INTELENCE Etravirine UPDATE Global Clinical Research Rekha Sinha

INTELENCE (Etravirine) UPDATE Global Clinical Research Rekha Sinha, MD January 30, 2009

Overview of Presentation l Etravirine (ETR) Safety and Tolerability - Pooled Data from Phase III Trials - Short-term safety in Pediatric Population l Planned Trials with Etravirine l Etravirine Resistance
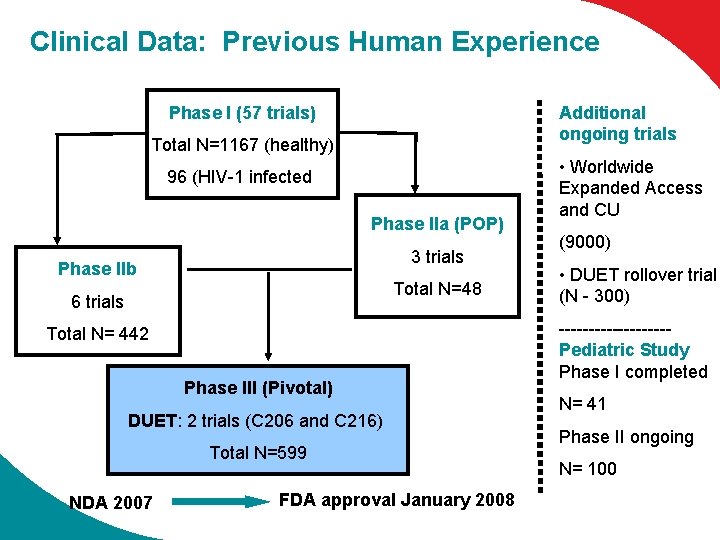
Clinical Data: Previous Human Experience Phase I (57 trials) Additional ongoing trials Total N=1167 (healthy) 96 (HIV-1 infected) Phase IIa (POP) 3 trials Phase IIb Total N=48 6 trials Total N= 442 Phase III (Pivotal) DUET: 2 trials (C 206 and C 216) Total N=599 NDA 2007 FDA approval January 2008 • Worldwide Expanded Access and CU (9000) • DUET rollover trial (N - 300) ---------Pediatric Study Phase I completed N= 41 Phase II ongoing N= 100

ETR Safety and Tolerability: DUET Trials (TMC 125 -C 206/C 216)

