Institute of Radiation Problems Azerbaijan National Academy of

Institute of Radiation Problems Azerbaijan National Academy of Sciences Laboratory of Radiochemistry INVESTIGATION OF URANIUM SORPTION LAWS IN SOLUTION BY GAMMA-SPECTROMETRIC METHOD WITH NATURAL AND MODIFIED ZEOLITES IN STATIC CONDITION Garibov A. A. , Mehdiyeva R. N. , Naghiyev J. A. Firudin Aghayev str. 9, Phone: (+994 12) 510 05 92, 439 33 89 fax: (+994 12) 439 83 18, calalnaghiyev@mail. ru ANKARA - 2008
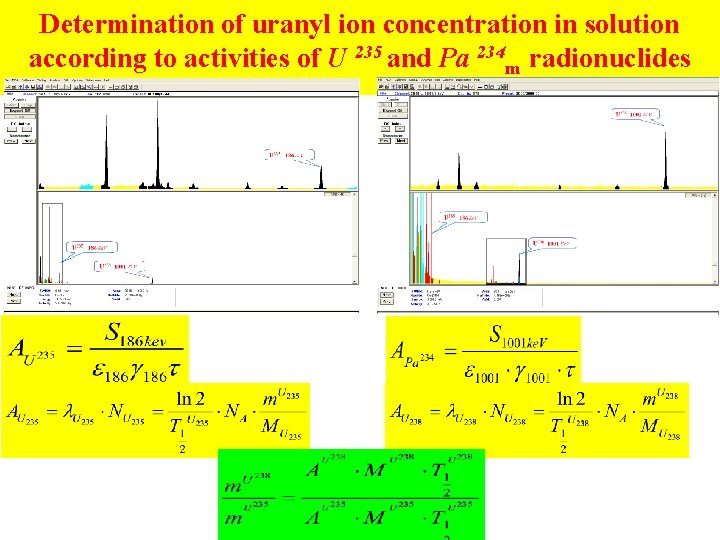
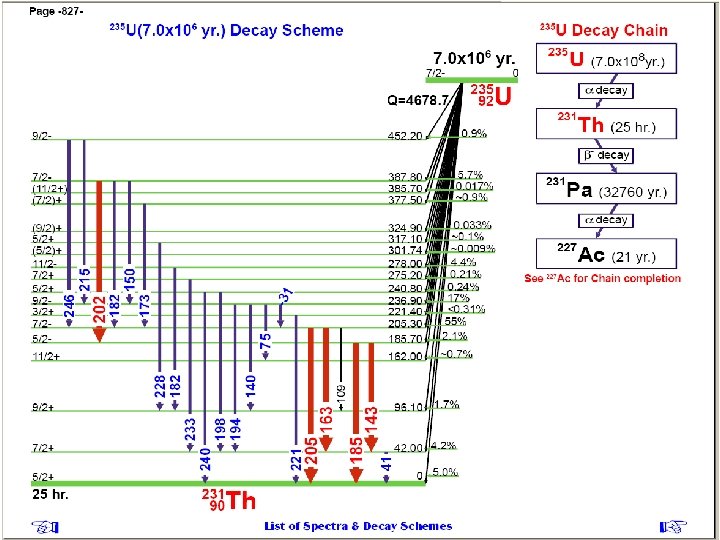
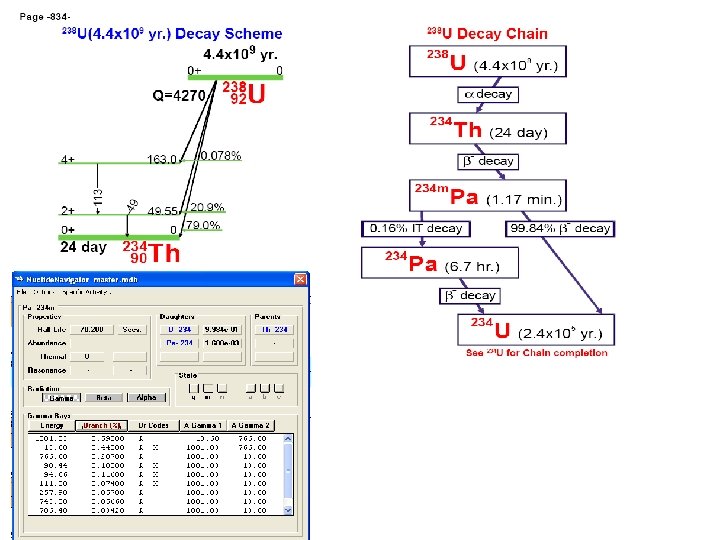
Determination of uranyl ion concentration in solution according to activities of U 235 and Pa 234 m radionuclides
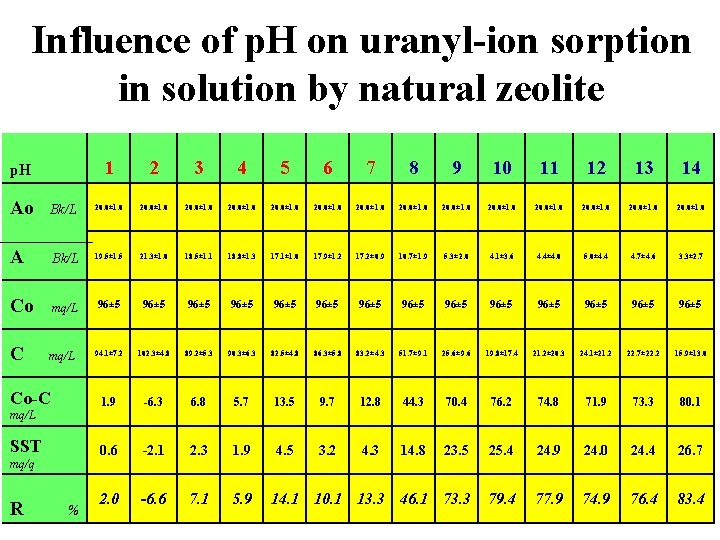
Influence of p. H on uranyl-ion sorption in solution by natural zeolite p. H 1 2 3 4 5 6 7 8 9 10 11 12 13 14 20. 0± 1. 0 20. 0± 1. 0 Ao Bk/L A Bk/L 19. 5± 1. 5 21. 3± 1. 0 18. 5± 1. 1 18. 8± 1. 3 17. 1± 1. 0 17. 9± 1. 2 17. 2± 0. 9 10. 7± 1. 9 5. 3± 2. 0 4. 1± 3. 6 4. 4± 4. 0 5. 0± 4. 4 4. 7± 4. 6 3. 3± 2. 7 Co mq/L 96± 5 96± 5 96± 5 96± 5 C mq/L 94. 1± 7. 2 102. 3± 4. 8 89. 2± 5. 3 90. 3± 6. 3 82. 5± 4. 8 86. 3± 5. 8 83. 2± 4. 3 51. 7± 9. 1 25. 