Interactions of Charged Particles with Matter Nick Harding





















- Slides: 38

Interactions of Charged Particles with Matter Nick Harding Clinical Scientist Radiotherapy Department Castle Hill Hospital Hull & East Yorkshire Hospitals NHS Trust email: nicholas. harding@hey. nhs. uk

CHARGED PARTICLES § Charged particles (e. g. α+2, β±, p+) interact with matter electromagnetically (i. e. via Coulombic forces) § They lose energy via: 1. Excitation; 2. Ionisation; 3. Radiative losses (electrons/positrons only). § Most interactions are with the electron cloud (due to small size of nucleus);

INDIVIDUAL INTERACTIONS §Charged particles § suffer many interactions along their path § energy loss considered a continuous process §At each interaction § charged particles are deflected/scattered §Charged particles may pass near a nucleus § undergoes larger deflection § most pronounced for light particles

• Introduce concepts of: • • • Elastic Collisions Inelastic Collisions Specific Ionisation Linear Energy Transfer Range Path

TYPES OF RADIATION • • Radiation Mass Electric Charge Speed α 4 +2 15, 000 km/s β 1/1800 ± 1 270, 000 km/s γ/X 0 0 300, 000 km/s α – radiation: Helium nuclei β – radiation: Negatrons (electrons) and positrons γ/X – radiation: Electromagnetic radiation Other forms of charged particles e. g. protons and heavy-ions. Considered similar to alpha in terms of interaction (heavy charged particles).

Heavy Charged Particles • Heavy CPs lose energy primarily through ionisation and excitation of atoms. • The moving CP interacts electromagnetically with the matter it is travelling in (i. e. via Coulombic forces) ; • Most interactions are with the electron cloud; • In most interactions the heavy CP only transfers a small amount of energy and hence does not scatter • Can occasionally interact with the nucleus (elastic scatter – large deflection);

Heavy Particle Elastic Collision with Nucleus Elastic collision = Kinetic energy of colliding particles unchanged

INELASTIC COLLISIONS §Colliding particles have less energy after collision; §Ionisation of the atom §Excitation of the atom

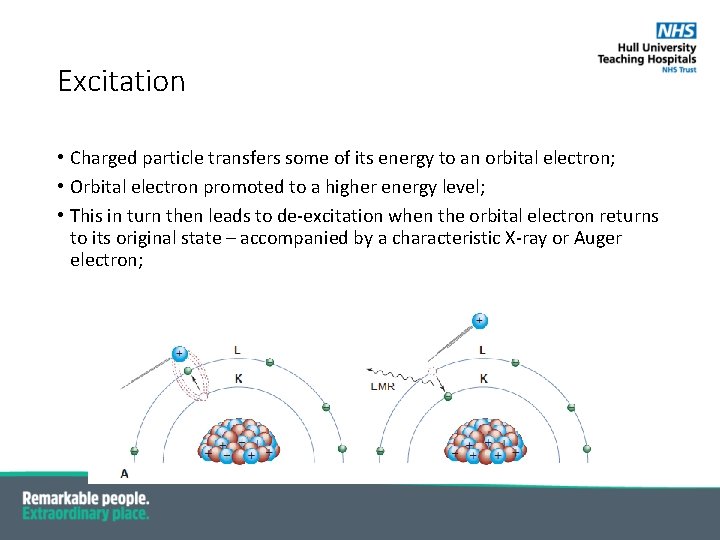
Excitation • Charged particle transfers some of its energy to an orbital electron; • Orbital electron promoted to a higher energy level; • This in turn then leads to de-excitation when the orbital electron returns to its original state – accompanied by a characteristic X-ray or Auger electron;

