Integrated Basic Science Review Introductory Section1 B Dr

Integrated Basic Science Review Introductory Section-1 B Dr. Paul S. Anderson Copyright – Medical Board Review Services
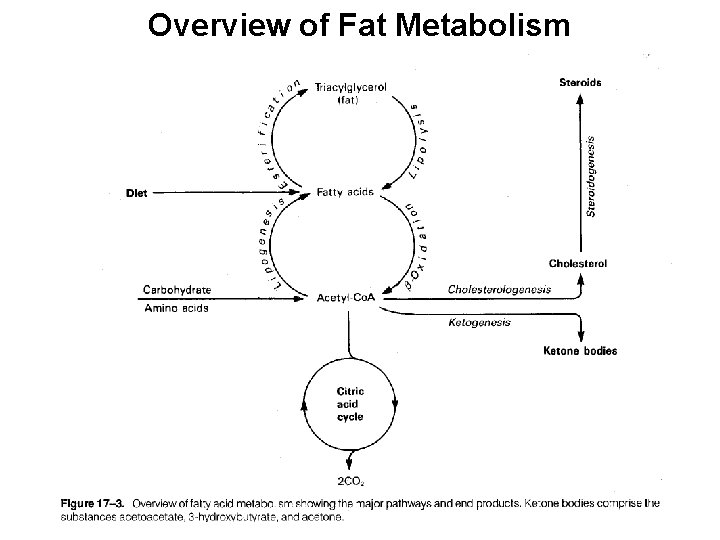
Overview of Fat Metabolism
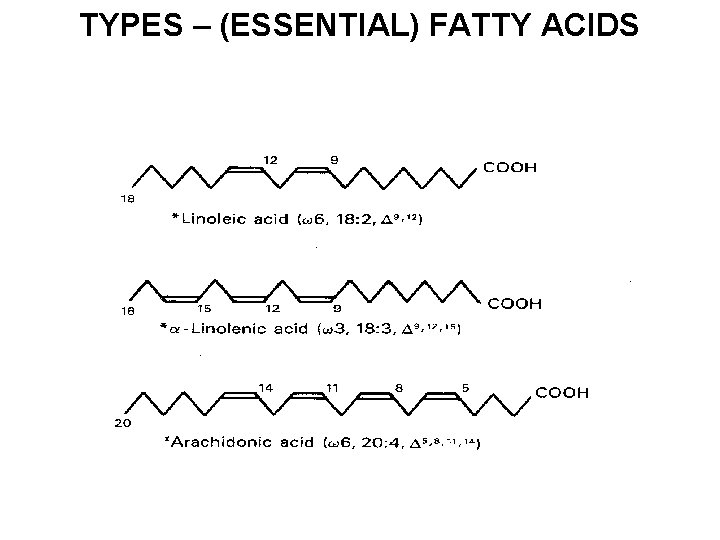
TYPES – (ESSENTIAL) FATTY ACIDS
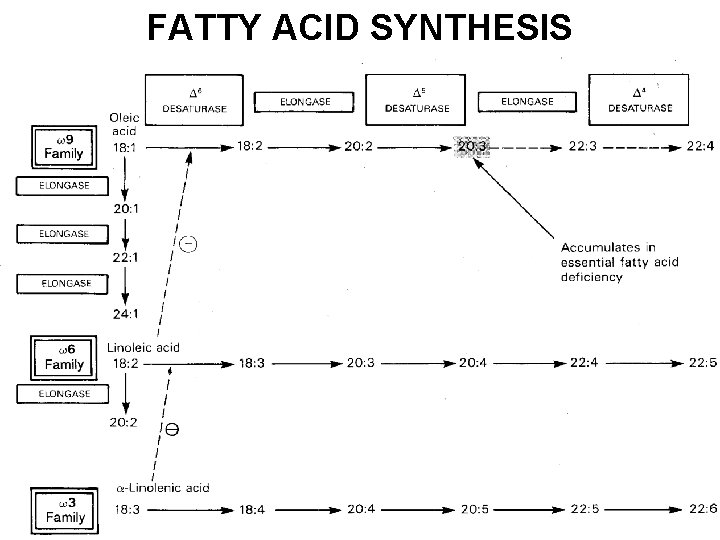
FATTY ACID SYNTHESIS

TYPES – (TRANS) FATTY ACIDS (Underlines added: ) Trans fats compete for cell membrane sites, effect fluidity, and aggravate essential fatty acid deficiency. They increase LDL and lower HDL. Generally these effects are pro-inflammatory.
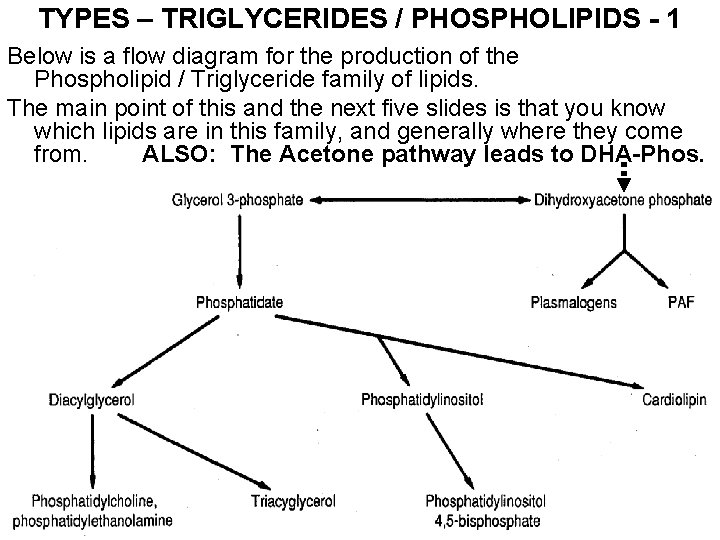
TYPES – TRIGLYCERIDES / PHOSPHOLIPIDS - 1 Below is a flow diagram for the production of the Phospholipid / Triglyceride family of lipids. The main point of this and the next five slides is that you know which lipids are in this family, and generally where they come from. ALSO: The Acetone pathway leads to DHA-Phos.
