Institute of Food and Agricultural Sciences IFAS Biogeochemistry

Institute of Food and Agricultural Sciences (IFAS) Biogeochemistry of Wetlands Science and Applications Carbon Cycling Processes Wetland Biogeochemistry Laboratory Soil and Water Science Department University of Florida Instructor K. Ramesh Reddy krr@ufl. edu 10/3/2020 WBL 1 1

Institute of Food and Agricultural Sciences (IFAS) Carbon Cycling Processes CO 2 OM CH 4 10/3/2020 WBL 2

Carbon Cycling Processes Lecture Outline v v v v 10/3/2020 Introduction Major components of carbon cycle Organic matter accumulation Characteristics of organic matter Decomposition processes Regulators of organic matter decomposition Greenhouse gases Summary WBL 3

Carbon Cycling Processes Learning Objectives Describe major components of carbon cycle v Develop an understanding of the chemical composition of plant litter and soil organic matter v Long-term accumulation of organic matter v Describe the role of enzymes and microbial communities involved in decomposition v Determine organic matter turnover v Indentify the role biogeochemical controls and regulators v Understand the global significance of carbon cycle v Draw a carbon cycle and identify storages and fluxes within and between soil and water column v 10/3/2020 WBL 4
![Oxidation States of Carbon [+4] [0] CO 2 C 6 H 12 O 6 Oxidation States of Carbon [+4] [0] CO 2 C 6 H 12 O 6](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-5.jpg)
Oxidation States of Carbon [+4] [0] CO 2 C 6 H 12 O 6 [-4] CH 4 10/3/2020 WBL 5
![Carbon Reservoirs [1014 kg] v Atmospheric CO 2 v Biomass v Fresh water v Carbon Reservoirs [1014 kg] v Atmospheric CO 2 v Biomass v Fresh water v](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-6.jpg)
Carbon Reservoirs [1014 kg] v Atmospheric CO 2 v Biomass v Fresh water v Marine v Soil organic matter 10/3/2020 WBL 7 4. 8 2. 5 5 -8 30 -50 6
![Soil Organic Matter [SOM] v Undecayed plant and animal tissues v Partially decomposed material Soil Organic Matter [SOM] v Undecayed plant and animal tissues v Partially decomposed material](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-7.jpg)
Soil Organic Matter [SOM] v Undecayed plant and animal tissues v Partially decomposed material v Soil biomass Sources of SOM v External: Particulate (inputs) v Internal: detrital material (macrophytes, algal mats, roots) 10/3/2020 WBL 7
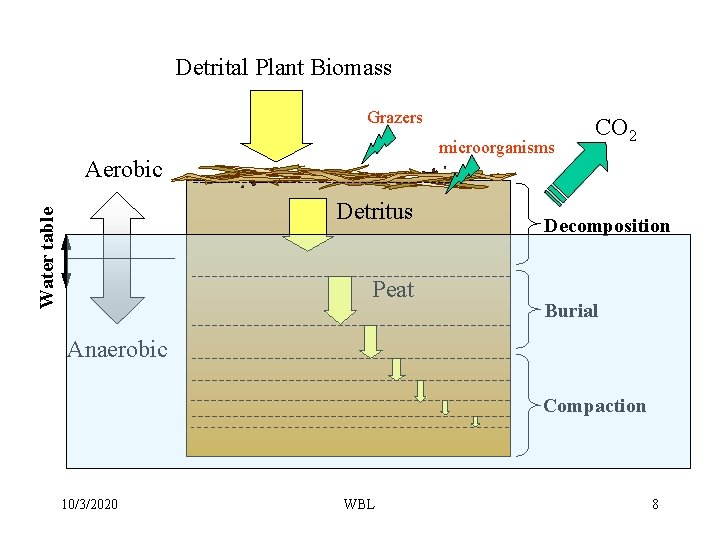
Detrital Plant Biomass Grazers microorganisms Aerobic Water table Detritus Peat CO 2 Decomposition Burial Anaerobic Compaction 10/3/2020 WBL 8
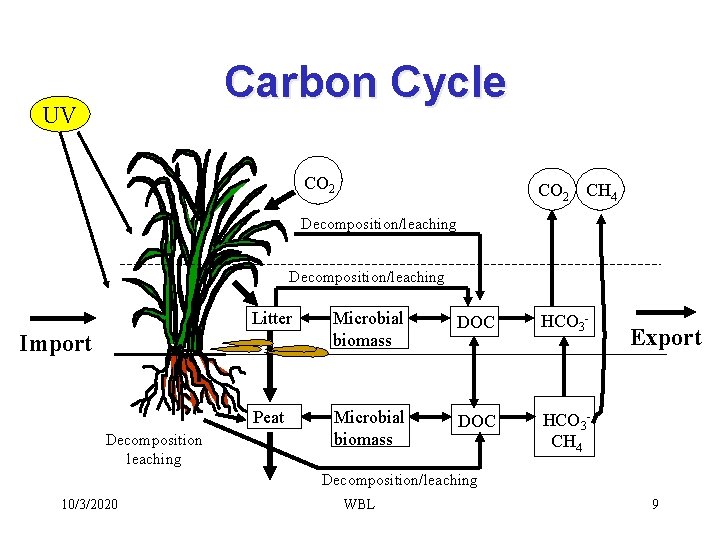
Carbon Cycle UV CO 2 CH 4 Decomposition/leaching Litter Microbial biomass DOC HCO 3 - Peat Microbial biomass DOC HCO 3 CH 4 Import Decomposition leaching Export Decomposition/leaching 10/3/2020 WBL 9

Organic Matter Ø Storages Ø Ø Soil organic matter Plant detritus/litter Dissolved organic matter Microbial biomass Ø Transformations Ø Outputs Ø Greenhouse gases Ø Nutrient export Ø Ecological/Environment al Significance Ø Ø Ø Microbial respiration Ø Methanogenesis 10/3/2020 WBL Carbon sequestration Global warming Water quality Ecosystem productivity 10
![Net Primary Productivity [g/m 2 - year] [Craft, 2001] Bog Marsh Riverine Fresh tidal Net Primary Productivity [g/m 2 - year] [Craft, 2001] Bog Marsh Riverine Fresh tidal](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-11.jpg)
Net Primary Productivity [g/m 2 - year] [Craft, 2001] Bog Marsh Riverine Fresh tidal Brackish Salt Mangroves 10/3/2020 380 -800 500 -1100 400 -1150 500 -1600 600 -1600 950 -2000 600 -1200 WBL 11
![Carbon Accumulation in Wetlands [g C/m 2 year] Alaska - Sphagnum Finland - Sphagnum Carbon Accumulation in Wetlands [g C/m 2 year] Alaska - Sphagnum Finland - Sphagnum](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-12.jpg)
Carbon Accumulation in Wetlands [g C/m 2 year] Alaska - Sphagnum Finland - Sphagnum - Carex Ontario - Sphagnum bog Georgia - Taxodium Florida - Cladium 10/3/2020 WBL 11 -61 20 -28 30 -32 23 70 -105 12
![Organic Matter Accumulation Soil Depth [cm] 0 Organic matter accumulation 10 1964 marker 20 Organic Matter Accumulation Soil Depth [cm] 0 Organic matter accumulation 10 1964 marker 20](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-13.jpg)
Organic Matter Accumulation Soil Depth [cm] 0 Organic matter accumulation 10 1964 marker 20 Cs-137 Activity 10/3/2020 WBL 13
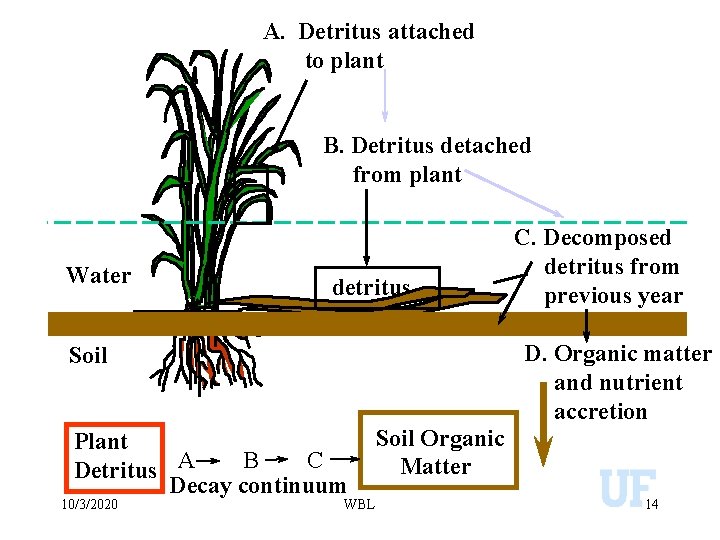
A. Detritus attached to plant B. Detritus detached from plant Water detritus D. Organic matter and nutrient accretion Soil Plant B C Detritus A Decay continuum 10/3/2020 C. Decomposed detritus from previous year WBL Soil Organic Matter 14
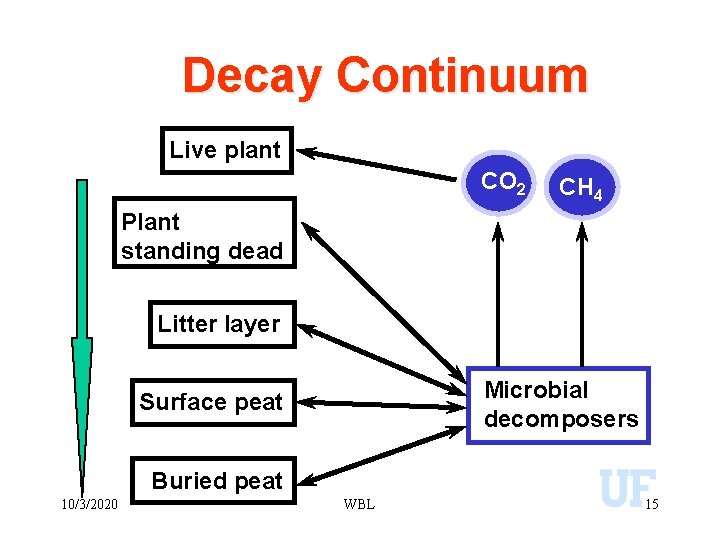
Decay Continuum Live plant CO 2 CH 4 Plant standing dead Litter layer Microbial decomposers Surface peat Buried peat 10/3/2020 WBL 15