6± 9. 6 19. 8± 17. 4 21. 2± 20. 3 24. 1± 21. 2 22. 7± 22. 2 15. 9± 13. 0 1. 9 -6. 3 6. 8 5. 7 13. 5 9. 7 12. 8 44. 3 70. 4 76. 2 74. 8 71. 9 73. 3 80. 1 0. 6 -2. 1 2. 3 1. 9 4. 5 3. 2 4. 3 14. 8 23. 5 25. 4 24. 9 24. 0 24. 4 26. 7 2. 0 -6. 6 7. 1 5. 9 14. 1 10. 1 13. 3 46. 1 73. 3 79. 4 77. 9 74. 9 76. 4 83. 4 Co-C mq/L SST mq/q R %
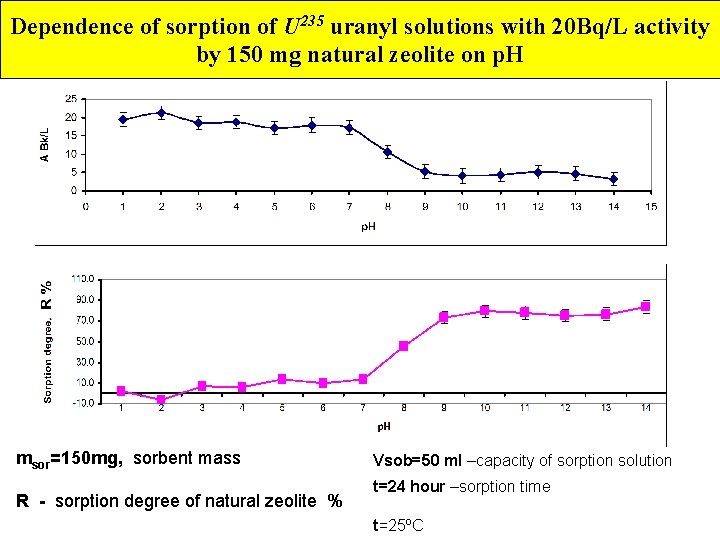
Dependence of sorption of U 235 uranyl solutions with 20 Bq/L activity by 150 mg natural zeolite on p. H msor=150 mg, sorbent mass R - sorption degree of natural zeolite % Vsob=50 ml –capacity of sorption solution t=24 hour –sorption time t=25ºC
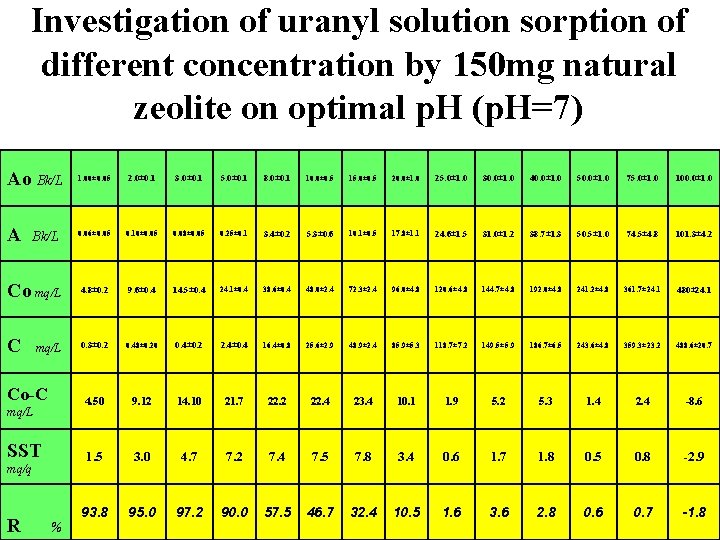
Investigation of uranyl solution sorption of different concentration by 150 mg natural zeolite on optimal p. H (p. H=7) Ao Bk/L 1. 00± 0. 05 2. 0± 0. 1 3. 0± 0. 1 5. 0± 0. 1 8. 0± 0. 1 10. 0± 0. 5 15. 0± 0. 5 20. 0± 1. 0 25. 0± 1. 0 30. 0± 1. 0 40. 0± 1. 0 50. 0± 1. 0 75. 0± 1. 0 100. 0± 1. 0 A 0. 06± 0. 05 0. 10± 0. 05 0. 08± 0. 05 0. 25± 0. 1 3. 4± 0. 2 5. 3± 0. 6 10. 1± 0. 5 17. 8± 1. 1 24. 6± 1. 5 31. 0± 1. 2 38. 7± 1. 3 50. 5± 1. 0 74. 5± 4. 8 101. 3± 4. 2 Co mq/L 4. 8± 0. 2 9. 6± 0. 4 14. 5± 0. 4 24. 1± 0. 4 38. 6± 0. 4 48. 0± 2. 4 72. 