Interaction electron solid Elastic electron scattering Electromagnetic interaction



- Slides: 19

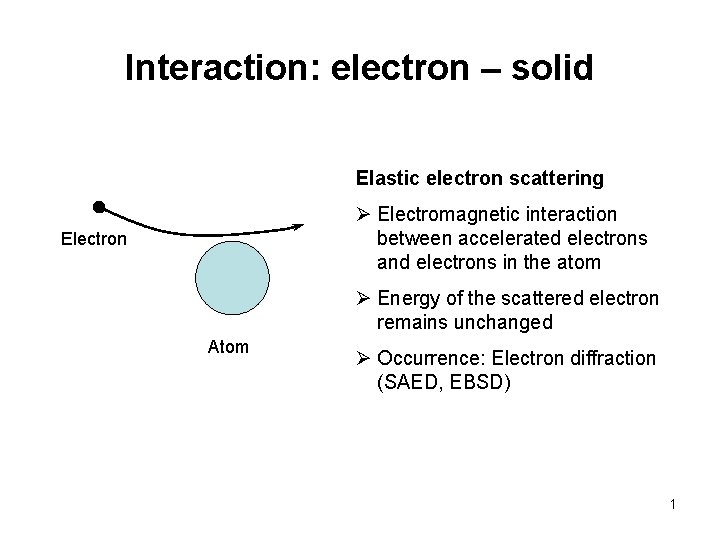
Interaction: electron – solid Elastic electron scattering Ø Electromagnetic interaction between accelerated electrons and electrons in the atom Electron Ø Energy of the scattered electron remains unchanged Atom Ø Occurrence: Electron diffraction (SAED, EBSD) 1

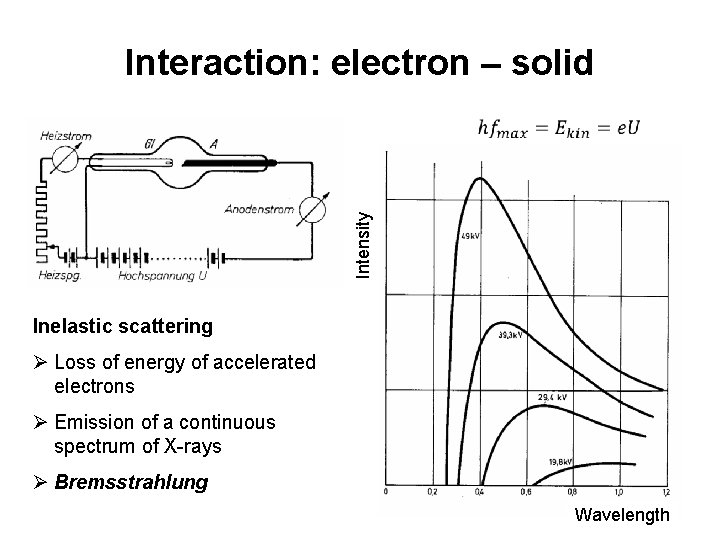
Intensity Interaction: electron – solid Inelastic scattering Ø Loss of energy of accelerated electrons Ø Emission of a continuous spectrum of X-rays Ø Bremsstrahlung 2 Wavelength

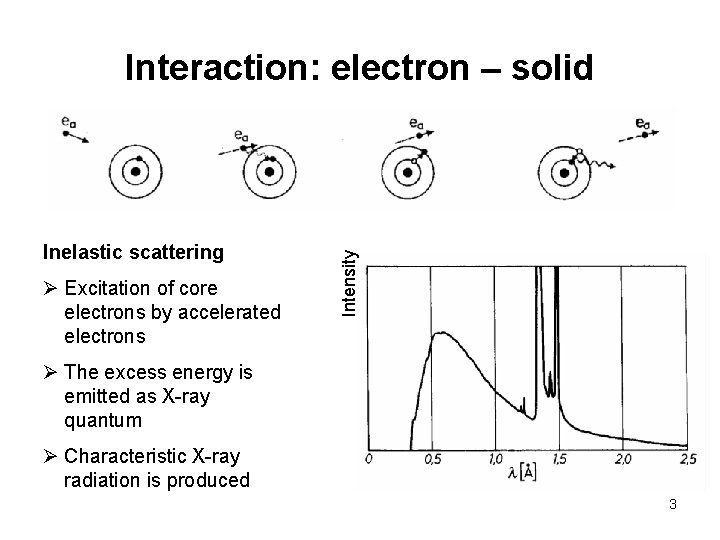
Inelastic scattering Ø Excitation of core electrons by accelerated electrons Intensity Interaction: electron – solid Ø The excess energy is emitted as X-ray quantum Ø Characteristic X-ray radiation is produced 3