Design and Major Inclusion Criteria Screening 6 weeks 48 -week treatment period with optional 48 -week extension Follow up 4 weeks 24 -week primary analysis 600 patients target per trial TMC 125 + BR* Placebo + BR* *BR = darunavir/ritonavir with optimized NRTIs and optional enfuvirtide l l l DUET-1 and -2 differed only in geographical location; pooled analysis was pre-specified Major inclusion criteria: – Plasma viral load >5, 000 HIV-1 RNA copies/m. L and stable therapy for ≥ 8 weeks – ≥ 1 NNRTI RAM*, at screening or in documented historical genotype – ≥ 3 primary PI mutations at screening Patients recruited from Thailand, Australia, Europe and the Americas *From extended list of 41 NNRTI RAMs (Tambuyzer et al. Abstract 67 EHDRW 2007); BR = background regimen; RAM = resistance-associated mutation
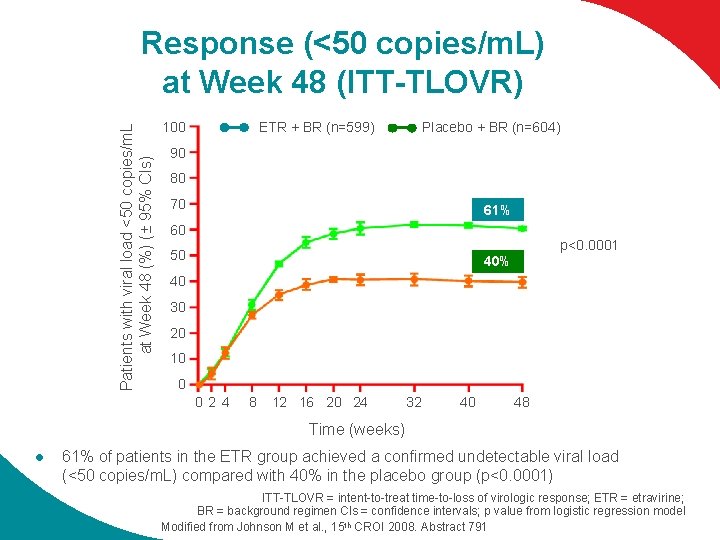
Patients with viral load <50 copies/m. L at Week 48 (%) (± 95% CIs) Response (<50 copies/m. L) at Week 48 (ITT-TLOVR) 100 Placebo + BR (n=604) ETR + BR (n=599) 90 80 70 61% 60 p<0. 0001 50 40% 40 30 20 10 0 02 4 8 12 16 20 24 32 40 48 Time (weeks) l 61% of patients in the ETR group achieved a confirmed undetectable viral load (<50 copies/m. L) compared with 40% in the placebo group (p<0. 0001) ITT-TLOVR = intent-to-treat time-to-loss of virologic response; ETR = etravirine; BR = background regimen CIs = confidence intervals; p value from logistic regression model Modified from Johnson M et al. , 15 th CROI 2008. Abstract 791
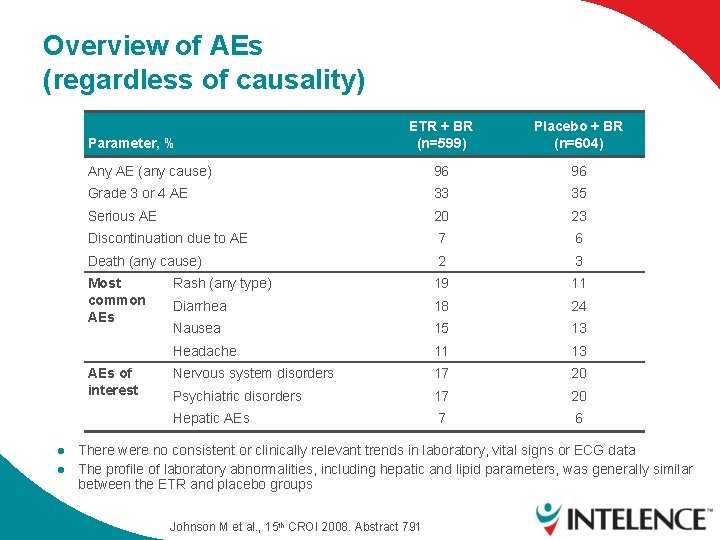
Overview of AEs (regardless of causality) ETR + BR (n=599) Placebo + BR (n=604) Any AE (any cause) 96 96 Grade 3 or 4 AE 33 35 Serious AE 20 23 Discontinuation due to AE 7 6 Death (any cause) 2 3 Most common AEs Rash (any type) 19 11 Diarrhea 18 24 Nausea 15 13 Headache 11 13 Nervous system disorders 17 20 Psychiatric disorders 17 20 Hepatic AEs 7 6 Parameter, % AEs of interest l l There were no consistent or clinically relevant trends in laboratory, vital signs or ECG data The profile of laboratory abnormalities, including hepatic and lipid parameters, was generally similar between the ETR and placebo groups Johnson M et al. , 15 th CROI 2008. Abstract 791

Rash (1) Investigator assessment of cause of rash, % Any cause l ETR group (n = 599) 19. 2 Placebo group (n = 604) 10. 9 Significance P = 0. 0001 In the ETR group – – early onset: median 14 days limited duration: median 15 days low severity: mostly mild-to-moderate; 1. 3% grade 3, no grade 4 ● mostly maculopapular; no mucosal involvement infrequently led to permanent discontinuation: 2. 2% of ETR group, 0% in placebo group ● mostly resolved with continued treatment Rash with etravirine: usually mild-to-moderate and infrequently led to permanent discontinuation

