atom it is sharing with → exact angle of bonding. Callister: Fundamentals of Material Science and Engineering, 4 th Edition, Fig. 2 -10 Covalent bonding in methane 10

Covalent bonding The number of covalent bonds possible for an atom depends on the number of valence electrons NV, i. e. , an atom can covalently bound with at most 8 -NV other atoms. Covalent, non-polar bond But in a covalent bonding maximum 3 electron pairs can take part. Covalent bonds can be very strong as in diamond, which is very hard and has a high melting point > 3550°C or it can be very week. Polymeric materials are covalent bonded. Covalent, polar bond Most compounds are partially ionic and partially covalent. Covalent bonding can be polar or non-polar. The greater the difference in electronegativity the more ionic the bonds. 11
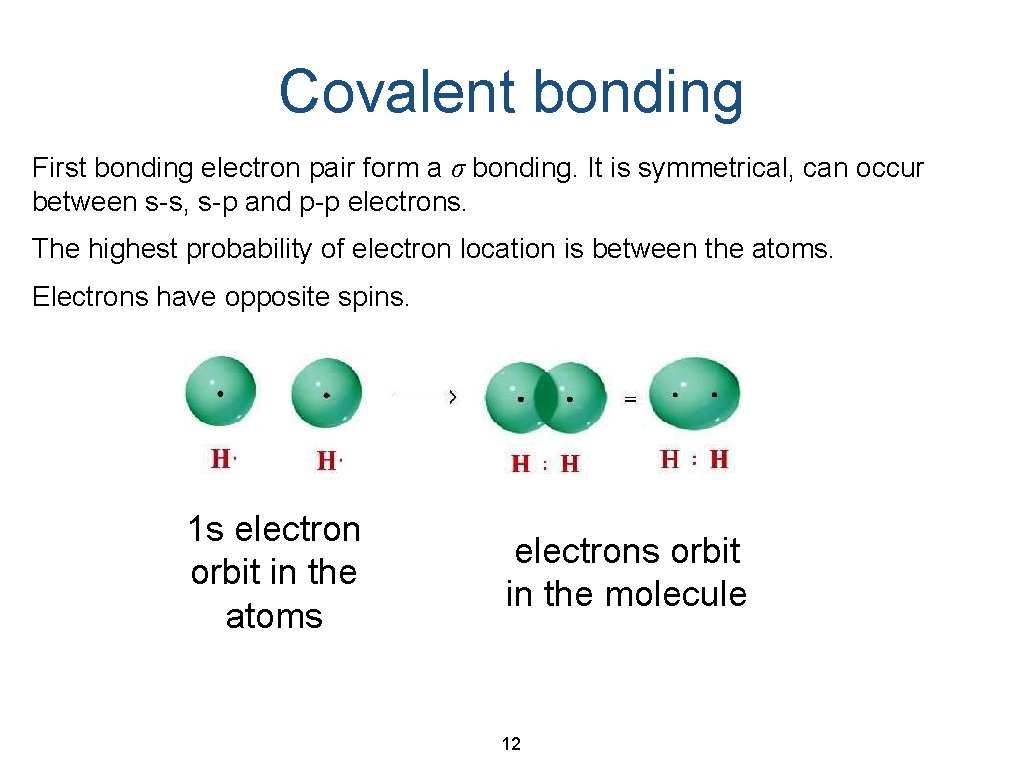
Covalent bonding First bonding electron pair form a σ bonding. It is symmetrical, can occur between s-s, s-p and p-p electrons. The highest probability of electron location is between the atoms. Electrons have opposite spins. 1 s electron orbit in the atoms electrons orbit in the molecule 12
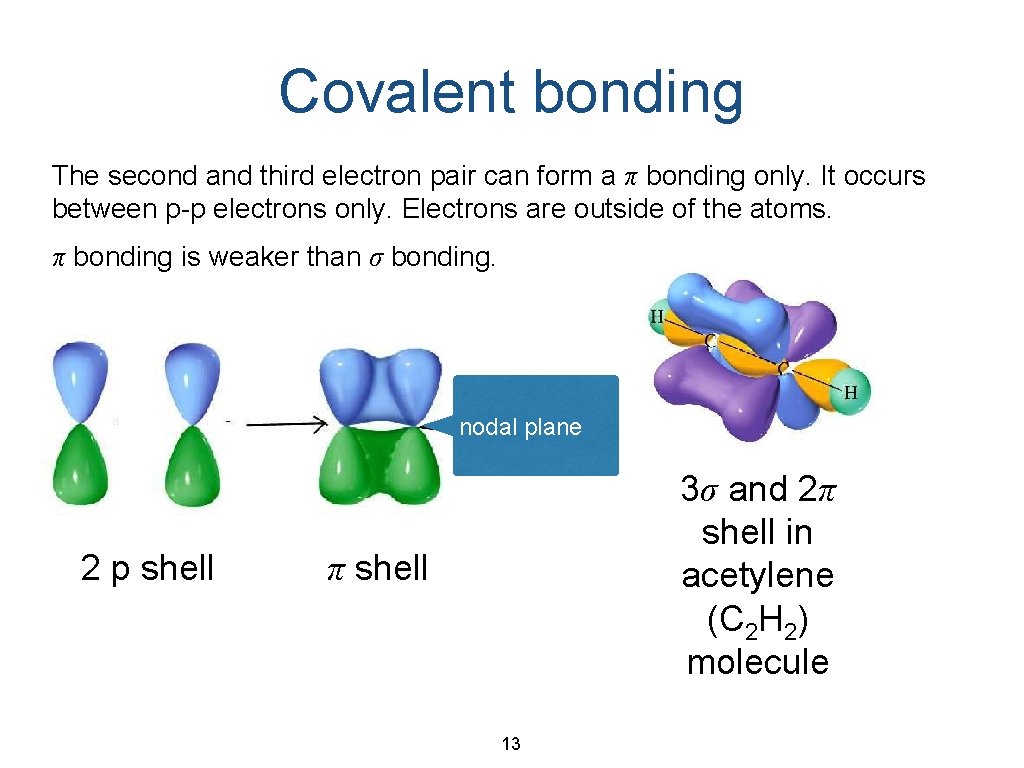
Covalent bonding The second and third electron pair can form a π bonding only. It occurs between p-p electrons only. Electrons are outside of the atoms. π bonding is weaker than σ bonding. nodal plane 2 p shell 3σ and 2π shell in acetylene (C 2 H 2) molecule π shell 13
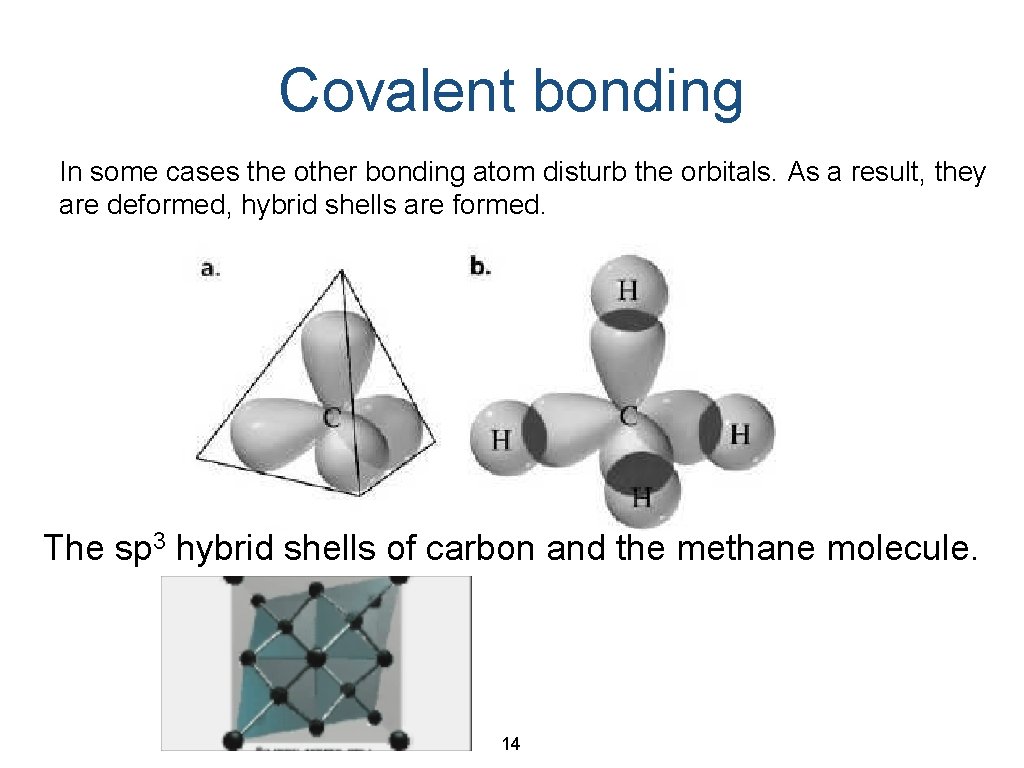
