Insulin Pump Therapy Evidence Based Medicine Saeid Kalbasi

Insulin Pump Therapy: Evidence Based Medicine Saeid Kalbasi, MD Associate Prof. of Endocrinology and Metabolism Shahid Beheshti University of Medical Sciences Research Institute of Endocrine Sciences
Insulin Pump Therapy: Evidence Based Medicine
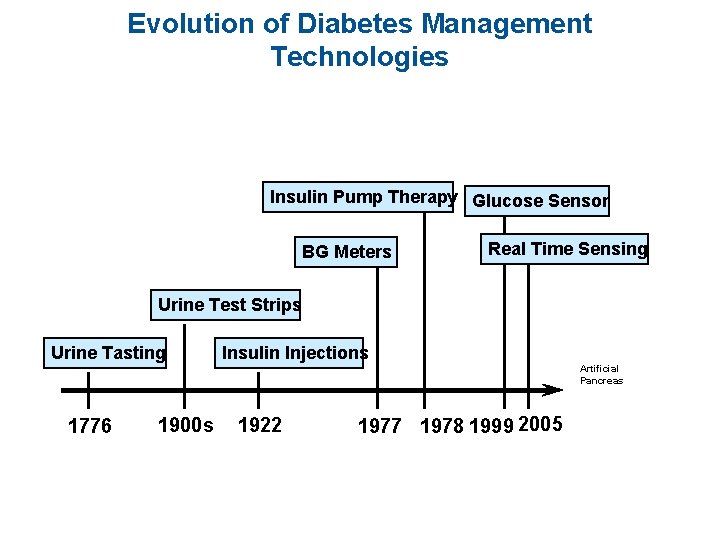
Evolution of Diabetes Management Technologies Insulin Pump Therapy Glucose Sensor BG Meters Real Time Sensing Urine Test Strips Urine Tasting Insulin Injections Artificial Pancreas 1776 1900 s 1922 1977 1978 1999 2005
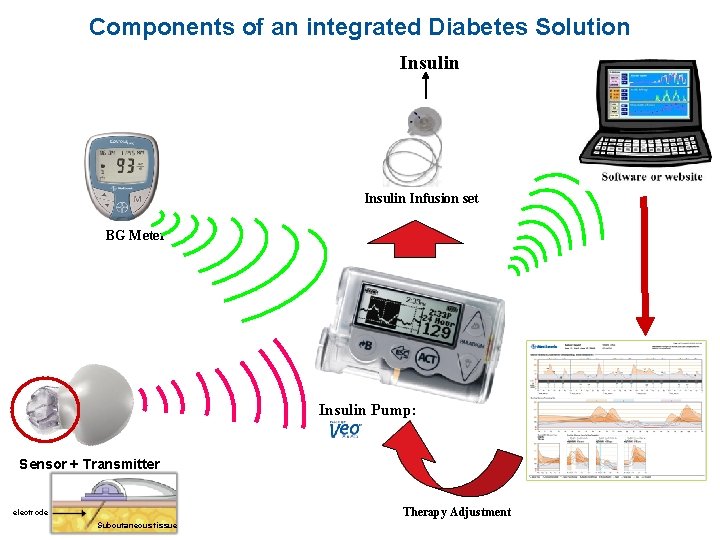
Components of an integrated Diabetes Solution Insulin Infusion set BG Meter Insulin Pump: Sensor + Transmitter Therapy Adjustment electrode Subcutaneous tissue
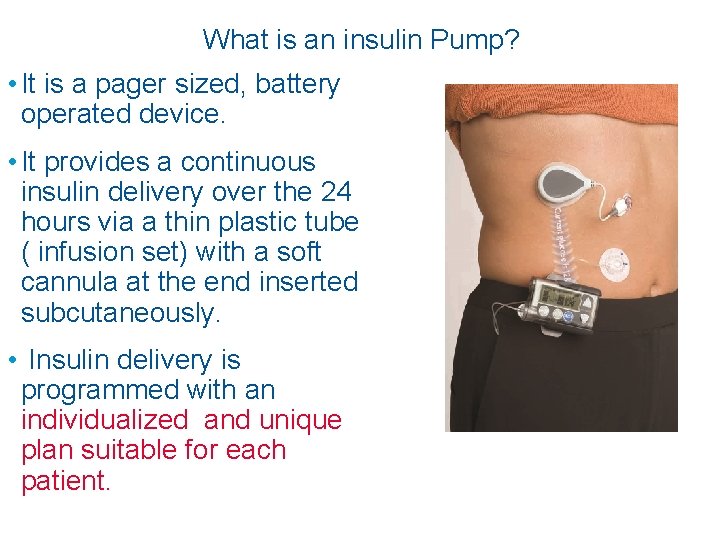
What is an insulin Pump? • It is a pager sized, battery operated device. • It provides a continuous insulin delivery over the 24 hours via a thin plastic tube ( infusion set) with a soft cannula at the end inserted subcutaneously. • Insulin delivery is programmed with an individualized and unique plan suitable for each patient.

Pump Therapy Basics Basal Rate • Preprogrammed • Continuous subcutaneous flow of fast-acting insulin • Matching variable metabolic needs • Meal Bolus – Matching insulin to carbohydrates in meal • Correction bolus – For high BG 6 5 Meal Bolus 4 3 Basal Rate 2 1 12 am 12 pm 12 am

Benefits of Insulin Pump Therapy • Improved Control Hb. A 1 c Hypoglycemia • Improved Quality of Life • Decreased Cost of Care

Pharmacokinetic Advantages • Uses only Short Acting Insulin – More predictable absorption than with modified insulins (variation 3 -7% vs 20 -50%*) • Uses one injection site for 2 to 3 days – Reduces variations in absorption due to site rotation • Eliminates most of the subcutaneous insulin depot • Programmable delivery allows closest match with physiologic needs * Lauritzen: Diabetologia 1983; 24: 326 -9

Indications In detail: • Dawn-Phenomenon • planned pregnancy • Dusk-Phenomenon • long fasting periods • recurrent hypoglycemia • shift worker • hypoglycemic unawareness • sporting activities • instable diabetic control • “time zone jumper” • low insulin amounts • Type 2

Patient Selection Criteria • Is motivated • Wants to improve glucose control • Desires to live a more normal life • Has family support • Has realistic expectations • Willing to perform at least 4 -6 BG tests a day • Psychologically stable Tanenberg: The Insulin Pump Book, Mini. Med 1995: 21 -30

Clinical studies

DCCT / EDIC n DCCT (1993): Diabetes Control and Complications Trial EDIC (2000, 2005): Epidemiology of Diabetes Interventions and Complications n

Major steps: DCCT / EDIC DCCT enrollment: 1983 -1989 n End of DCCT 1993 n – A 10 -year study n EDIC Nephropathy & Retinopathy: – 4 years post DCCT end. n EDIC CVD: – 11 years post DCCT end