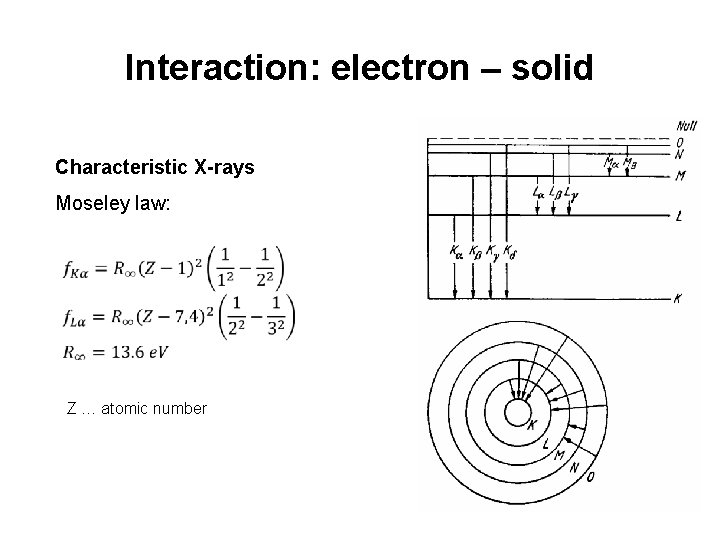
Interaction: electron – solid Characteristic X-rays Moseley law: Z … atomic number 4

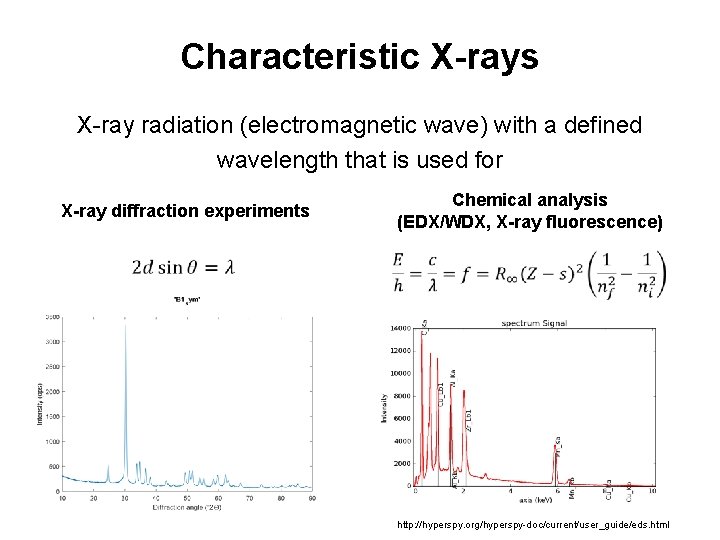
Characteristic X-rays X-ray radiation (electromagnetic wave) with a defined wavelength that is used for X-ray diffraction experiments Chemical analysis (EDX/WDX, X-ray fluorescence) 5 http: //hyperspy. org/hyperspy-doc/current/user_guide/eds. html

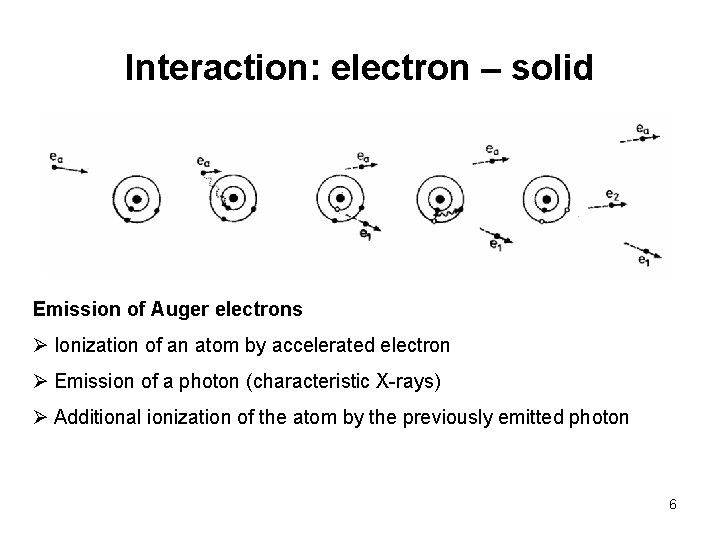
Interaction: electron – solid Emission of Auger electrons Ø Ionization of an atom by accelerated electron Ø Emission of a photon (characteristic X-rays) Ø Additional ionization of the atom by the previously emitted photon 6

