INTER 111 Graduate Biochemistry Bioenergetics Bioenergetics Learning objectives

INTER 111: Graduate Biochemistry Bioenergetics

Bioenergetics: Learning objectives � The change in free energy for a reaction predicts the direction in which it will spontaneously proceed. do positive, negative, and zero values for DG signify for a chemical reaction? � What should know the relationship of DG between the forward and back reactions and understand the coupling of standard free energy changes (DGo) for multiple reactions. � You � There is a thermodynamic relationship between free energy, enthalpy, and entropy.

Bioenergetics � Studies how energy is utilized and transferred in cells. � Is concerned with energy involved in making/breaking of molecular bonds within biological organisms. � includes the study of different cellular processes (cellular respiration, metabolism, growth, and development) that lead to production and utilization of energy in forms such as ATP molecules.

Bioenergetics � It is primarily concerned with the initial and final energy states of a chemical reaction. A � The B change in free energy between the initial and final states of a reaction allows prediction if the reaction is possible.
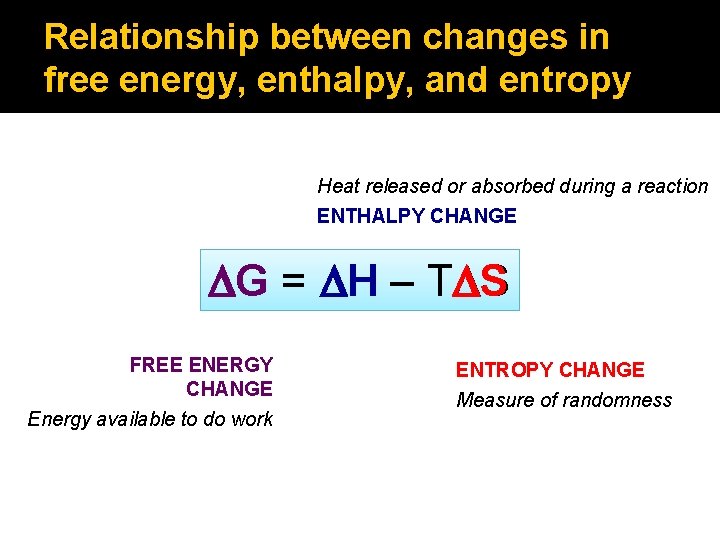