Carbon Accumulation in Wetlands v Potential energy source (reduced carbon, electron donor v Long-term storage of nutrients, heavy metals, and toxic organic compounds v Major component of global carbon cycles 10/3/2020 WBL 16

Carbon Forms v Particulate organic carbon (POC) v Microbial biomass carbon (MBC) v Dissolved organic carbon (DOC) v Dissolved inorganic carbon (DIC) v CO 2 + H 2 O = H 2 CO 3 v H 2 CO 3 = HCO 3 - + H+ v HCO 3 - = CO 32 - + H+ 10/3/2020 WBL 17

Chemical constituents of organic matter u Non Humic compounds: u Carbohydrates (Simple sugars) u Monosaccharides: glucose. u Polysaccharides: Starch, Cellulose, and Hemicellulose Proteins u Lipids etc u u Phenolic compounds: Lignin (branched random polymer of phenyl propanoid unit) u Tannins (heterogeneous groups of phenolic compounds) u 10/3/2020 WBL 18
![Organic Matter (Plant and Soil) • Water soluble components [<10%] – Sugars, amino acids Organic Matter (Plant and Soil) • Water soluble components [<10%] – Sugars, amino acids](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-19.jpg)
Organic Matter (Plant and Soil) • Water soluble components [<10%] – Sugars, amino acids and fatty acids • • • Cellulose [15 -60%] Hemicellulose [10 -30%] Lignin [5 -30%] Proteins [2 -15%] Lipids and Waxes [1 -8%] Ash (mineral) [1 -13%] 10/3/2020 WBL 19
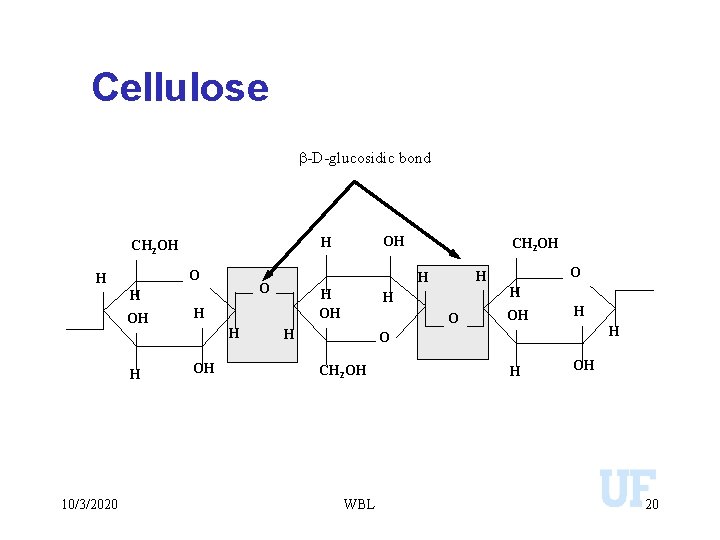
Cellulose b-D-glucosidic bond O H H 10/3/2020 H OH H H OH CH 2 OH H H O H OH OH H CH 2 OH OH H H O CH 2 OH WBL H OH 20
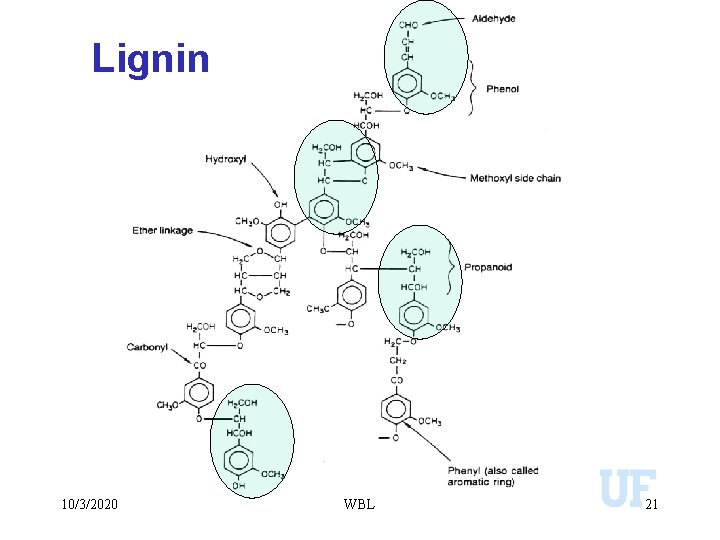
Lignin 10/3/2020 WBL 21
![Soil Organic Matter [SOM] SOM Extract with Alkali [alkali-soluble] Humin [alkali-insoluble] Treat with Acid Soil Organic Matter [SOM] SOM Extract with Alkali [alkali-soluble] Humin [alkali-insoluble] Treat with Acid](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-22.jpg)
Soil Organic Matter [SOM] SOM Extract with Alkali [alkali-soluble] Humin [alkali-insoluble] Treat with Acid Humic Acid Fulvic Acid [acid-insoluble] [acid-soluble] 10/3/2020 WBL 22

Fulvic Acid • • • More ‘O’ and less ‘C’. MW 1000 -30, 000. Less advanced stage of decomposition. More COOH group per unit mass. Functional group acidity (11. 2 mol/kg). Alkali and acid soluble. 10/3/2020 WBL 23

Humic Acid • • • More ‘C’ and less ‘O’. MW 10, 000 -100, 000. Advanced stage of decomposition. Less COOH group per unit mass. Functional group acidity (6. 7 mol/kg). Alkali soluble. 10/3/2020 WBL 24

Available Carbon Pool § Represents small but biologically active fraction of DOC § Immediately available for microbial utilization § Extremely small in C-limited system § Rapid turnover § May not be directly measurable § Affects short-term community metabolism 10/3/2020 WBL 25

Microbial Biomass 10/3/2020 WBL 26
![Microorganisms [Percent weight] • 70% water • Macromolecules • 15% protein • 3% polysaccharide Microorganisms [Percent weight] • 70% water • Macromolecules • 15% protein • 3% polysaccharide](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-27.jpg)
Microorganisms [Percent weight] • 70% water • Macromolecules • 15% protein • 3% polysaccharide • 2% lipids • 5% RNA • 1 % DNA • • q Total weight of actively growing cell of Escherichia coli Wet wt = 9. 5 x 10 -13 g Dry wt = 2. 8 x 10 -13 g 1 % Inorganic ions 3 % others 10/3/2020 WBL 27

Microbial Decomposers v. Typically 1 -5% of total C mass in soil v. Process most of the ecosystem net production v. Principal transformers of organic carbon v. Recycle carbon and nutrients in recalcitrant biopolymers v. Regulate energy flow and nutrient retention 10/3/2020 WBL 28

Techniques to Measure MICROBIAL BIOMASS Direct cell count : abundance Lipid based : live microbial biomass CHCl 3 Fumigation-extraction based: estimate of Carbon Metabolic activity based: Enzyme activities 10/3/2020 WBL 29