Interaction: electron – solid Summary Ø Elastic scattering of electrons Ø Inelastic scattering Ø Ø Emission of Bremsstrahlung Emission of secondary electron Emission of characteristic X-rays Emission of Auger electron 7

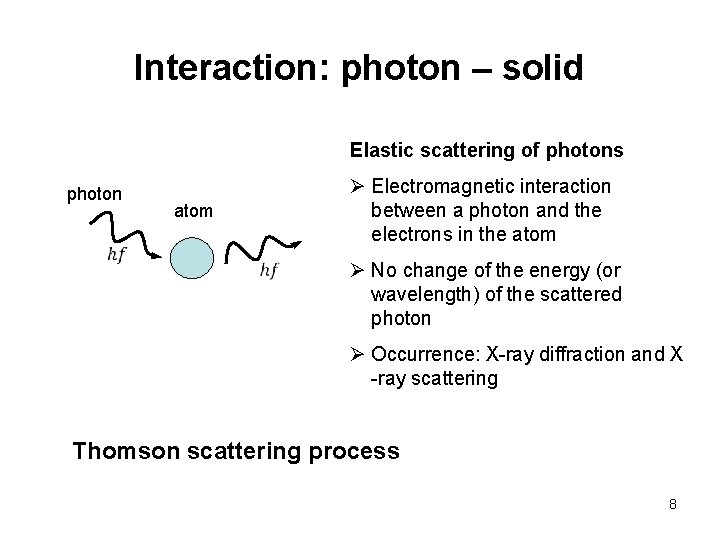
Interaction: photon – solid Elastic scattering of photons photon atom Ø Electromagnetic interaction between a photon and the electrons in the atom Ø No change of the energy (or wavelength) of the scattered photon Ø Occurrence: X-ray diffraction and X -ray scattering Thomson scattering process 8

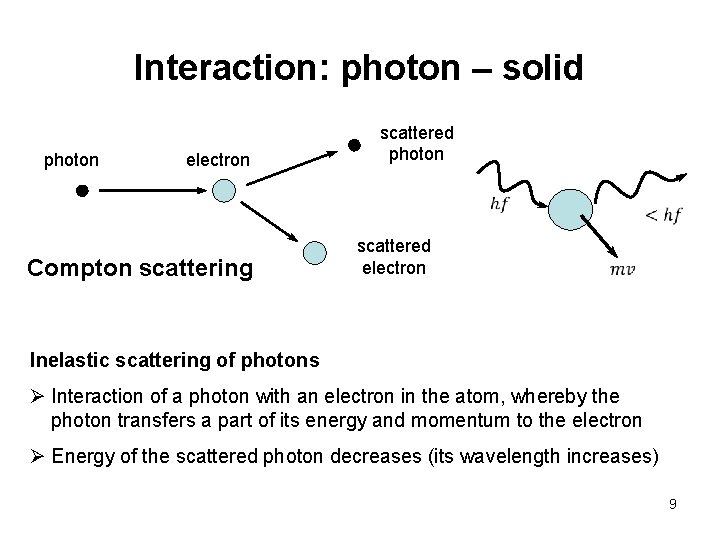
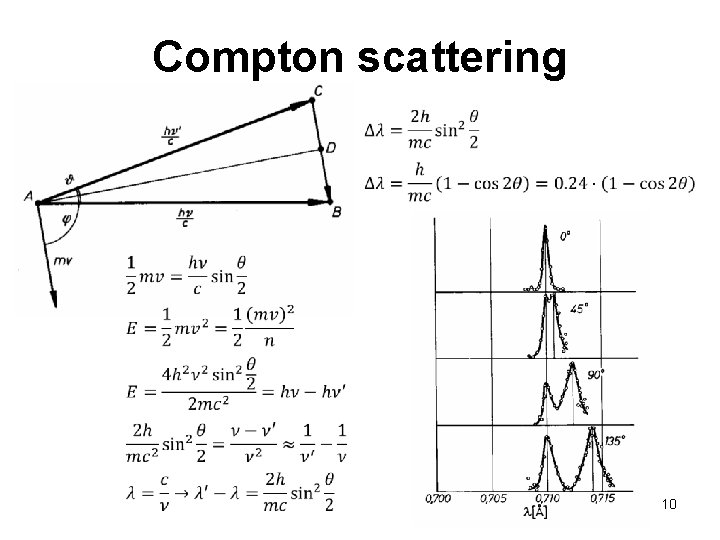
Interaction: photon – solid photon electron Compton scattering scattered photon scattered electron Inelastic scattering of photons Ø Interaction of a photon with an electron in the atom, whereby the photon transfers a part of its energy and momentum to the electron Ø Energy of the scattered photon decreases (its wavelength increases) 9