3± 2. 4 96. 0± 4. 8 120. 6± 4. 8 144. 7± 4. 8 192. 0± 4. 8 241. 2± 4. 8 361. 7± 24. 1 480± 24. 1 C 0. 3± 0. 2 0. 48± 0. 20 0. 4± 0. 2 2. 4± 0. 4 16. 4± 0. 8 25. 6± 2. 9 48. 9± 2. 4 85. 9± 5. 3 118. 7± 7. 2 149. 5± 5. 9 186. 7± 6. 5 243. 6± 4. 8 359. 3± 23. 2 488. 6± 20. 7 4. 50 9. 12 14. 10 21. 7 22. 2 22. 4 23. 4 10. 1 1. 9 5. 2 5. 3 1. 4 2. 4 -8. 6 1. 5 3. 0 4. 7 7. 2 7. 4 7. 5 7. 8 3. 4 0. 6 1. 7 1. 8 0. 5 0. 8 -2. 9 93. 8 95. 0 97. 2 90. 0 57. 5 46. 7 32. 4 10. 5 1. 6 3. 6 2. 8 0. 6 0. 7 -1. 8 Bk/L mq/L Co-C mq/L SST mq/q R %
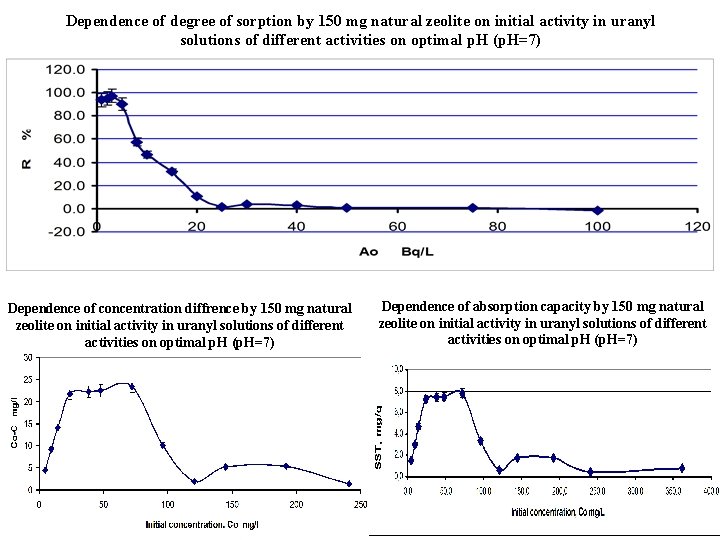
Dependence of degree of sorption by 150 mg natural zeolite on initial activity in uranyl solutions of different activities on optimal p. H (p. H=7) Dependence of concentration diffrence by 150 mg natural zeolite on initial activity in uranyl solutions of different activities on optimal p. H (p. H=7) Dependence of absorption capacity by 150 mg natural zeolite on initial activity in uranyl solutions of different activities on optimal p. H (p. H=7)
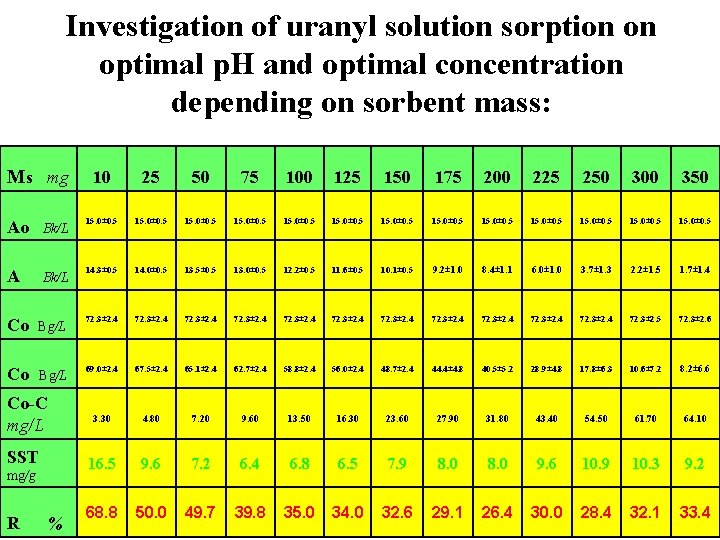
