Insulation and Heat Transfer Taking a look at













- Slides: 15

Insulation and Heat Transfer Taking a look at how heat can be transferred in various mediums and how it can be stopped or slowed down.

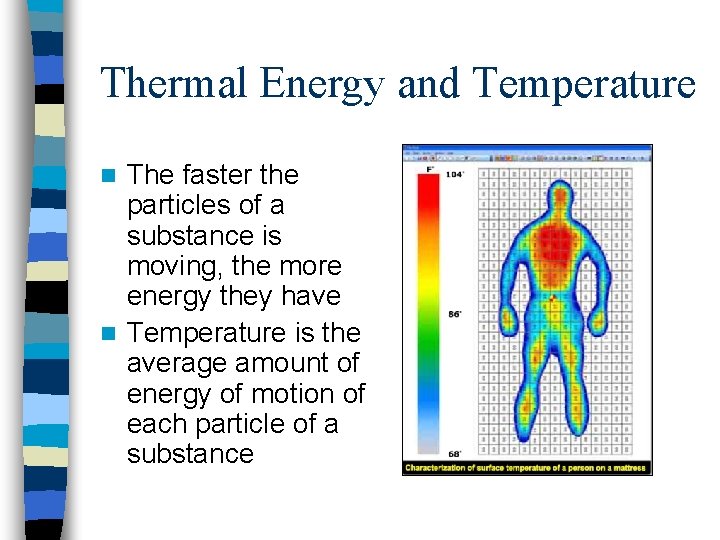
Thermal Energy and Temperature The faster the particles of a substance is moving, the more energy they have n Temperature is the average amount of energy of motion of each particle of a substance n

Temperature and Thermal Energy n Temperature measures the amount of heat in an object n Measured in o. F, o. C, and K

Temperature and Thermal Energy n What is Thermal Energy? – “The total energy of the particles that make up a material. ” • Includes both Kinetic & Potential energy – Ex: A cold spoon on ice cream • Nothing happens (same temp. ) – Ex: Hot spoon on ice cream • Ice cream melts • Thermal energy transferred from spoon to ice cream – Ex: 2 hot spoons on ice cream • Ice cream melts 2 x faster (2 x as much thermal energy)

How can Heat be Transferred? n Three methods of heat transfer: – 1) Conduction: The transfer of thermal energy by the direct contact of molecules. • Caused by the vibration of molecules • Substance does not move • Different materials conduct heat at different rates.

How can Heat be Transferred? n 2) Convection: Transfer of thermal energy by the direct movement of a fluid. (Gas or Liquid) – Fluids will rise when heated, sink when cooled. – Hot air balloons function via convection • Air is heated up to cause balloon to rise. • Air is cooled when balloon needs to descend. • Heated air is less dense, so it rises.

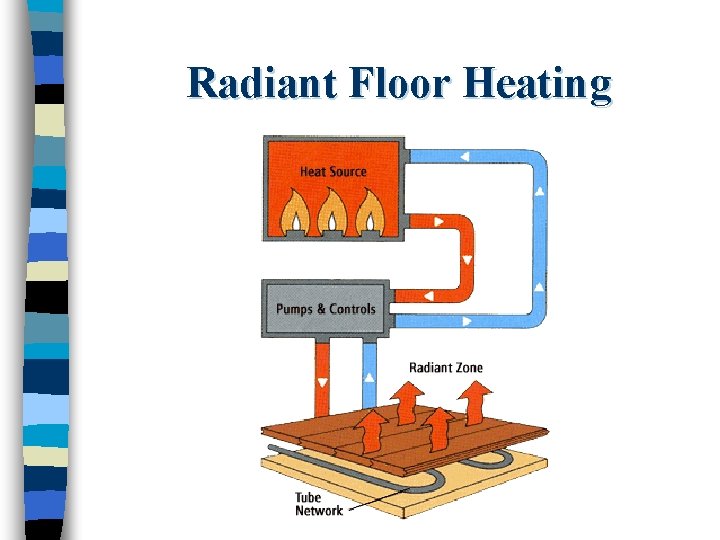
How can Heat be Transferred? n 3) Radiation: The emission of thermal energy as particles or waves. – Does not need a medium to move through. • Radiant energy from the sun travels through space to earth. – Gets absorbed better by dark objects • Radiate more heat than shiny objects. • Engine-cooling mantles are black for this reason. • Thermos is shiny to keep heat in.

Radiant Floor Heating

Thermos - Shiny on outside and inside to not allow any heat to radiate or be absorbed. -Prevents Radiation - Air pocket in between 2 layers to insulate liquid. -Prevents Conduction -Firm seal also prevents fluids from exiting container. -Prevents Convection

Measuring Temperature n Temperature is one of the most important factors affecting weather n Air temperature is measured with a thermometer n A thermometer is a thin glass tube with a bulb on one end that contains a liquid, usually mercury or colored alcohol

Temperature Scales and Conversions n Three temperature scales: Celsius, Fahrenheit, Kelvin n Measured n Being in the unit degrees able to convert from one scale to another is important!!!!

Converting Between Scales n Fahrenheit to Celsius – °C=5 (°F-32) 9 n Celsius to Fahrenheit – °F=(°C* 9) + 32 5 n Celsius to Kelvin – K= °C + 273

Practice Problems n 32°C to °F – °F=(°C* 9) + 32 5 – °F=(32* 9) + 32 5 – °F=57. 6 + 32 – 89. 6 °F

Practice Problems n 150°F to °C – °C=5 (°F-32) 9 – °C=5 (150 -32) 9 – °C=5 (118) 9 – 65. 6 °C

Practice Problems n 25°C to K – K= °C + 273 – K= 25 + 273 – 298 K