DCCT: Study design Patients with type 1 diabetes (n = 1441) Primary prevention* (n = 726) Secondary intervention+ (n = 715) Randomise Conventional Intensive DCCT: N Engl J Med 1993; 329: 977– 86 Conventional * No retinopathy Intensive + mild retinopathy
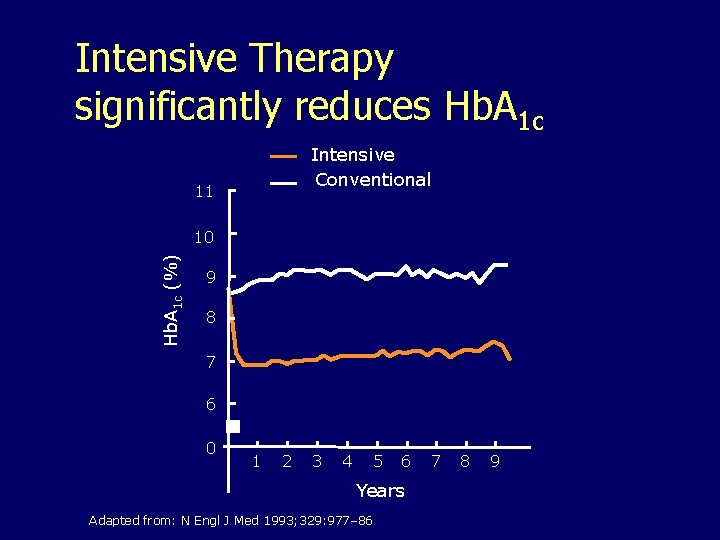
Intensive Therapy significantly reduces Hb. A 1 c Intensive Conventional 11 Hb. A 1 c (%) 10 9 8 7 6 0 1 2 3 4 5 6 Years Adapted from: N Engl J Med 1993; 329: 977– 86 7 8 9
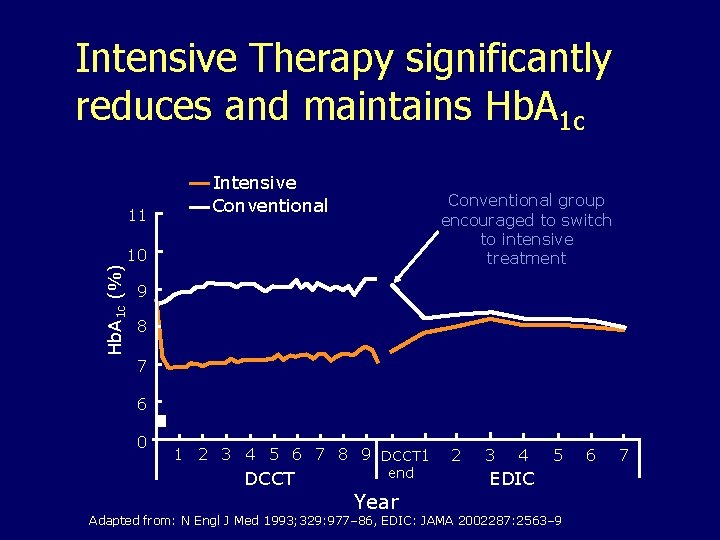
Intensive Therapy significantly reduces and maintains Hb. A 1 c 11 Intensive Conventional group encouraged to switch to intensive treatment Hb. A 1 c (%) 10 9 8 7 6 0 1 2 3 4 5 6 7 8 9 DCCT 1 DCCT end Year 2 3 4 5 EDIC Adapted from: N Engl J Med 1993; 329: 977– 86, EDIC: JAMA 2002287: 2563– 9 6 7
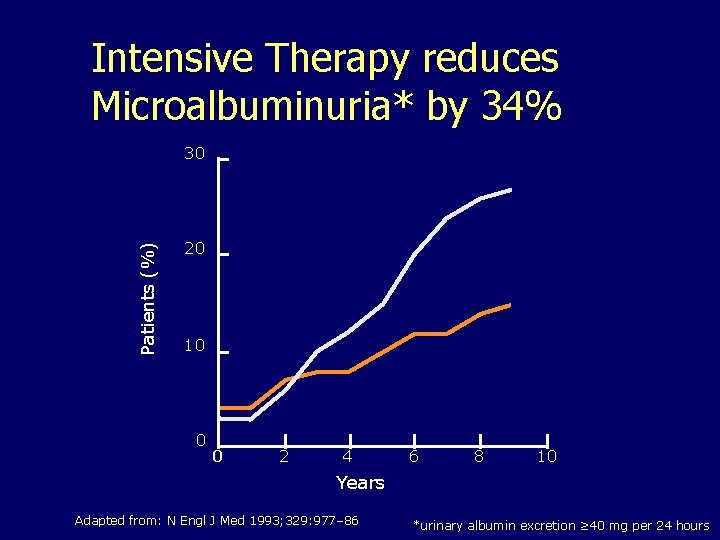
Intensive Therapy reduces Microalbuminuria* by 34% Patients (%) 30 20 10 0 0 2 4 6 8 10 Years Adapted from: N Engl J Med 1993; 329: 977– 86 *urinary albumin excretion ≥ 40 mg per 24 hours
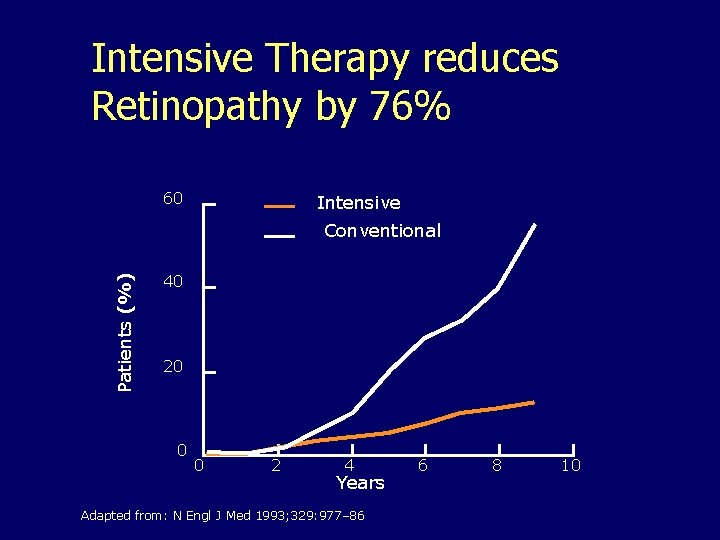
Intensive Therapy reduces Retinopathy by 76% 60 Intensive Patients (%) Conventional 40 20 0 0 2 4 Years Adapted from: N Engl J Med 1993; 329: 977– 86 6 8 10

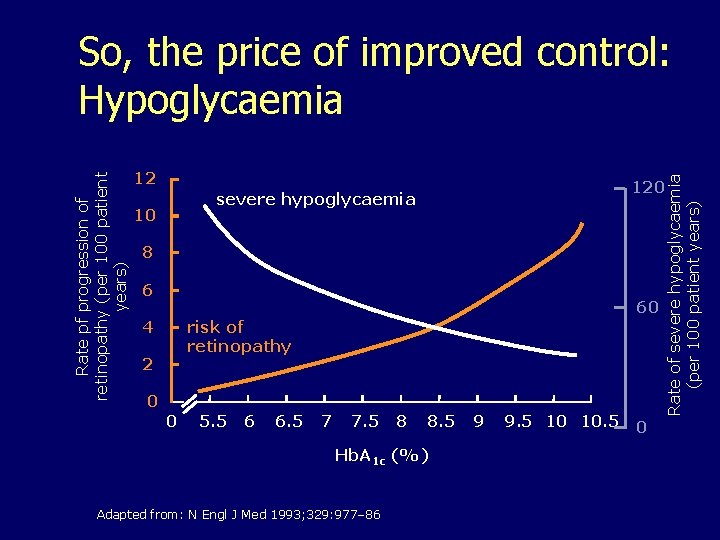
12 120 severe hypoglycaemia 10 8 6 60 risk of retinopathy 4 2 0 0 5. 5 6 6. 5 7 7. 5 8 8. 5 Hb. A 1 c (%) Adapted from: N Engl J Med 1993; 329: 977– 86 Rate of severe hypoglycaemia (per 100 patient years) Rate pf progression of retinopathy (per 100 patient years) So, the price of improved control: Hypoglycaemia 9 9. 5 10 10. 5 0
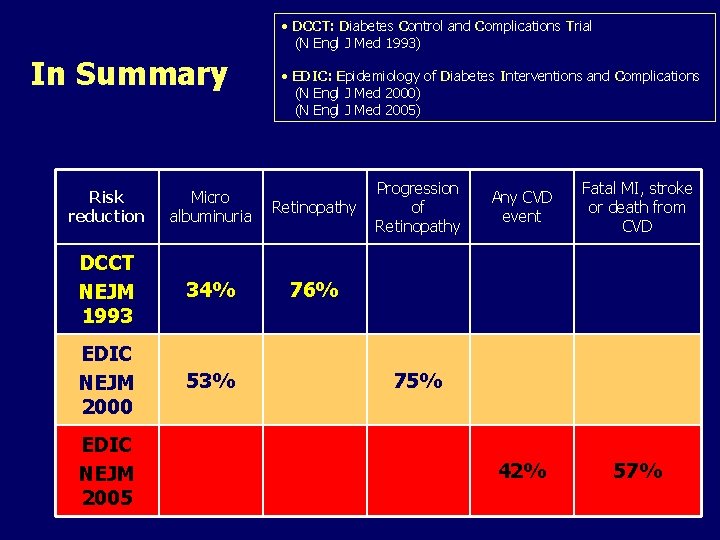
• DCCT: Diabetes Control and Complications Trial (N Engl J Med 1993) In Summary • EDIC: Epidemiology of Diabetes Interventions and Complications (N Engl J Med 2000) (N Engl J Med 2005) Risk reduction Micro albuminuria Retinopathy DCCT NEJM 1993 34% 76% EDIC NEJM 2000 53% EDIC NEJM 2005 Progression of Retinopathy Any CVD event Fatal MI, stroke or death from CVD 42% 57% 75%

Summary DCCT/EDIC • DCCT: Diabetes Control and Complications Trial (N Engl J Med 1993) • EDIC: Epidemiology of Diabetes Interventions and Complications (N Engl J Med 2000) (N Engl J Med 2005) Intensified insulin therapy: n Reduces Hb. A 1 C n Increases number of Hypoglycemia events n Reduces long term complications: – Retinopathy – Nephropathy – CVD

Conclusion Important q Good glycemic control reduces long term complications significantly !!! q Intensified therapy increases the risk of Hypoglycemia

UKPDS UK Prospective Diabetes Study Lancet - 1998
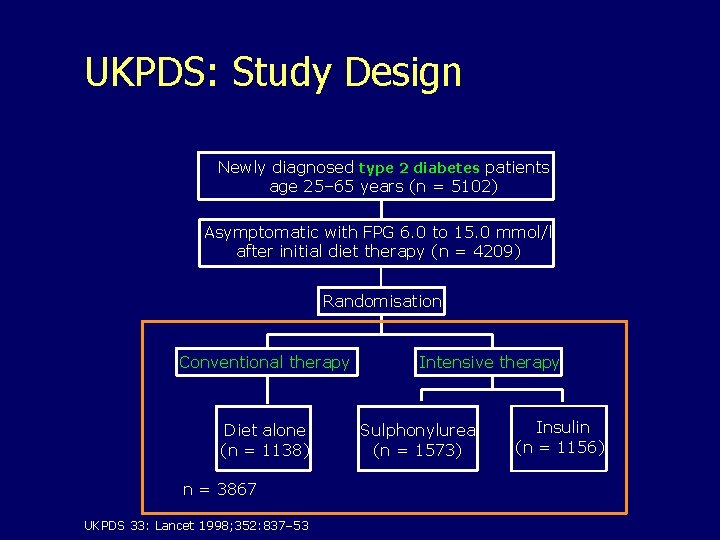
UKPDS: Study Design Newly diagnosed type 2 diabetes patients age 25– 65 years (n = 5102) Asymptomatic with FPG 6. 0 to 15. 0 mmol/l after initial diet therapy (n = 4209) Randomisation Conventional therapy Diet alone (n = 1138) n = 3867 UKPDS 33: Lancet 1998; 352: 837– 53 Intensive therapy Sulphonylurea (n = 1573) Insulin (n = 1156)
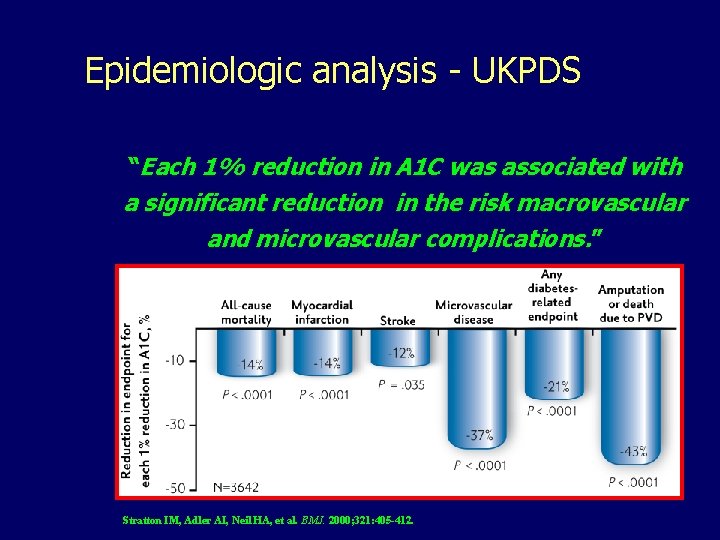
Epidemiologic analysis - UKPDS “Each 1% reduction in A 1 C was associated with a significant reduction in the risk macrovascular and microvascular complications. ” Stratton IM, Adler AI, Neil HA, et al. BMJ. 2000; 321: 405 -412.