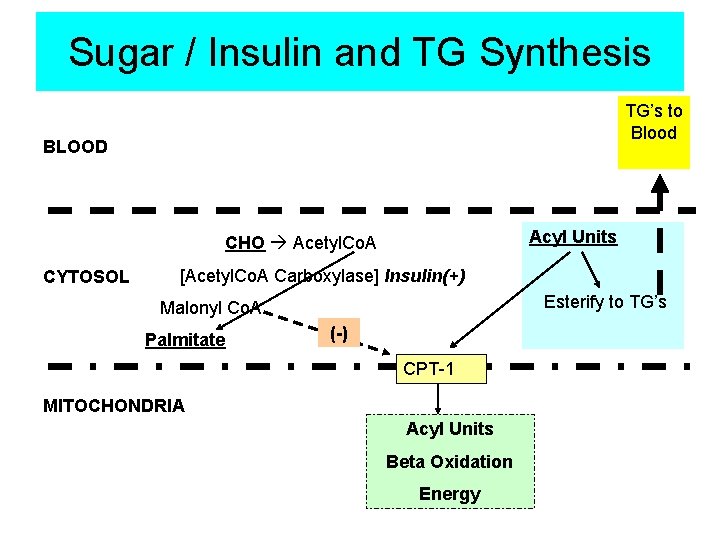
Sugar / Insulin and TG Synthesis TG’s to Blood BLOOD Acyl Units CHO Acetyl. Co. A CYTOSOL [Acetyl. Co. A Carboxylase] Insulin(+) Esterify to TG’s Malonyl Co. A Palmitate (-) CPT-1 MITOCHONDRIA Acyl Units Beta Oxidation Energy
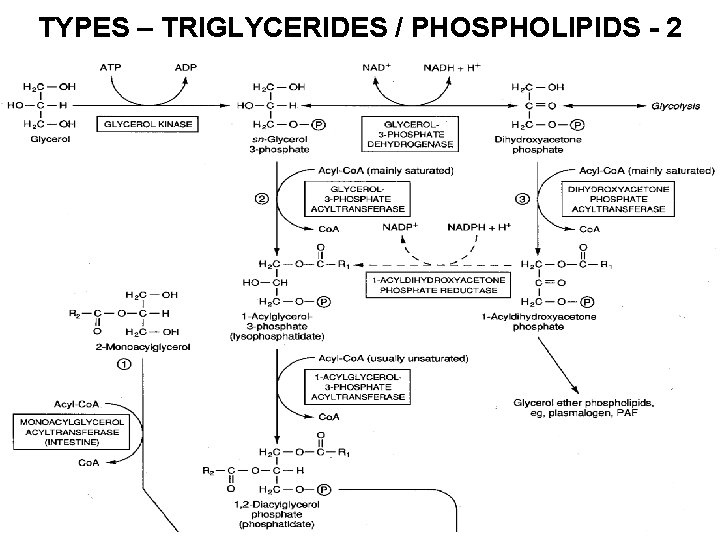
TYPES – TRIGLYCERIDES / PHOSPHOLIPIDS - 2
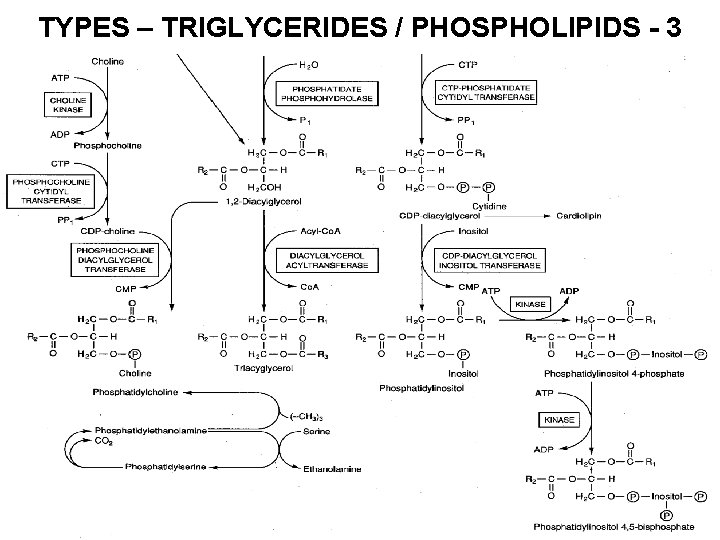
TYPES – TRIGLYCERIDES / PHOSPHOLIPIDS - 3
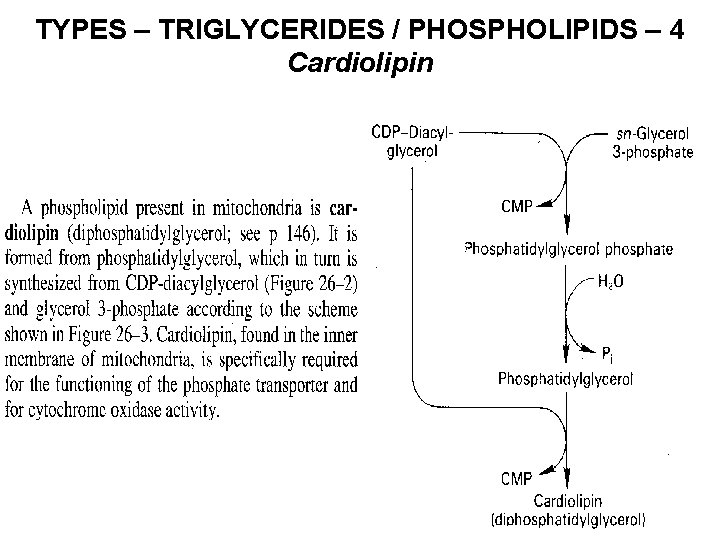
TYPES – TRIGLYCERIDES / PHOSPHOLIPIDS – 4 Cardiolipin
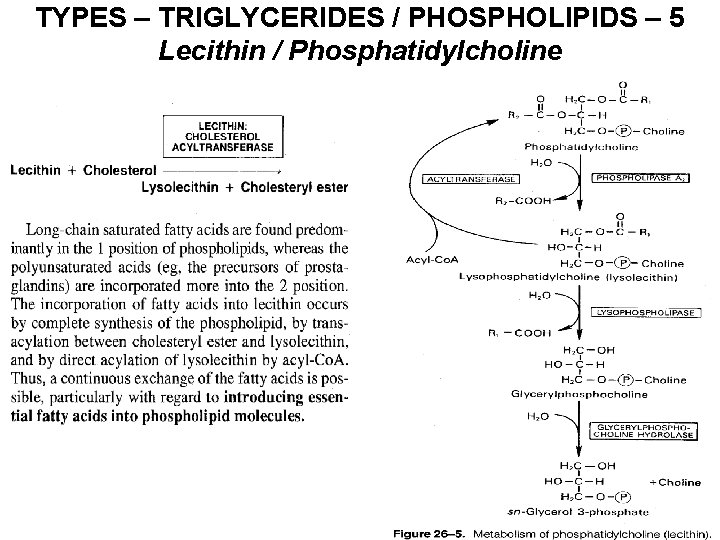
TYPES – TRIGLYCERIDES / PHOSPHOLIPIDS – 5 Lecithin / Phosphatidylcholine

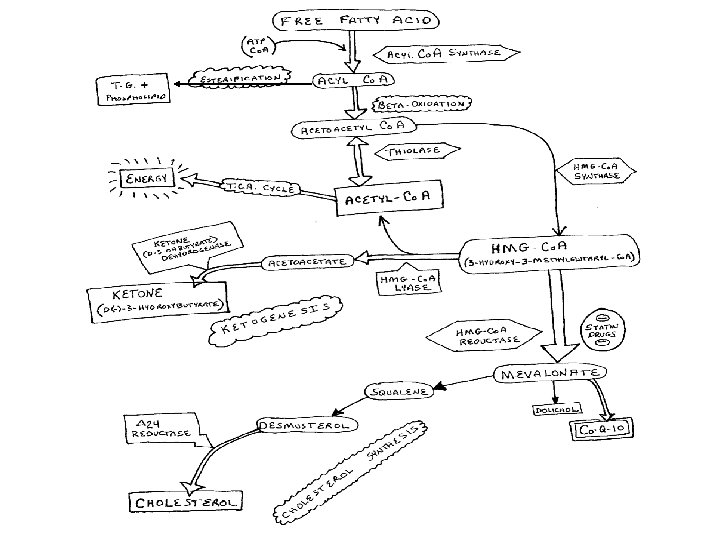
CHOLESTEROL Synthesis: Tissues occurs in: The liver is the major site of cholesterol synthesis although other tissues are also active (intestines, adrenals, gonads, skin, neural tissue, aorta). Precursors of Pathway & Intermediates: All the carbon atoms in cholesterol are provided by acetate & NADPH provides the reducing equivalents. Synthesis occurs in the cytoplasm. 1. B-hydroxy-B-methylgutaryl Co. A (HMG Co. A) is formed in the cytosol from acetyl Co. A in two steps. 2. The rate-limiting step of cholesterol synthesis is the conversion of HMG Co. A to mevalonate by HMG Co. A reductase. This rate-limiting step is inhibited by dietary cholesterol as well as endogenously synthesized cholesterol. 3. Melvalonate is activated & then decarboxylated to form 5 -carbon isoprenoid isomers> isopentenyl pyrophosphate (IPP) & 3, 3 dimethylallyl pyrophosphate (DPP). 