Institutional Review Board IRB Human Subjects Research Update

Institutional Review Board (IRB) Human Subjects Research Update New CITI Training Modules Child Clearance Requirements We. Pay Secrets Christopher M. Ryan, Ph. D. IRB Director ryancm@upmc. edu

Institutional Review Board (IRB) Collaborative Initiative Training Institute (CITI): Why the Change? • Federal regulations mandate training in human subject protections • CITI Program: “Gold standard” for internet-based research ethics training – Accepted by virtually all institutions/agencies – Single source for all training • New courses continually developed and existing courses updated 2

Institutional Review Board (IRB) General Information • Pitt/UPMC: Continue to use HSConnect authentication – Every user must have a HSConnect account – Special portal to access CITI courses • www. citi. pitt. edu • External Investigators with CITI accounts – Obtain credit for previously completed CITI modules that match Pitt requirements (if taken within 2 years) – Eliminates redundant training across institutions 3

Institutional Review Board (IRB) • New users required to take the CITI courses prior to conducting human subject research or to access OSIRIS • Existing users have one year to complete the CITI courses – After March 31, 2013, access to OSIRIS will be denied • Cumulative passing score of 80% • 3 -5 hours to complete all modules (some report 7 hours) • Re-certification every 3 years – CITI ‘Refresher Course’ plus other requirements – Reminder notifications sent out automatically 4