Conclusion Similar findings as compared to DCCT: n Improved control: – Reduces Hb. A 1 C – Reduces any long term complications – Increases chance of Hypoglycemia

Continuous Subcutaneous Insulin Infusion vs. Multiple Daily Injections CSII vs. MDI

“CSII is the most physiological method of insulin delivery currently available” — Consensus statement on use of insulin pumps in pediatrics endorsed by the ADA and European Association for the Study of Diabetes (EASD) When compared to multiple daily injections (MDI), CSII results in • Better metabolic control (improved A 1 C and fewer excursions) • Less hypoglycemia • Better patients’ satisfaction and quality of life Phillip M, Battelino T, Rodriguez H, et al. Diabetes Care. 2007; 30: 1653 -1662.
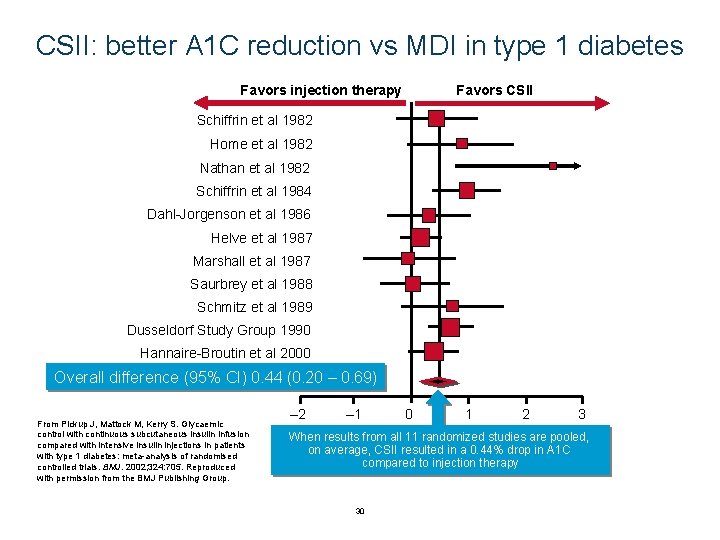
CSII: better A 1 C reduction vs MDI in type 1 diabetes Favors injection therapy Favors CSII Schiffrin et al 1982 Home et al 1982 Nathan et al 1982 Schiffrin et al 1984 Dahl-Jorgenson et al 1986 Helve et al 1987 Marshall et al 1987 Saurbrey et al 1988 Schmitz et al 1989 Dusseldorf Study Group 1990 Hannaire-Broutin et al 2000 Overall difference (95% CI) 0. 44 (0. 20 – 0. 69) From Pickup J, Mattock M, Kerry S. Glycaemic control with continuous subcutaneous insulin infusion compared with intensive insulin injections in patients with type 1 diabetes: meta-analysis of randomised controlled trials. BMJ. 2002; 324: 705. Reproduced with permission from the BMJ Publishing Group. – 2 – 1 0 1 2 3 When results from all 11 randomized studies are pooled, on average, CSII resulted in a 0. 44% drop in A 1 C compared to injection therapy 30
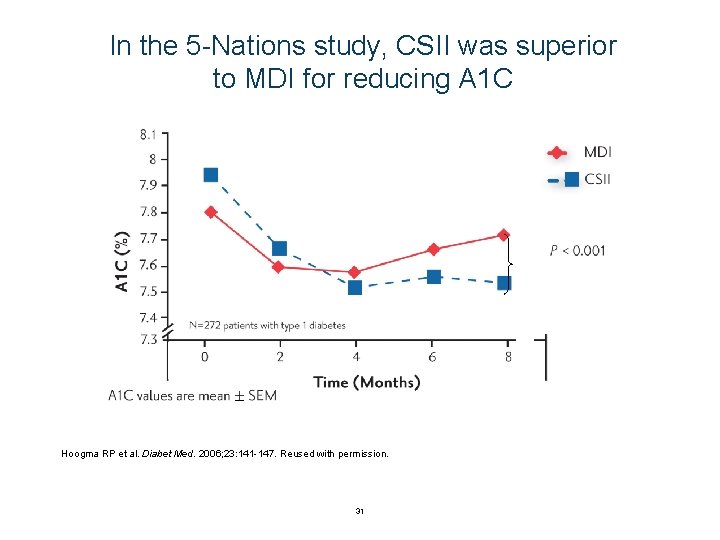
In the 5 -Nations study, CSII was superior to MDI for reducing A 1 C Hoogma RP et al. Diabet Med. 2006; 23: 141 -147. Reused with permission. 31
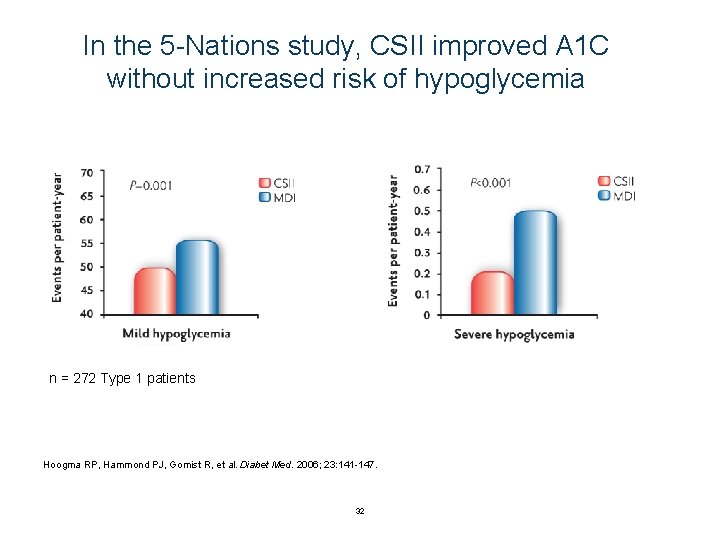
In the 5 -Nations study, CSII improved A 1 C without increased risk of hypoglycemia n = 272 Type 1 patients Hoogma RP, Hammond PJ, Gomist R, et al. Diabet Med. 2006; 23: 141 -147. 32
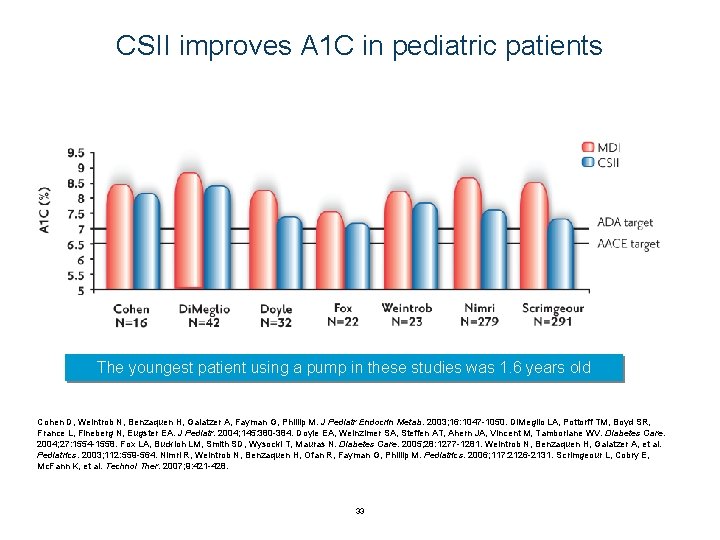
CSII improves A 1 C in pediatric patients The youngest patient using a pump in these studies was 1. 6 years old Cohen D, Weintrob N, Benzaquen H, Galatzer A, Fayman G, Phillip M. J Pediatr Endocrin Metab. 2003; 16: 1047 -1050. Di. Meglio LA, Pottorff TM, Boyd SR, France L, Fineberg N, Eugster EA. J Pediatr. 2004; 145: 380 -384. Doyle EA, Weinzimer SA, Steffen AT, Ahern JA, Vincent M, Tamborlane WV. Diabetes Care. 2004; 27: 1554 -1558. Fox LA, Buckloh LM, Smith SD, Wysocki T, Mauras N. Diabetes Care. 2005; 28: 1277 -1281. Weintrob N, Benzaquen H, Galatzer A, et al. Pediatrics. 2003; 112: 559 -564. Nimri R, Weintrob N, Benzaquen H, Ofan R, Fayman G, Phillip M. Pediatrics. 2006; 117: 2126 -2131. Scrimgeour L, Cobry E, Mc. Fann K, et al. Technol Ther. 2007; 9: 421 -428. 33
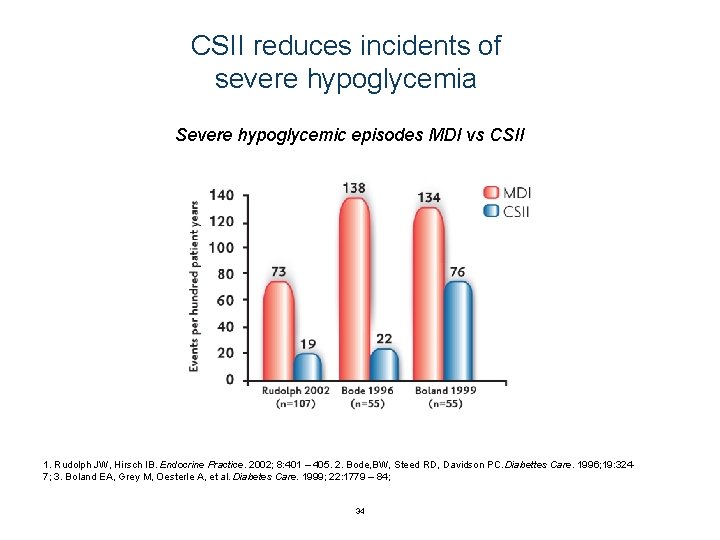
CSII reduces incidents of severe hypoglycemia Severe hypoglycemic episodes MDI vs CSII 1. Rudolph JW, Hirsch IB. Endocrine Practice. 2002; 8: 401 – 405. 2. Bode, BW, Steed RD, Davidson PC. Diabettes Care. 1996; 19: 3247; 3. Boland EA, Grey M, Oesterle A, et al. Diabetes Care. 1999; 22: 1779 – 84; 34
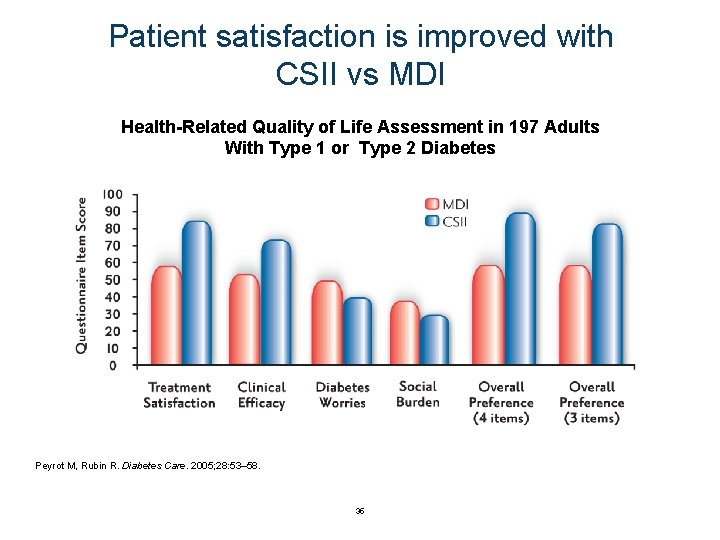
Patient satisfaction is improved with CSII vs MDI Health-Related Quality of Life Assessment in 197 Adults With Type 1 or Type 2 Diabetes Peyrot M, Rubin R. Diabetes Care. 2005; 28: 53– 58. 35