Nervous system* & Psychiatric disorders Most common Nervous system events (reported in ≥ 1. 0% of patients in the ETR group) Headache 10. 9 12. 7 Dizziness 3. 2 4. 3 Somnolence 1. 8 2. 3 *Selected based on the nervous system events commonly associated with approved NNRTIs. Most common psychiatric events (reported in ≥ 1. 0% of patients in the ETR group) Insomnia Depression Anxiety Sleep disorder l l l 7. 2 4. 2 3. 8 1. 3 8. 3 6. 6 4. 1 0. 8 No increased risk in patients with a history of psychiatric disorders Abnormal dreams/nightmares were similar in incidence to placebo (0. 8% vs 1. 0% for the ETR and Placebo groups respectively) No episodes of hallucinations, suicidal ideation, or manic symptoms in the ETR group AEs: similar incidence to placebo, low severity, did not lead to discontinuation

Hepatic AEs and laboratory abnormalities ETR group (n = 599) Placebo group (n = 604) Any cause or severity Grade 3/4 6. 5 3. 0 6. 1 3. 0 Leading to discontinuation 1. 0 0. 7 Laboratory abnormalities ALT elevated (grade 3/4) AST elevated (grade 3/4) 1. 6/0. 8 1. 2/0. 2 1. 8/0. 4 1. 2/0. 2 Hepatic AEs, % Hepatic disorders: similar incidence to placebo, low severity, infrequently led to discontinuation ALT = alanine aminotransferase; AST = aspartate aminotransferase.

Safety conclusions l Safety and tolerability of ETR were generally comparable to placebo, except for the incidence of rash (any type) l Overall, most AEs were of low severity and infrequently led to discontinuation l Rash, the only AE to occur more frequently with ETR – generally mild-to-moderate – most often resolved with continuing treatment – infrequently led to discontinuation l Nature and incidence of nervous system and psychiatric AEs were similar to placebo l ETR was not associated with any increase in laboratory abnormalities, including hepatic and lipid parameters

Safety in Pediatric population • Phase I trial in children (6 -17 years)

TMC 125 -C 126 Trial Design 40 treatment experienced children on a Stable ARV & Virologically suppressed Group 1 6 to 12 years Screening up to 28 days Stage I 20 subjects (10/Group) Dose: 4 mg/kg b. i. d Group 2 12 to 17 years Treatment Phase 7 days Interim analysis Follow-up 1 month Stage II 20 subjects (10/Group) Dose: 5. 2 mg/kg b. i. d Stable ARV: LPV/rtv and a minimum of 2 NRTIs +/- ENF
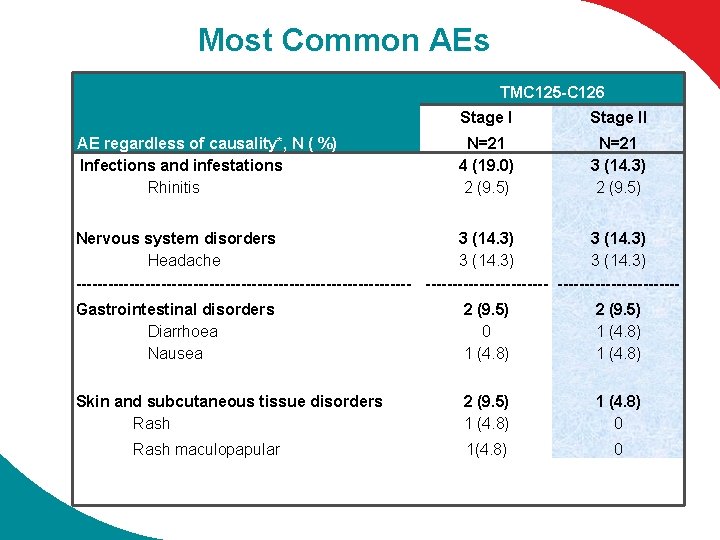
Most Common AEs TMC 125 -C 126 AE regardless of causality*, N ( %) Infections and infestations Rhinitis Stage II N=21 4 (19. 0) 2 (9. 5) N=21 3 (14. 3) 2 (9. 5) Nervous system disorders 3 (14. 3) Headache 3 (14. 3) --------------------------------Gastrointestinal disorders Diarrhoea Nausea 2 (9. 5) 0 1 (4. 8) 2 (9. 5) 1 (4. 8) Skin and subcutaneous tissue disorders Rash 2 (9. 5) 1 (4. 8) 0 Rash maculopapular 1(4. 8) 0 * 10. 0% in TMC 125 group