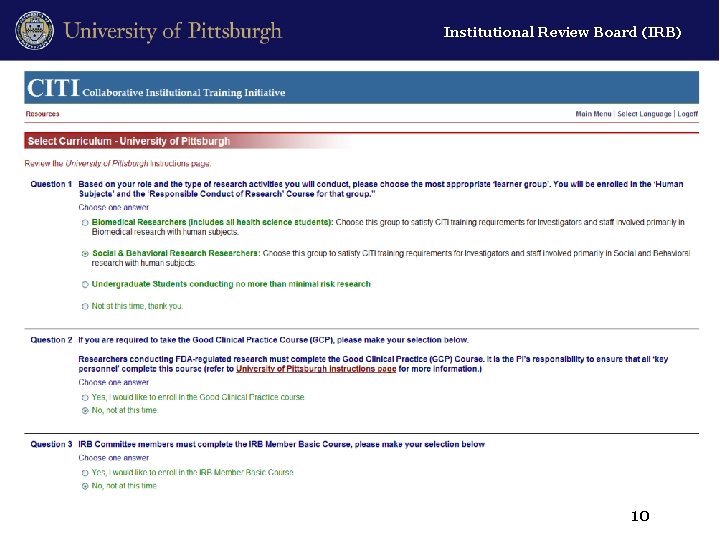
Institutional Review Board (IRB) Designated “User Groups” • Biomedical Researcher (including all health science students) • Social & Behavioral Researcher • Undergraduate Student Researcher • Optional course for IRB Members 5
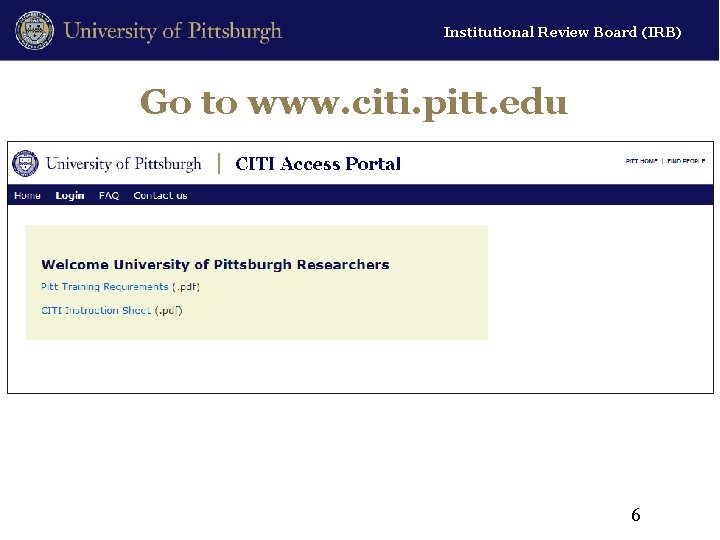
Institutional Review Board (IRB) Go to www. citi. pitt. edu 6
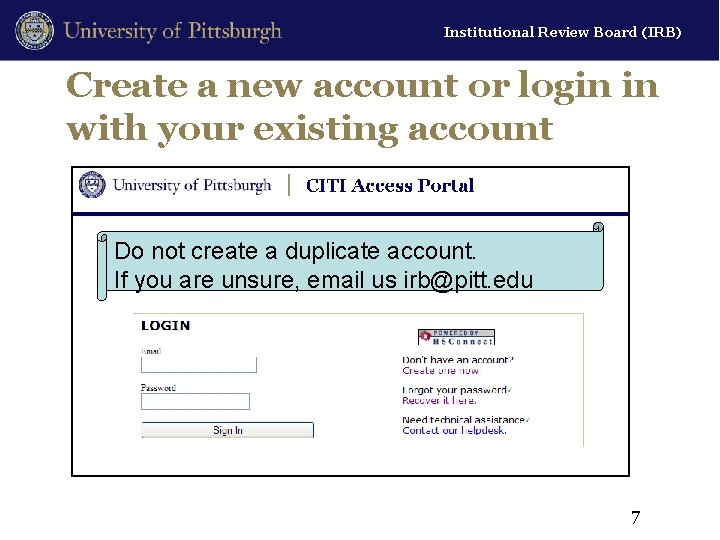
Institutional Review Board (IRB) Create a new account or login in with your existing account Do not create a duplicate account. If you are unsure, email us irb@pitt. edu 7
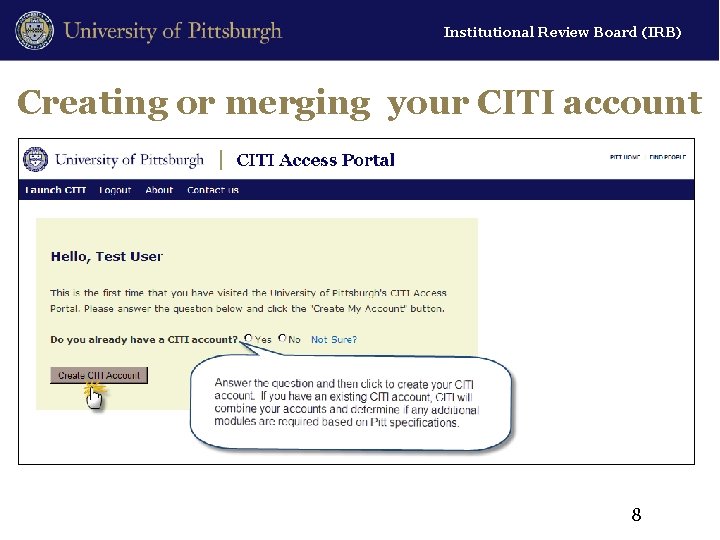
Institutional Review Board (IRB) Creating or merging your CITI account 8
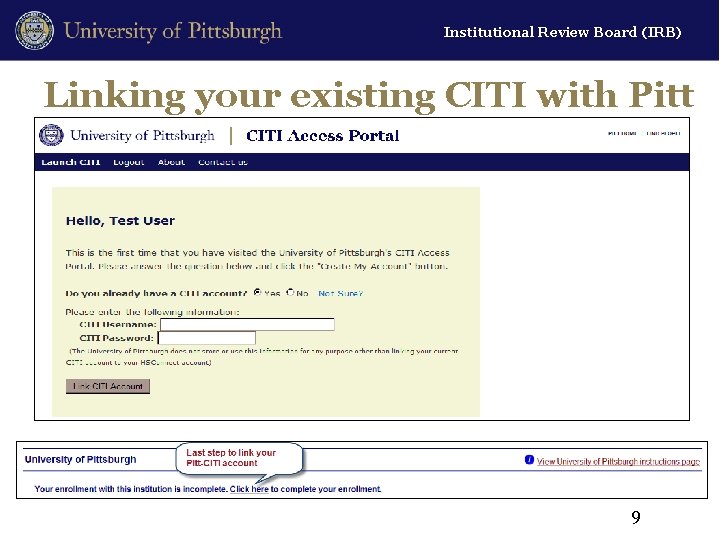
Institutional Review Board (IRB) Linking your existing CITI with Pitt 9
Institutional Review Board (IRB) 10