Benefits of CSII vs MDI in type 2 diabetes • Improved glycemic control 1, 2 • Fewer postprandial glucose excursions 1 • Less glycemic variability 3 • Improved patient satisfaction 4 • Weight neutral 2, 5, 6 1. Berthe E, Lireux B, Coffin C, et al. Horm Metab Res. 2007; 39: 224 -229; 2. Wainstein J, Metzger M, Boaz M, et al. Diabet Med. 2005; 22: 1037 -1046; 3. Hirsch IB, Brownlee M. J Diabetes Complications. 2005; 19: 178 -181; 4. Raskin P, Bode BW, Marks JB, et al. Diabetes Care. 2003; 26: 2598 -2603; 5. Looker HC, Knowler WC, Hanson RL. Diabetes Care. 2001; 24: 1917 -1922; 6. Wainstein J, Metzger M, Wexler ID, Cohen J, Raz I. Diabetes Care. 2001; 24: 1299. 36

Efficacy and safety of CSII and MDI are comparable in type 2 diabetes Raskin P, Bode BW, Marks JB, et al. Diabetes Care. 2003; 26: 2598 -2603; Herman WH, Ilag LL, Johnson SL, et al. Diabetes Care. 2005; 28: 1568 -1573. Wainstein J, Metzger M, Boaz M, et al. Diabet Med. 2005; 22: 1037 -1046; Berthe E, Lireux B, Coffin C, et al. Horm Metab Res. 2007; 39: 224 -229. 37

CSII minimizes glucose excursions • Insulin pumps use only rapid-acting insulin – Using an insulin pump eliminates unpredictable effects of intermediate- or long-acting insulin • Using an insulin pump usually results in fewer large fluctuations in blood glucose levels American Diabetes Association. Available at: http: //www. diabetes. org/type-1 -diabetes/insulin-pumps. jsp. 38

Indications for insulin pump therapy per AACE guidelines Type 1 Diabetes Patients • Uncontrolled on MDI • Frequent hypoglycemia and/or hypoglycemia unawareness • Pregnancy • Extreme insulin sensitivity • Dawn phenomenon • Microvascular complications • On MDI who are willing and able to do frequent SMBG, carbohydrate counting, and insulin adjustment Type 2 Diabetes Patients • Consider use of CSII in insulin-treated patients • The efficacy and safety of continuous subcutaneous insulin infusion with an insulin pump are comparable to multiple daily injection insulin therapy AACE Diabetes Mellitus Guidelines. Endocr Pract. 2007; 13(suppl 1): 3 -68. 39

CSII is safe and beneficial in pediatric patients with diabetes “All children with diabetes, regardless of age, should be considered to be potentially eligible candidates for insulin pump therapy” — Position statement of the American Academy of Pediatrics “CSII is the most physiological method of insulin delivery currently available” — Consensus statement endorsed by the American Diabetes Association (ADA) and European Association for the Study of Diabetes (EASD) Eugster EA, Francis G. Pediatrics. 2006; 118: e 1244 -e 1249. Phillip M, Battelino T, Rodriguez H, Danne T, Kaufman F. Diabetes Care. 2007; 30: 1653 -1662. 40

Specific pediatric subgroups in whom CSII may be of particular benefit From the consensus statement on CSII in pediatrics, endorsed by ADA and the EASD • Recurrent severe hypoglycemia • Wide fluctuations in blood glucose levels regardless of A 1 C • Suboptimal diabetes control • Microvascular complications or risk factors for macrovascular complications • Good metabolic control but insulin regimen that compromises lifestyle • Young children, especially infants and neonates • Adolescents with eating disorders • Children and adolescents with a pronounced dawn phenomenon • Children with needle phobia • Pregnant adolescents, ideally preconception • Ketosis-prone individuals • Competitive athletes Phillip M, Battelino T, Rodriguez H, Danne T, Kaufman F. Diabetes Care. 2007; 30: 1653 -1662. 41

Recent Clinical Trials Evaluating CSII in Pregnancy Misso et al. In 2010, Cochrane review of RCT • Comparing CSII with MDI (with at least three insulin injections per day) • In non pregnant participants with T 1 DM • Included 23 studies with a total of 976 participants. • Misso ML, Egberts KJ, Page M, et al. Continuous subcutaneous insulin infusion (CSII) versus multiple insulin injections for type 1 diabetes mellitus. Cochrane Database Syst Rev. 2010; 1: CD 005103.

Cochrane review of Misso et al Systematic review found that • There was a statistically significant difference (-0. 3% CI, − 0. 1 to − 0. 4) in Hb. A 1 c that favored CSII • Reduction in severe hypoglycemia with CSII • Quality-of-life were better for CSII. • Authors concluded that CSII offers superior glycemic control when compared with MDI for non pregnant patients with T 1 DM • Misso ML, Egberts KJ, Page M, et al. Continuous subcutaneous insulin infusion (CSII) versus multiple insulin injections for type 1 diabetes mellitus. Cochrane Database Syst Rev. 2010; 1: CD 005103.

Clinical Trials: Petrovski et al. • Evaluated CSII with constant or intermittent CGM. • CSII started at least 3 months prior to conception. • Concluded : insulin pump therapy together with constant or intermittent CGM can improve diabetes control and pregnancy outcome in T 1 DM • glucose profiles at conception were an important factor for pregnancy outcome. Petrovski G, Dimitrovski C, Bogoev M, et al. : Is there a difference in pregnancy and glycemic outcome in patients with type 1 diabetes on insulin pump with constant or intermittent glucose monitoring? A pilot study. Diabetes Technol Ther 2011

Is There a Difference in Pregnancy and Glycemic Outcome in Patients with Type 1 Diabetes on Insulin Pump with Constant or Intermittent Glucose Monitoring? A Pilot Study Goran Petrovski, M. D. , Ph. D. , Cedomir Dimitrovski, M. D. , Ph. D. , Milco Bogoev, M. D. , Ph. D. , Tatjana Milenkovic, M. D. , Ph. D. , Irfan Ahmeti, M. D. , and Iskra Bitovska, M. D. , M. S.