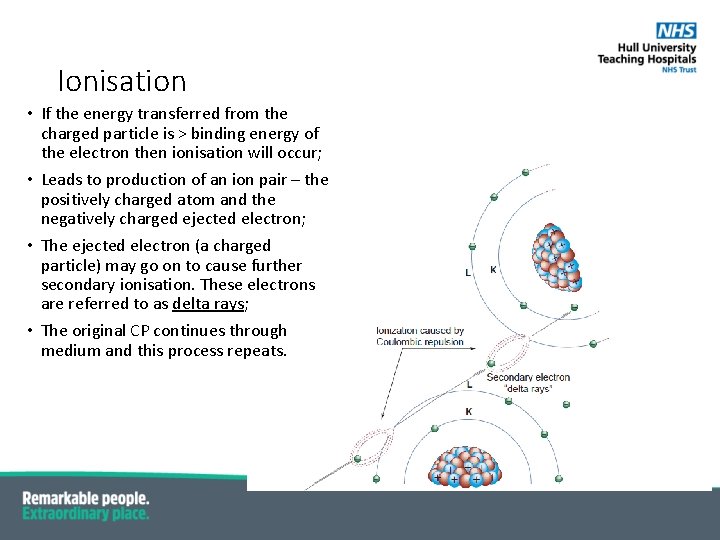
Ionisation • If the energy transferred from the charged particle is > binding energy of the electron then ionisation will occur; • Leads to production of an ion pair – the positively charged atom and the negatively charged ejected electron; • The ejected electron (a charged particle) may go on to cause further secondary ionisation. These electrons are referred to as delta rays; • The original CP continues through medium and this process repeats.

