Institutional Review Board IRB Tsukegee Syphilis Experiment 3

Institutional Review Board (IRB)

Tsukegee Syphilis Experiment

3 Pillars of Ethical Research ► What constitutes ethical research practices as it pertains to the use of human subjects? ► Respect for persons ► Beneficence - Causing no harm (minimizing harm and maximizing benefits) ► Justice - Ensuring benefits are equal to all 3

Additional Guidelines for Ethical Research Practices ► Even without having to go through the IRB process, there is a duty to protect the rights of people in the study as well as their privacy and sensitivity. ► Researchers should secure the permission and maintain the interests of all those involved in the study. ► The confidentiality of those involved in the research must be met, keeping their anonymity and privacy secure. ► Researchers should not misuse any of the information discovered. ► Receiving Institutional Review Board (IRB) approval provides protection against individuals engaging in unethical research practices. 4

Institutional Review Board (IRB) ► What is an IRB? ► A committee of specific composition at an affiliated fair, high school or institution that reviews research plans involving human subjects to determine potential physical and/or psychological risk. ► The IRB reviews and approves ALL research involving human subjects BEFORE experimentation begins. (This includes surveys, professional tests, questionnaires, and studies in which the researcher is the subject of his/her own research. ) ► The IRB determines the level of RISK involved in the project. 5

Risk Assessment and Safe Research Practices ► What is risk? • Exercise or exertion other than ordinarily encountered in daily life • Emotional stress to questioning or activity or stress resulting from an invasion of privacy • Ingestion of and/or physical contact with any substance ► Who is at risk? • Any participant involved in experimentation with toxic chemicals, radiation, or known pathogens and carcinogens • Any member of a risk group (e. g. , anyone with a disease, cardiac disorder, pregnant women, etc. ) • Any member of special groups covered by federal/national regulations (e. g. , Singapore military, prisoners, special needs persons, including the disabled and gifted, children, economically or educationally disadvantaged persons) 6

Questions the IRB Will Ask Question 1. Does this project or study involve collection of data that identifies individuals (e. g. , SSN# data on individuals, surveys, or interviews identifiable by name or student number etc. )? 2. Will data identifiable by individual be shared with anyone (such as in conference presentations, published articles and reports, etc. )? Correct Response This question should be answered with a “NO” and should be made clear how you are avoiding/correcting for this via your method. 3. Are the participants being offered one or more of the incentives to participate (such as money, extra credit for the class, etc. )? List the incentive(s) here: This question could be answered YES or NO but it should be clear that participants all receive the benefit even if they leave the study before its finished. 4. Is participation in this project or study voluntary for the individuals participating in the program or study? This question should be answered with a “YES” and should be made clear how you are addressing this via your method. 5. Will participants be fully informed about the benefits and any risks? This question should be answered with a “YES” and should be made clear how you are addressing this via your method. 7
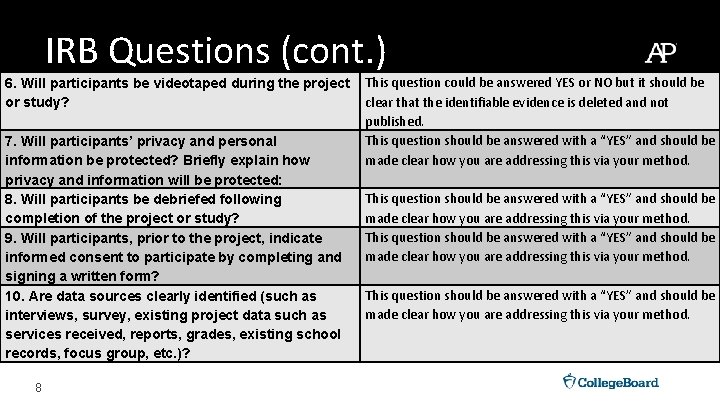
IRB Questions (cont. ) 6. Will participants be videotaped during the project or study? 7. Will participants’ privacy and personal information be protected? Briefly explain how privacy and information will be protected: 8. Will participants be debriefed following completion of the project or study? 9. Will participants, prior to the project, indicate informed consent to participate by completing and signing a written form? 10. Are data sources clearly identified (such as interviews, survey, existing project data such as services received, reports, grades, existing school records, focus group, etc. )? 8 This question could be answered YES or NO but it should be clear that the identifiable evidence is deleted and not published. This question should be answered with a “YES” and should be made clear how you are addressing this via your method.
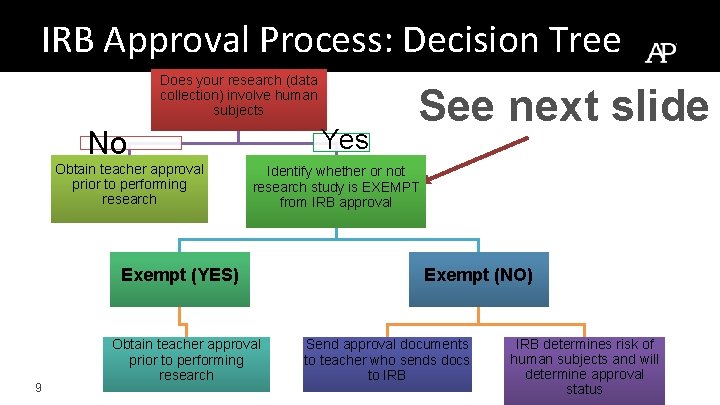
IRB Approval Process: Decision Tree Does your research (data collection) involve human subjects Yes No Obtain teacher approval prior to performing research Identify whether or not research study is EXEMPT from IRB approval Exempt (YES) 9 See next slide Obtain teacher approval prior to performing research Exempt (NO) Send approval documents to teacher who sends docs to IRB determines risk of human subjects and will determine approval status