Conclusion

CGM (Continuous Glucose Monitoring)
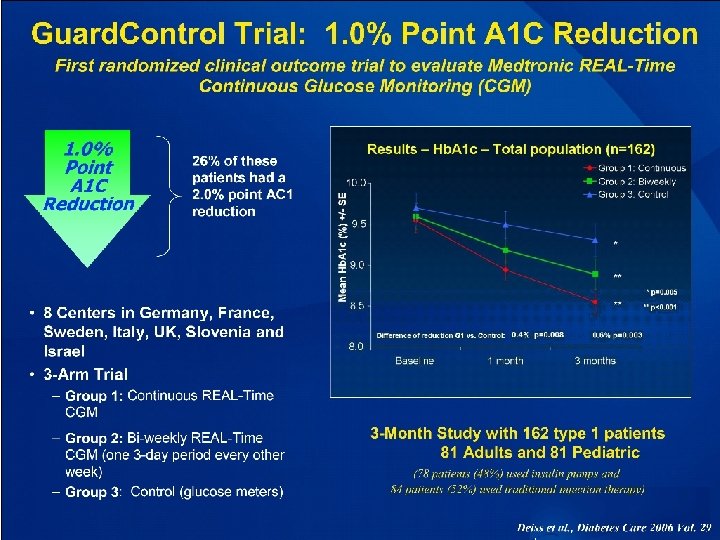

JDRF CGM Study (2008) Impact of Real-Time CGM on Hb. A 1 c In T 1 D patients (conventional or pump therapy)




Continuous Glucose Monitoring and Intensive Treatment of Type 1 Diabetes The JDRF CGM Study Group. N Engl J Med 2008; 359: 1464 -76. UC 200902547 EN

JDRF CGM Study – NEJM 2008 JDRF Randomized Clinical Trial to Assess the Efficacy of Real-Time Continuous Glucose Monitoring in the Management of Type 1 Diabetes Three studies in one (n = 322) – Children: 8 -14 years of age (n = 114) – Adolescents: 15 -24 years of age (n = 110) – Adults: ≥ 25 years of age (n = 98) Primary outcome – Change in mean A 1 C level from baseline to 26 weeks Secondary outcomes – Relative A 1 C decrease of ≥ 10. 0% – Absolute A 1 C decrease of ≥ 0. 5% – 26 week level of A 1 C < 7. 0% with no severe hypoglycemia The Juvenile Diabetes Research Foundation Study Group, N Engl JMed 2008, 359. Published at www. nejm. org on September 8, 2008.


STAR 3 Clinical Trial Sensor-augmented pump Therapy for A 1 C Reduction Supported by Medtronic & Novo Nordisk Richard M. Bergenstal, M. D. , William V. Tamborlane, M. D. , Andrew Ahmann, M. D. , John B. Buse, M. D. , Ph. D. , George Dailey, M. D. , Stephen N. Davis, M. D. , Carol Joyce, M. D. , Bruce A. Perkins, M. D. , M. P. H. , Steve M. Willi, M. D. , Michael A. Wood, M. D. ; for the STAR 3 Study Group 9301119 -011 20100625

The landmark trial STAR 3 is a large-scale, multicenter, randomized, controlled trial – the longest and largest study of its kind.

Design • Robustly designed study – Prospective, multicenter (30 sites), randomized control trial – Largest and longest study of its kind • 485 type 1 diabetes patients • 1 year study duration with 6 -month continuation phase • Primary endpoint – Change in A 1 C between SAP and MDI arms from baseline to 1 year • Secondary endpoints – Rate of severe hypoglycemia – Percentage of patients with A 1 C ≤ 7% – AUC > 180 mg/d. L and AUC < 70 mg/d. L