MICROBIAL COMMUNITY STRUCTURE v Pure culture approach v Microscopy v Community level physiological profile (CLPP): Substrate utilization: BIOLOG v Measurement of cellular component (physiological status, functional groups): PLFA v Methods based on nucleic acids analysis (abundance, diversity and phylogeny of organisms): gene specific analysis (16 S r. DNA, DGGE, Trflp) 10/3/2020 WBL 30
![MICROBIAL BIOMASS [Site = WCA-2 A - Everglades] % of Total Carbon 10 9 MICROBIAL BIOMASS [Site = WCA-2 A - Everglades] % of Total Carbon 10 9](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-31.jpg)
MICROBIAL BIOMASS [Site = WCA-2 A - Everglades] % of Total Carbon 10 9 8 7 LITTER 6 0 -10 cm 5 4 10 -30 cm 3 2 1 0 0 2 4 6 8 10 Distance from Inflow, km 10/3/2020 WBL 31
![MICROBIAL NUMBERS [MPN/g soil] [Site = WCA-2 A - Everglades] Substrate Eutrophic Oligotrophic Lactate MICROBIAL NUMBERS [MPN/g soil] [Site = WCA-2 A - Everglades] Substrate Eutrophic Oligotrophic Lactate](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-32.jpg)
MICROBIAL NUMBERS [MPN/g soil] [Site = WCA-2 A - Everglades] Substrate Eutrophic Oligotrophic Lactate 9. 3 x 105 9. 2 x 103 Acetate 2. 3 x 105 3. 6 x 103 Propionate 4. 3 x 105 9. 2 x 103 Butyrate 4. 3 x 105 < 3. 0 x 103 Formate 2. 3 x 105 < 3. 0 x 103 Hector et al. 2003 10/3/2020 WBL 32
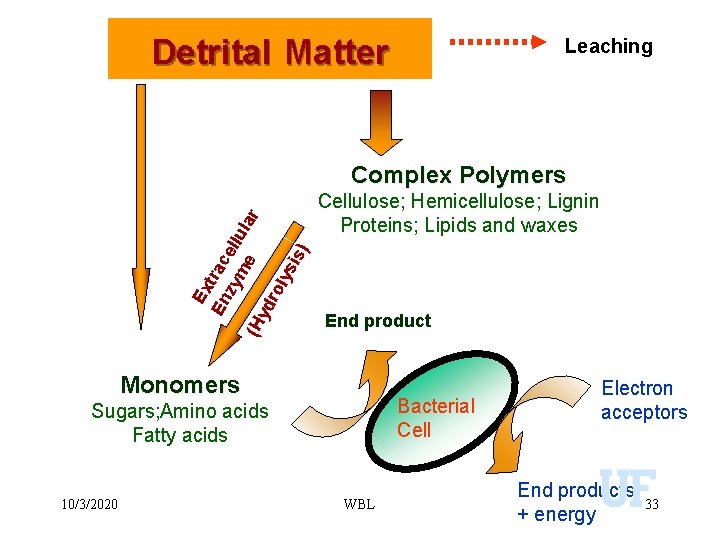
Detrital Matter Leaching Ex En trac zy ell me ula r (H yd ro lys is) Complex Polymers Cellulose; Hemicellulose; Lignin Proteins; Lipids and waxes End product Monomers Bacterial Cell Sugars; Amino acids Fatty acids 10/3/2020 WBL Electron acceptors End products + energy 33

Extracellular Enzymes 10/3/2020 WBL 34

Extracellular Enzymes • An extracellular enzyme is involved in transformation or degradation of polymeric substances external to cell membrane. – Enzyme can be bound to the cell membrane or are periplasmic (ectoenzyme) (Chrost, 1990) – Enzyme occurs free in the water or adsorbed to surface other than its producers e. g. , detrital particles or clay material (extracellular enzyme) Bacterial cell Periplasmic space Detrital/clay material • Most of these are hydrolases 10/3/2020 WBL 35

Enzymes • Cellulose degradation – Exocellulase - Cellulose – B-glucosidase - Cellobiose • Hemicellulose degradation – Exoxylanase - Xylan – B-xylosidase - Xylobiose • Lignin degradation – Phenol oxidase - Lignin and Phenols 10/3/2020 WBL 36
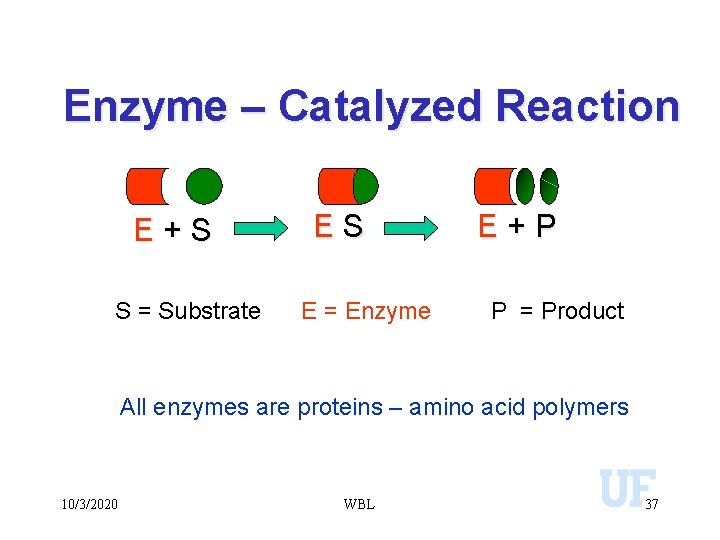
Enzyme – Catalyzed Reaction E+S S = Substrate ES E = Enzyme E+P P = Product All enzymes are proteins – amino acid polymers 10/3/2020 WBL 37
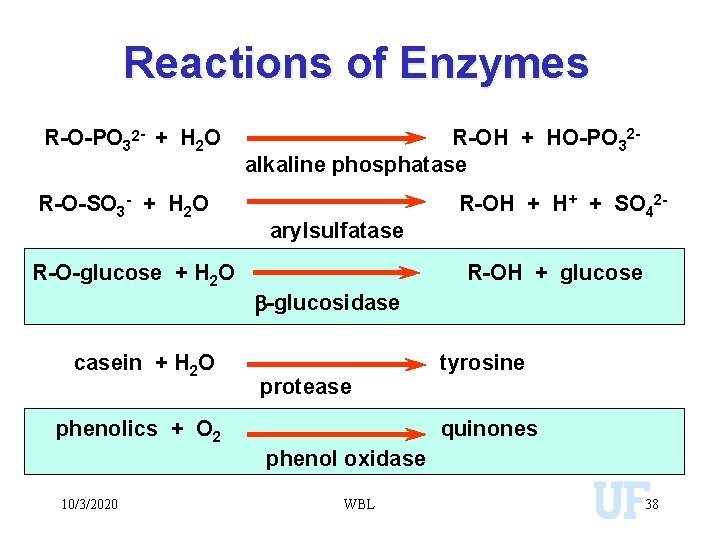
Reactions of Enzymes R-O-PO 32 - + H 2 O R-O-SO 3 - + H 2 O R-O-glucose + H 2 O casein + H 2 O R-OH + HO-PO 32 alkaline phosphatase arylsulfatase R-OH + + SO 42 R-OH + glucose b-glucosidase protease phenolics + O 2 tyrosine quinones phenol oxidase 10/3/2020 WBL 38
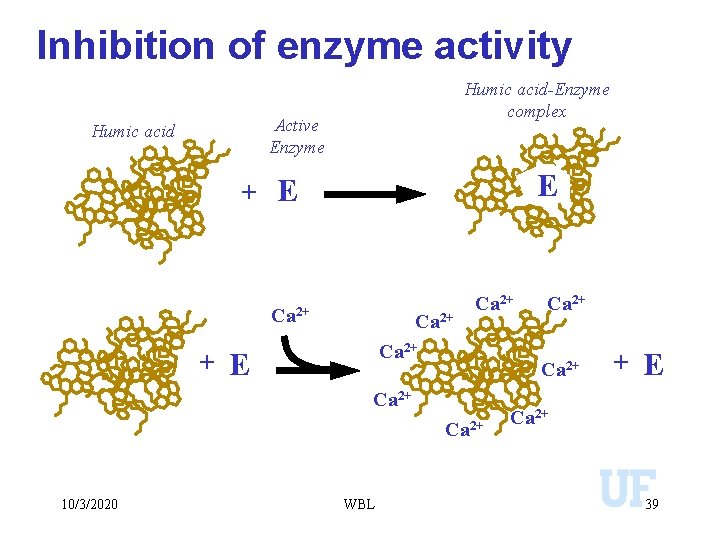
Inhibition of enzyme activity Humic acid-Enzyme complex Active Enzyme Humic acid E + E Ca 2+ Ca 2+ 10/3/2020 WBL Ca 2+ + E Ca 2+ 39

Measurement of Enzymes • Spectroscopic – p-nitrophenol phosphate (p. NPP) • Fluorescence – Methylumbelliferyl phosphate (MUF) – Enzyme Labeled Fluorescence (ELF) P MUF-P 10/3/2020 APase P MUF WBL Pi 40
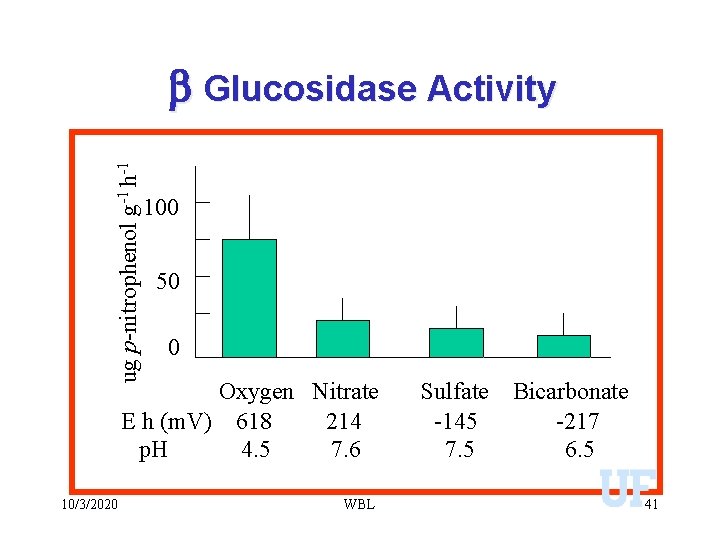
ug p-nitrophenol g-1 h-1 b Glucosidase Activity 100 50 0 Oxygen Nitrate E h (m. V) 618 214 p. H 4. 5 7. 6 10/3/2020 WBL Sulfate -145 7. 5 Bicarbonate -217 6. 5 41
![b Glucosidase Activity B-D-Glucosidase Activity (mg p- nitrophenol g-1 h-1) 4 [Everglades -WCA-2 A] b Glucosidase Activity B-D-Glucosidase Activity (mg p- nitrophenol g-1 h-1) 4 [Everglades -WCA-2 A]](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-42.jpg)
b Glucosidase Activity B-D-Glucosidase Activity (mg p- nitrophenol g-1 h-1) 4 [Everglades -WCA-2 A] February 2 impacted transitional unimpacted 0 4 May 2 0 4 August 2 0 Detritus 10/3/2020 Wright and Reddy, 2001 0 -10 cm WBL 10 -30 cm 42
![Pheno oxidase Activity Phenol Oxidase Activity (umole [DQC]g-1 min-1) [Everglades -WCA-2 A] Wright and Pheno oxidase Activity Phenol Oxidase Activity (umole [DQC]g-1 min-1) [Everglades -WCA-2 A] Wright and](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-43.jpg)
Pheno oxidase Activity Phenol Oxidase Activity (umole [DQC]g-1 min-1) [Everglades -WCA-2 A] Wright and Reddy, 2001 5 4 3 2 1 0 May August Detritus 0 -10 cm impacted transitional unimpacted 10 -30 cm DQC = dihydroindole quinone carboxylate 10/3/2020 WBL 43