4. The pyrophosphorylated isoprenoid units condense to form squalene. 5. Cyclization of squalene to form lanosterol. 6. Conversion of lanosterol to cholesterol. Functions: - Component of all cell membranes - Precursor of bile acids, steroid hormones (cortisol, aldosterone, testosterone, estradiol, progesterone) & vitamin D.
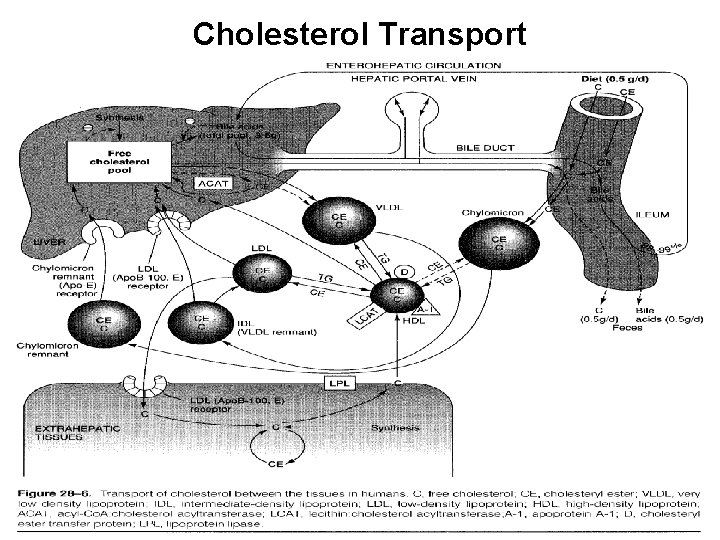
Cholesterol Transport
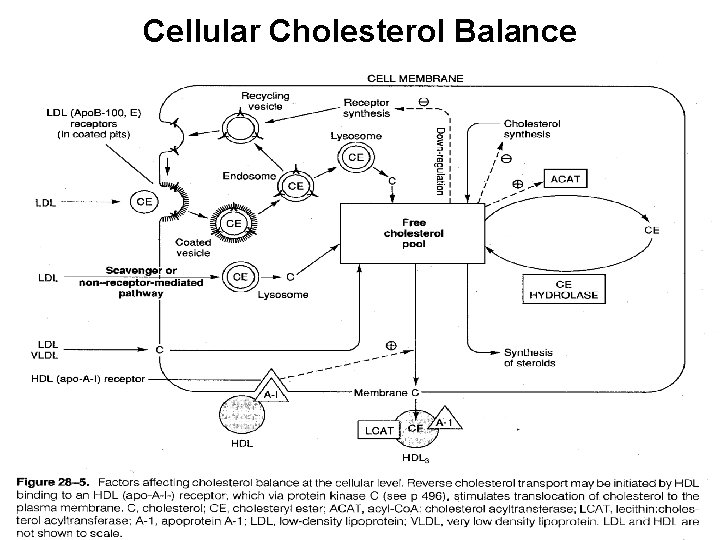
Cellular Cholesterol Balance
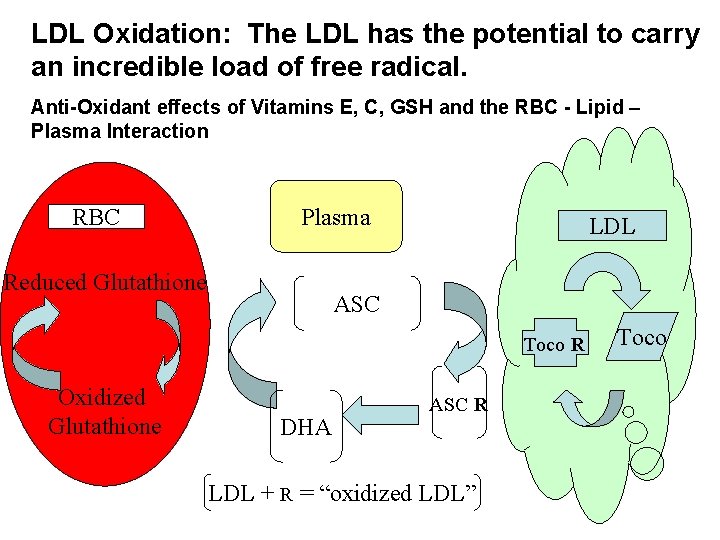
LDL Oxidation: The LDL has the potential to carry an incredible load of free radical. Anti-Oxidant effects of Vitamins E, C, GSH and the RBC - Lipid – Plasma Interaction RBC Plasma Reduced Glutathione LDL ASC Toco R Oxidized Glutathione DHA ASC R LDL + R = “oxidized LDL” Toco

EICOSANOIDS Biologically active 20 -carbon (eicosa-) fatty acids. These fatty acids give rise to the Prostanoids which specifically include the Prostaglandins, Thromboxanes, and Leukotrienes. These substances act as “local hormones” (paracrine substances) via G-protein linked cellular receptors. Of the three series of eicosanoids, two are completely essential (Linoleic and alpha Linolenic)fatty acids, and one is conditionally essential (Arachodonate). Each series gives rise to an individual set of prostaglandin, leukotriene and thromboxane. Each set is noted by series through the notation PG-1, LK-1, TX-1; PG-2, LK-2, TX-2; and PG-3, LK-3, TX-3. Each of these series of prostanoids has a slightly different physiologic role, and the three eicosanoid series work in concert to create inflammatory and anti-inflammatory balance.