Covalent bonding In some cases the other bonding atom disturb the orbitals. As a result, they are deformed, hybrid shells are formed. The sp 3 hybrid shells of carbon and the methane molecule. 14
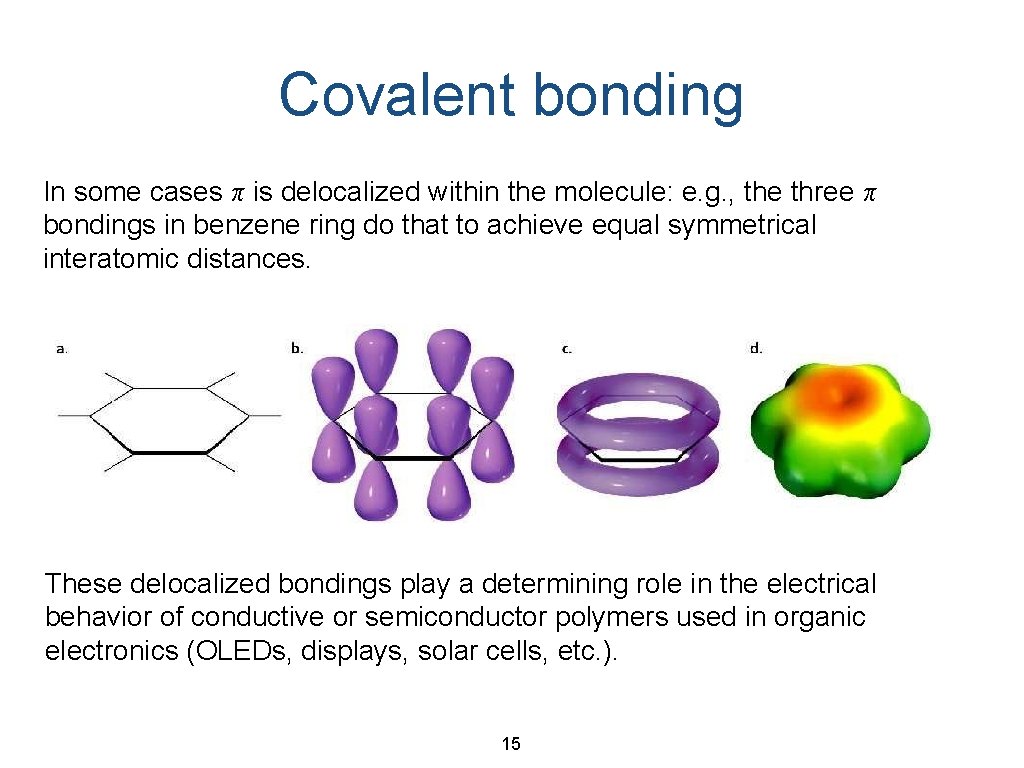
Covalent bonding In some cases π is delocalized within the molecule: e. g. , the three π bondings in benzene ring do that to achieve equal symmetrical interatomic distances. These delocalized bondings play a determining role in the electrical behavior of conductive or semiconductor polymers used in organic electronics (OLEDs, displays, solar cells, etc. ). 15
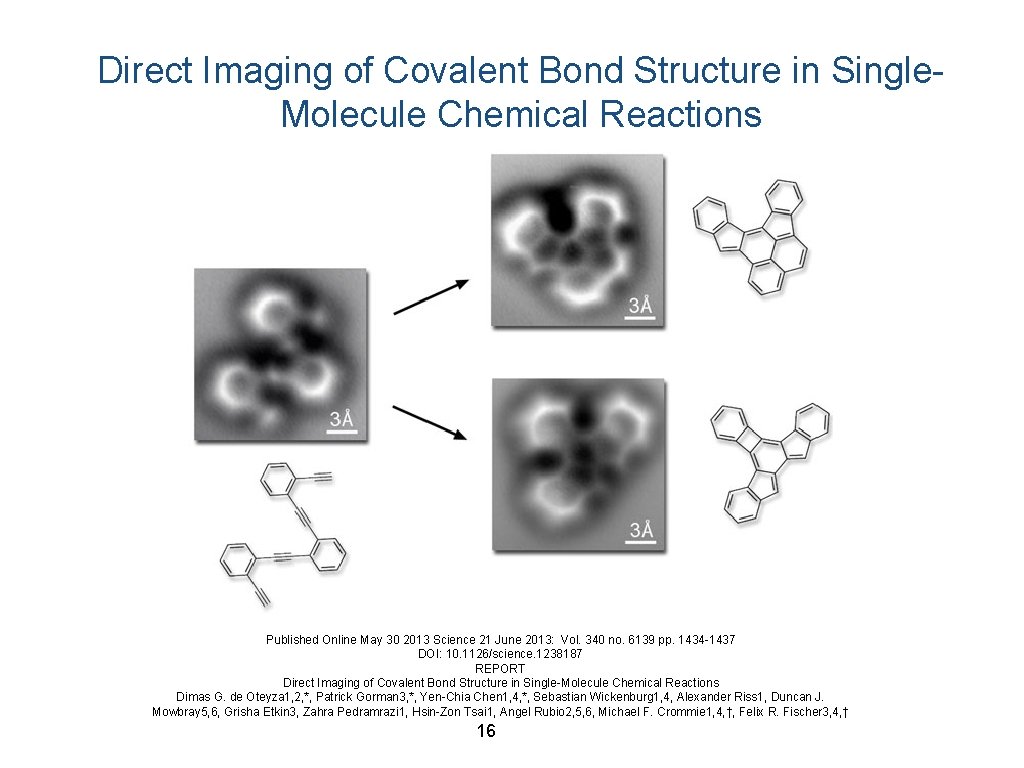
Direct Imaging of Covalent Bond Structure in Single. Molecule Chemical Reactions Published Online May 30 2013 Science 21 June 2013: Vol. 340 no. 6139 pp. 1434 -1437 DOI: 10. 1126/science. 