Conclusion Based on the comparable exposures of TMC 125 in children to that seen in adults from the DUET trials and the overall safety of TMC 125 in Stage II of TMC 125 -C 126 The recommended dose per weight band for children and adolescents aged between 6 and 17, inclusive, will be based on 5. 2 mg/kg b. i. d.

Planned trials with ETR - New target population - New ARV regimen

ETR, RAL plus 3 TC in HIV-infected Early Treatment-Experienced Patients Phase IV Single arm open label design Population: First or Second line failure, VL > 500 c/m. L, Sensitive to ETR, Naïve to integrase N= 50 ARV regimen: ETR 200 mg/ RAL 400 mg/ lamivudine 150 mg each b. i. d Duration: 48 week study Objective: To assess the percentage of early treatmentexperienced HIV-infected patients that have achieved an HIV RNA <50 copies/m. L at week 24

Study of Efavirenz Neuropsychiatric Symptoms versus Etravirine (SENSE) Double blind, active controlled trial Population: Treatment naive, VL > 5000 cp/m. L, sensitive to ETR and background regimen (2 NRTIs), no NNRTI resistance N= 150 ARV regimen: ETR 400 mg q. d ( 4 ETR tablets 100 mg each) versus efavirenz 600 mg q. d Duration: 48 week study Objective: To compare neuropsychiatric adverse event profile of ETR versus efavirenz in combination with 2 N(t)RTIs as assessed at Wk 12

ETR in a Nucleoside Sparing Regimen Open label, active controlled trial Population: Treatment experienced, NNRTI resistant, VL > 500 cp/m. L, sensitive to ETR and background regimen N= 520 ARV regimen: ETR 200 mg b. i. d plus PI/rtv versus PI/rtv plus 2 N(t)RTIs Duration: 48 week study Objective: Efficacy of ETR given in a PI-containing N(t)RTI-sparing regimen in terms of the proportion of subjects achieving a plasma viral load < 50 HIV-1 RNA copies/m. L at Week 24.