Relationship between changes in free energy, enthalpy, and entropy Heat released or absorbed during a reaction ENTHALPY CHANGE DG = DH – TDS DS FREE ENERGY CHANGE Energy available to do work ENTROPY CHANGE Measure of randomness
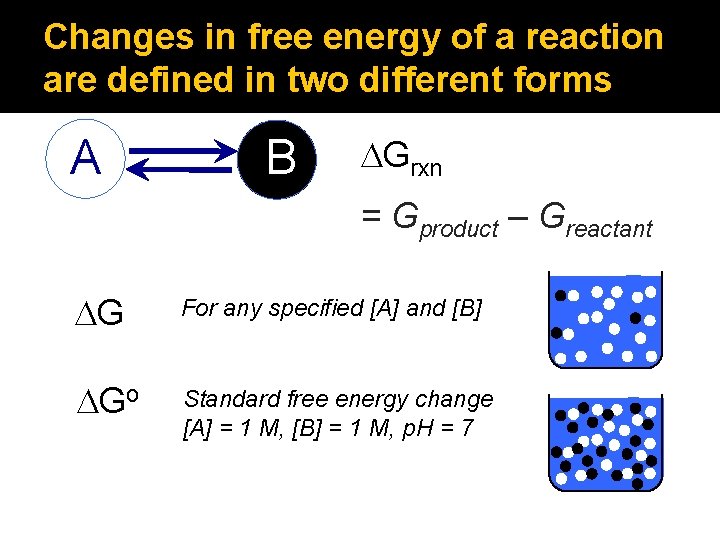
Changes in free energy of a reaction are defined in two different forms A B DGrxn = Gproduct – Greactant DG For any specified [A] and [B] DGo Standard free energy change [A] = 1 M, [B] = 1 M, p. H = 7

A negative DG indicates the reaction is spontaneous as written A B DGforward rxn = Gproduct – Greactant = GB – GA
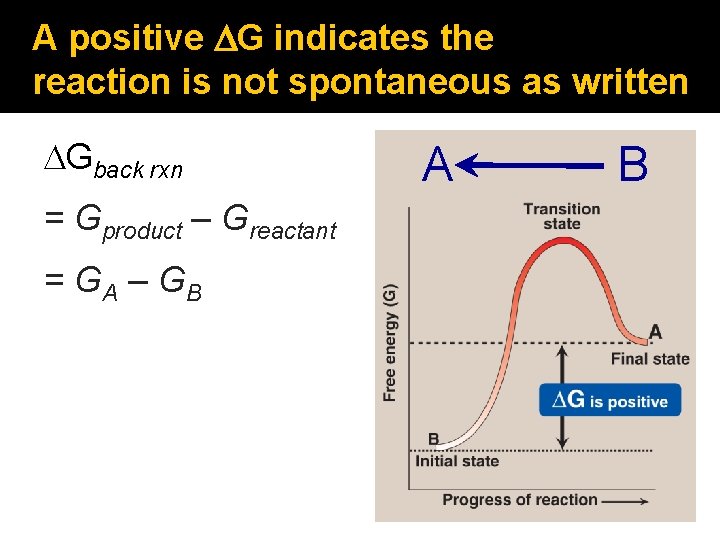
A positive DG indicates the reaction is not spontaneous as written DGback rxn = Gproduct – Greactant = GA – GB A B
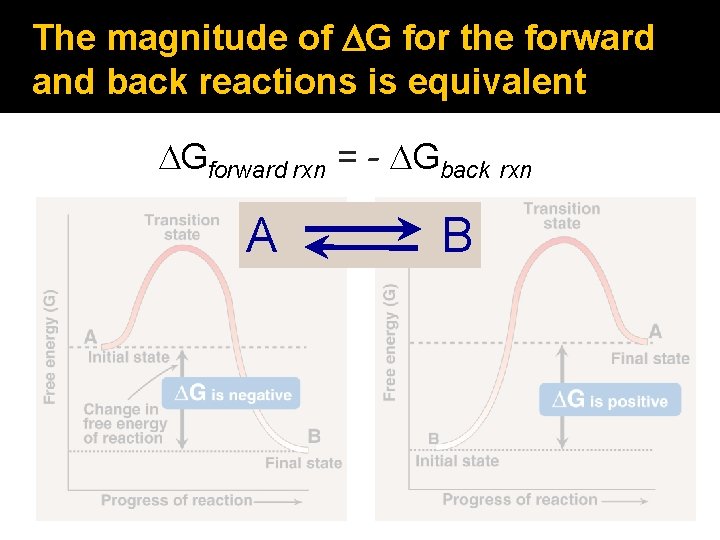
The magnitude of DG for the forward and back reactions is equivalent DGforward rxn = - DGback rxn A B
![At constant temperature and pressure, DG and DGo are related. DG = DGo [B] At constant temperature and pressure, DG and DGo are related. DG = DGo [B]](http://slidetodoc.com/presentation_image_h2/ecf093bfc3de80f51fc943bbc363719e/image-10.jpg)
At constant temperature and pressure, DG and DGo are related. DG = DGo [B] + RT ln [A] glucose 6 -phosphate A fructose 6 -phosphate B