IRB Exempt Decision Tree • If you are performing your research as a class assignment (such is the case for the AP Research course) and are engaged in interviewing, surveying, etc. types of research, this is not classified as human research so long as it is NOT PUBLISHED. • This type of research is exempt from IRB approval • YOU MUST STILL include safety measures in your method. 10 ► http: //www. hhs. gov/ohrp/policy/checklists/decisioncharts. html#c 2

IRB Exempt Decision Tree • If you plan on publishing your research in which you interact with humans, you must submit your proposal to an IRB for approval. • You will need: • SAS Informed Consent Form • Confidentiality agreements • IRB application ► 11 http: //www. hhs. gov/ohrp/policy/checklists/decisioncharts. html#c 2

Common Documents Needs for IRB Approval ► Common documents for IRB Approval • Application for IRB Approval • Informed Consent Form(s) • Letters of approval from cooperating entities (organizations or institutions -where applicable) • Research methods (research design, data source, sampling strategy, etc. ) • Questionnaires, surveys, or other data-gathering forms • Sample emails, letters, flyers etc. , that will be distributed to the study subjects 12

When are Informed Consent Forms for Under 18 Research Subjects not required? ► Research conducted in established settings: ► Involving normal educational practices; ► Research on individual or group behavior or characteristics of individuals, such as studies of perception, cognition, game theory, or test development, where the investigator does not manipulate subjects’ behavior and the research will not involve stress to subject. ► Research involving observation of legal public behavior. ► Research involving collection or study of existing publicly available data. 13
Rule of Thumb ► ► Do not come in contact with or use harmful/hazardous substances or organisms ► If you are working at an institution or organization performing scientific experiments where you will come in contact with harmful/hazardous substances or organisms, your supervising scientist will not allow you to perform research without engaging in IRB approval with their institution/organization and should work with you to develop these documents. ► You will need copies of these documents for your PREP Do not experiment on yourself 14

Rule of Thumb ► Do not experiment on other human beings with anything other engaging in surveys, focus groups, interviews, or questionnaires ► When doing so: ► Include ethical/safety measures in your method ► See table on IRB questions ► Do not collect identifiable information ► If you have to do interviews/surveys face to face then: ► Code all information so that it is “de-identified” ► Do not publish identifiable information ► Destroy all identifiable information collected 15
- Slides: 15