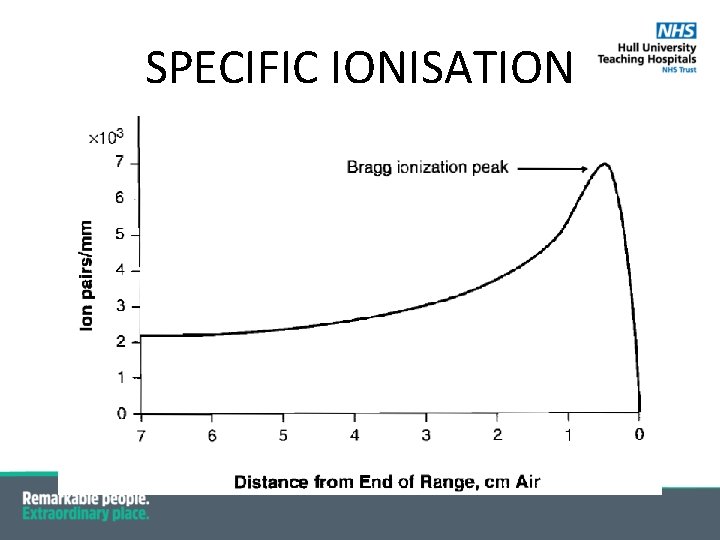
Specific Ionisation •

SPECIFIC IONISATION

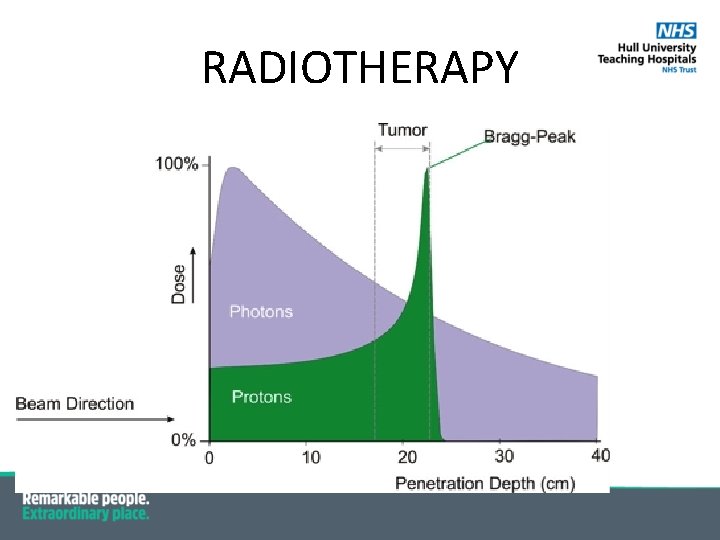
RADIOTHERAPY

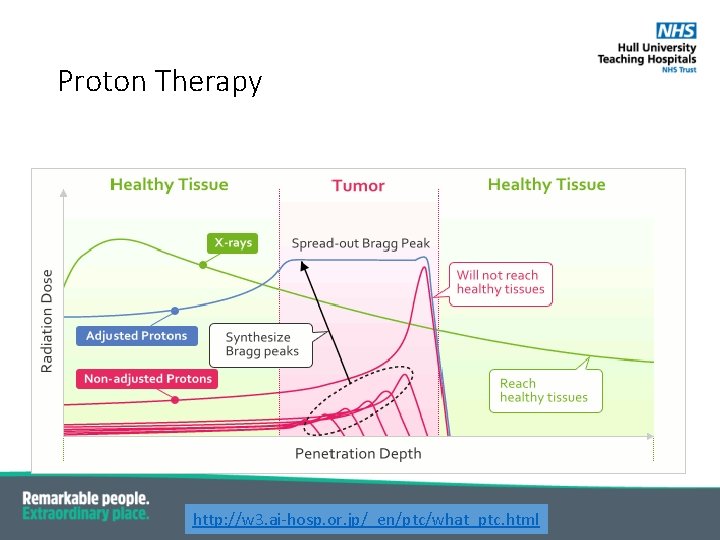
Proton Therapy http: //w 3. ai-hosp. or. jp/_en/ptc/what_ptc. html

LINEAR ENERGY TRANSFER §The linear energy transfer (LET) is defined as: § the amount of energy deposited per unit length (e. V/mm) §The LET of a particular type of radiation determines § the biologic consequence of radiation exposure § high LET radiations (α-particles, protons) § low LET radiations (electrons, γ-rays and X-rays)

PATH VERSUS RANGE § Particle path length § the actual distance the particle travels § is dependent on the mass of the particle § Particle range § the actual depth of penetration of the particle in matter § is dependent § on the mass and kinetic energy of the particle § the traversing material (i. e. atomic number Z)

PATH LENGTH (α) § a-particles § large mass particles § dense linear ionisation track § Path = Range

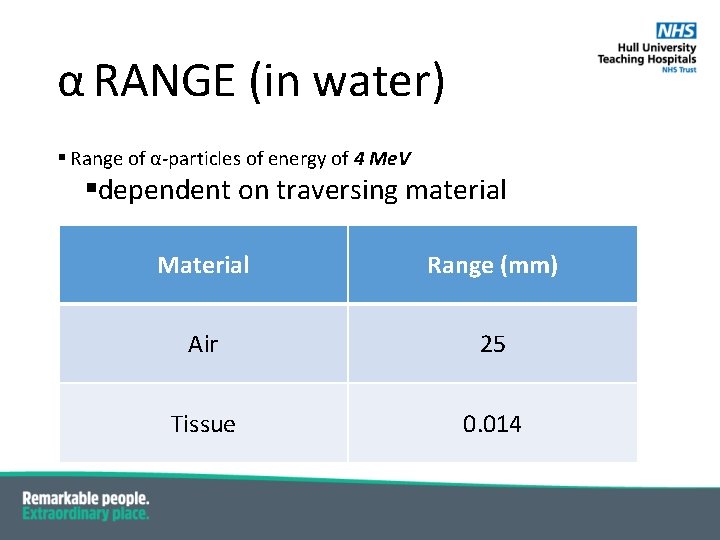
α RANGE (in water) § Range of α-particles of energy of 4 Me. V §dependent on traversing material Material Range (mm) Air 25 Tissue 0. 014

α RANGE (in water) § Range of α-particles of energy of 8 Me. V §dependent on traversing material Material Range (mm) Air 70 Tissue 0. 042

