Integ Science Location Due Date Textbook Notebook Today
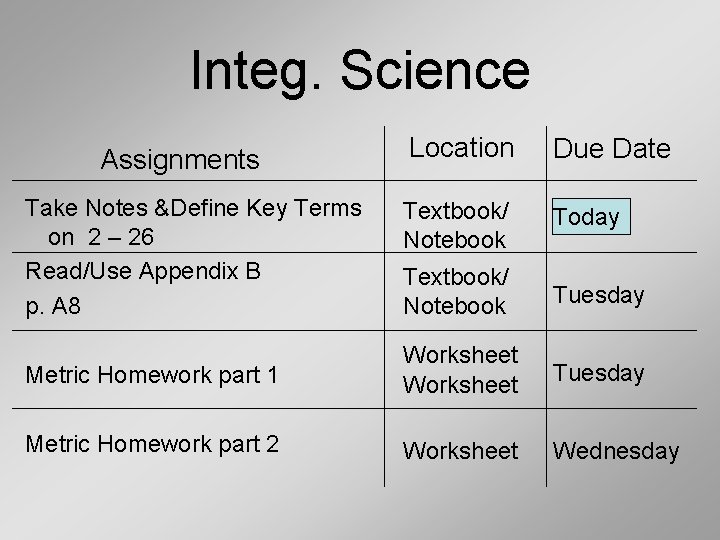
Integ. Science Location Due Date Textbook/ Notebook Today Textbook/ Notebook Tuesday Metric Homework part 1 Worksheet Tuesday Metric Homework part 2 Worksheet Wednesday Assignments Take Notes &Define Key Terms on 2 – 26 Read/Use Appendix B p. A 8

Precision and Accuracy Precision indicates degree of reproducibility of a measured number. Accuracy indicates how close your measurements are to the true value.

Precision and Accuracy When making measurements in science you want them to be both precise and accurate.

SI Base Units Physical Quantity Time Length Temperature Mass Volume Amount Unit (standard) Symbol
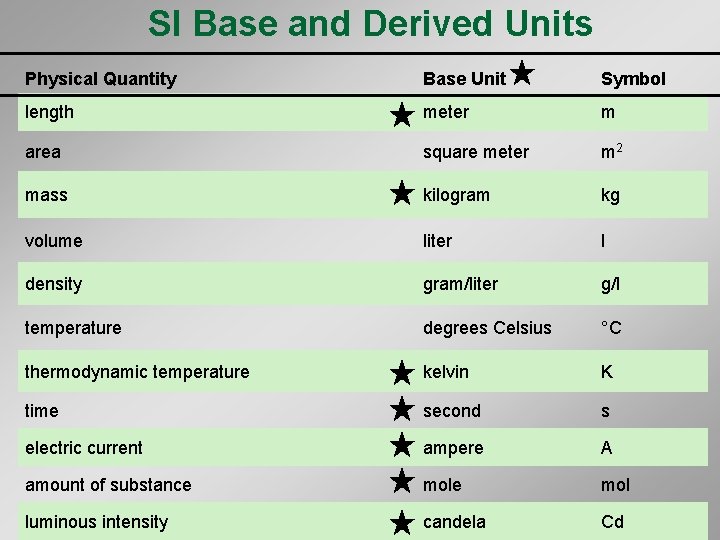
SI Base and Derived Units Physical Quantity Base Unit Symbol length meter m area square meter m 2 mass kilogram kg volume liter l density gram/liter g/l temperature degrees Celsius °C thermodynamic temperature kelvin K time second s electric current ampere A amount of substance mol luminous intensity candela Cd

Metric System Designed during the French Revolution of the 1790's, the metric system brought order out of the conflicting and confusing traditional systems of weights and measures then being used in Europe. Prior to the introduction of the metric system, it was common for units of length, land area, and weight to vary, not just from one country to another but from one region to another within the same country.

Metric System • The metric system replaces all the traditional units, except the units of time and of angle measure, with units satisfying three conditions: • (1) One fundamental unit is defined for each quantity. These units are now defined precisely in the International System of Units. • (2) Multiples and fractions of these fundamental units are created by adding prefixes to the names of the defined units. • (3) The fundamental units are defined rationally and are related to each other in a rational fashion. • The metric units were defined in an elegant way unlike any traditional units of measure. The Earth itself was selected as the measuring stick. The meter was defined to be one ten-millionth of the distance from the Equator to the North Pole
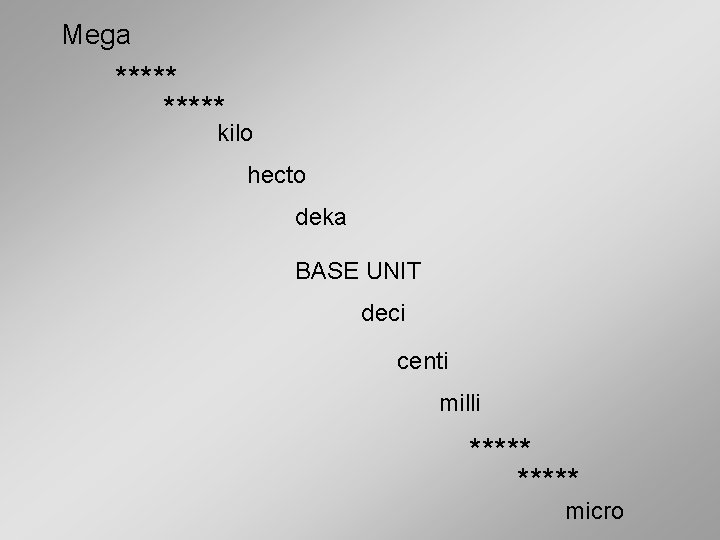
Mega ***** kilo hecto deka BASE UNIT deci centi milli ***** micro