1238187 REPORT Direct Imaging of Covalent Bond Structure in Single-Molecule Chemical Reactions Dimas G. de Oteyza 1, 2, *, Patrick Gorman 3, *, Yen-Chia Chen 1, 4, *, Sebastian Wickenburg 1, 4, Alexander Riss 1, Duncan J. Mowbray 5, 6, Grisha Etkin 3, Zahra Pedramrazi 1, Hsin-Zon Tsai 1, Angel Rubio 2, 5, 6, Michael F. Crommie 1, 4, †, Felix R. Fischer 3, 4, † 16

Secondary bonding Secondary = van der Waals = physical (as opposite to chemical bonding that involves electron transfer) bonding results from interaction of atomic or molecular dipoles. It is weak, ~0. 1 e. V/atom or ~10 k. J/mol. Permanent dipole bonds Permanent dipole moments exist in some molecules (called polar molecules) due to the asymmetrical arrangement of positively and negatively charged regions (HCl, H 2 O). Bonds between adjacent polar molecules – permanent dipole bonds – are the strongest among secondary bonds. 17

Secondary bonding Induced dipole bonds Polar molecules can induce dipoles in adjacent non-polar molecules and bond is formed due to the attraction between the permanent and induced dipoles. Fluctuating Induced Dipole Even in electrically symmetric molecules/atoms an electric dipole can be created by fluctuations of electron density distribution. Fluctuating electric field in one atom is felt by the electrons of an adjacent atom, and induce a dipole momentum in the adjacent atom. This bond due to fluctuating induced dipoles is the weakest one (inert gases, H 2, Cl 2 molecules). 18
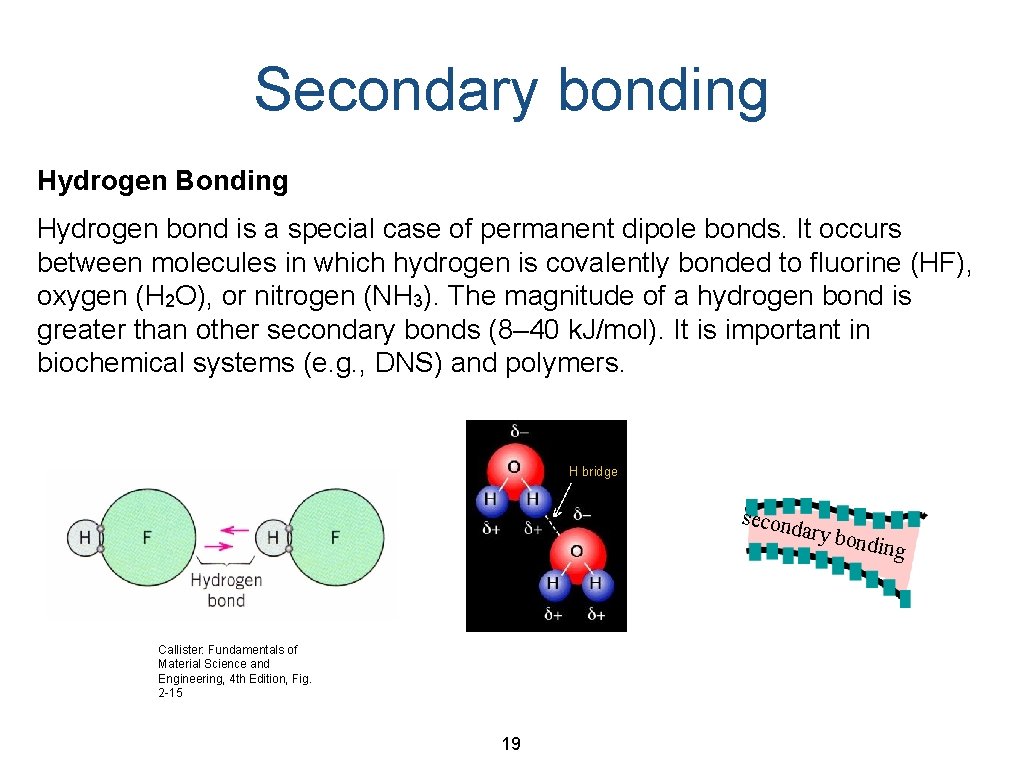
Secondary bonding Hydrogen Bonding Hydrogen bond is a special case of permanent dipole bonds. It occurs between molecules in