Compton scattering 10

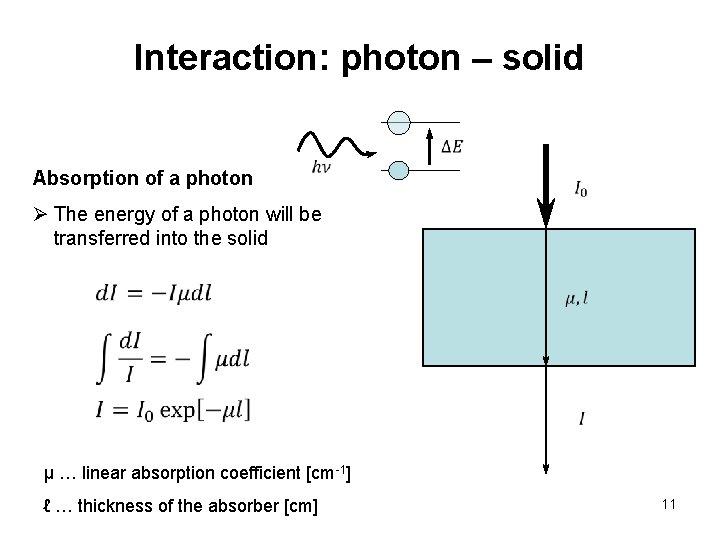
Interaction: photon – solid Absorption of a photon Ø The energy of a photon will be transferred into the solid µ … linear absorption coefficient [cm-1] ℓ … thickness of the absorber [cm] 11

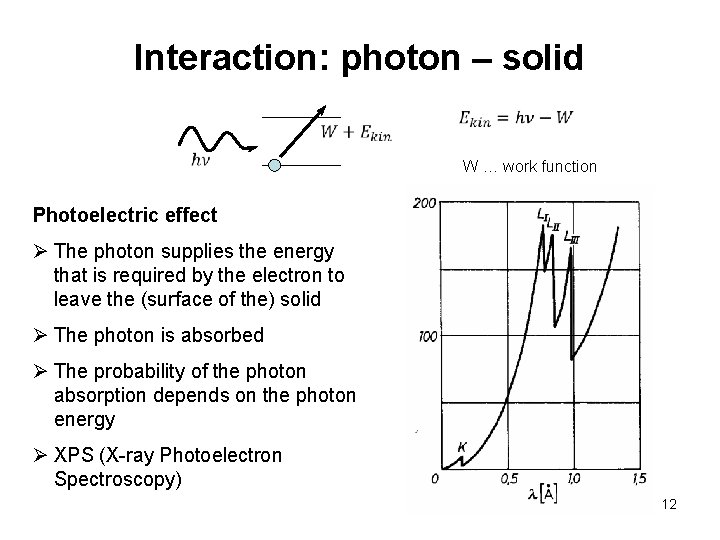
Interaction: photon – solid W … work function Photoelectric effect Ø The photon supplies the energy that is required by the electron to leave the (surface of the) solid Ø The photon is absorbed Ø The probability of the photon absorption depends on the photon energy Ø XPS (X-ray Photoelectron Spectroscopy) 12