Institutional Review Board (IRB) Access the course modules Example: Student Researchers 11

Institutional Review Board (IRB) Social/Behavioral Human Subjects Curriculum • • • Belmont Report History and Ethical Principles Defining Research with Human Subjects The Regulations & Social/Behavioral Science Assessing Risk in Social/Behavioral Research Informed Consent Privacy & Confidentiality Conflicts of Interest Two Additional Electives 12

Institutional Review Board (IRB) Social/Behavioral Responsible Conduct of Research Curriculum • Responsible Mentoring • Introduction to the Responsible Conduct of Research • Research Misconduct • Data Acquisition, management & Sharing • Publication Practices and Responsible Authorship • Collaborative Research • + 1 Elective module 13

Institutional Review Board (IRB) Additional Continuing Education Requirements • Required to attend at least 3 continuing education hours (CEUs) in a 3 -year period specifically related to human subject protection or ethics – Examples may include: • IRB programs (i. e. , Ask the IRB) • IRB board member attendance • ACRP programs • Keep records of attendance in your file 14

Institutional Review Board (IRB) FAQs 1. I took the VA version of CITI last year. Is that acceptable at Pitt or should I take it again? 2. As an administrator, how do I verify which courses have been completed by my faculty? 3. I conduct both biomedical and behavioral research. Do I have to complete both courses in CITI? 4. Will we receive reminders when our CITI courses are close to expiring? 5. According to the Provost, all faculty must take the Research Integrity Module and the COI module. If I’ve taken the CITI modules, is that sufficient? 15

Institutional Review Board (IRB) Child Clearance Requirements

Institutional Review Board (IRB) New Question Added to OSIRIS • It is the responsibility of the principal investigator to ensure that all research staff have these clearances prior to any interaction with children. • Act 33; Act 34; FBI Criminal Background Check • Contact Human Resources at 412 -624 -8150 for assistance with this process. • OGC Contact: Alan Pittler 17

Institutional Review Board (IRB) FAQs 1. I called Human Resources and they said I was “grandfathered. ” Does that mean that no clearances are required? 2. I have an ongoing study. Do I have to stop the study until everyone has their clearances? 3. We are starting a new study. Do I have to wait until all staff have these clearances? 4. We provide ‘babysitting’ for our subjects; does that staff member need these clearances? 18

Institutional Review Board (IRB) FAQs, cont. 5. My secretary greets the kids when they come in but otherwise isn’t involved with the research. Clearances Required? 6. I am interviewing children over the telephone. Clearances Required? 7. I am conducting on-line surveys of children. Clearances Required? 8. I am doing a medical record review on children. Clearances Required? 9. Do these clearances ever expire? 19

Institutional Review Board (IRB) We. Pay Facts You May Not Know • I am the ONLY person at the IRB who addresses We. Pay issues • SSNs must be collected unless a ‘waiver’ is requested – E-mail me with request – Subjects cannot receive more than $100 in a year • Exception: certain studies (e. g. , spousal abuse) where receipt of 1099 could lead to harm 20

Institutional Review Board (IRB) • Gift cards ordinarily cannot be used – E-mail me for an exception (granted infrequently) • ‘Man in the Street’ option – ‘Petty cash’ approach • Can be used to purchase gift cards (but…) – Investigators are required to maintain records documenting who received what when – Names may not be required, but info is needed to ensure that subjects actually participated • Amazon gift codes may be possible… 21

Institutional Review Board (IRB) Upcoming Education Programs Building Bridges: Research Around The World – National conference on international research cosponsored by Pitt, CMU, VA Healthcare System, and the federal Office of Human Research Protections – Sheraton Station Square – Friday, June 22, 2012 22
- Slides: 22