which hydrogen is covalently bonded to fluorine (HF), oxygen (H 2 O), or nitrogen (NH 3). The magnitude of a hydrogen bond is greater than other secondary bonds (8– 40 k. J/mol). It is important in biochemical systems (e. g. , DNS) and polymers. H bridge second ary bo Callister: Fundamentals of Material Science and Engineering, 4 th Edition, Fig. 2 -15 19 nding

Real-Space Identification of Intermolecular Bonding with Atomic Force Microscopy Published Online September 26 2013 Science DOI: 10. 1126/science. 1242603 REPORT Real-Space Identification of Intermolecular Bonding with Atomic Force Microscopy Jun Zhang 1, *, Pengcheng Chen 1, *, Bingkai Yuan 1, Wei Ji 2, †, Zhihai Cheng 1, †, Xiaohui Qiu 1, † 20
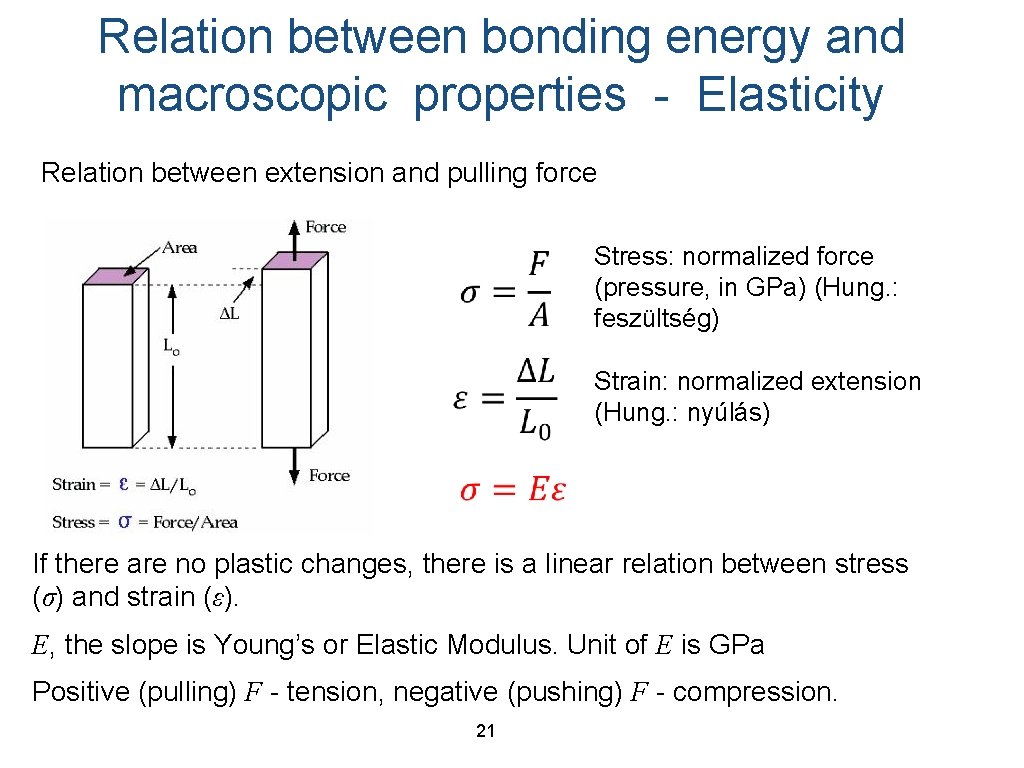