Light Charged Particles • Light = positrons and electrons; • Will refer to electrons below but same applies to positrons; • As with heavy particles, electrons can excite and ionise; • Electrons can be scattered by large angles, elastically by the electron cloud (and the nucleus) • Also undergo radiative losses - bremsstrahlung;

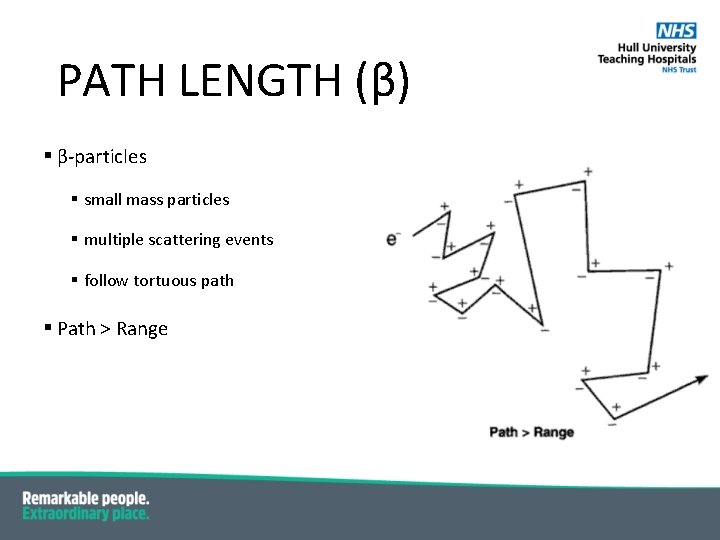
PATH LENGTH (β) § β-particles § small mass particles § multiple scattering events § follow tortuous path § Path > Range

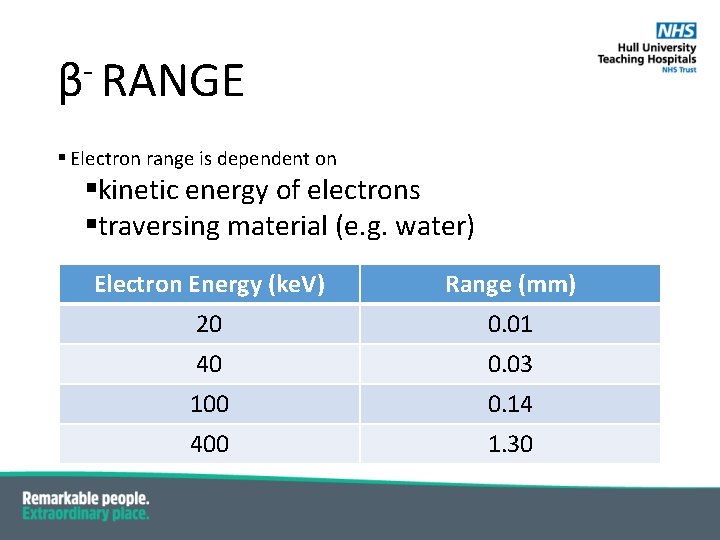
β RANGE § Electron range is dependent on §kinetic energy of electrons §traversing material (e. g. water) Electron Energy (ke. V) Range (mm) 20 0. 01 40 0. 03 100 0. 14 400 1. 30