Investigation of uranyl solution sorption on optimal p. H and optimal concentration depending on sorbent mass: Ms mg 10 25 50 75 100 125 150 175 200 225 250 300 350 Ao Bk/L 15. 0± 0. 5 15. 0± 0. 5 A Bk/L 14. 3± 0. 5 14. 0± 0. 5 13. 5± 0. 5 13. 0± 0. 5 12. 2± 0. 5 11. 6± 0. 5 10. 1± 0. 5 9. 2± 1. 0 8. 4± 1. 1 6. 0± 1. 0 3. 7± 1. 3 2. 2± 1. 5 1. 7± 1. 4 Co Bg/L 72. 3± 2. 4 72. 3± 2. 5 72. 3± 2. 6 Co Bg/L 69. 0± 2. 4 67. 5± 2. 4 65. 1± 2. 4 62. 7± 2. 4 58. 8± 2. 4 56. 0± 2. 4 48. 7± 2. 4 44. 4± 4. 8 40. 5± 5. 2 28. 9± 4. 8 17. 8± 6. 3 10. 6± 7. 2 8. 2± 6. 6 3. 30 4. 80 7. 20 9. 60 13. 50 16. 30 23. 60 27. 90 31. 80 43. 40 54. 50 61. 70 64. 10 16. 5 9. 6 7. 2 6. 4 6. 8 6. 5 7. 9 8. 0 9. 6 10. 9 10. 3 9. 2 68. 8 50. 0 49. 7 39. 8 35. 0 34. 0 32. 6 29. 1 26. 4 30. 0 28. 4 32. 1 33. 4 Co-C mg/L SST mg/g R %
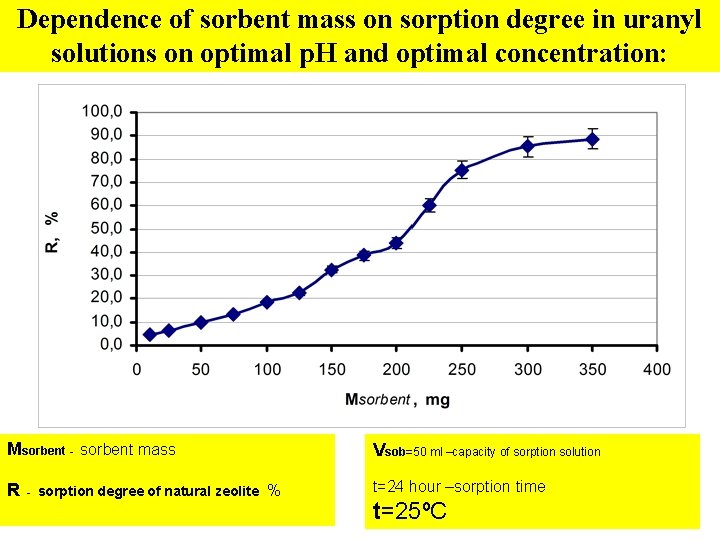
Dependence of sorbent mass on sorption degree in uranyl solutions on optimal p. H and optimal concentration: Msorbent - sorbent mass R - sorption degree of natural zeolite Vsob=50 ml –capacity of sorption solution % t=24 hour –sorption time t=25ºC
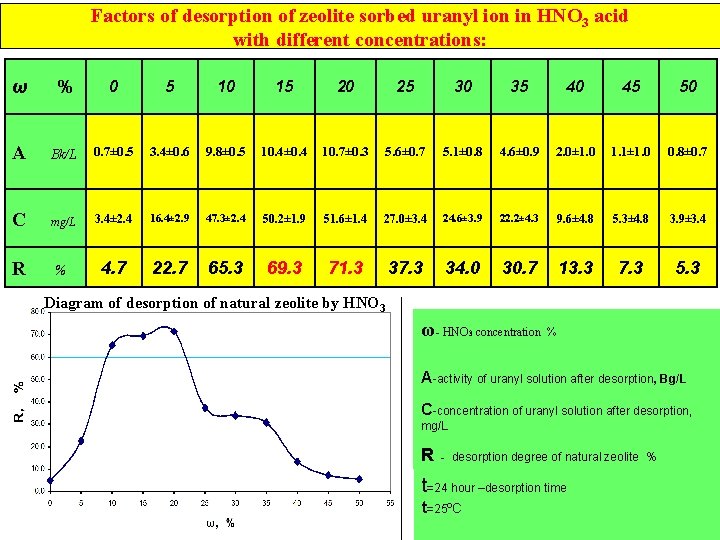
Factors of desorption of zeolite sorbed uranyl ion in HNO 3 acid with different concentrations: ω % 0 5 10 15 20 25 30 35 40 45 50 A Bk/L 0. 7± 0. 5 3. 4± 0. 6 9. 8± 0. 5 10. 4± 0. 4 10. 7± 0. 3 5. 6± 0. 