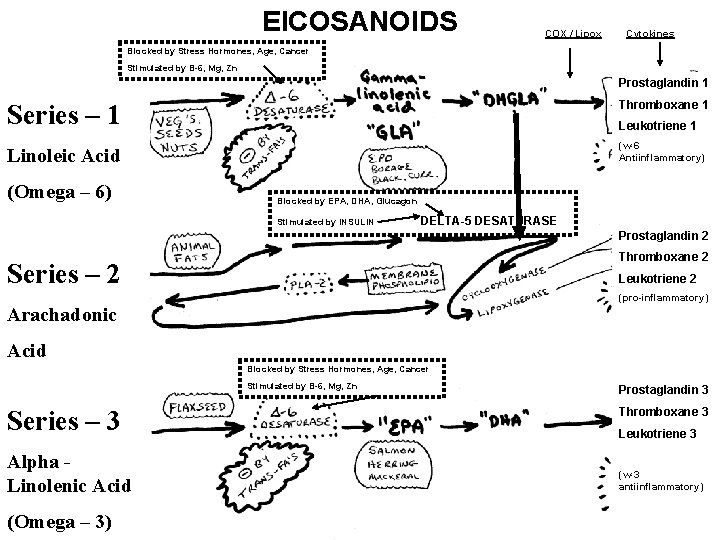
EICOSANOIDS COX / Lipox Cytokines Blocked by Stress Hormones, Age, Cancer Stimulated by B-6, Mg, Zn Prostaglandin 1 Series – 1 Thromboxane 1 Linoleic Acid (w-6 Antiinflammatory) (Omega – 6) Leukotriene 1 Blocked by EPA, DHA, Glucagon Stimulated by INSULIN DELTA-5 DESATURASE Prostaglandin 2 Thromboxane 2 Series – 2 Leukotriene 2 (pro-inflammatory) Arachadonic Acid Blocked by Stress Hormones, Age, Cancer Stimulated by B-6, Mg, Zn Series – 3 Alpha Linolenic Acid (Omega – 3) Prostaglandin 3 Thromboxane 3 Leukotriene 3 (w-3 antiinflammatory)
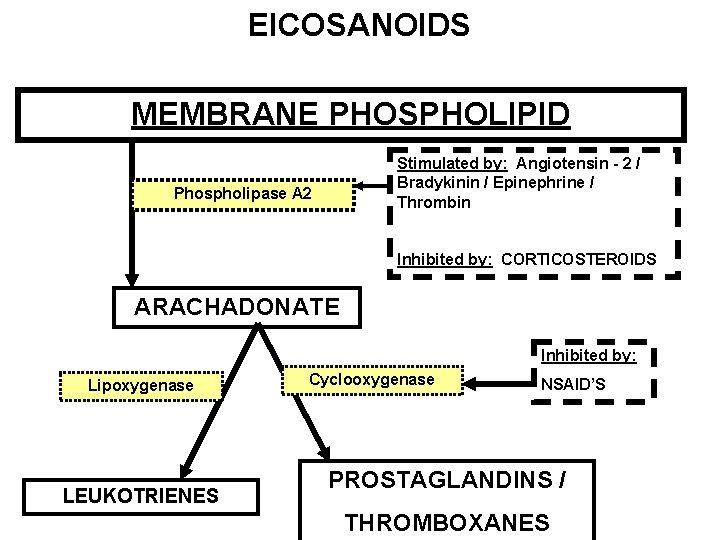
EICOSANOIDS MEMBRANE PHOSPHOLIPID Stimulated by: Angiotensin - 2 / Bradykinin / Epinephrine / Thrombin Phospholipase A 2 Inhibited by: CORTICOSTEROIDS ARACHADONATE Inhibited by: Lipoxygenase LEUKOTRIENES Cyclooxygenase NSAID’S PROSTAGLANDINS / THROMBOXANES

DIGESTION / ABSORPTION OF LIPIDS - 1 • ORAL – LINGUAL LIPASE • STOMACH – RENIN : Active in infants. Turns milk to paracasein – which is digested by pepsin) – HEAT: from stomach liquefies lipid – GASTRIC LIPASE : Hydrolyzes TG, Short and Medium chain FA’s. These absorb directly from the stomach into the portal circulation. • SMALL INTESTINE – BILE : Micelle Formation – CHOLESTEROL ESTERASE : Hydrolyzes Cholysteryl Esters to free cholesterol (absorbed intact) – PHOSPHOLIPASES : Attacks phospholipids, produces FFA, Glycerol, Choline (bases), and phosphoric acid.
