Insulin degludec Tresiba ULTRALONG ACTING BASAL INSULIN 1

Insulin degludec (Tresiba®) ULTRA-LONG ACTING BASAL INSULIN 1 Andre Mc. Mahon Pharm. D Candidate, University of Florida College of Pharmacy
Overview 2 Approval status Pharmacology Efficacy Type 1 DM Type 2 DM Safety

Approval Status 3 Japan: Approved USA: Pending Approval (projected early 2013 launch) Nov 8 – Advisory panel to the U. S. FDA voted to recommend approval 1

Pharmacology – Structure 4 Retained sequence of human insulin Depletion of B 30 residue No amino acid substitutions Fatty acid (hexadecanedioic acid) coupled to lysine at B 29 position via glutamic acid ‘spacer’
Pharmacology – Kinetics 2 5 T 1/2 : 25 hours Glucose-lowering duration : > 42 hours Time to steady-state : 3 days of once-daily dosing Peak : peakless

Recent Trial Highlights – BEGIN Type 1 6 Type 1 DM insulin degludec vs. glargine Study Trial design & Treatment arms Trial population Results BEGIN Basal. Bolus Type 1 3 • 52 weeks, randomized, controlled, open-label, multinational, parallel design, treat-to-target, non -inferiority trial • 629 patients with type 1 DM • Hb. A 1 c fall at year 1 • 0. 40% degludec • 0. 39% glargine • Treatment difference: -0. 01% [95% CI -0. 14 to 0. 11]; p<0. 0001 for non-inferiority testing • Basal insulin: insulin degludec or insulin glargine • Bolus insulin: insulin aspart • Diabetes duration for > 1 year, basal insulin use for > 1 year, Hb. A 1 c of 10% or less, and BMI of 35 kg/m 2 or less • 472 patients to degludec • 157 patients to glargine
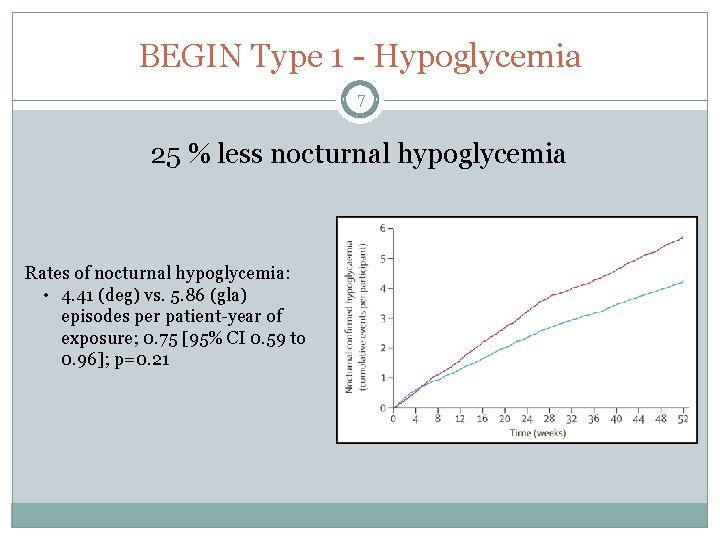
BEGIN Type 1 - Hypoglycemia 7 25 % less nocturnal hypoglycemia Rates of nocturnal hypoglycemia: • 4. 41 (deg) vs. 5. 86 (gla) episodes per patient-year of exposure; 0. 75 [95% CI 0. 59 to 0. 96]; p=0. 21
Recent Trial Highlights – BEGIN Type 2 8 Type 2 DM insulin degludec vs. glargine Study Trial design & Treatment arms Trial population Results BEGIN Basal. Bolus Type 2 4 • 52 weeks, randomized, controlled, open-label, treat-to-target, multinational, noninferiority trial • 1006 patients with type 2 DM • Hb. A 1 c fall at year 1 • 1. 1 % degludec • 1. 2 % glargine • Treatment difference: 0. 08% [95% CI -0. 05 to 0. 21], confirming non-inferiority • Diabetes duration for > 6 months, any insulin use for at least 3 months, Hb. A 1 c of 7. 0 – 10. 0%, and BMI of 40. 0 kg/m 2 or less, with or without oral antidiabetic drugs • 744 patients to degludec • 248 patients to glargine • Excluded: glp-1 agonist or rosiglitazone use within previous 3 months