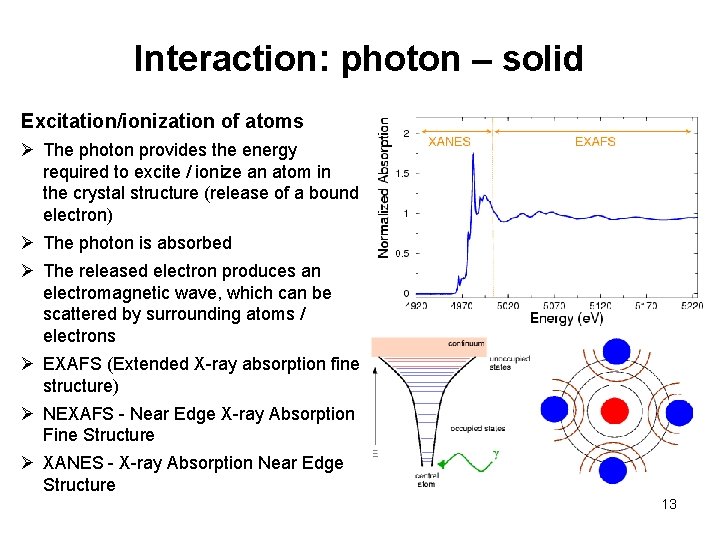
Interaction: photon – solid Excitation/ionization of atoms Ø The photon provides the energy required to excite / ionize an atom in the crystal structure (release of a bound electron) Ø The photon is absorbed Ø The released electron produces an electromagnetic wave, which can be scattered by surrounding atoms / electrons Ø EXAFS (Extended X-ray absorption fine structure) Ø NEXAFS - Near Edge X-ray Absorption Fine Structure Ø XANES - X-ray Absorption Near Edge Structure 13

Interaction: photon – solid X-ray fluorescence Ø A photon excites an electron in the solid Ø The photon is absorbed Ø XRF (X-ray fluorescence analysis) Ø The recorded energy spectrum contains the spectral lines of all atoms in the analyzed material 14

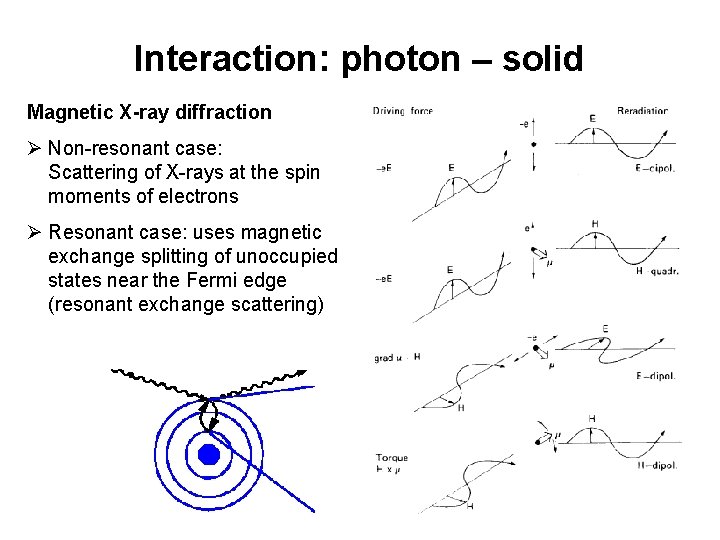
Interaction: photon – solid Magnetic X-ray diffraction Ø Non-resonant case: Scattering of X-rays at the spin moments of electrons Ø Resonant case: uses magnetic exchange splitting of unoccupied states near the Fermi edge (resonant exchange scattering) 15

Interaction: photon – solid Summary Ø Elastic X-ray diffraction and X-ray scattering (Thomson) Ø Inelastic X-ray scattering (Compton) Ø Absorption of photons Ø Photoelectric effect Ø EXAFS, NEXAFS, XANES Ø X-ray fluorescence Ø Magnetic X-ray diffraction 16