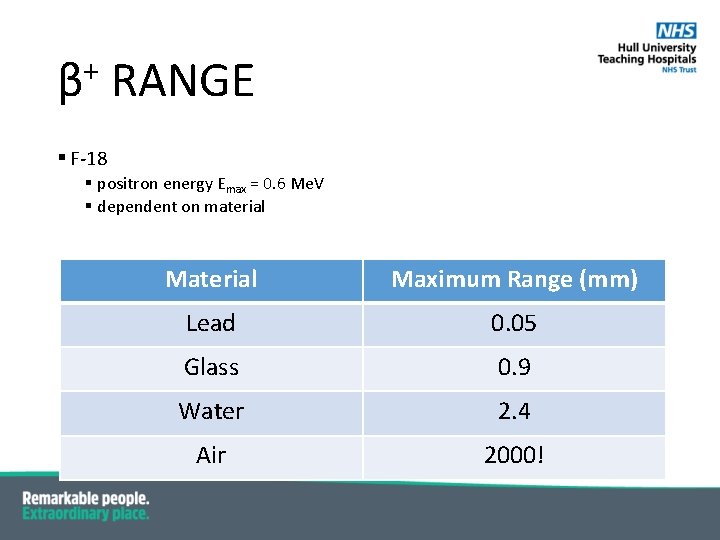
+ β RANGE § F-18 § positron energy Emax = 0. 6 Me. V § dependent on material Maximum Range (mm) Lead 0. 05 Glass 0. 9 Water 2. 4 Air 2000!

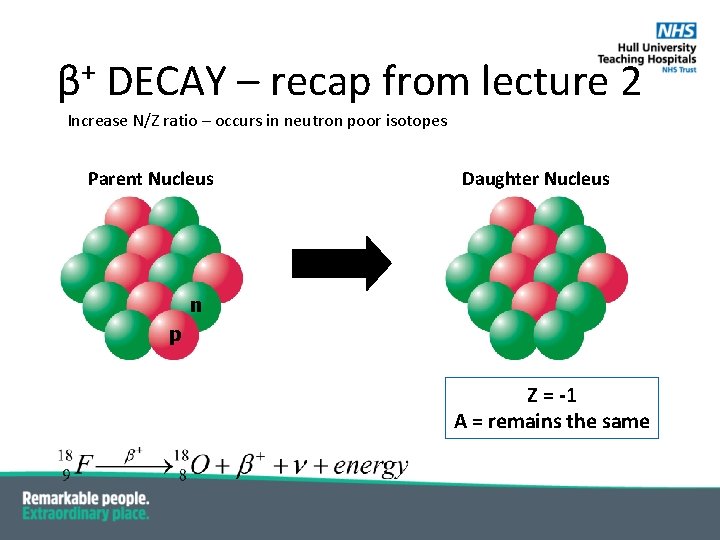
β+ DECAY – recap from lecture 2 Increase N/Z ratio – occurs in neutron poor isotopes Parent Nucleus Daughter Nucleus n p β+ n p p Ζ = -1 Α = remains the same

POSITRON ANNIHILATION

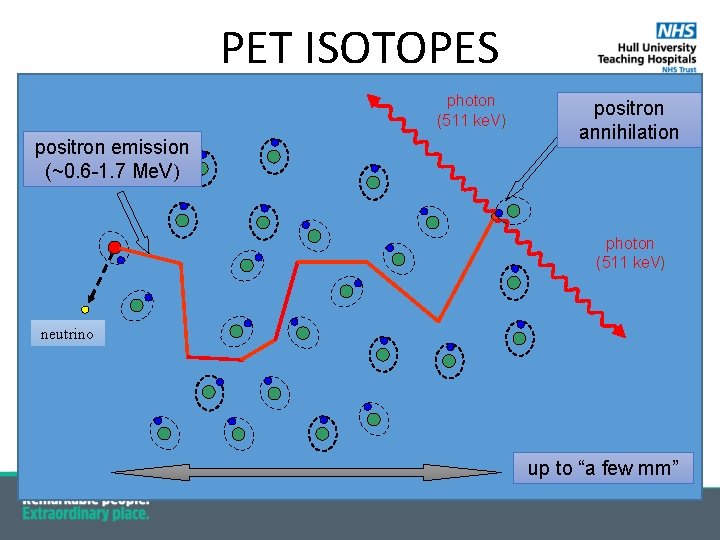
PET ISOTOPES photon (511 ke. V) positron emission (~0. 6 -1. 7 Me. V) positron annihilation photon (511 ke. V) neutrino up to “a few mm”