7 5. 1± 0. 8 4. 6± 0. 9 2. 0± 1. 0 1. 1± 1. 0 0. 8± 0. 7 C mg/L 3. 4± 2. 4 16. 4± 2. 9 47. 3± 2. 4 50. 2± 1. 9 51. 6± 1. 4 27. 0± 3. 4 24. 6± 3. 9 22. 2± 4. 3 9. 6± 4. 8 5. 3± 4. 8 3. 9± 3. 4 R % 4. 7 22. 7 65. 3 69. 3 71. 3 37. 3 34. 0 30. 7 13. 3 7. 3 5. 3 Diagram of desorption of natural zeolite by HNO 3 ω- HNO 3 concentration % A-activity of uranyl solution after desorption, Bg/L C-concentration of uranyl solution after desorption, mg/L ms=450 mq -desorbsiya olunan sorbentin kütləsi R - desorption degree ofgedən naturalməhlulun zeolite % Vdes=50 ml -desorbsiya həcmi t=24 hour –desorption time t=25ºC
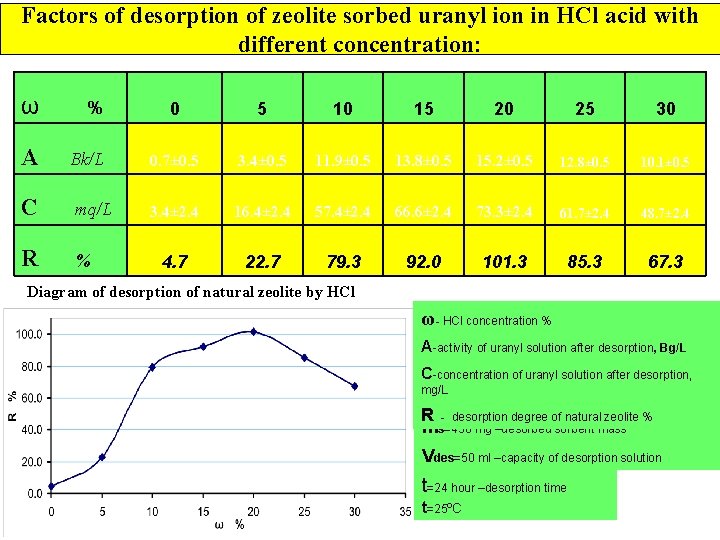
Factors of desorption of zeolite sorbed uranyl ion in HCl acid with different concentration: ω % A 0 5 10 15 20 25 30 Bk/L 0. 7± 0. 5 3. 4± 0. 5 11. 9± 0. 5 13. 8± 0. 5 15. 2± 0. 5 12. 8± 0. 5 10. 1± 0. 5 C mq/L 3. 4± 2. 4 16. 4± 2. 4 57. 4± 2. 4 66. 6± 2. 4 73. 3± 2. 4 61. 7± 2. 4 48. 7± 2. 4 R % 4. 7 22. 7 79. 3 92. 0 101. 3 85. 3 67. 3 Diagram of desorption of natural zeolite by HCl ω- HCl concentration % A-activity of uranyl solution after desorption, Bg/L C-concentration of uranyl solution after desorption, mg/L R - desorption degree of natural zeolite % ms=450 mg –desorbed sorbent mass Vdes=50 ml –capacity of desorption solution t=24 hour –desorption time t=25ºC