Metric System Prefixes giga – G 1, 000, 000 = 1*10 9 mega – M 1, 000 = 1*10 6 kilo – k 1, 000 = 1*10 3 hecto – h 100 = 1*10 2 deka – da 10 = 1*10 1 Base Unit – (meter, gram, liter, second) = 1*10 0 deci – d 0. 1 = 1*10 -1 centi – c 0. 01 = 1*10 -2 milli – m 0. 001 = 1*10 -3 micro - µ 0. 000 001 = 1*10 -6 nano – n 0. 000 001 = 1*10 -9

Metric System Understanding prefixes Prefixes are short names and letter symbols for numbers (powers of ten). A prefix is attached to the front of a unit, without a space. Prefixes are easier to write and say than powers of ten, ordinary notation, or traditional number names. Compare: 25 MW(pronounced and spelled out: 25 megawatts) 25 X 106 (the 106 is a power of ten) 25 000 W (ordinary notation) 25 million watts (traditional number name)

Metric System As you go up the "ladder" of these prefixes, the unit is multiplied in steps of 1000, or 103. km = 1000 X m Mm = 1000 X km Gm = 1000 X Mm [kilometer] [megameter] [gigameter] Going down the prefix scale, a unit is divided in steps of 1000. In other words, it is multiplied in steps of 0. 001 (= 1/1000). mm = 0. 001 X m µm = 0. 001 X mm nm = 0. 001 X µm [millimeter] [micrometer] [nanometer]

Metric System Changing prefixes by moving the decimal point Choose a prefix that will simplify an expression by eliminating unnecessary placeholding zeros (non-significant digits). To switch to the next larger prefix, move the decimal point three places to the left. – 4 000 m = 4 km – 1 500 mg = 1. 5 g – 500 m. L = 0. 5 L – 76 000 kg = 76 Mg – 2 300 µs = 2. 3 ms To switch to the next smaller prefix, move the decimal point three places to the right. – 0. 005 m = 5 mm – 0. 009 kg = 9 g – 0. 003 2 mm = 3. 2 µm When moving the decimal point to the right, you may have to add one or two place holding zeros at the end of the number to show where the (unexpressed) decimal point goes. – 0. 03 g = 30 mg – 0. 2 L = 200 m. L

Practice Problems 1) 120 mm = ________cm 2) 48. 6 g = ________ cg 3) 84, 000 cm = ________ Mm 4) 19. 7 mm = ________m 5) 23. 89 km = ________cm 6). 098 mg = ________kg 7) 29. 9 Ms = ________µs

Practice Problems 1. 421 m = ________cm 2. 486 cg = ________ Mg 3. 17, 000 km = ________ Mm 4. 17 mm = ________dam 5. 23 km = ________cm 6. 225, 081 mg = ________kg 7. 53 Ms = ________µs

English – Metric Conversion Tables
![Linear Measure Imperial Metric 1 inch [in] 2. 54 cm 1 foot [ft] 12 Linear Measure Imperial Metric 1 inch [in] 2. 54 cm 1 foot [ft] 12](http://slidetodoc.com/presentation_image_h2/6b675e77f8be50c12f9862ea793f2d82/image-16.jpg)
Linear Measure Imperial Metric 1 inch [in] 2. 54 cm 1 foot [ft] 12 in 0. 3048 m 1 yard [yd] 3 ft 0. 9144 m 1 mile 1760 yd 1. 6093 km 1 nautical mile 2025. 4 yd 1. 852 km