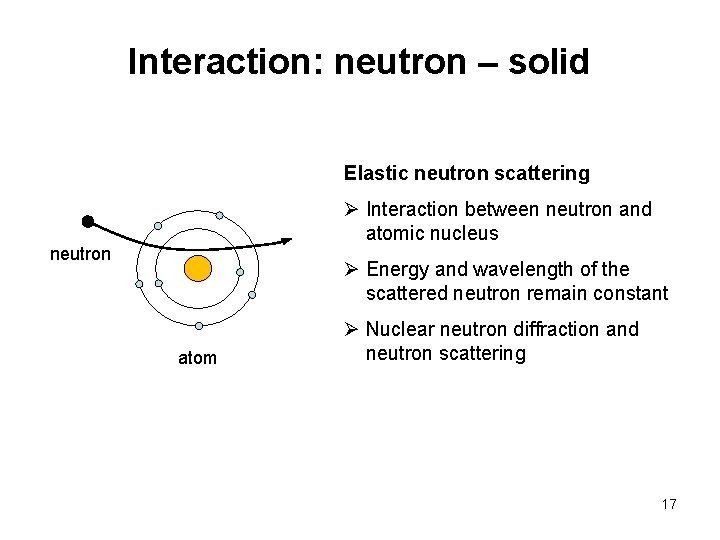
Interaction: neutron – solid Elastic neutron scattering Ø Interaction between neutron and atomic nucleus neutron Ø Energy and wavelength of the scattered neutron remain constant atom Ø Nuclear neutron diffraction and neutron scattering 17

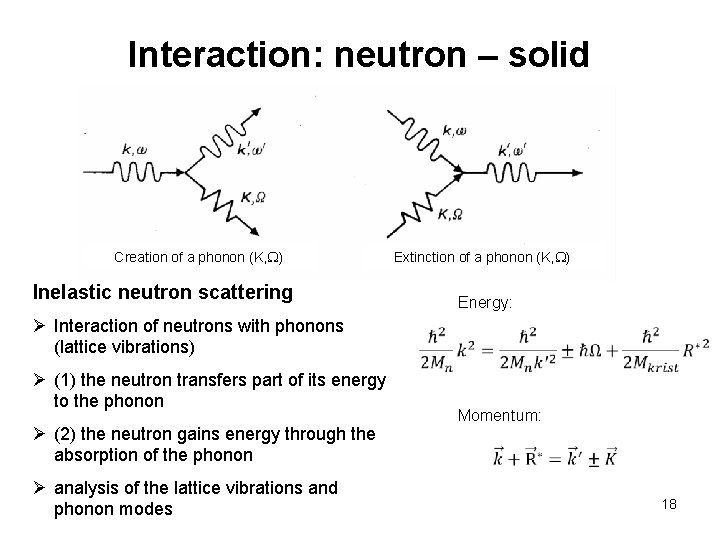
Interaction: neutron – solid Creation of a phonon (K, W) Inelastic neutron scattering Extinction of a phonon (K, W) Energy: Ø Interaction of neutrons with phonons (lattice vibrations) Ø (1) the neutron transfers part of its energy to the phonon Ø (2) the neutron gains energy through the absorption of the phonon Ø analysis of the lattice vibrations and phonon modes Momentum: 18

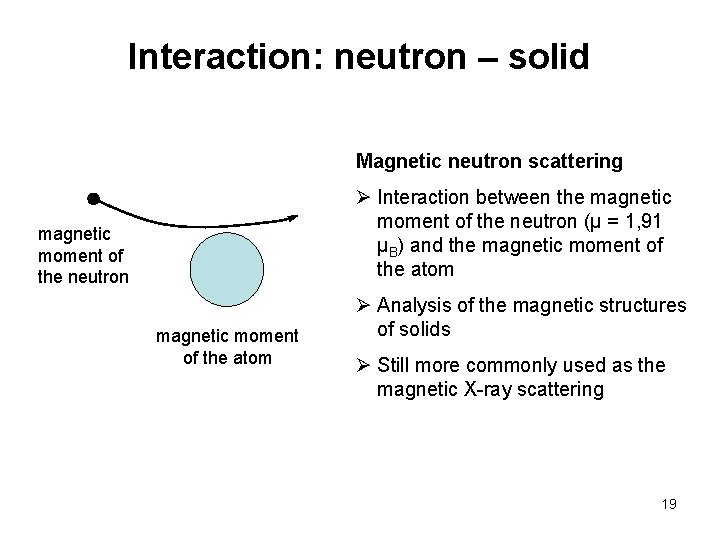
Interaction: neutron – solid Magnetic neutron scattering Ø Interaction between the magnetic moment of the neutron (µ = 1, 91 µB) and the magnetic moment of the atom magnetic moment of the neutron magnetic moment of the atom Ø Analysis of the magnetic structures of solids Ø Still more commonly used as the magnetic X-ray scattering 19