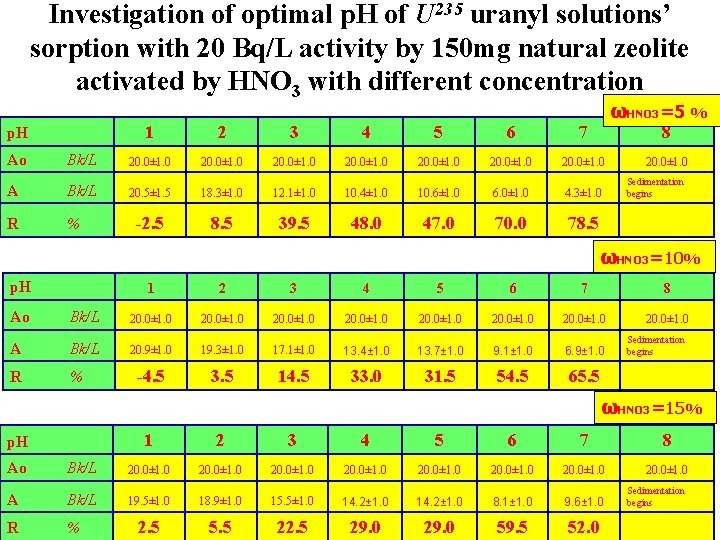
Investigation of optimal p. H of U 235 uranyl solutions’ sorption with 20 Bq/L activity by 150 mg natural zeolite activated by HNO 3 with different concentration p. H 1 2 3 4 5 6 7 ωHNO 3=5 % 8 Ao Bk/L 20. 0± 1. 0 20. 0± 1. 0 A Bk/L 20. 5± 1. 5 18. 3± 1. 0 12. 1± 1. 0 10. 4± 1. 0 10. 6± 1. 0 6. 0± 1. 0 4. 3± 1. 0 R % -2. 5 8. 5 39. 5 48. 0 47. 0 70. 0 78. 5 20. 0± 1. 0 Sedimentation begins ωHNO 3=10% p. H 1 2 3 4 5 6 7 8 20. 0± 1. 0 Ao Bk/L 20. 0± 1. 0 20. 0± 1. 0 A Bk/L 20. 9± 1. 0 19. 3± 1. 0 17. 1± 1. 0 13. 4± 1. 0 13. 7± 1. 0 9. 1± 1. 0 6. 9± 1. 0 R % -4. 5 3. 5 14. 5 33. 0 31. 5 54. 5 65. 5 Sedimentation begins ωHNO 3=15% p. H 1 2 3 4 5 6 7 8 20. 0± 1. 0 Ao Bk/L 20. 0± 1. 0 20. 0± 1. 0 A Bk/L 19. 5± 1. 0 18. 9± 1. 0 15. 5± 1. 0 14. 2± 1. 0 8. 1± 1. 0 9. 6± 1. 0 R % 2. 5 5. 5 22. 5 29. 0 59. 5 52. 0 Sedimentation begins
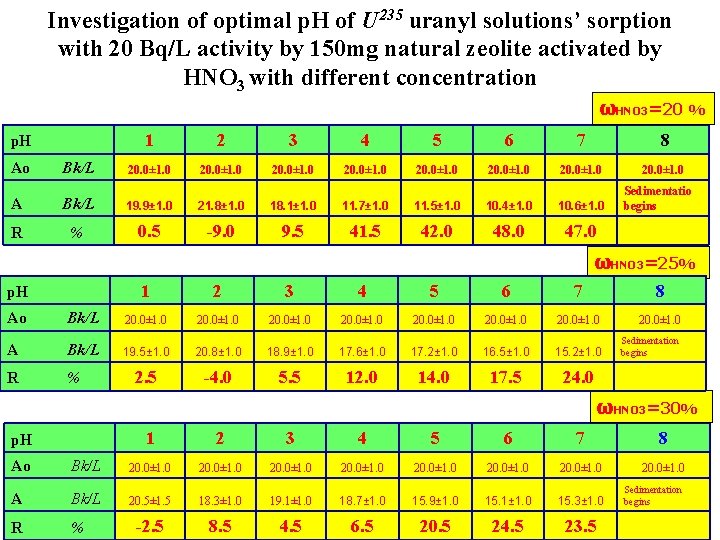