Eligibility Criteria • Clinical Criteria Adults, teens & children – Type 1 patients ages 7 - 70 – 7. 4% > Hb. A 1 C < 9. 5% – MDI: ≥ 3 injections, including a long-acting analogue (Lantus – Glargine) for ≥ 3 months – No prior pump therapy within the past 3 years – SMBG: mean ≥ 4 per/day – Severe hypoglycemia: < 2 events in the prior 12 months • Other Patient Criteria – Diabetes Care: by study investigator or referring physician for ≥ 6 months – Access: to computer and internet
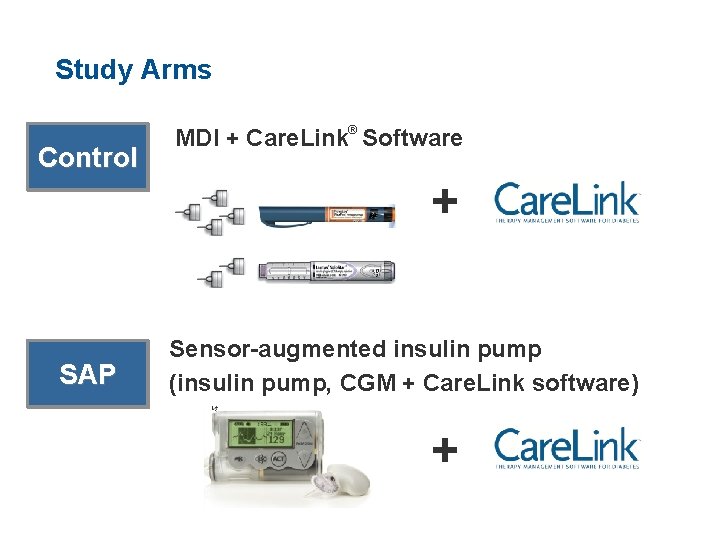
Study Arms Control MDI + Care. Link® Software + SAP Sensor-augmented insulin pump (insulin pump, CGM + Care. Link software) +
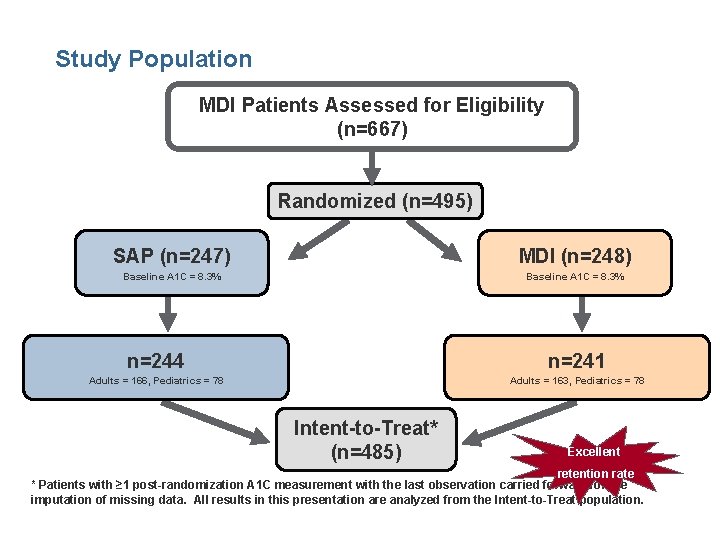
Study Population MDI Patients Assessed for Eligibility (n=667) Randomized (n=495) SAP (n=247) MDI (n=248) Baseline A 1 C = 8. 3% n=244 n=241 Adults = 166, Pediatrics = 78 Adults = 163, Pediatrics = 78 Intent-to-Treat* (n=485) Excellent retention rate * Patients with ≥ 1 post-randomization A 1 C measurement with the last observation carried forward for the imputation of missing data. All results in this presentation are analyzed from the Intent-to-Treat population.
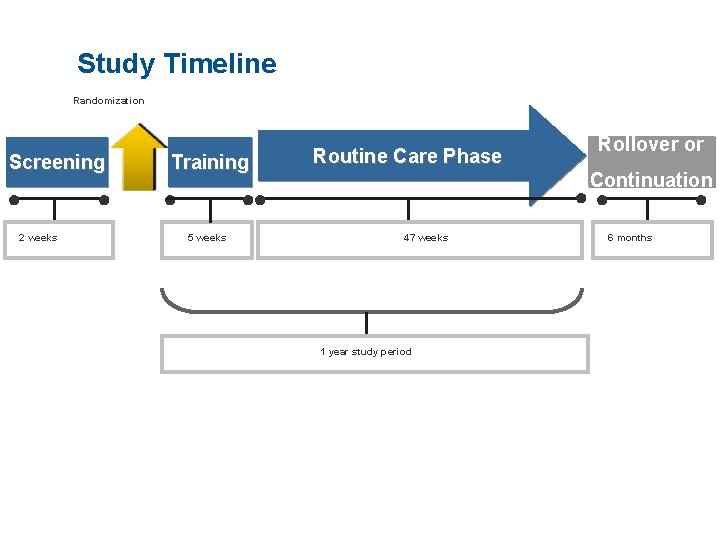
Study Timeline Randomization Screening ● 2 weeks ● Training ● Routine Care Phase Continuation ●● ●● 5 weeks Rollover or 47 weeks 1 year study period 6 months ●
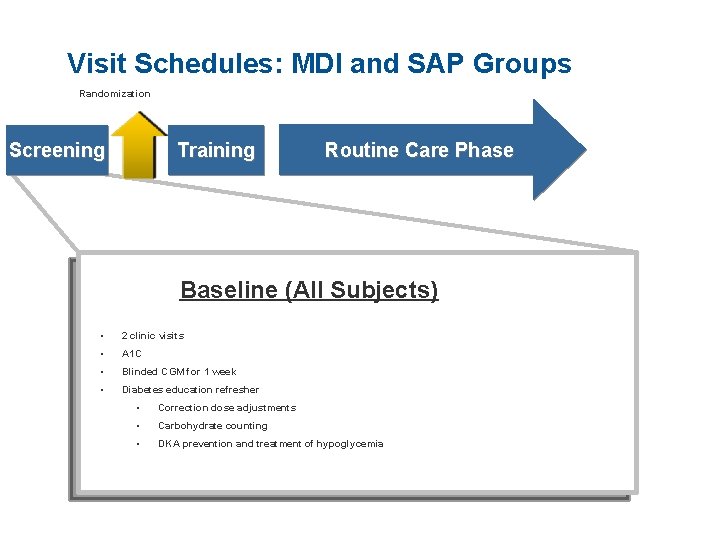
Visit Schedules: MDI and SAP Groups Randomization Screening Training Routine Care Phase Baseline (All Subjects) • 2 clinic visits • A 1 C • Blinded CGM for 1 week • Diabetes education refresher • Correction dose adjustments • Carbohydrate counting • DKA prevention and treatment of hypoglycemia
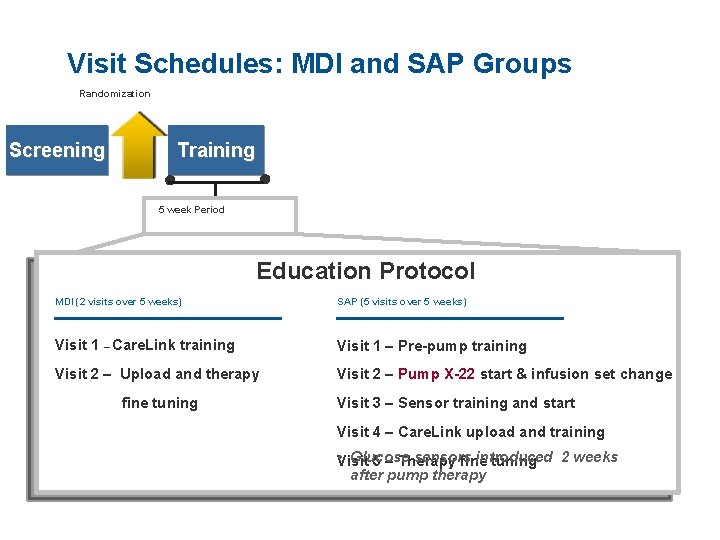
Visit Schedules: MDI and SAP Groups Randomization Screening Training ● ● 5 week Period Education Protocol MDI (2 visits over 5 weeks) SAP (5 visits over 5 weeks) Visit 1 – Care. Link training Visit 1 – Pre-pump training Visit 2 – Upload and therapy Visit 2 – Pump X-22 start & infusion set change fine tuning Visit 3 – Sensor training and start Visit 4 – Care. Link upload and training • Visit Glucose sensors introduced 2 weeks 5 – Therapy fine tuning after pump therapy
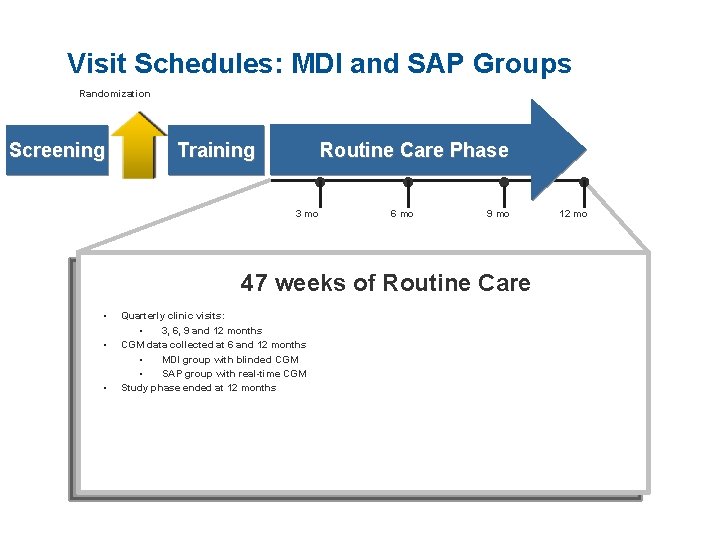
Visit Schedules: MDI and SAP Groups Randomization Screening Routine Care Phase Training ● 3 mo ● ● 6 mo 9 mo 47 weeks of Routine Care • • • Quarterly clinic visits: • 3, 6, 9 and 12 months CGM data collected at 6 and 12 months • MDI group with blinded CGM • SAP group with real-time CGM Study phase ended at 12 months ● 12 mo
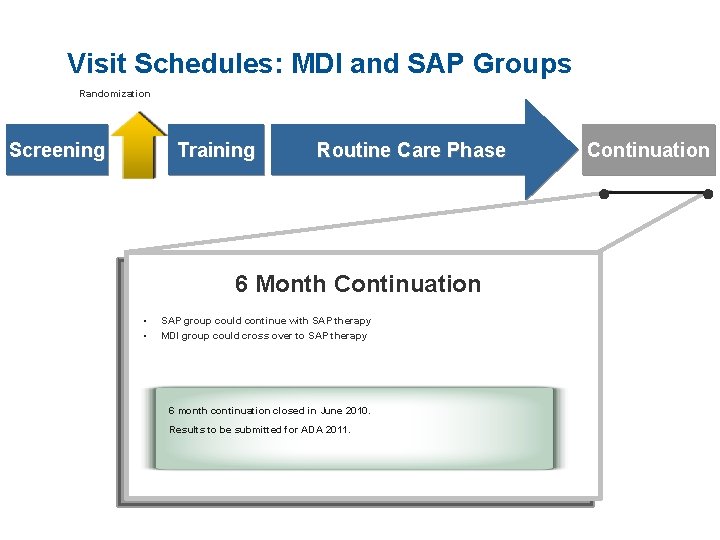
Visit Schedules: MDI and SAP Groups Randomization Screening Training Routine Care Phase Continuation ● 6 Month Continuation • • SAP group could continue with SAP therapy MDI group could cross over to SAP therapy 6 month continuation closed in June 2010. Results to be submitted for ADA 2011. ●
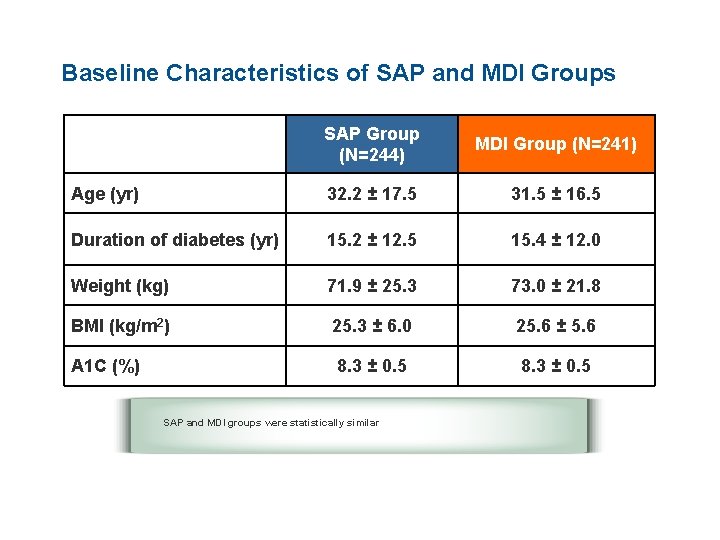
Baseline Characteristics of SAP and MDI Groups SAP Group (N=244) MDI Group (N=241) Age (yr) 32. 2 ± 17. 5 31. 5 ± 16. 5 Duration of diabetes (yr) 15. 2 ± 12. 5 15. 4 ± 12. 0 Weight (kg) 71. 9 ± 25. 3 73. 0 ± 21. 8 BMI (kg/m 2) 25. 3 ± 6. 0 25. 6 ± 5. 6 A 1 C (%) 8. 3 ± 0. 5 SAP and MDI groups were statistically similar
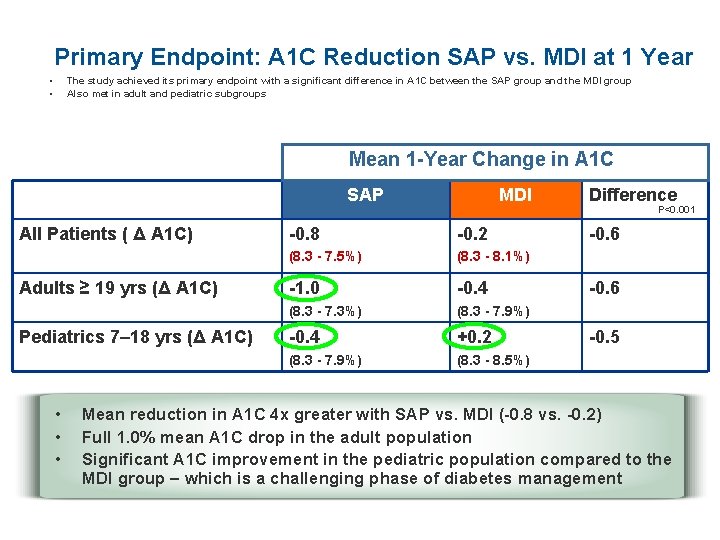
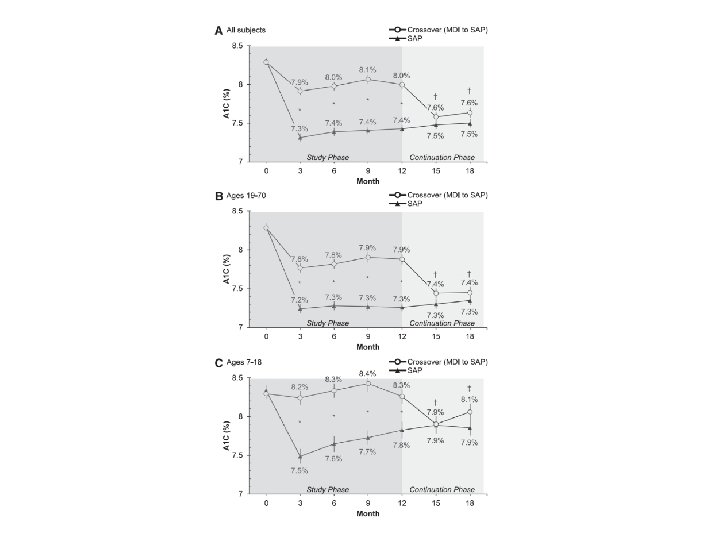
Primary Endpoint: A 1 C Reduction SAP vs. MDI at 1 Year • • The study achieved its primary endpoint with a significant difference in A 1 C between the SAP group and the MDI group Also met in adult and pediatric subgroups Mean 1 -Year Change in A 1 C SAP All Patients ( Δ A 1 C) Adults ≥ 19 yrs (Δ A 1 C) Pediatrics 7– 18 yrs (Δ A 1 C) • • • MDI -0. 8 -0. 2 (8. 3 - 7. 5%) (8. 3 - 8. 1%) -1. 0 -0. 4 (8. 3 - 7. 3%) (8. 3 - 7. 9%) -0. 4 +0. 2 (8. 3 - 7. 9%) (8. 3 - 8. 5%) Difference P<0. 001 -0. 6 -0. 5 Mean reduction in A 1 C 4 x greater with SAP vs. MDI (-0. 8 vs. -0. 2) Full 1. 0% mean A 1 C drop in the adult population Significant A 1 C improvement in the pediatric population compared to the MDI group – which is a challenging phase of diabetes management
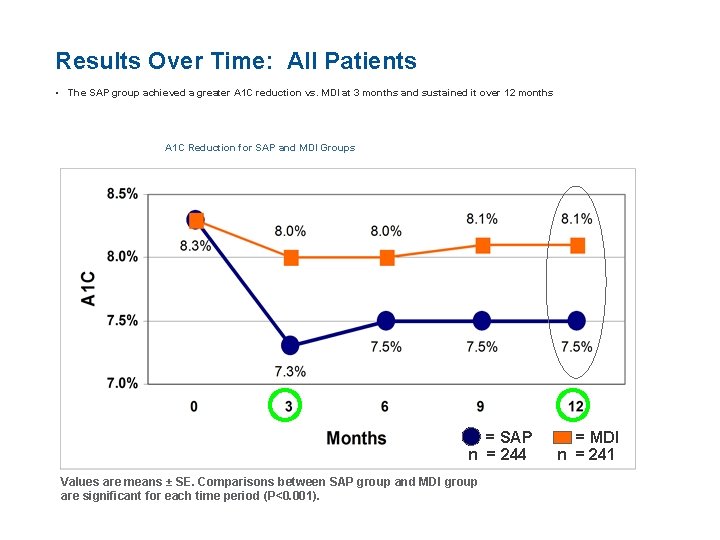