Microbial Activity 10/3/2020 WBL 44
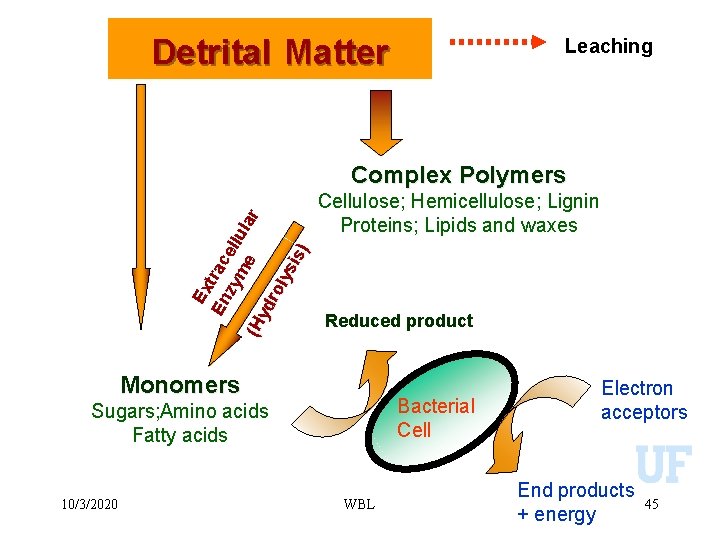
Detrital Matter Leaching Ex En trac zy ell me ula r (H yd ro lys is) Complex Polymers Cellulose; Hemicellulose; Lignin Proteins; Lipids and waxes Reduced product Monomers Bacterial Cell Sugars; Amino acids Fatty acids 10/3/2020 WBL Electron acceptors End products + energy 45
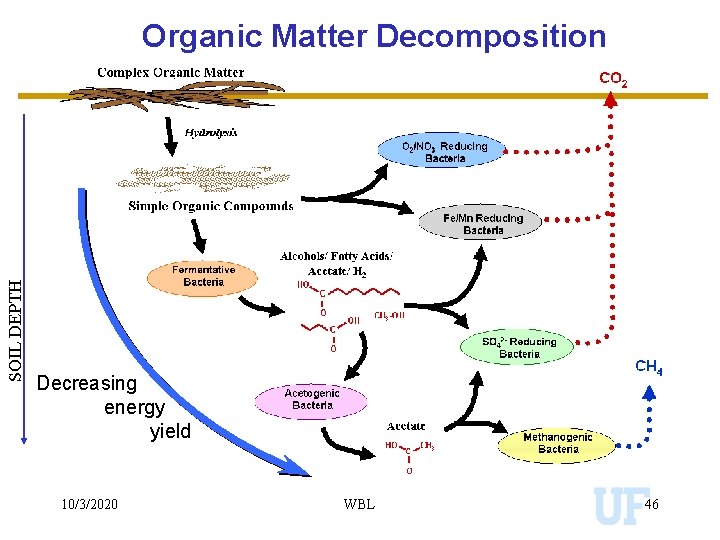
SOIL DEPTH Organic Matter Decomposition Decreasing energy yield 10/3/2020 WBL 46

Metabolism • Catabolism • Anabolism • Types of energy source • Light … Phototrophs • Inorganic … Lithotrophs • Organic …. Heterotrophs • Oxidation of organic compounds • Fermentation • Respiration 10/3/2020 WBL 47