![DGo is predictive only standard conditions glucose fructose [B] o 6 -phosphate DG = DGo is predictive only standard conditions glucose fructose [B] o 6 -phosphate DG =](http://slidetodoc.com/presentation_image_h2/ecf093bfc3de80f51fc943bbc363719e/image-11.jpg)
DGo is predictive only standard conditions glucose fructose [B] o 6 -phosphate DG = DG + RT ln [A] A B DGo = standard free energy change DG = DGo standard conditions A =1 M B =1 M
![DG depends on the concentration of reactants and products glucose fructose [B] o 6 DG depends on the concentration of reactants and products glucose fructose [B] o 6](http://slidetodoc.com/presentation_image_h2/ecf093bfc3de80f51fc943bbc363719e/image-12.jpg)
DG depends on the concentration of reactants and products glucose fructose [B] o 6 -phosphate DG = DG + RT ln [A] A B Can the reaction proceed in the forward direction, i. e. produce fructose 6 -phosphate? DG = DGo = + 0. 4 kcal/mol nonequilibrium conditions A = 0. 90 M B = 0. 09 M
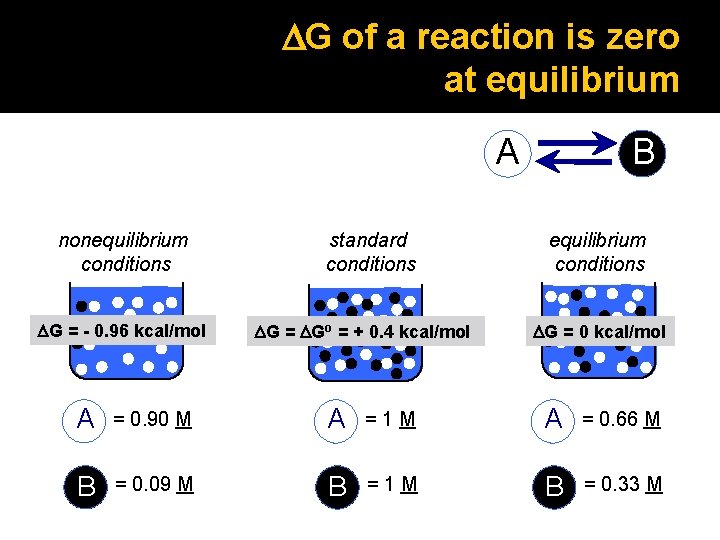
DG of a reaction is zero at equilibrium A nonequilibrium conditions DG = - 0. 96 kcal/mol standard conditions DG = DGo = + 0. 4 kcal/mol B equilibrium conditions DG = 0 kcal/mol A = 0. 90 M A =1 M A = 0. 66 M B = 0. 09 M B =1 M B = 0. 33 M