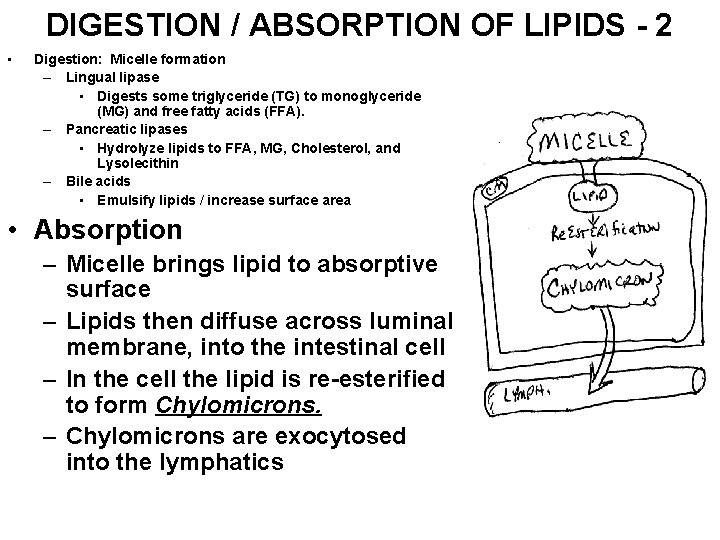
DIGESTION / ABSORPTION OF LIPIDS - 2 • Digestion: Micelle formation – Lingual lipase • Digests some triglyceride (TG) to monoglyceride (MG) and free fatty acids (FFA). – Pancreatic lipases • Hydrolyze lipids to FFA, MG, Cholesterol, and Lysolecithin – Bile acids • Emulsify lipids / increase surface area • Absorption – Micelle brings lipid to absorptive surface – Lipids then diffuse across luminal membrane, into the intestinal cell – In the cell the lipid is re-esterified to form Chylomicrons. – Chylomicrons are exocytosed into the lymphatics
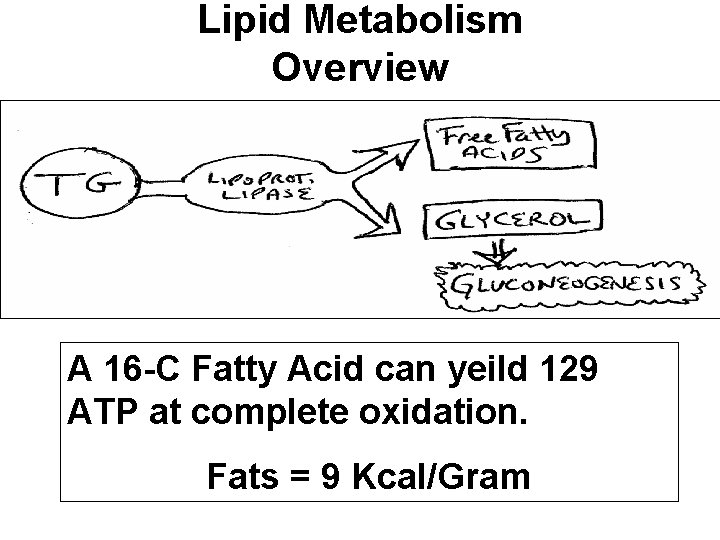
Lipid Metabolism Overview A 16 -C Fatty Acid can yeild 129 ATP at complete oxidation. Fats = 9 Kcal/Gram
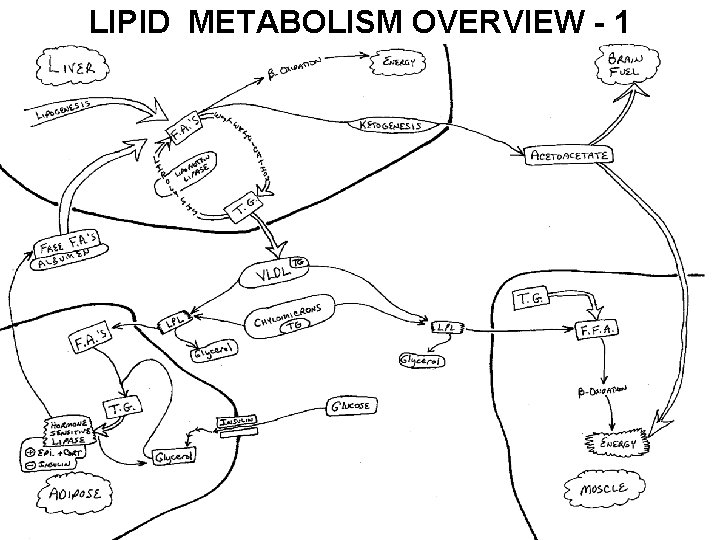
LIPID METABOLISM OVERVIEW - 1
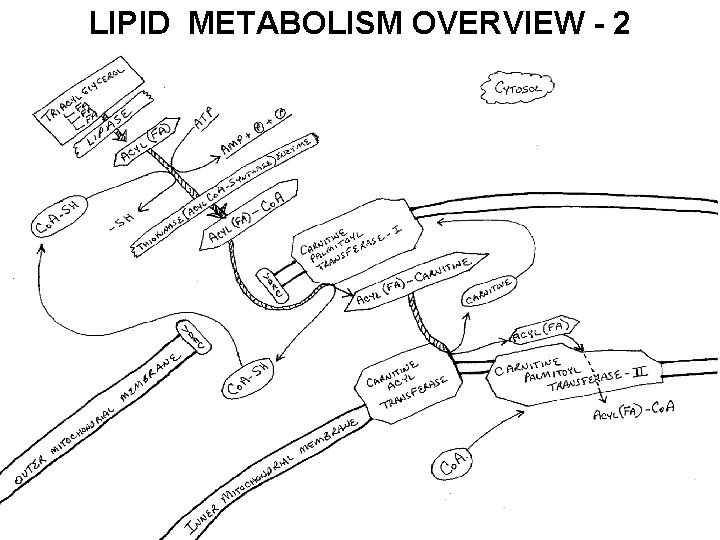
LIPID METABOLISM OVERVIEW - 2
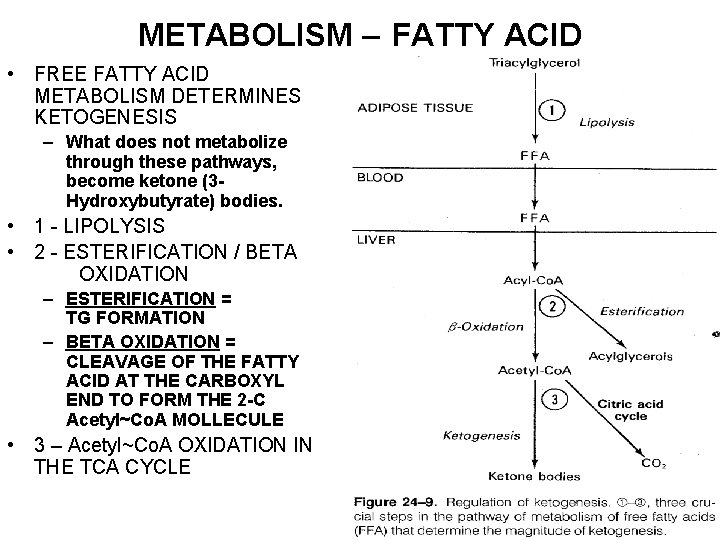
METABOLISM – FATTY ACID • FREE FATTY ACID METABOLISM DETERMINES KETOGENESIS – What does not metabolize through these pathways, become ketone (3 Hydroxybutyrate) bodies. • 1 - LIPOLYSIS • 2 - ESTERIFICATION / BETA OXIDATION – ESTERIFICATION = TG FORMATION – BETA OXIDATION = CLEAVAGE OF THE FATTY ACID AT THE CARBOXYL END TO FORM THE 2 -C Acetyl~Co. A MOLLECULE • 3 – Acetyl~Co. A OXIDATION IN THE TCA CYCLE

PROTEINS – STRUCTURE - 1 • PRIMARY: (all body proteins have) – Amino acid sequence. Covalently bonded. – Must have an enzyme to break. • SECONDARY: (all body proteins have) – First polypeptide folding, use disulfide bonds. • TERTIARY: – More folding, with secondary structure, s~s bonds • QUATERNARY: – Made with more than one polypeptide subunit – Subunits bonded by weak forces – Example: HEMOGLOBIN • NOTE: A protein is at least 10 amino acids in length, or it is considered a peptide.