Linear Measure Practice • Inches to Centimeters and cm to in 34. 3 in = ? ? ? cm 94 cm = ? ? ? in • Feet to Meters and m to ft 8 ft = ? ? ? m 323 m = ? ? ? ft • Yards to Meters and m to yd 100 yd = ? ? ? m 7. 24 m = ? ? ? yd • Miles to Kilometers and km to mi. 51. 8 mi = ? ? ? km 5 km = ? ? ? mi
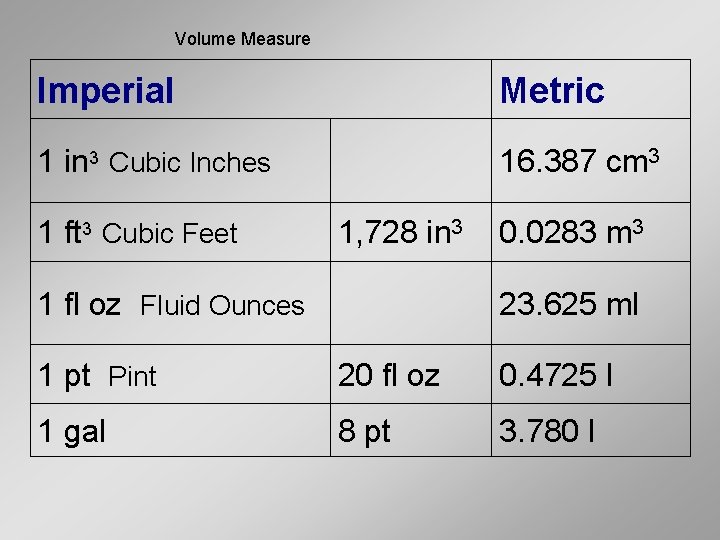
Volume Measure Imperial Metric 1 in 3 Cubic Inches 16. 387 cm 3 1 ft 3 Cubic Feet 1, 728 in 3 1 fl oz Fluid Ounces 0. 0283 m 3 23. 625 ml 1 pt Pint 20 fl oz 0. 4725 l 1 gal 8 pt 3. 780 l

Volume Practice • Cubic inches to cubic centimeters 15 in 3 = ? ? ? cm 3 31. 7 cm 3 = ? ? ? in 3 • Cubic feet to cubic meters 3 ft 3 = ? ? ? m 3 894 m 3 = ? ? ? ft 3 • Ounces to milliliters 89 oz = ? ? ? ml 89 ml = ? ? ? oz • Gallons to liters 4 gal = ? ? ? L 63 L = ? ? ? gal
![Mass Measure Imperial Metric 1 ounce [oz] 437. 5 grain 28. 35 g 1 Mass Measure Imperial Metric 1 ounce [oz] 437. 5 grain 28. 35 g 1](http://slidetodoc.com/presentation_image_h2/6b675e77f8be50c12f9862ea793f2d82/image-20.jpg)
Mass Measure Imperial Metric 1 ounce [oz] 437. 5 grain 28. 35 g 1 pound [lb] 16 oz 0. 4536 kg 1 stone 14 lb 6. 3503 kg 1 hundredweight [cwt] 112 lb 50. 802 kg 1 long ton (UK) 20 cwt 1. 016 t 1 short ton (US) 2, 000 lb 0. 907 t

Physical Properties Mass, Volume, and Density
Mass What do you know about mass?

Mass • Measure of the amount of matter that makes up an object. • Units used to designate mass are kilograms (kg) • You can measure an objects mass using a balance (triple beam, electronic, spring).

Volume What is Volume?

Volume • Volume is a measurement of the threedimensional space occupied by an object. • Units include cm 3 and m. L. • Solids, liquids, and gases all have volume, but you measure each differently. – Solid – calculate geometrically or displacement – Liquid – measure using a graduated cylinder

Density How does Density relate to Mass and Volume?

Density • The amount of matter in a given space. – Does this sound familiar? • Concentration or Compactness • The unit for density is or .

Mass, Volume, and Density • Mass volume and density are directly related. Practice Exercise: 1. Measure the mass and volume of an object in the room. 2. Calculate the Density of the Object. 3. What are the units associated with this calculation?

Formulae Density = Mass / Volume Mass = Density x Volume = Mass / Density

Practice Problems 1. Calculate the volume of an object that is 34 cm by 25 cm by 8 cm. 2. Given: V = 50 m. L Calculate: Mass D =. 75 g/m. L 3. Given: M = 55 g Calculate: Volume D = 2. 3 g/cm 3 4. Given: M =. 13 kg Calculate: Density V = 20 m. L

Significant Figures • It is important to record the precision of your measurements so that other people can understand interpret your results. • A common convention used in science to indicate precision is known as significant figures. • Significant figures are those digits in a measurement that are known with certainty plus the first digit that is uncertain.

Significant Figures Even though this ruler is marked in only centimeters and half-centimeters, if you estimate, you can use it to report measurements to a precision of a millimeter.

Rules for Sig Fig Rule 1 Zeros between other nonzero digits are significant. Examples a. 50. 3 m has three significant figures b. 3. 0025 s has five significant figures

Rules for Sig Fig Rule 2 Zeros in front of nonzero digits are not significant. Examples a. 0. 892 has three significant figures b. 0. 0008 s has one significant figure

Rules for Sig Fig Rule 3 Zeros that are at the end of a number and also to the right of a decimal point are significant. Examples a. 57. 00 g has four significant figures b. 2. 000 kg has seven significant figure

Rules for Sig Fig Rule 4 Zeros that are at the end of a number but left of the decimal point are not significant. Examples a. 100 m has ONE significant figure b. 20 m has ONE significant figure

Rules for Sig. Fig. Extra Rule Zeros that are at the end of a number but left of the decimal point that are measured to be significant are indeed significant. Examples a. A scale measures 1200. kg has four significant figures and is written in scientific notation: 3 1. 200 x 10 kg so Rule 3 applies
- Slides: 37