BEGIN Type 2 - Hypoglycemia 9 Lower rates of hypoglycemia: overall and nocturnal (A) Overall confirmed hypoglycemic episodes. (B) Nocturnal confirmed hypoglycemic episodes. (C) Diurnal Confirmed hypoglycemic episodes. (D) Cumulative # of hypoglycemic episodes per participant during 24 h

Cardiovascular Safety 10 MACE (Major Adverse Cardiovascular Events) Composite of CV death, stroke, myocardial infarction (MI), and unstable angina pectoris (UAP) In the 16 phase 3 trials included in the NDA 80 patients experienced a MACE (76/80 patients Type 2 DM) Similar incidence rates 53 In the degludec group vs. 27 in comparator group 1. 48 degludec vs. 1. 44 comparator group Estimated hazard ratio 1. 097; 95% CI: 0. 681 – 1. 768

Cardiovascular Safety 11 FDA Requested Post Hoc Analyses of MACE Excluded UAP from MACE composite Estimated hazard ratio 1. 393; 95% CI: 0. 757 – 2. 565 Additional Post Hoc Analyses of MACE Included data from 9 additional completed trials (6 extension trials and 3 phase 3 trials) Contributed 742 degludec 149 comparator patients Excluded UAP and included MACE reported within 30 days after drug discontinuation Estimated hazard ratio 1. 614; 95% CI: 0. 999 – 2. 609

Cardiovascular Safety 12 In summary: Data neither confirms nor excludes increased CV risk Post-approval studies planned

Insulin degludec (Tresiba®) 13 Insulin degludec is an ultra long-acting insulin formulation with several advantages: Lower risk of hypoglycemia True 24 hour insulin Allows flexibility in dosing; especially with missed doses Can be coformulated with other proteins Combination of degludec with insulin aspart planned to allow effective mealtime coverage

Notes 14 1. 2. 3. 4. 5. Pierson, Ransdell. "FDA Panel Recommends Approval of Novo Degludec Insulin. "Reuters. Thomson Reuters, 08 Nov. 2012. <http: //www. reuters. com/article/2012/11/08/us-novo-vote-id. USBRE 8 A 71 HP 20121108>. Jonassen I, Havelund S, ribel U, et al. Insulin degludec: Multi-hexamer formation is the underlying basis for this new generation ultra-long acting basal insulin. Paper presented at: European Association for the Study of Diabetes Annual Meeting; September 20 -24, 2010; Stockholm, Sweden. Heller S, Buse J, Fisher M, et al; BEGIN Basal-Bolus Type 1 Trial Investigators. Insulin degludec, an ultra-longacting basal insulin, versus insulin glargine in basal-bolus treatment with mealtime insulin aspart in type 1 diabetes (BEGIN Basal-Blus Type 1): a phase 3, randomised, open-label, treat-to-target non-inferiority trial. Lancet. 2012; 379(9825): 1489– 1497. Garber AJ, King AB, Del Prato S, et al; NN 1250 -3582 (BEGIN BB T 2 D) Trial Investigators. Insulin degludec, an ultralongacting basal insulin, versus insulin glargine in basal-bolus treatment with mealtime insulin aspart in type 2 diabetes (BEGIN Basal-Bolus Type 2): a phase 3, randomised, open-label, treat-to-target non-inferiority trial. Lancet. 2012; 379(9825): 1498– 1507. "Insulin Degludec and Insulin Degludec/Insulin Aspart Treatment to Improve Glycemic Control in Patients with Diabetes Mellitus - Briefing Document. " FDA, 8 Nov. 2012. Web. <http: //www. fda. gov/downloads/Advisory. Committees/Committees. Meeting. Materials/Drugs/Endocrinologicand. Meta bolic. Drugs. Advisory. Committee/UCM 327017. pdf>.
- Slides: 14