Chemolithotrophy v Inorganic compound as energy source veg. H 2 S, Hydrogen gas, Fe(II), and NH 3 v Source of carbon for biosynthesis cannot be organic therefore use CO 2 and hence are autotrophs v Hydrogen oxidation v Sulfur oxidation v Ferrous iron oxidation v Annamox v Nitrification 10/3/2020 WBL 48
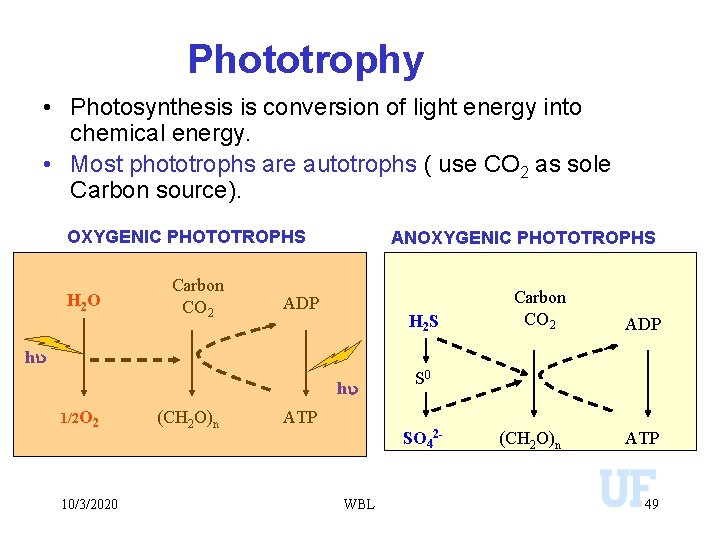
Phototrophy • Photosynthesis is conversion of light energy into chemical energy. • Most phototrophs are autotrophs ( use CO 2 as sole Carbon source). OXYGENIC PHOTOTROPHS H 2 O Carbon CO 2 ANOXYGENIC PHOTOTROPHS ADP H 2 S Carbon CO 2 ADP (CH 2 O)n ATP hu hu 1/2 O 2 10/3/2020 (CH 2 O)n S 0 ATP SO 42 - WBL 49
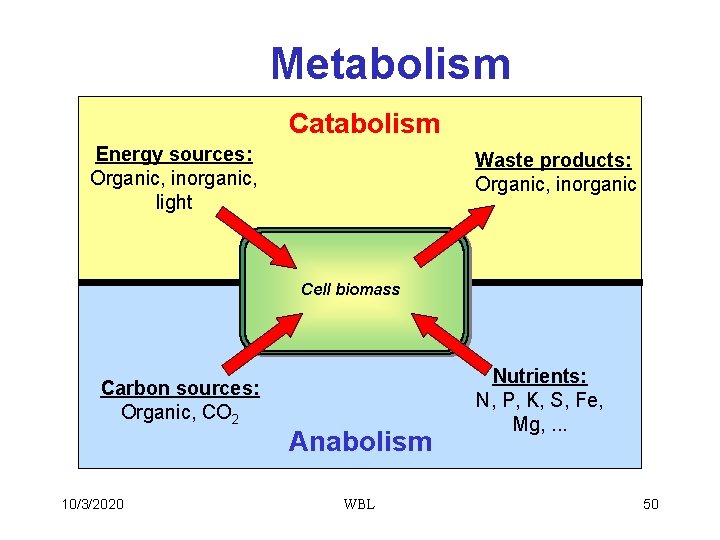
Metabolism Catabolism Energy sources: Organic, inorganic, light Waste products: Organic, inorganic Cell biomass Carbon sources: Organic, CO 2 10/3/2020 Anabolism WBL Nutrients: N, P, K, S, Fe, Mg, . . . 50
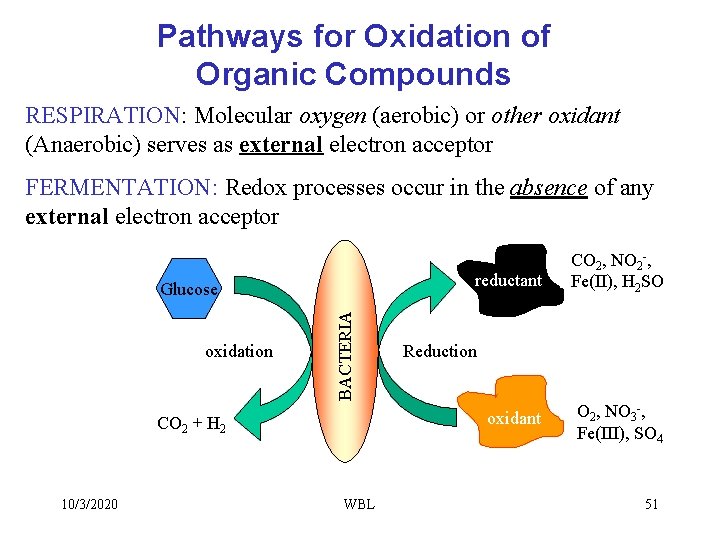
Pathways for Oxidation of Organic Compounds RESPIRATION: Molecular oxygen (aerobic) or other oxidant (Anaerobic) serves as external electron acceptor FERMENTATION: Redox processes occur in the absence of any external electron acceptor reductant oxidation BACTERIA Glucose 10/3/2020 Reduction oxidant CO 2 + H 2 WBL CO 2, NO 2 -, Fe(II), H 2 SO O 2, NO 3 -, Fe(III), SO 4 51
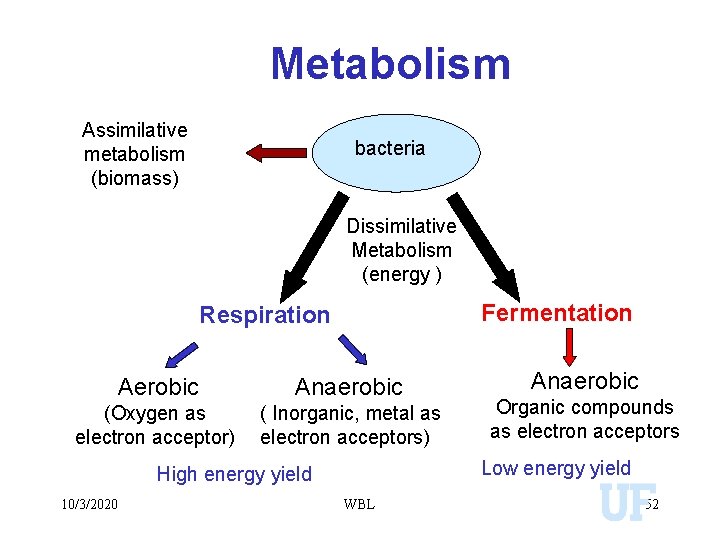
Metabolism Assimilative metabolism (biomass) bacteria Dissimilative Metabolism (energy ) Fermentation Respiration Aerobic Anaerobic (Oxygen as electron acceptor) ( Inorganic, metal as electron acceptors) Organic compounds as electron acceptors Low energy yield High energy yield 10/3/2020 Anaerobic WBL 52
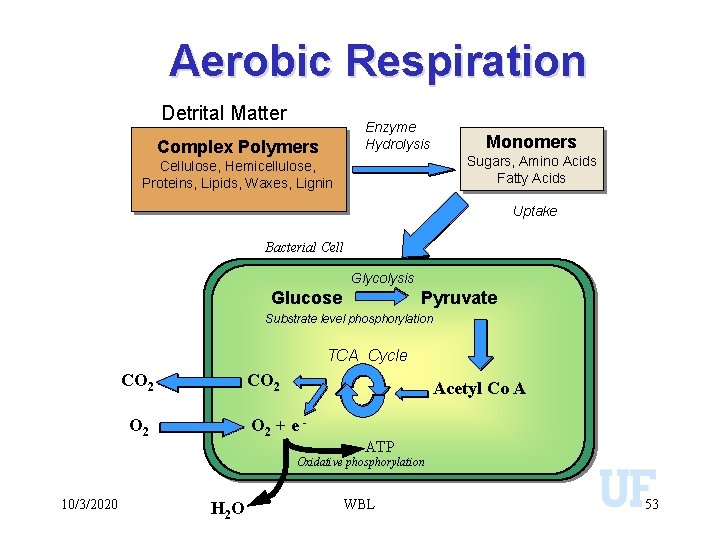
Aerobic Respiration Detrital Matter Enzyme Hydrolysis Complex Polymers Monomers Sugars, Amino Acids Fatty Acids Cellulose, Hemicellulose, Proteins, Lipids, Waxes, Lignin Uptake Bacterial Cell Glycolysis Glucose Pyruvate Substrate level phosphorylation TCA Cycle CO 2 Acetyl Co A O 2 + e - O 2 ATP Oxidative phosphorylation 10/3/2020 H 2 O WBL 53