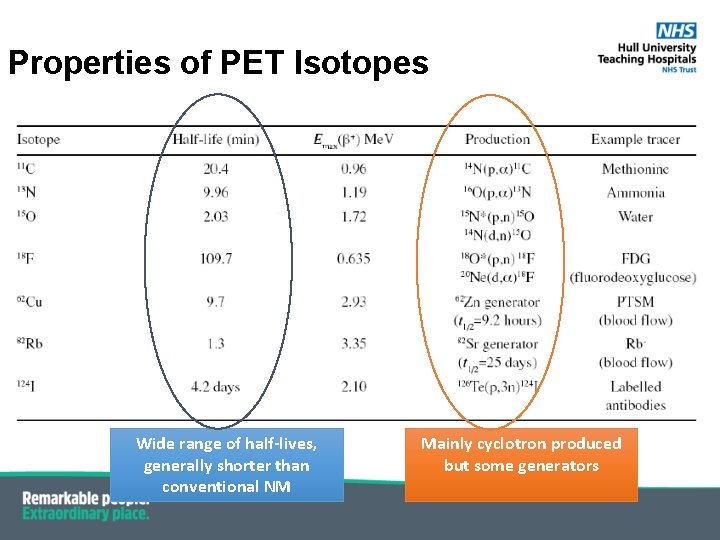
Properties of PET Isotopes Wide range of half-lives, generally shorter than conventional NM Mainly cyclotron produced but some generators

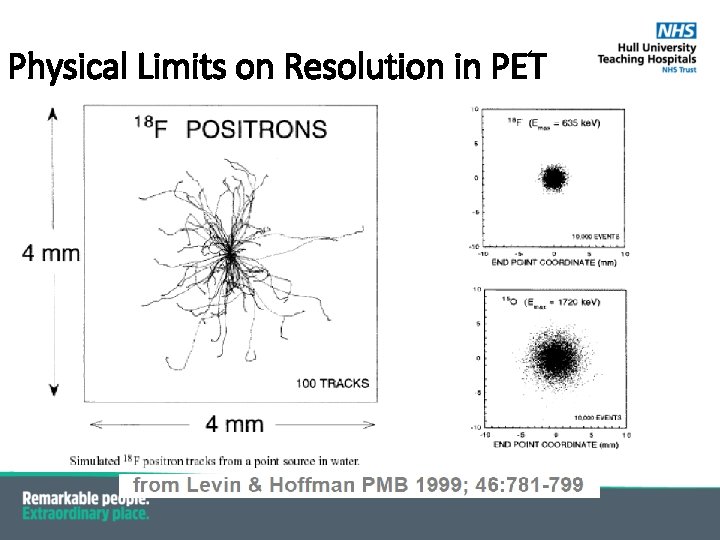
Variation of Resolution with Positron Energy Wide range of positron energies higher energy → worse spatial resolution

Physical Limits on Resolution in PET

INTERACTION MECHANISMS – Radiative Losses • Radiative interactions with atomic nucleus • Involves the emission of radiation when a charged particle is accelerated/decelerated