Relation between bonding energy and macroscopic properties - Elasticity Relation between extension and pulling force Stress: normalized force (pressure, in GPa) (Hung. : feszültség) Strain: normalized extension (Hung. : nyúlás) If there are no plastic changes, there is a linear relation between stress (σ) and strain (ε). E, the slope is Young’s or Elastic Modulus. Unit of E is GPa Positive (pulling) F - tension, negative (pushing) F - compression. 21
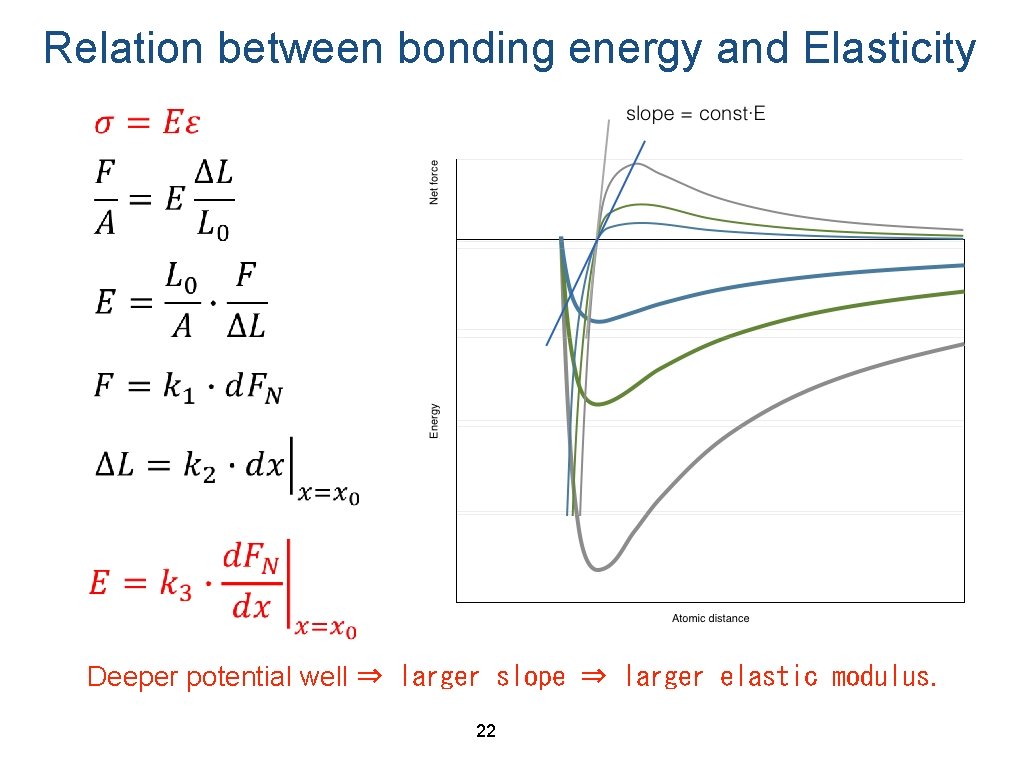
Relation between bonding energy and Elasticity Deeper potential well ⇒ larger slope ⇒ larger elastic modulus. 22
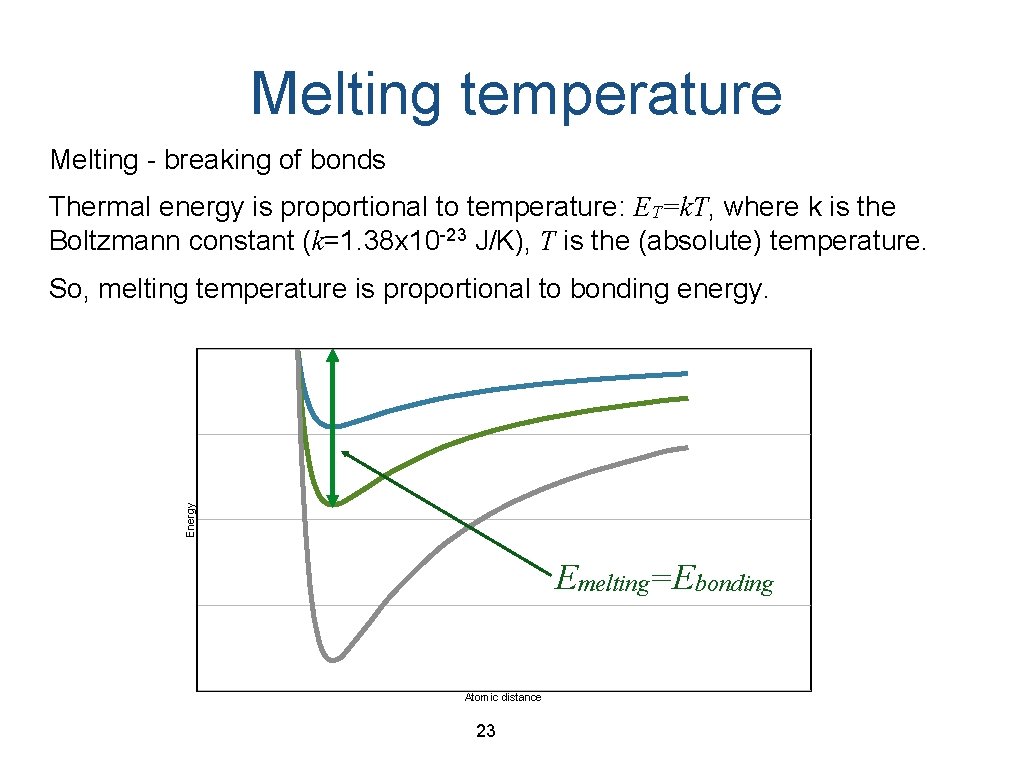
Melting temperature Melting - breaking of bonds Thermal energy is proportional to temperature: ET=k. T, where k is the Boltzmann constant (k=1. 38 x 10 -23 J/K), T is the (absolute) temperature. Energy So, melting temperature is proportional to bonding energy. Emelting=Ebonding Atomic distance 23