Investigation of optimal p. H of U 235 uranyl solutions’ sorption with 20 Bq/L activity by 150 mg natural zeolite activated by HNO 3 with different concentration ωHNO 3=20 % p. H Ao Bk/L 1 2 3 4 5 6 7 8 20. 0± 1. 0 20. 0± 1. 0 A Bk/L 19. 9± 1. 0 21. 8± 1. 0 18. 1± 1. 0 11. 7± 1. 0 11. 5± 1. 0 10. 4± 1. 0 10. 6± 1. 0 R % 0. 5 -9. 0 9. 5 41. 5 42. 0 48. 0 47. 0 Sedimentatio begins ωHNO 3=25% p. H 1 2 3 4 5 6 7 8 Ao Bk/L 20. 0± 1. 0 20. 0± 1. 0 A Bk/L 19. 5± 1. 0 20. 8± 1. 0 18. 9± 1. 0 17. 6± 1. 0 17. 2± 1. 0 16. 5± 1. 0 15. 2± 1. 0 Sedimentation begins R % 2. 5 -4. 0 5. 5 12. 0 14. 0 17. 5 24. 0 ωHNO 3=30% p. H 1 2 3 4 5 6 7 8 Ao Bk/L 20. 0± 1. 0 20. 0± 1. 0 A Bk/L 20. 5± 1. 5 18. 3± 1. 0 19. 1± 1. 0 18. 7± 1. 0 15. 9± 1. 0 15. 1± 1. 0 15. 3± 1. 0 Sedimentation begins R % -2. 5 8. 5 4. 5 6. 5 20. 5 24. 5 23. 5
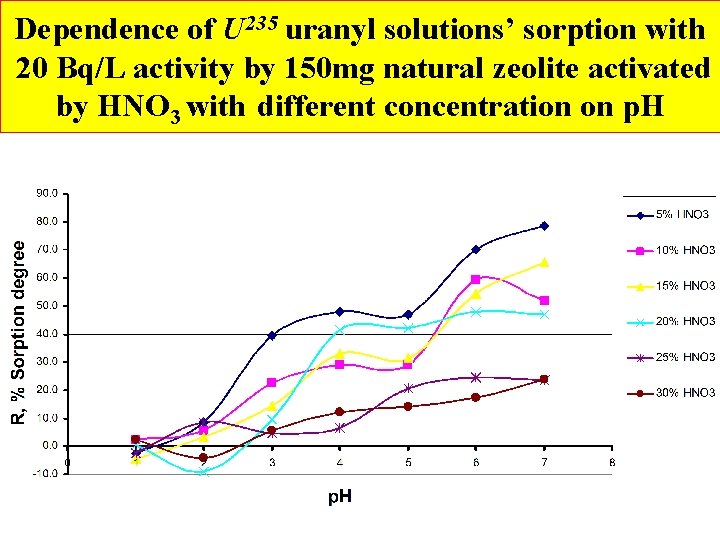
Dependence of U 235 uranyl solutions’ sorption with 20 Bq/L activity by 150 mg natural zeolite activated by HNO 3 with different concentration on p. H
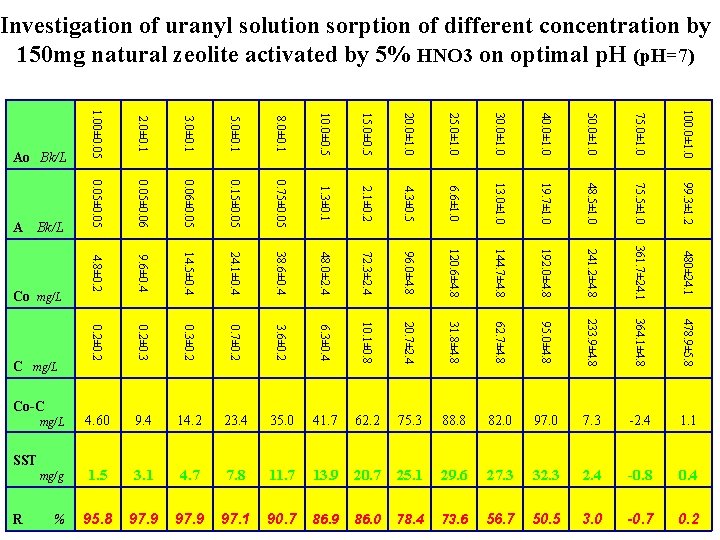
Investigation of uranyl solution sorption of different concentration by 150 mg natural zeolite activated by 5% HNO 3 on optimal p. H (p. H=7) 100. 0± 1. 0 75. 0± 1. 0 50. 0± 1. 0 40. 0± 1. 0 30. 0± 1. 0 25. 0± 1. 0 20. 0± 1. 0 15. 0± 0. 5 10. 0± 0. 5 8. 0± 0. 1 5. 0± 0. 1 3. 0± 0. 1 99. 3± 1. 2 75. 5± 1. 0 48. 5± 1. 