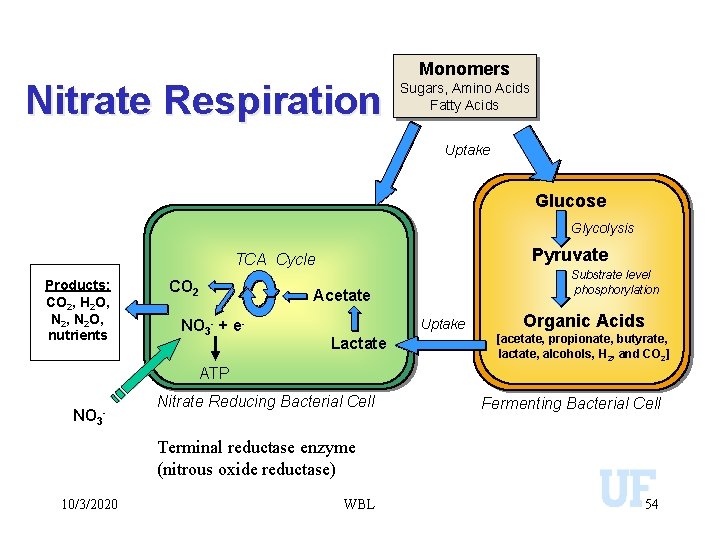
Nitrate Respiration Monomers Sugars, Amino Acids Fatty Acids Uptake Glucose Glycolysis Pyruvate TCA Cycle Products: CO 2, H 2 O, N 2 O, nutrients CO 2 NO 3 - + e- Substrate level phosphorylation Acetate Uptake Lactate Organic Acids [acetate, propionate, butyrate, lactate, alcohols, H 2, and CO 2] ATP NO 3 - Nitrate Reducing Bacterial Cell Fermenting Bacterial Cell Terminal reductase enzyme (nitrous oxide reductase) 10/3/2020 WBL 54
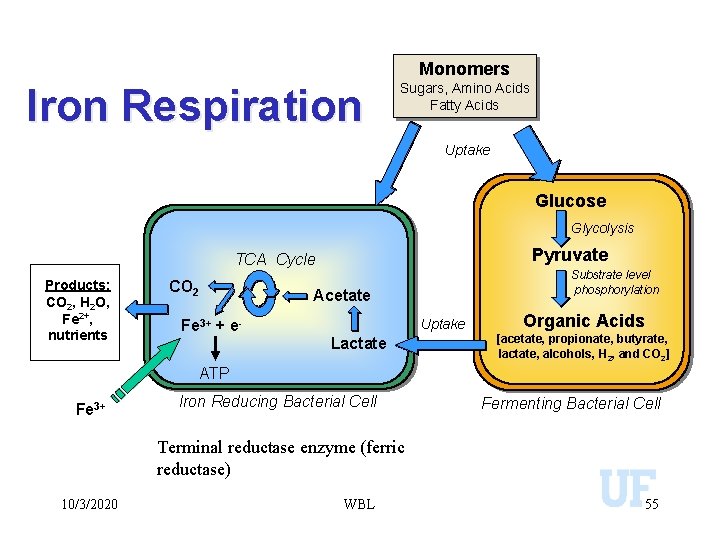
Monomers Iron Respiration Sugars, Amino Acids Fatty Acids Uptake Glucose Glycolysis Pyruvate TCA Cycle Products: CO 2, H 2 O, Fe 2+, nutrients CO 2 Fe 3+ + e- Substrate level phosphorylation Acetate Uptake Lactate Organic Acids [acetate, propionate, butyrate, lactate, alcohols, H 2, and CO 2] ATP Fe 3+ Iron Reducing Bacterial Cell Fermenting Bacterial Cell Terminal reductase enzyme (ferric reductase) 10/3/2020 WBL 55
![Fermentation Organic compound Bacterial Cell Oxidation Oxidized Organic compounds [Pyruvate] Reduction Electron carriers Reduced Fermentation Organic compound Bacterial Cell Oxidation Oxidized Organic compounds [Pyruvate] Reduction Electron carriers Reduced](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-56.jpg)
Fermentation Organic compound Bacterial Cell Oxidation Oxidized Organic compounds [Pyruvate] Reduction Electron carriers Reduced Organic compounds [Ethanol] 10/3/2020 WBL 56
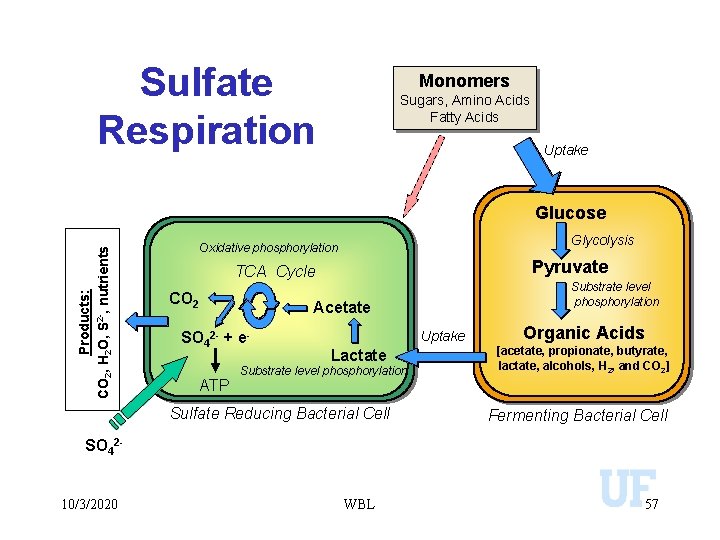
Sulfate Respiration Monomers Sugars, Amino Acids Fatty Acids Uptake Products: CO 2, H 2 O, S 2 -, nutrients Glucose Glycolysis Oxidative phosphorylation Pyruvate TCA Cycle CO 2 Substrate level phosphorylation Acetate SO 42 - + e- Uptake Organic Acids Lactate [acetate, propionate, butyrate, lactate, alcohols, H 2, and CO 2] Sulfate Reducing Bacterial Cell Fermenting Bacterial Cell ATP Substrate level phosphorylation SO 4210/3/2020 WBL 57