Results Over Time: All Patients • The SAP group achieved a greater A 1 C reduction vs. MDI at 3 months and sustained it over 12 months A 1 C Reduction for SAP and MDI Groups = SAP n = 244 Values are means ± SE. Comparisons between SAP group and MDI group are significant for each time period (P<0. 001). = MDI n = 241
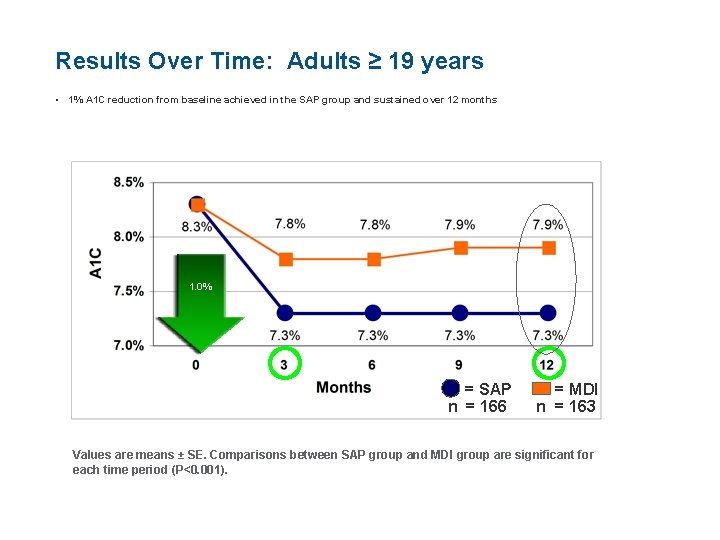
Results Over Time: Adults ≥ 19 years • 1% A 1 C reduction from baseline achieved in the SAP group and sustained over 12 months 1. 0% = SAP n = 166 = MDI n = 163 Values are means ± SE. Comparisons between SAP group and MDI group are significant for each time period (P<0. 001).
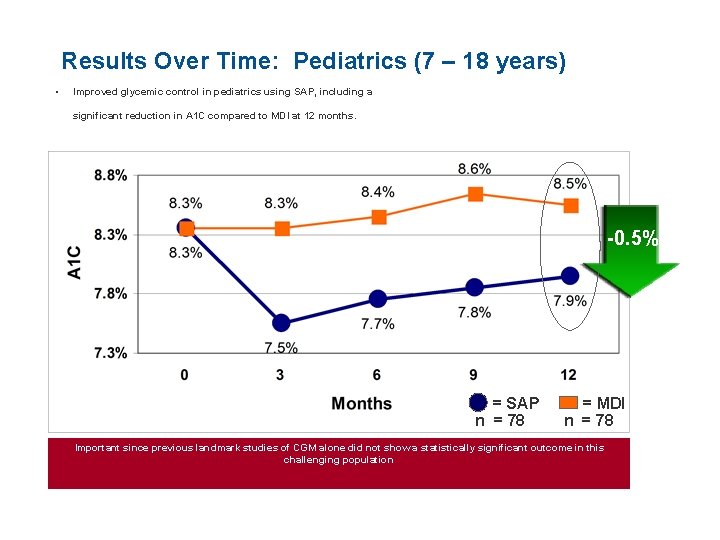
Results Over Time: Pediatrics (7 – 18 years) • Improved glycemic control in pediatrics using SAP, including a significant reduction in A 1 C compared to MDI at 12 months. -0. 5% = SAP n = 78 = MDI n = 78 Important since previous landmark studies of CGM alone did not show a statistically significant outcome in this challenging population Values are means ± SE. Comparisons between SAP group and MDI group are significant for each time period (P<0. 001).
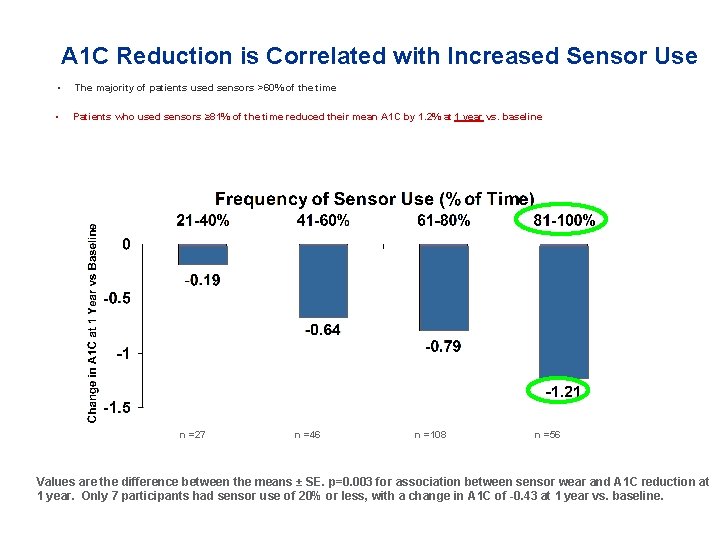
A 1 C Reduction is Correlated with Increased Sensor Use • The majority of patients used sensors >60% of the time • Patients who used sensors ≥ 81% of the time reduced their mean A 1 C by 1. 2% at 1 year vs. baseline n =27 n =46 n =108 n =56 Values are the difference between the means ± SE. p=0. 003 for association between sensor wear and A 1 C reduction at 1 year. Only 7 participants had sensor use of 20% or less, with a change in A 1 C of -0. 43 at 1 year vs. baseline.
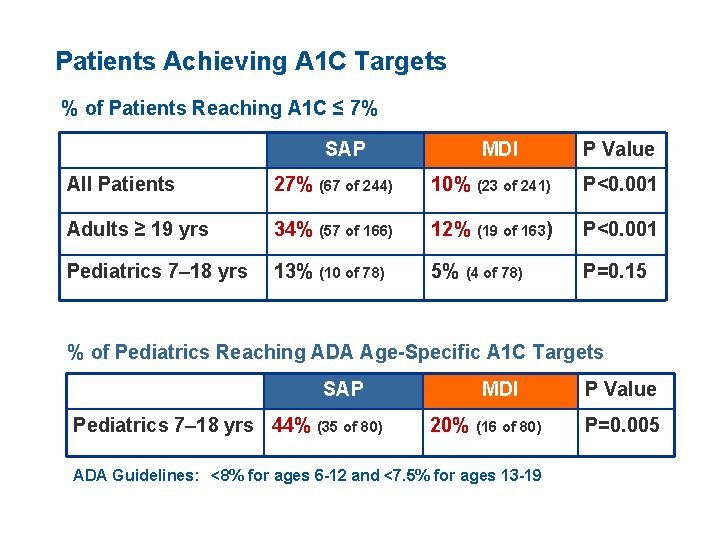
Patients Achieving A 1 C Targets % of Patients Reaching A 1 C ≤ 7% SAP MDI P Value All Patients 27% (67 of 244) 10% (23 of 241) P<0. 001 Adults ≥ 19 yrs 34% (57 of 166) 12% (19 of 163) P<0. 001 Pediatrics 7– 18 yrs 13% (10 of 78) 5% (4 of 78) P=0. 15 % of Pediatrics Reaching ADA Age-Specific A 1 C Targets SAP Pediatrics 7– 18 yrs 44% (35 of 80) MDI 20% (16 of 80) ADA Guidelines: <8% for ages 6 -12 and <7. 5% for ages 13 -19 P Value P=0. 005
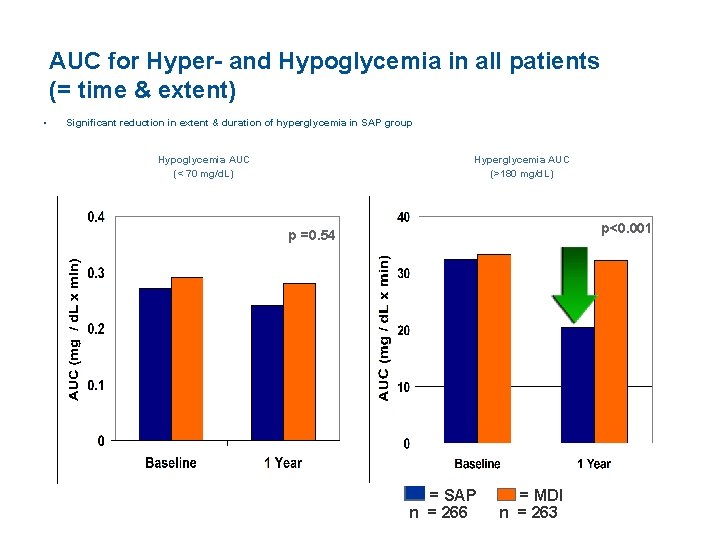
AUC for Hyper- and Hypoglycemia in all patients (= time & extent) • Significant reduction in extent & duration of hyperglycemia in SAP group Hypoglycemia AUC (< 70 mg/d. L) Hyperglycemia AUC (>180 mg/d. L) p<0. 001 p =0. 54 = SAP n = 266 = MDI n = 263
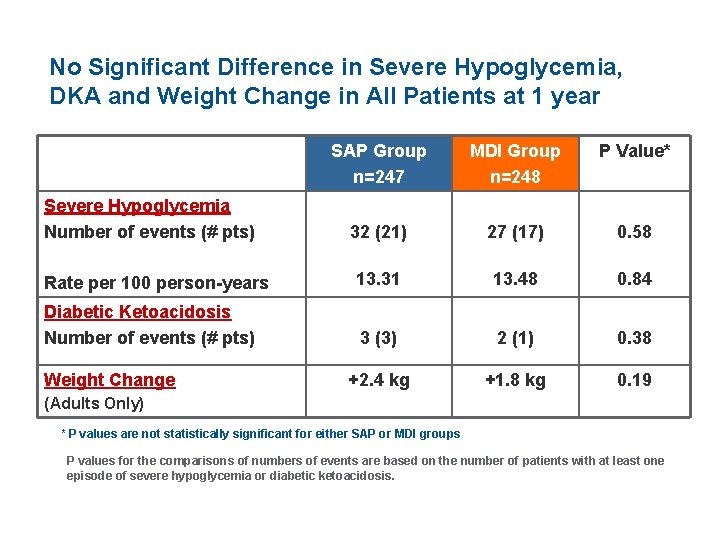
No Significant Difference in Severe Hypoglycemia, DKA and Weight Change in All Patients at 1 year SAP Group n=247 MDI Group n=248 P Value* 32 (21) 27 (17) 0. 58 Rate per 100 person-years 13. 31 13. 48 0. 84 Diabetic Ketoacidosis Number of events (# pts) 3 (3) 2 (1) 0. 38 +2. 4 kg +1. 8 kg 0. 19 Severe Hypoglycemia Number of events (# pts) Weight Change (Adults Only) * P values are not statistically significant for either SAP or MDI groups P values for the comparisons of numbers of events are based on the number of patients with at least one episode of severe hypoglycemia or diabetic ketoacidosis.
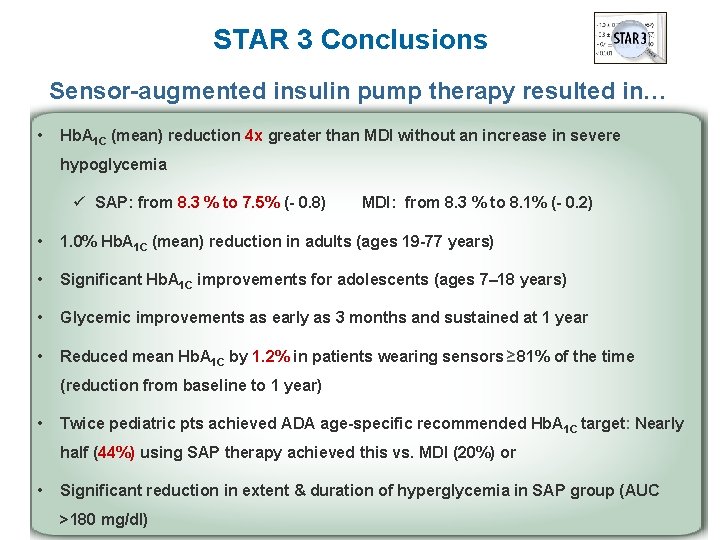
STAR 3 Conclusions Sensor-augmented insulin pump therapy resulted in… • Hb. A 1 C (mean) reduction 4 x greater than MDI without an increase in severe hypoglycemia ü SAP: from 8. 3 % to 7. 5% (- 0. 8) MDI: from 8. 3 % to 8. 1% (- 0. 2) • 1. 0% Hb. A 1 C (mean) reduction in adults (ages 19 -77 years) • Significant Hb. A 1 C improvements for adolescents (ages 7– 18 years) • Glycemic improvements as early as 3 months and sustained at 1 year • Reduced mean Hb. A 1 C by 1. 2% in patients wearing sensors ≥ 81% of the time (reduction from baseline to 1 year) • Twice pediatric pts achieved ADA age-specific recommended Hb. A 1 C target: Nearly half (44%) using SAP therapy achieved this vs. MDI (20%) or • Significant reduction in extent & duration of hyperglycemia in SAP group (AUC >180 mg/dl)
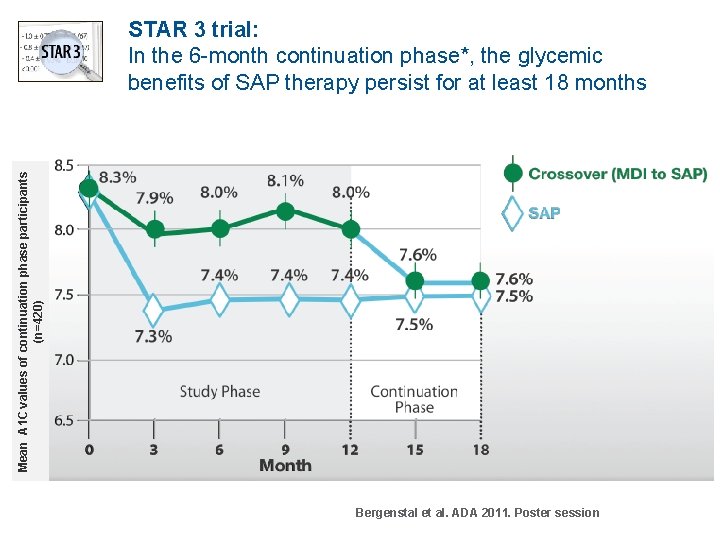
Mean A 1 C values of continuation phase participants (n=420) STAR 3 trial: In the 6 -month continuation phase*, the glycemic benefits of SAP therapy persist for at least 18 months Bergenstal et al. ADA 2011. Poster session