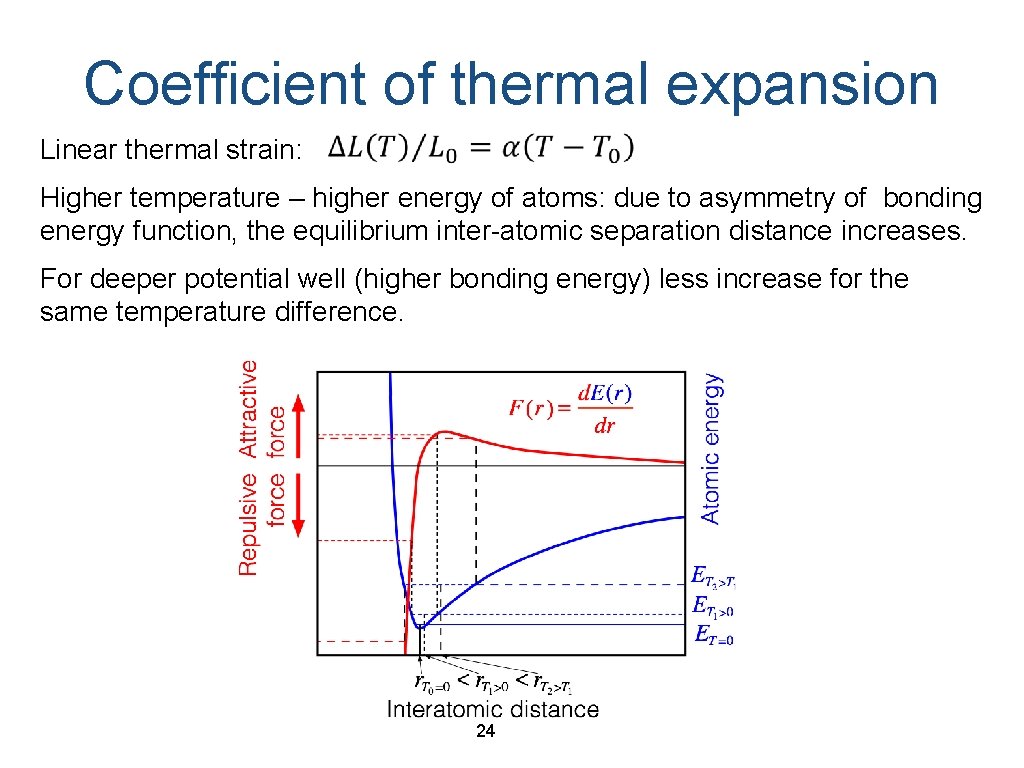
Coefficient of thermal expansion Linear thermal strain: Higher temperature – higher energy of atoms: due to asymmetry of bonding energy function, the equilibrium inter-atomic separation distance increases. For deeper potential well (higher bonding energy) less increase for the same temperature difference. 24
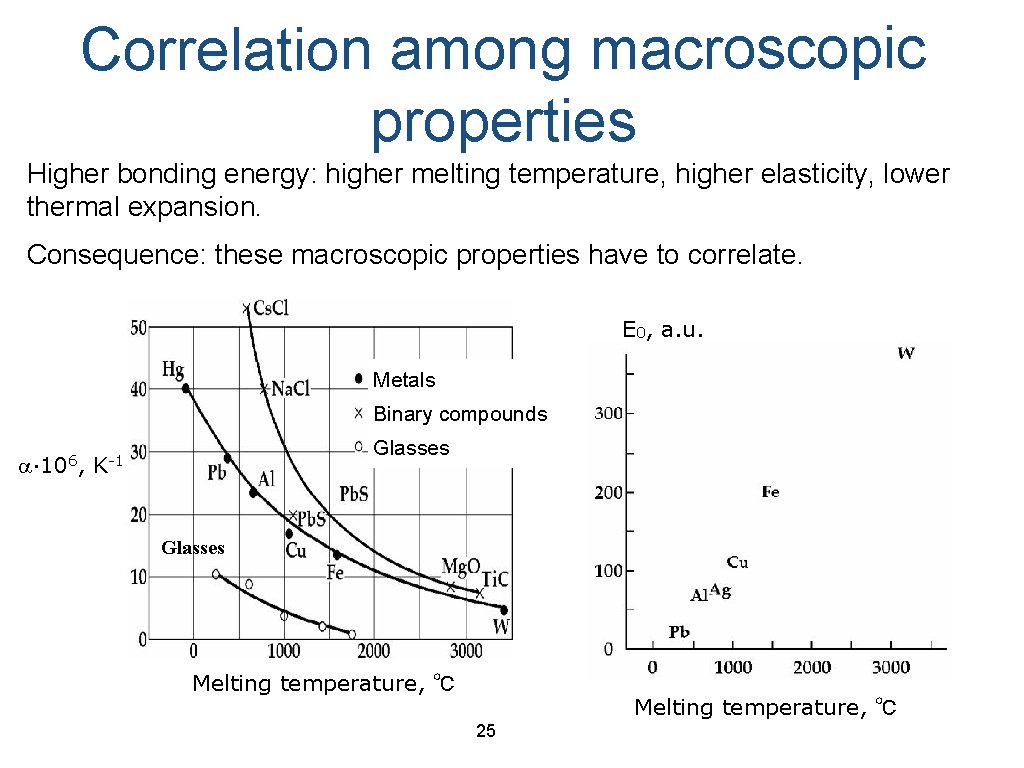
Correlation among macroscopic properties Higher bonding energy: higher melting temperature, higher elasticity, lower thermal expansion. Consequence: these macroscopic properties have to correlate. E 0, a. u. Metals Binary compounds a∙ 106, Glasses K-1 Glasses Melting temperature, ℃ 25
- Slides: 25