Methanogens v. Archaea…not bacteria v H 2 is electron donor and CO 2 is electron acceptor and reduced to CH 4 (autotrophic, chemolithotrophy) 131 k. J/mol v Respiration, not fermentation v Some other substrates that can yield electrons are: v. Hydrogen vmethanol v. Formate 10/3/2020 WBL 58

Methanogens v Hydrogenotrophic methanogens: use H 2 (as electron donor) and CO 2 v Acetotrophic methanogens: oxidation of acetate results in CO 2 and CH 4. 10/3/2020 WBL 59
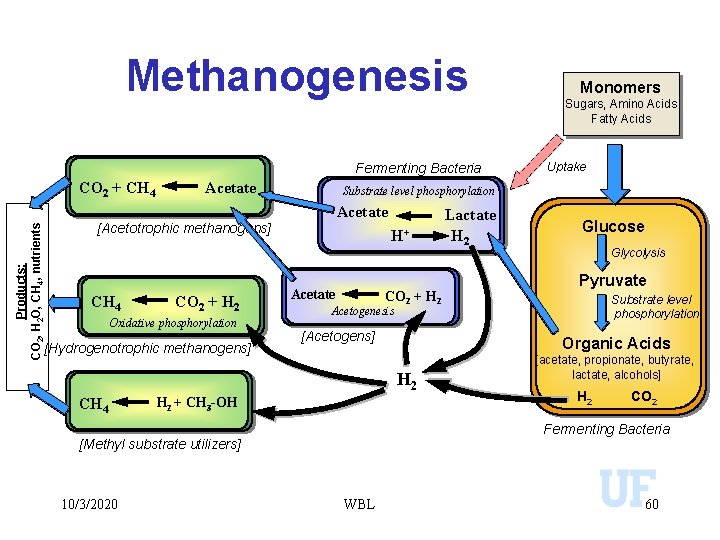
Methanogenesis Fermenting Bacteria CO 2 + CH 4 Acetate Products: CO 2, H 2 O, CH 4, nutrients [Acetotrophic methanogens] CO 2 + H 2 Oxidative phosphorylation [Hydrogenotrophic methanogens] H+ Acetate CO 2 + H 2 [Acetogens] H 2 + CH 3 -OH Lactate H 2 Glucose Glycolysis Pyruvate Substrate level phosphorylation Organic Acids [acetate, propionate, butyrate, lactate, alcohols] H 2 CO 2 Fermenting Bacteria [Methyl substrate utilizers] 10/3/2020 Uptake Acetogenesis H 2 CH 4 Sugars, Amino Acids Fatty Acids Substrate level phosphorylation Acetate CH 4 Monomers WBL 60

Other Terminal Electron Acceptors Inorganic Terminal Electron Acceptors Heavy metals as electron acceptors e. g. • Chromate Cr(VI) Chromium Cr(III) • Arsenate (As. O 43 -) Arsenite (As. O 33 -) • Selenate (Se. O 42 -) Selenite (Se. O 32 -) inorg. Se Organic Terminal Electron Acceptors Fumarate succinate Trimethyl amine oxide (TMAO) trimethlamine(TMA) Dimethyl sulfoxide (DMSO) Dimethyl sulfide Reductive dechlorination 10/3/2020 WBL 61
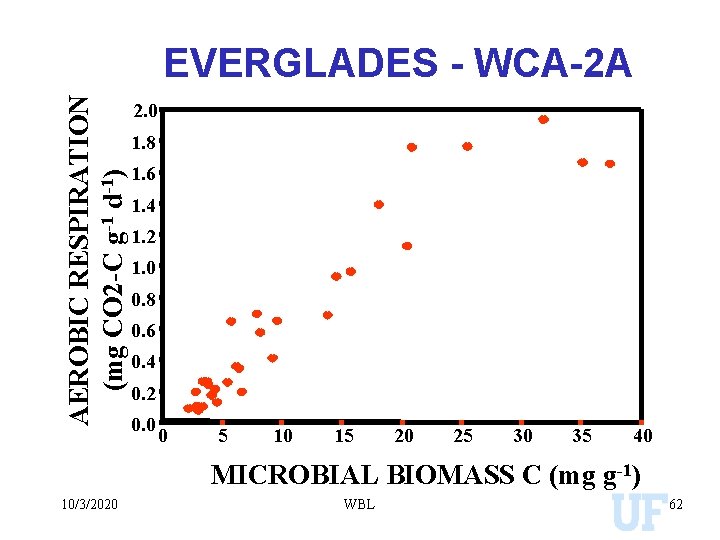
AEROBIC RESPIRATION (mg CO 2 -C g-1 d-1) EVERGLADES - WCA-2 A 2. 0 1. 8 1. 6 1. 4 1. 2 1. 0 0. 8 0. 6 0. 4 0. 2 0. 0 0 5 10 15 20 25 30 35 40 MICROBIAL BIOMASS C (mg g-1) 10/3/2020 WBL 62
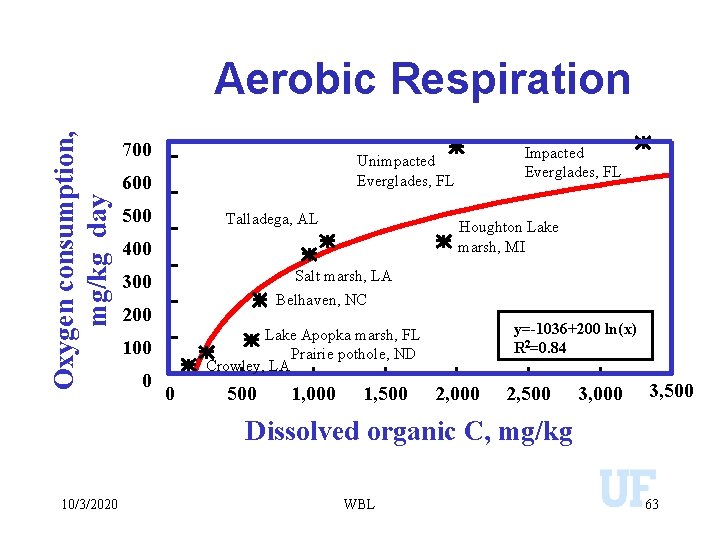
Oxygen consumption, mg/kg day Aerobic Respiration 700 600 500 Talladega, AL Houghton Lake marsh, MI 400 Salt marsh, LA 300 Belhaven, NC 200 y=-1036+200 ln(x) R 2=0. 84 Lake Apopka marsh, FL Prairie pothole, ND Crowley, LA 100 0 Impacted Everglades, FL Unimpacted Everglades, FL 0 500 1, 000 1, 500 2, 000 2, 500 3, 000 3, 500 Dissolved organic C, mg/kg 10/3/2020 WBL 63
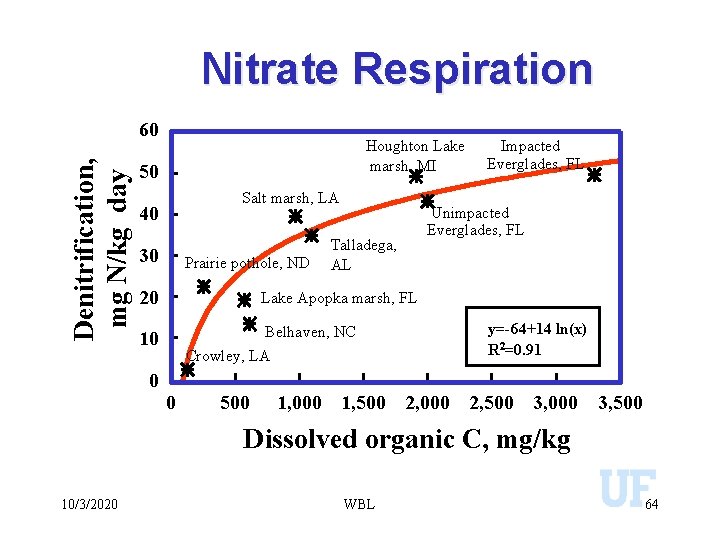
Nitrate Respiration Denitrification, mg N/kg day 60 Houghton Lake marsh, MI 50 Salt marsh, LA 40 30 Prairie pothole, ND 20 Unimpacted Everglades, FL Lake Apopka marsh, FL Belhaven, NC Crowley, LA 10 0 Talladega, AL Impacted Everglades, FL 0 500 y=-64+14 ln(x) R 2=0. 91 1, 000 1, 500 2, 000 2, 500 3, 000 3, 500 Dissolved organic C, mg/kg 10/3/2020 WBL 64
![Microbial Respiration [Everglades Soils] [mg kg-1 hour-1] Denitrifying/Sulfate reducing conditions 60 50 Denitrifying 40 Microbial Respiration [Everglades Soils] [mg kg-1 hour-1] Denitrifying/Sulfate reducing conditions 60 50 Denitrifying 40](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-65.jpg)
Microbial Respiration [Everglades Soils] [mg kg-1 hour-1] Denitrifying/Sulfate reducing conditions 60 50 Denitrifying 40 y = 0. 41 x + 1. 1 r 2 = 0. 89; n = 24 y = 0. 33 x + 1. 3 r 2 = 0. 88; n = 24 30 20 10/3/2020 Sulfate reducing 10 20 30 40 50 Aerobic [mg kg-1 hour-1] WBL 60 65
![[Everglades Soils] 10 [mg kg-1 hour-1] Methanogenic conditions Microbial Respiration 10/3/2020 8 CO 2 [Everglades Soils] 10 [mg kg-1 hour-1] Methanogenic conditions Microbial Respiration 10/3/2020 8 CO 2](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-66.jpg)
[Everglades Soils] 10 [mg kg-1 hour-1] Methanogenic conditions Microbial Respiration 10/3/2020 8 CO 2 y = 0. 13 x + 0. 3 r 2 = 0. 85, n = 24 6 4 2 y = 0. 08 x - 0. 2 CH 4 r 2 = 0. 70, n = 24 0 10 20 30 40 50 Aerobic, [mg kg-1 hour-1] WBL 60 66
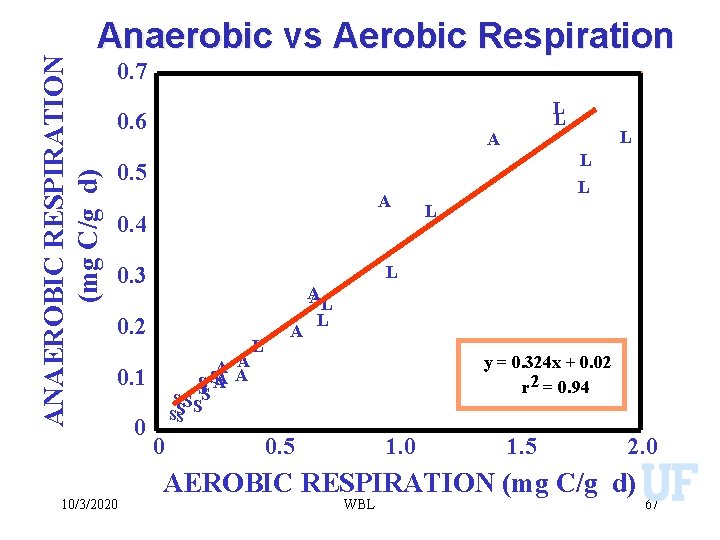
ANAEROBIC RESPIRATION (mg C/g d) Anaerobic vs Aerobic Respiration 0. 7 0. 6 L L A L 0. 5 A 0. 4 0. 2 A S SS A SS S 0. 1 10/3/2020 L L L 0. 3 0 L 0 A A L A A AL L y = 0. 324 x + 0. 02 r 2 = 0. 94 0. 5 1. 0 1. 5 2. 0 AEROBIC RESPIRATION (mg C/g d) WBL 67