ETR Resistance
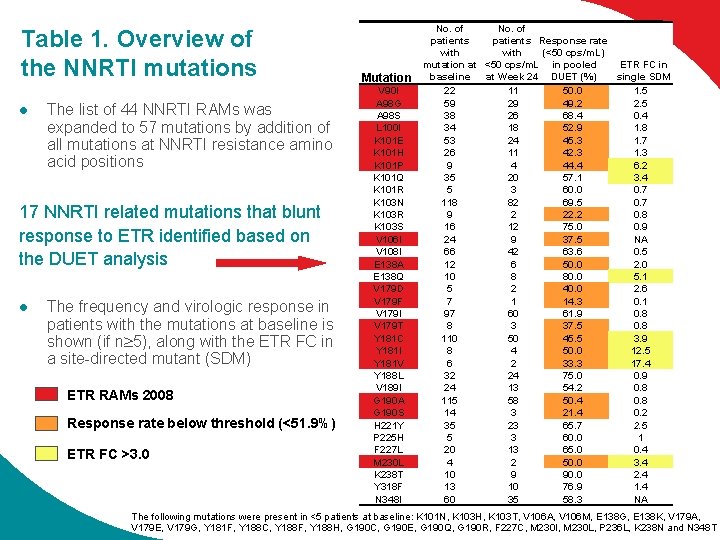
Table 1. Overview of the NNRTI mutations l The list of 44 NNRTI RAMs was expanded to 57 mutations by addition of all mutations at NNRTI resistance amino acid positions 17 NNRTI related mutations that blunt response to ETR identified based on the DUET analysis l The frequency and virologic response in patients with the mutations at baseline is shown (if n 5), along with the ETR FC in a site-directed mutant (SDM) ETR RAMs 2008 Response rate below threshold (<51. 9%) ETR FC >3. 0 Mutation V 90 I A 98 G A 98 S L 100 I K 101 E K 101 H K 101 P K 101 Q K 101 R K 103 N K 103 R K 103 S V 106 I V 108 I E 138 A E 138 Q V 179 D V 179 F V 179 I V 179 T Y 181 C Y 181 I Y 181 V Y 188 L V 189 I G 190 A G 190 S H 221 Y P 225 H F 227 L M 230 L K 238 T Y 318 F N 348 I No. of patients Response rate (<50 cps/m. L) with ETR FC in mutation at <50 cps/m. L in pooled single SDM baseline at Week 24 DUET (%) 22 11 50. 0 1. 5 59 29 49. 2 2. 5 38 26 68. 4 0. 4 34 18 52. 9 1. 8 53 24 45. 3 1. 7 26 11 42. 3 1. 3 9 4 44. 4 6. 2 35 20 57. 1 3. 4 5 3 60. 0 0. 7 118 82 69. 5 0. 7 9 2 22. 2 0. 8 16 12 75. 0 0. 9 24 9 37. 5 NA 66 42 63. 6 0. 5 12 6 50. 0 2. 0 10 8 80. 0 5. 1 5 2 40. 0 2. 6 7 1 14. 3 0. 1 97 60 61. 9 0. 8 8 3 37. 5 0. 8 110 50 45. 5 3. 9 8 4 50. 0 12. 5 6 2 33. 3 17. 4 32 24 75. 0 0. 9 24 13 54. 2 0. 8 115 58 50. 4 0. 8 14 3 21. 4 0. 2 35 23 65. 7 2. 5 5 3 60. 0 1 20 13 65. 0 0. 4 4 2 50. 0 3. 4 10 9 90. 0 2. 4 13 10 76. 9 1. 4 60 35 58. 3 NA The following mutations were present in <5 patients at baseline: K 101 N, K 103 H, K 103 T, V 106 A, V 106 M, E 138 G, E 138 K, V 179 A, V 179 E, V 179 G, Y 181 F, Y 188 C, Y 188 F, Y 188 H, G 190 C, G 190 E, G 190 Q, G 190 R, F 227 C, M 230 I, M 230 L, P 236 L, K 238 N and N 348 T