0 19. 7± 1. 0 13. 0± 1. 0 6. 6± 1. 0 4. 3± 0. 5 2. 1± 0. 2 1. 3± 0. 1 0. 75± 0. 05 0. 15± 0. 05 0. 06± 0. 05 480± 24. 1 361. 7± 24. 1 241. 2± 4. 8 192. 0± 4. 8 144. 7± 4. 8 120. 6± 4. 8 96. 0± 4. 8 72. 3± 2. 4 48. 0± 2. 4 38. 6± 0. 4 24. 1± 0. 4 14. 5± 0. 4 0. 2± 0. 3± 0. 2 0. 7± 0. 2 3. 6± 0. 2 6. 3± 0. 4 10. 1± 0. 8 20. 7± 2. 4 31. 8± 4. 8 62. 7± 4. 8 95. 0± 4. 8 233. 9± 4. 8 364. 1± 4. 8 478. 9± 5. 8 23. 4 35. 0 41. 7 62. 2 75. 3 88. 8 82. 0 97. 0 7. 3 -2. 4 1. 1 mg/g 1. 5 3. 1 4. 7 7. 8 11. 7 13. 9 20. 7 25. 1 29. 6 27. 3 32. 3 2. 4 -0. 8 0. 4 95. 8 97. 9 97. 1 90. 7 86. 9 86. 0 78. 4 73. 6 56. 7 50. 5 3. 0 -0. 7 0. 2 % 9. 6± 0. 4 14. 2 SST 0. 05± 0. 06 9. 4 Co-C 2. 0± 0. 1 0. 2± 0. 2 4. 60 C mg/L 4. 8± 0. 2 Co mg/L 0. 05± 0. 05 mg/L R Bk/L A 1. 00± 0. 05 Ao Bk/L
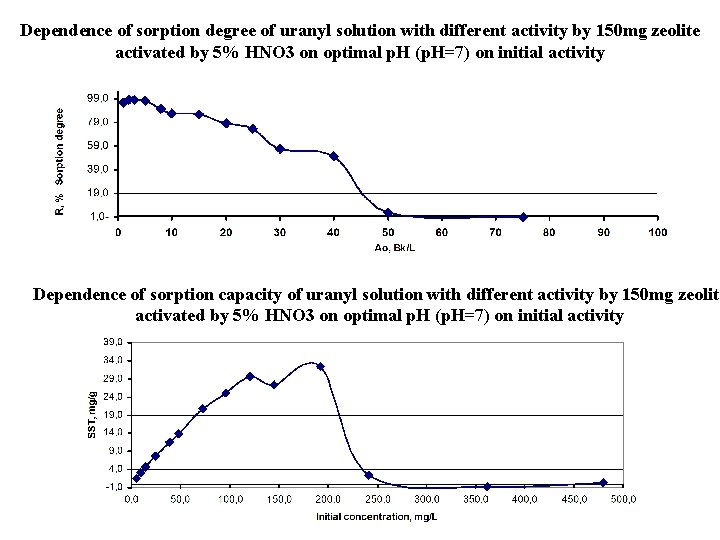
Dependence of sorption degree of uranyl solution with different activity by 150 mg zeolite activated by 5% HNO 3 on optimal p. H (p. H=7) on initial activity Dependence of sorption capacity of uranyl solution with different activity by 150 mg zeolite activated by 5% HNO 3 on optimal p. H (p. H=7) on initial activity

THANK YOU FOR YOUR ATTENTION !!! “ ANY QUESTIONS. . . ? ”
- Slides: 18