![DG of a reaction is zero at equilibrium A B equilibrium conditions [B]eq [A]eq DG of a reaction is zero at equilibrium A B equilibrium conditions [B]eq [A]eq](http://slidetodoc.com/presentation_image_h2/ecf093bfc3de80f51fc943bbc363719e/image-14.jpg)
DG of a reaction is zero at equilibrium A B equilibrium conditions [B]eq [A]eq = Keq = equilibrium constant DGo = - RT ln Keq DG = 0 kcal/mol A = 0. 66 M B = 0. 33 M
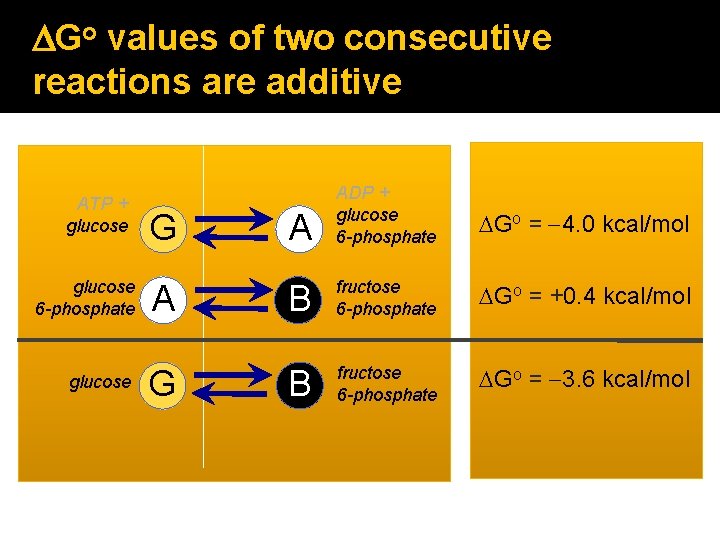
DGo values of two consecutive reactions are additive ATP + glucose 6 -phosphate glucose G A ADP + glucose 6 -phosphate DGo = -4. 0 kcal/mol B fructose 6 -phosphate DGo = +0. 4 kcal/mol B fructose 6 -phosphate DGo = -3. 6 kcal/mol

DG values of consecutive reactions are also additive � Additive property of free energy changes is central to biochemical pathways. sum of individual reaction DGs is negative, pathway can proceed as written. � If
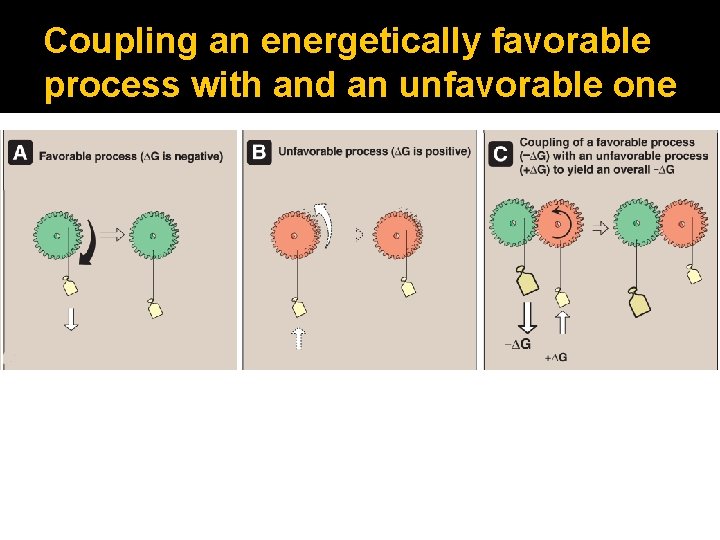
Coupling an energetically favorable process with and an unfavorable one
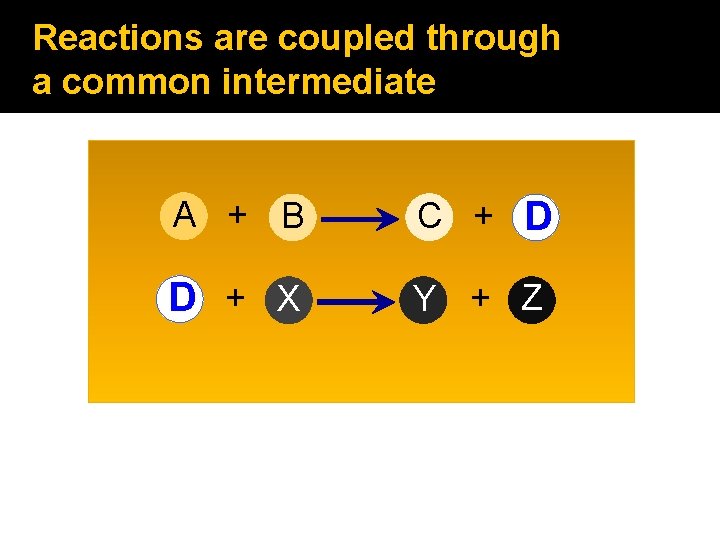
Reactions are coupled through a common intermediate A + B C + D D + X Y + Z
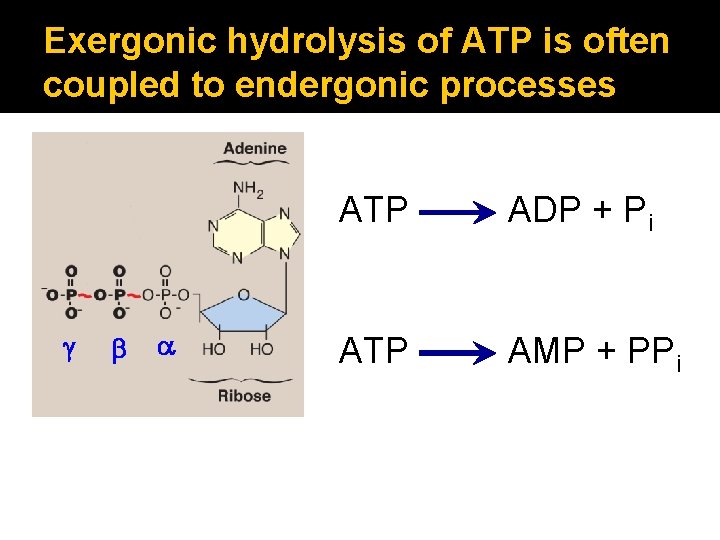
Exergonic hydrolysis of ATP is often coupled to endergonic processes g b a ATP ADP + Pi ATP AMP + PPi
- Slides: 19