PROTEINS – STRUCTURE (bonds) - 2 • Covalent: Strongest. Only broken by specific enzymes. 110 Kcal / energy • Weaker bonds: 1 to 5 Kcal / energy. Can be denatured by heat, acid… – Hydrogen: H and some electronegative atom. – Hydrophobic: Hydrophobic residues attract each other, within a protein. – Electrostatic: aka Salt bridges. Negative and positive charges on the same protein attract. – Van der Waals forces: Electrons attract one atom to another. – Disulfide (s~s): Covalent, but act like a weak bond. (Found in S-containing AA’s. ) CYS, MET.
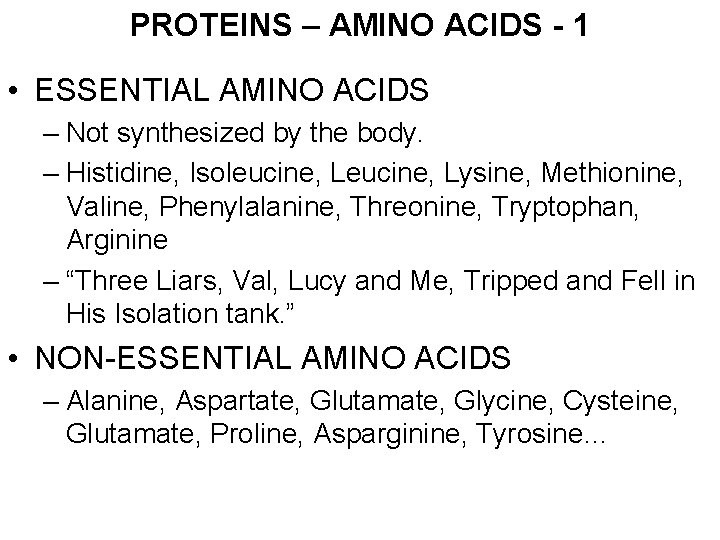
PROTEINS – AMINO ACIDS - 1 • ESSENTIAL AMINO ACIDS – Not synthesized by the body. – Histidine, Isoleucine, Lysine, Methionine, Valine, Phenylalanine, Threonine, Tryptophan, Arginine – “Three Liars, Val, Lucy and Me, Tripped and Fell in His Isolation tank. ” • NON-ESSENTIAL AMINO ACIDS – Alanine, Aspartate, Glutamate, Glycine, Cysteine, Glutamate, Proline, Asparginine, Tyrosine…

PROTEINS – AMINO ACIDS - 2 • FUNCTIONS: – Synthesis of structural proteins (ie. Collagen) – Base for Enzymes – Conversion to other molecules: • Tyrosine to Thyroxine, Histadine to Histamine… – FUEL (4 Kcal / gram) • Via transformation to pyruvate, acyl~Co. A, … • GLUCOGENIC TYPES – Can convert to Glucose • KETOGENIC TYPES (Ketogenic Amines) – Those that convert to acetyl~Co. A (and are too late to be Glucose)

PROTEINS – AMINO ACIDS - 3
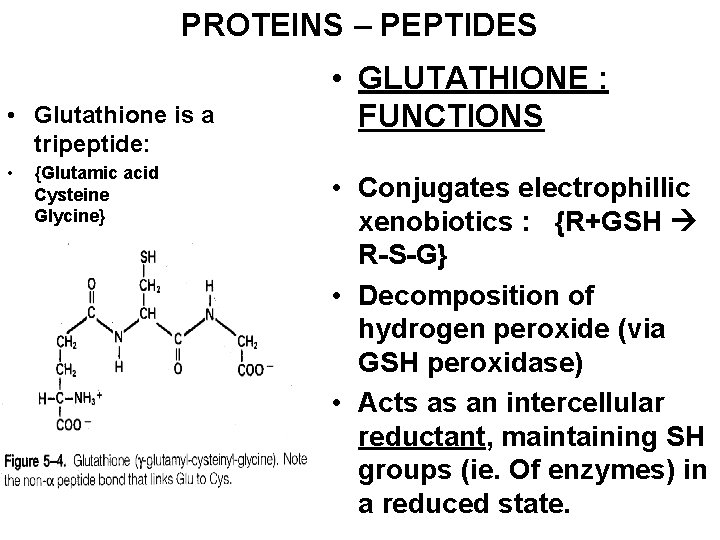
PROTEINS – PEPTIDES • Glutathione is a tripeptide: • {Glutamic acid Cysteine Glycine} • GLUTATHIONE : FUNCTIONS • Conjugates electrophillic xenobiotics : {R+GSH R-S-G} • Decomposition of hydrogen peroxide (via GSH peroxidase) • Acts as an intercellular reductant, maintaining SH groups (ie. Of enzymes) in a reduced state.