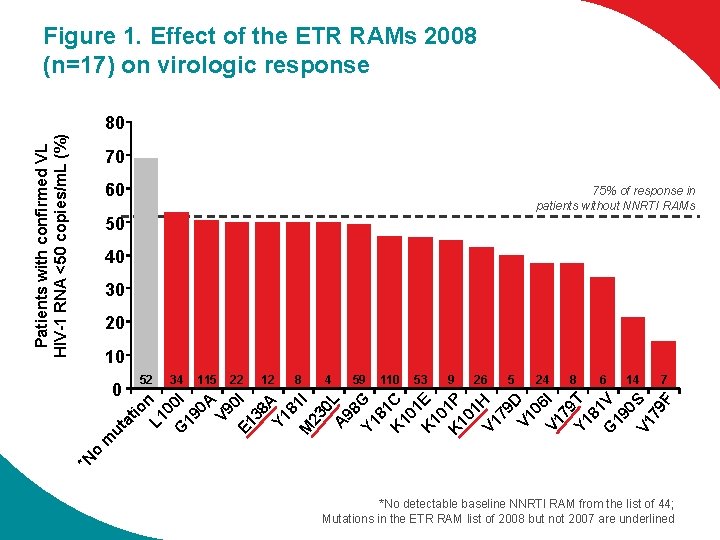
Figure 1. Effect of the ETR RAMs 2008 (n=17) on virologic response Patients with confirmed VL HIV-1 RNA <50 copies/m. L (%) 80 70 60 75% of response in patients without NNRTI RAMs 50 40 30 20 10 34 34 115 22 12 12 88 4 59 59 110 53 9 26 26 5 24 88 6 14 14 77 *N o m ut at io n L 1 0 G 0 l 19 0 A V 9 0 E 1 l 38 A Y 1 81 M l 23 0 L A 98 Y 1 G 81 K C 10 1 K E 10 K 1 P 10 1 H V 1 79 D V 1 06 V 1 l 79 Y 1 T 81 G V 19 0 S V 1 79 F 0 52 *No detectable baseline NNRTI RAM from the list of 44; Mutations in the ETR RAM list of 2008 but not 2007 are underlined
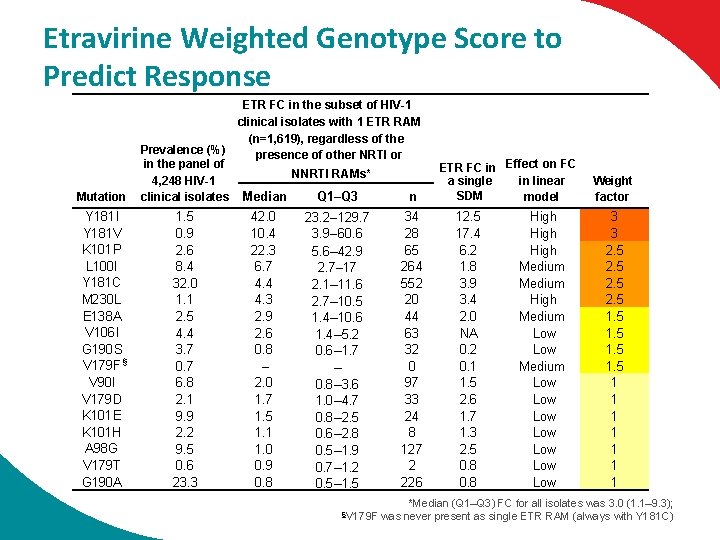
Etravirine Weighted Genotype Score to Predict Response Mutation Y 181 I Y 181 V K 101 P L 100 I Y 181 C M 230 L E 138 A V 106 I G 190 S V 179 F § V 90 I V 179 D K 101 E K 101 H A 98 G V 179 T G 190 A ETR FC in the subset of HIV-1 clinical isolates with 1 ETR RAM (n=1, 619), regardless of the Prevalence (%) presence of other NRTI or in the panel of NNRTI RAMs* 4, 248 HIV-1 Q 1–Q 3 n clinical isolates Median 1. 5 0. 9 2. 6 8. 4 32. 0 1. 1 2. 5 4. 4 3. 7 0. 7 6. 8 2. 1 9. 9 2. 2 9. 5 0. 6 23. 3 42. 0 10. 4 22. 3 6. 7 4. 4 4. 3 2. 9 2. 6 0. 8 – 2. 0 1. 7 1. 5 1. 1 1. 0 0. 9 0. 8 23. 2– 129. 7 3. 9– 60. 6 5. 6– 42. 9 2. 7– 17 2. 1– 11. 6 2. 7– 10. 5 1. 4– 10. 6 1. 4– 5. 2 0. 6– 1. 7 – 0. 8– 3. 6 1. 0– 4. 7 0. 8– 2. 5 0. 6– 2. 8 0. 5– 1. 9 0. 7– 1. 2 0. 5– 1. 