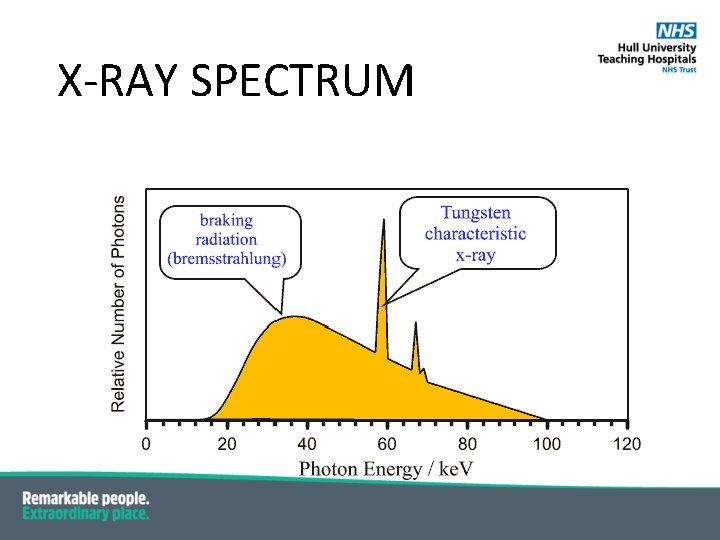
RADIATIVE COLLISIONS § Inelastic collision with electric field of nucleus § Charged particle is deflected § by the positive charge of nucleus § with a loss of kinetic energy of CP § Bremsstrahlung = braking radiation § X-rays § Energy of X-ray is equal to the energy lost by the electron: Photon energy hf = E 2 – E 1

X-RAY TUBE

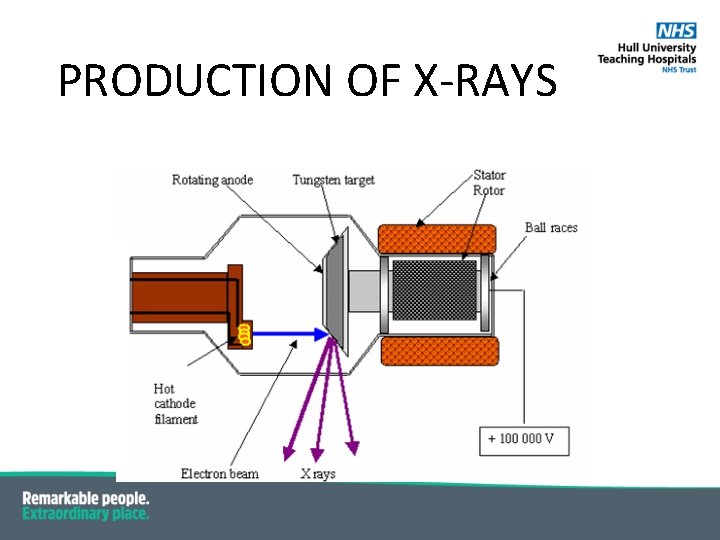
PRODUCTION OF X-RAYS

X-RAY SPECTRUM

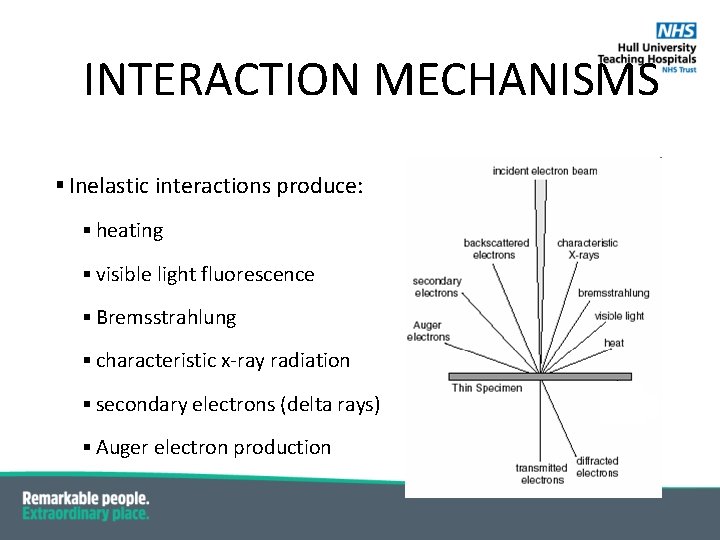
INTERACTION MECHANISMS § Inelastic interactions produce: § heating § visible light fluorescence § Bremsstrahlung § characteristic x-ray radiation § secondary electrons (delta rays) § Auger electron production

AURORA - Luminescence

AURORA

Thanks for listening