PROTEINS – ZYMOGEN SYSTEM • Zymogen: Inactive precursor of an Active enzyme. • Activity is induced by: – Cleavage of bonds or addition of phosphoryl or other group to the molecule. • Zymogen – Trypsinogen – Pepsinogen – Prothrombin Enzyme Trypsin Pepsin Thrombin • **** Non-Enzyme Examples: – Proinsulin – Procollagen Insulin Collagen
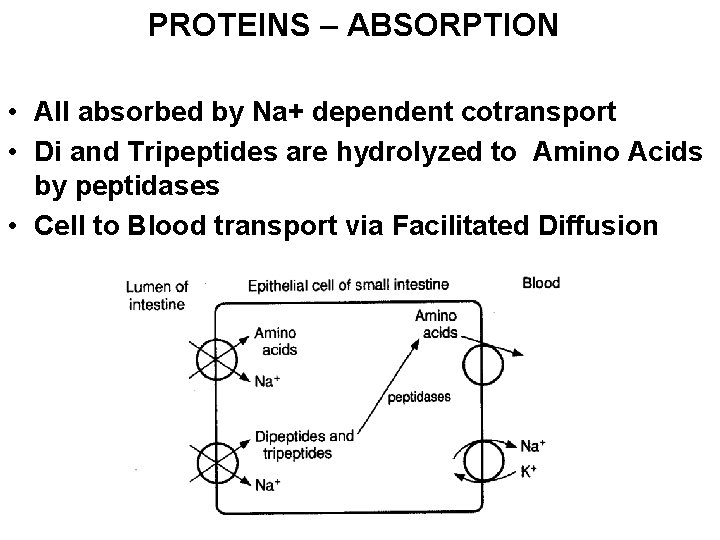
PROTEINS – ABSORPTION • All absorbed by Na+ dependent cotransport • Di and Tripeptides are hydrolyzed to Amino Acids by peptidases • Cell to Blood transport via Facilitated Diffusion
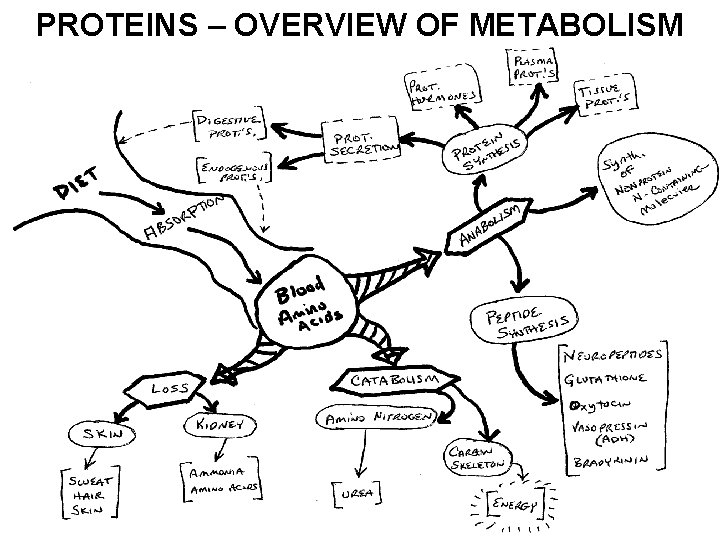
PROTEINS – OVERVIEW OF METABOLISM

ENZYMES – GENERAL INFORMATION • Speed up and already thermodynamically favorable reaction by lowering the energy of activation. – Enzyme is specific and powerful as a catalyst – Enzyme causes faster production of product • The enzyme must bind to a substrate for a product to be produced. – Enzyme is not part of any product. • Enzyme activity (reaction rate) is affected by temperature. – All enzymes have p. H and temperature optimums. • Cofactors (coenzymes) are small molecular weight substances (B-vitamins and trace minerals) that are needed to help some enzymes react. – Typically the cofactor is part of the substrate binding site, on the enzyme.

ENZYMES - 1 • INHIBITION – Competitive: attach at the substrate binding site, resemble the substrate. – Noncompetitive: reversibly bind to a non-substrate site. – Irreversible (poison): Heavy metals, etc. • KINETICS – Michaelis – Menten (and Hill) • Saturable quality of Enzyme reactions: • Estimating the speed of enzymatic reaction based on [substrate. ] • Once the enzyme system is saturated (by substrate) the kinetics (e. A) change.

ENZYMES – 2 : TYPES • Know the Digestive Enzymes, AND… • Hydrolase – Adds H 20 to break bonds (Hydrolysis) • Aldolase – Cleaves C-C bonds to form Aldehydes • Dehydrogenase – Removes H from substrate • Hydratase – Adds H 2 O to C-C bonds without breaking the bond – It can also remove H 2 O to create a double bond • Isomerase – Converts between Isomers: • Cis / Trans, D & L, or Aldolase and Ketolase • Esterase – Hydrolyzes Ester Linkages to form an Alcohol and Acid

CLINICAL BIOCHEMISTRY • Hepatic Detox • Metabolic Control • BLOOD CONSTITUENTS – Glucose – Cholesterol – Triglyceride – Non-protein Nitrogens – Carrier proteins – Enzymes – Non-steroidal Hormones
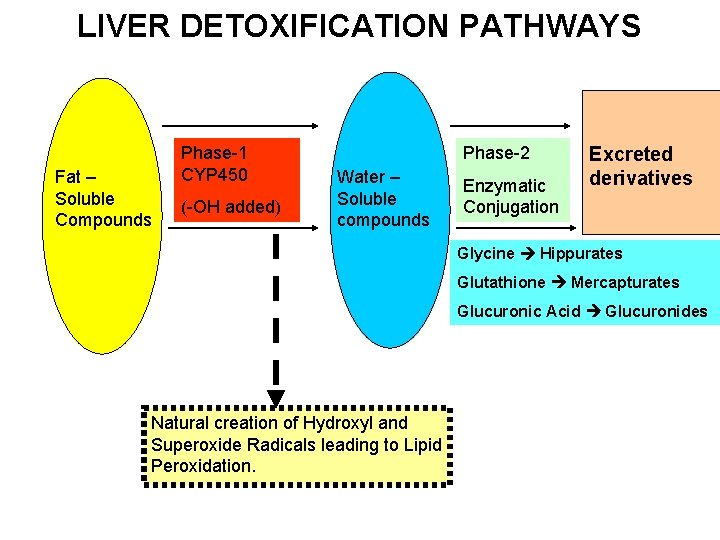
LIVER DETOXIFICATION PATHWAYS Fat – Soluble Compounds Phase-1 CYP 450 (-OH added) Phase-2 Water – Soluble compounds Enzymatic Conjugation Excreted derivatives Glycine Hippurates Glutathione Mercapturates Glucuronic Acid Glucuronides Natural creation of Hydroxyl and Superoxide Radicals leading to Lipid Peroxidation.