5 34 28 65 264 552 20 44 63 32 0 97 33 24 8 127 2 226 ETR FC in Effect on FC in linear a single SDM model 12. 5 17. 4 6. 2 1. 8 3. 9 3. 4 2. 0 NA 0. 2 0. 1 1. 5 2. 6 1. 7 1. 3 2. 5 0. 8 High Medium High Medium Low Low Low Weight factor 3 3 2. 5 1 1 1 1 *Median (Q 1–Q 3) FC for all isolates was 3. 0 (1. 1– 9. 3); §V 179 F was never present as single ETR RAM (always with Y 181 C)
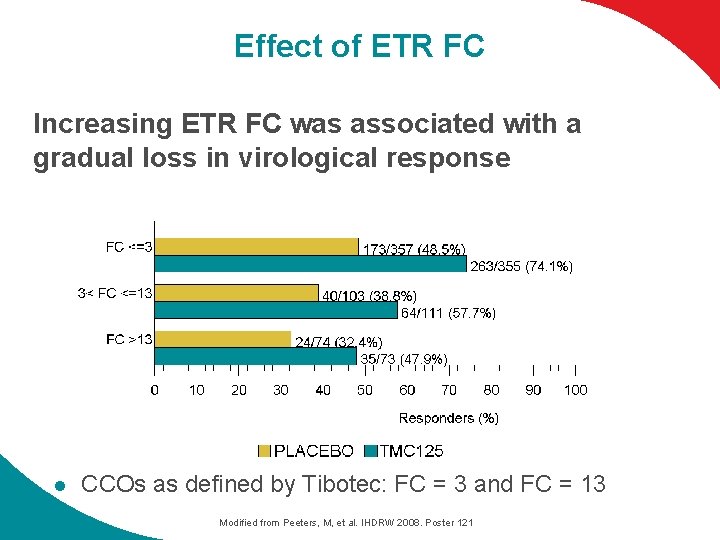
Effect of ETR FC Increasing ETR FC was associated with a gradual loss in virological response l CCOs as defined by Tibotec: FC = 3 and FC = 13 Modified from Peeters, M, et al. IHDRW 2008. Poster 121
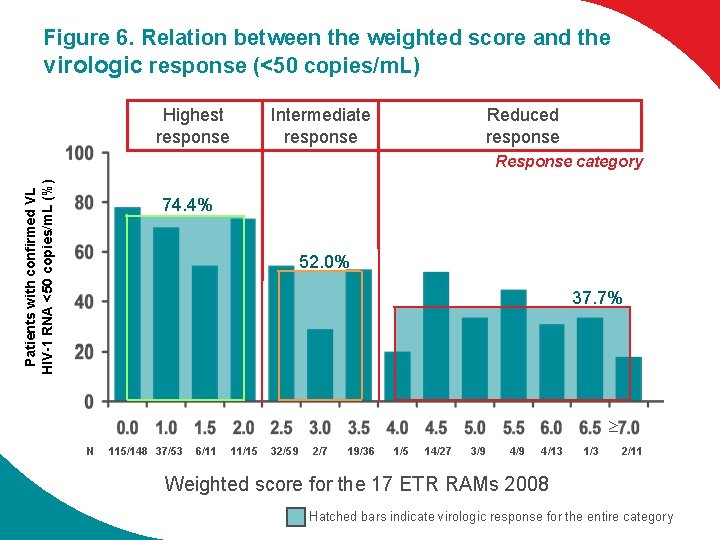
Figure 6. Relation between the weighted score and the virologic response (<50 copies/m. L) Highest response Intermediate response Reduced response Patients with confirmed VL HIV-1 RNA <50 copies/m. L (%) Response category 74. 4% 52. 0% 37. 7% N 115/148 37/53 6/11 11/15 32/59 2/7 19/36 1/5 14/27 3/9 4/13 1/3 2/11 Weighted score for the 17 ETR RAMs 2008 Hatched bars indicate virologic response for the entire category

Understanding Etravirine Susceptibility The virologic response is a function of the number and weight of the baseline ETR RAMs l Among the 17 ETR RAMs, Y 181 I and Y 181 V had the highest weight, followed by L 100 I, K 101 P, Y 181 C and M 230 L l Among the 17 ETR RAMs, mutations with the highest weight had a low prevalence Weighted mutation score of 0 -2 Highest response rates (74%) Weighted mutation score of 2. 5 -3. 5 Intermediate response rates (52%) Weighted mutation score of >/=4 Reduced response rates (74%) ·
- Slides: 26