STAR 3 trial: Health-Related Quality of Life and Treatment Satisfaction in the Sensor-Augmented Pump Therapy for A 1 C Reduction “In the first-ever large-scale study of SAPT compared with optimal conventional therapy, SAPT had significant advantages for hypoglycemia fear in adults and caregivers and for treatment satisfaction in adults, children, and caregivers (all p<0. 001)” Rubin R. , Peyrot M. and STAR 3 Study Group. Diabetes Technology & Therapeutics. February 2012, 14(2): 143 -151






What do Guidelines tell us?

Recommendation: Adults and children 12 years and older Insulin pump therapy is recommended as a treatment option for adults and children 12 years and older with type 1 diabetes provided: • attempts to achieve target haemoglobin A 1 c (Hb. A 1 c) levels with multiple daily injections (MDIs) result in the person experiencing disabling hypoglycaemia or • Hb. A 1 c levels have remained high (8. 5% or above) on MDI therapy despite a high level of care. NICE guidelines, 2008

Recommendation: Children younger than 12 years CSII therapy is recommended as a treatment option for children younger than 12 years with type 1 diabetes provided: • MDI therapy is considered to be impractical or inappropriate, and • children on insulin pumps would be expected to undergo a trial of MDI therapy between the ages of 12 and 18 years. NICE guidelines, 2008
- Slides: 95