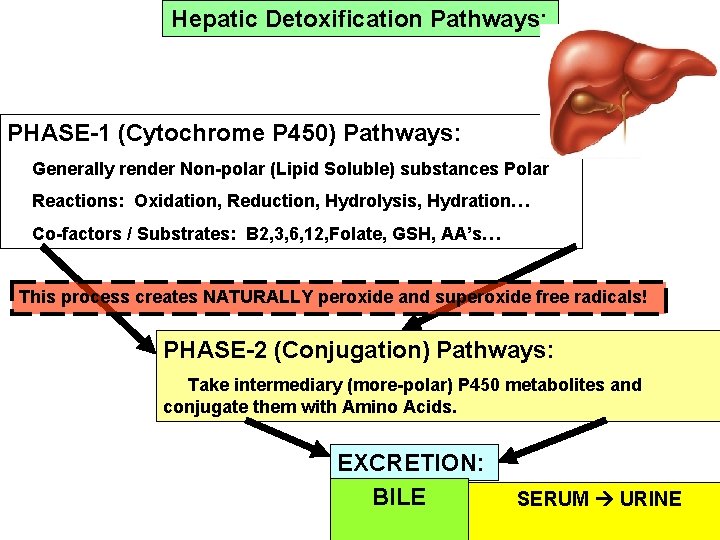
Hepatic Detoxification Pathways: PHASE-1 (Cytochrome P 450) Pathways: Generally render Non-polar (Lipid Soluble) substances Polar Reactions: Oxidation, Reduction, Hydrolysis, Hydration… Co-factors / Substrates: B 2, 3, 6, 12, Folate, GSH, AA’s… This process creates NATURALLY peroxide and superoxide free radicals! PHASE-2 (Conjugation) Pathways: Take intermediary (more-polar) P 450 metabolites and conjugate them with Amino Acids. EXCRETION: BILE SERUM URINE
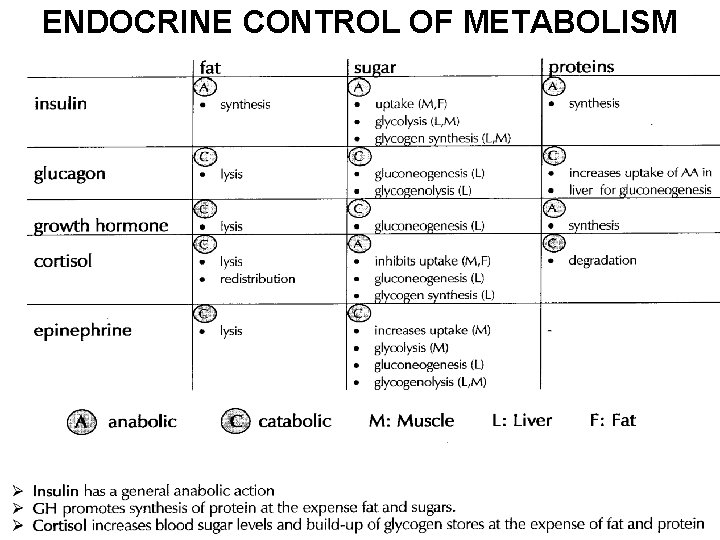
ENDOCRINE CONTROL OF METABOLISM
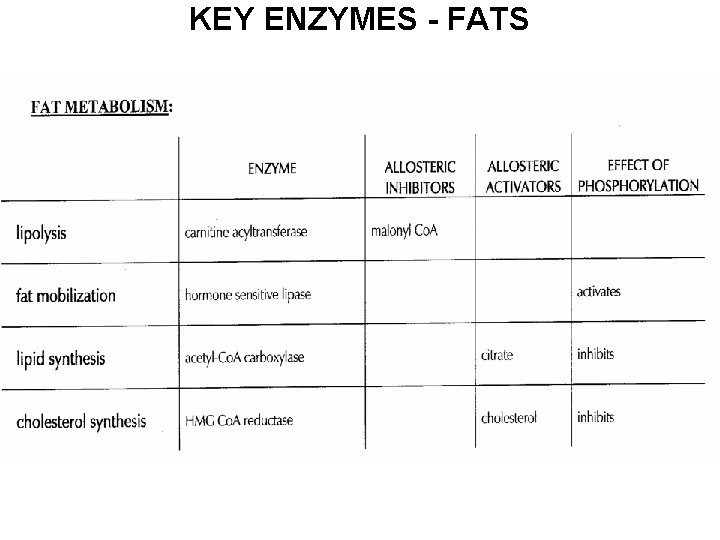
KEY ENZYMES - FATS
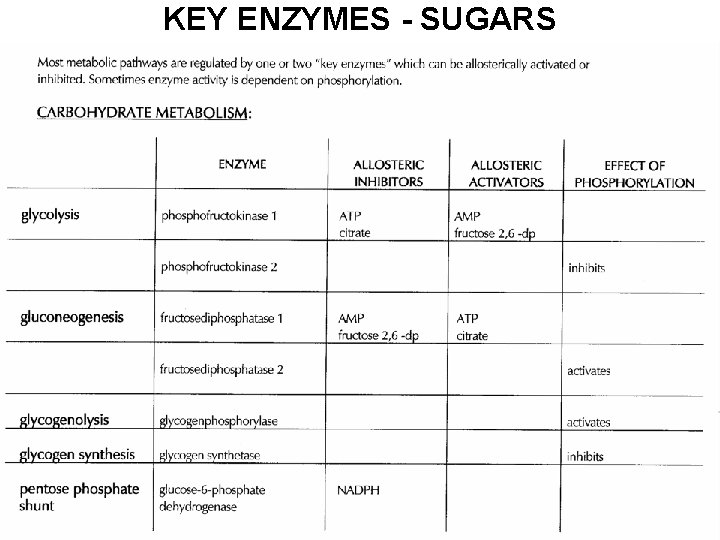
KEY ENZYMES - SUGARS
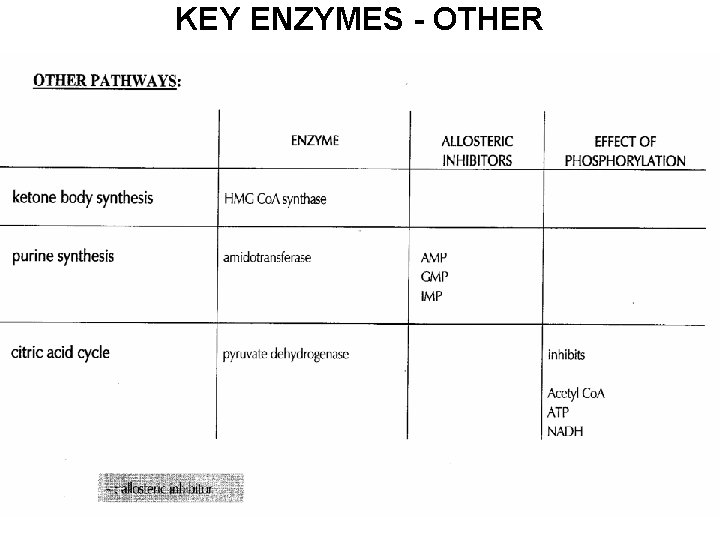
KEY ENZYMES - OTHER
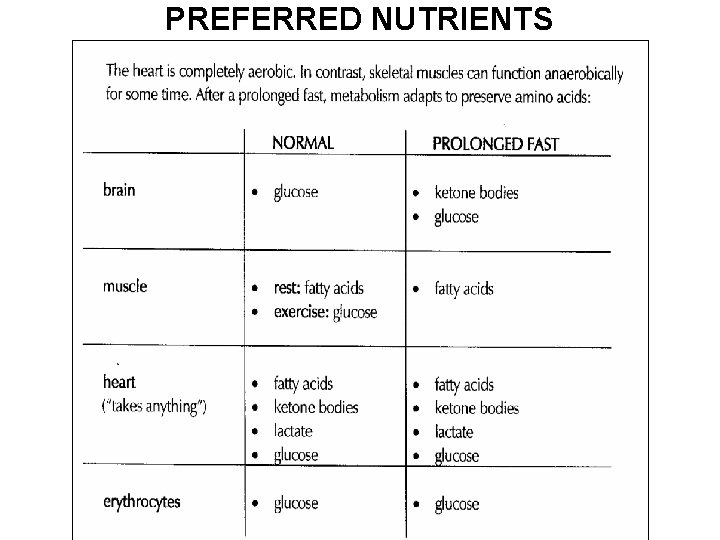
PREFERRED NUTRIENTS
- Slides: 45