Regulators 10/3/2020 WBL 68

Regulators of Organic Matter Decomposition v Substrate quality v carbon to nitrogen ratio or carbon to phosphorus ratio of the substrate v Temperature v Availability of electron acceptors v Microbial populations 10/3/2020 WBL 69
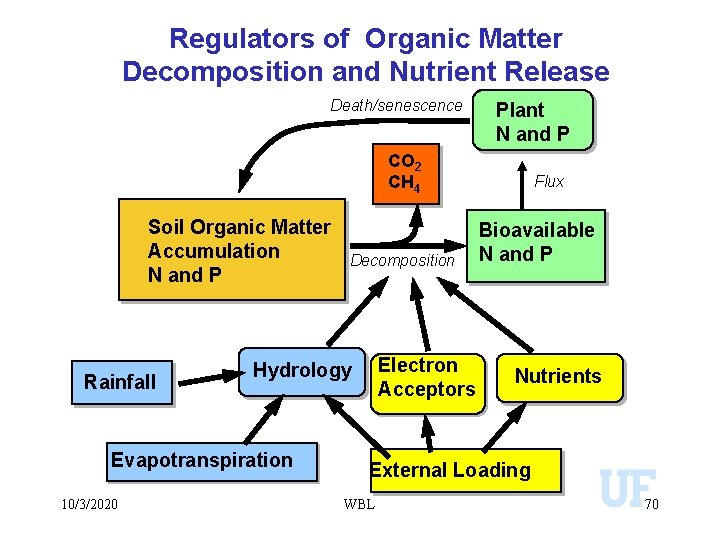
Regulators of Organic Matter Decomposition and Nutrient Release Death/senescence Plant N and P CO 2 CH 4 Soil Organic Matter Accumulation N and P Rainfall Electron Acceptors Hydrology Evapotranspiration 10/3/2020 Decomposition Flux Bioavailable N and P Nutrients External Loading WBL 70
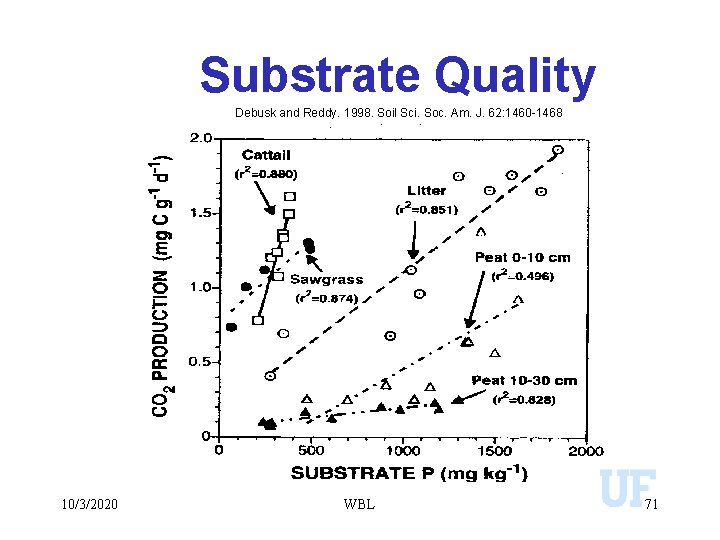
Substrate Quality Debusk and Reddy. 1998. Soil Sci. Soc. Am. J. 62: 1460 -1468 10/3/2020 WBL 71
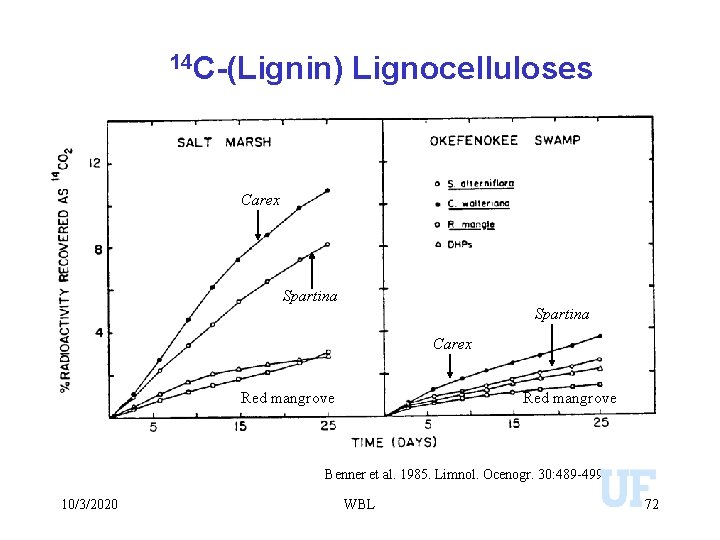
14 C-(Lignin) Lignocelluloses Carex Spartina Carex Red mangrove Benner et al. 1985. Limnol. Ocenogr. 30: 489 -499 10/3/2020 WBL 72
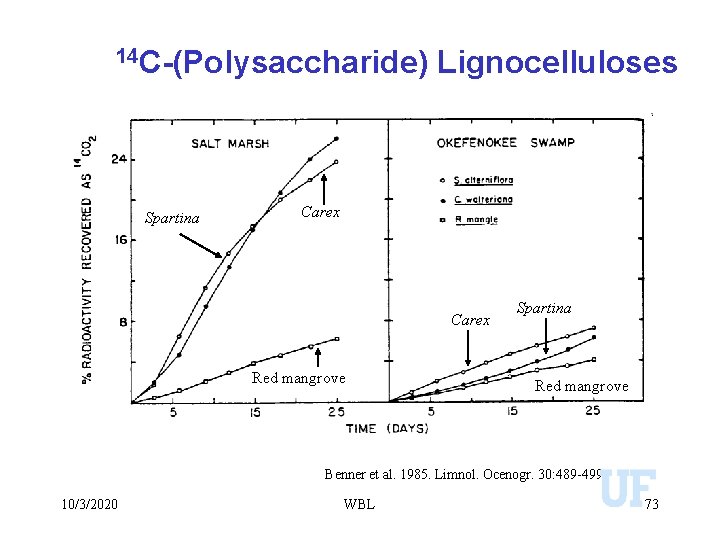
14 C-(Polysaccharide) Spartina Lignocelluloses Carex Red mangrove Spartina Red mangrove Benner et al. 1985. Limnol. Ocenogr. 30: 489 -499 10/3/2020 WBL 73
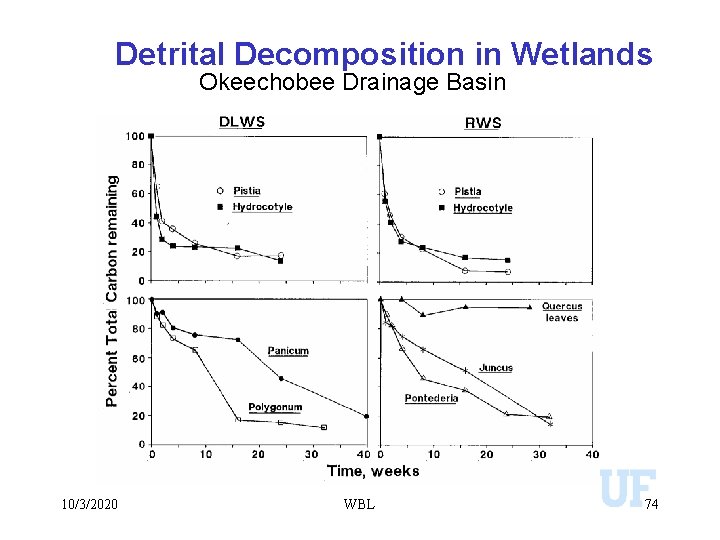
Detrital Decomposition in Wetlands Okeechobee Drainage Basin 10/3/2020 WBL 74
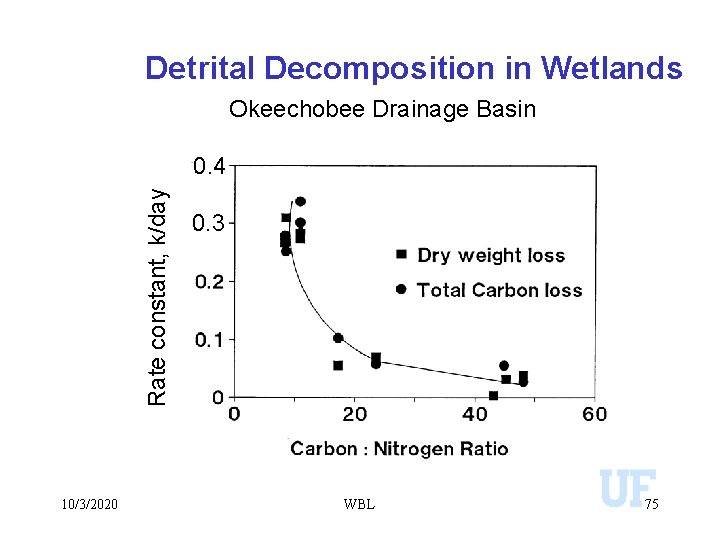
Detrital Decomposition in Wetlands Okeechobee Drainage Basin Rate constant, k/day 0. 4 10/3/2020 0. 3 WBL 75
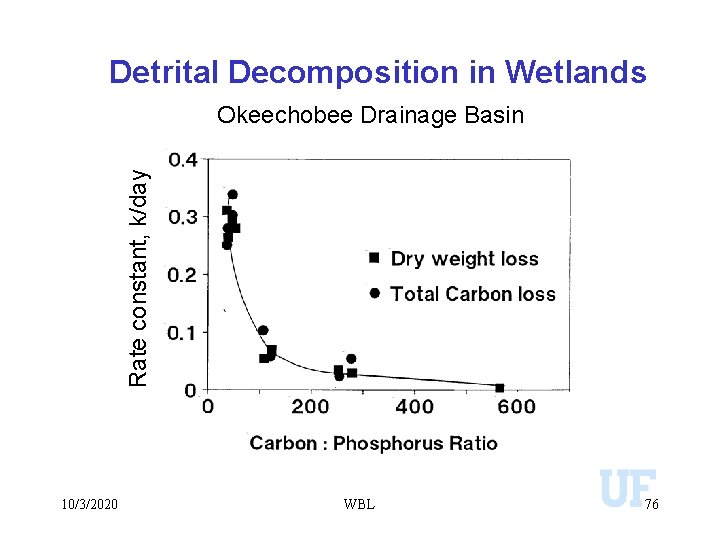
Detrital Decomposition in Wetlands Rate constant, k/day Okeechobee Drainage Basin 10/3/2020 WBL 76
![Relative Biodegradability of Substrates [Aerobic] [Time - half life, days] ê ê 10/3/2020 Sugars Relative Biodegradability of Substrates [Aerobic] [Time - half life, days] ê ê 10/3/2020 Sugars](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-77.jpg)
Relative Biodegradability of Substrates [Aerobic] [Time - half life, days] ê ê 10/3/2020 Sugars Hemicellulose Cellulose Lignin 0. 6 days 7 days 14 days 365 days WBL 77
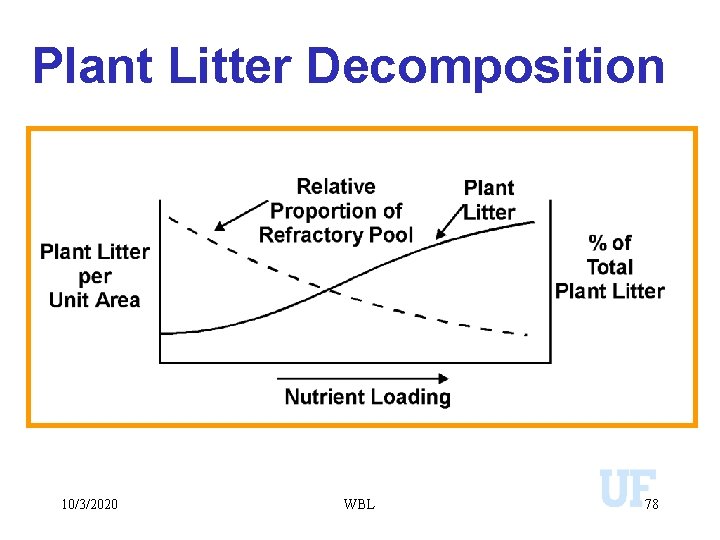
Plant Litter Decomposition 10/3/2020 WBL 78
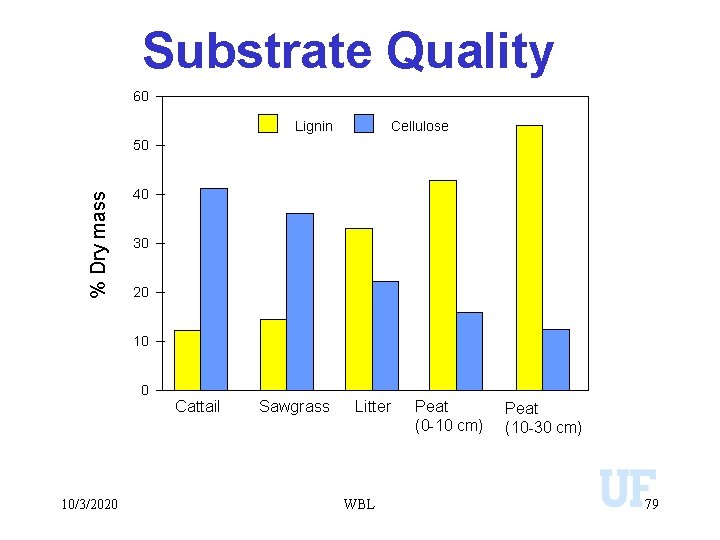
Substrate Quality 60 Lignin Cellulose % Dry mass 50 40 30 20 10/3/2020 Cattail Sawgrass Litter WBL Peat (0 -10 cm) Peat (10 -30 cm) 79
![A. Live Tissue [ LCI = 0. 14 -0. 17] LCI = [Lignin] [Lignin A. Live Tissue [ LCI = 0. 14 -0. 17] LCI = [Lignin] [Lignin](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-80.jpg)
A. Live Tissue [ LCI = 0. 14 -0. 17] LCI = [Lignin] [Lignin + Cellulose] B. Detritus attached to the plant [LCI = 0. 23 -0. 29] C. Detritus [LCI = 0. 6] Water 0 -10 cm soil [LCI = 0. 73] Soil 10 -30 cm soil [LCI = 0. 81] 10/3/2020 WBL 80
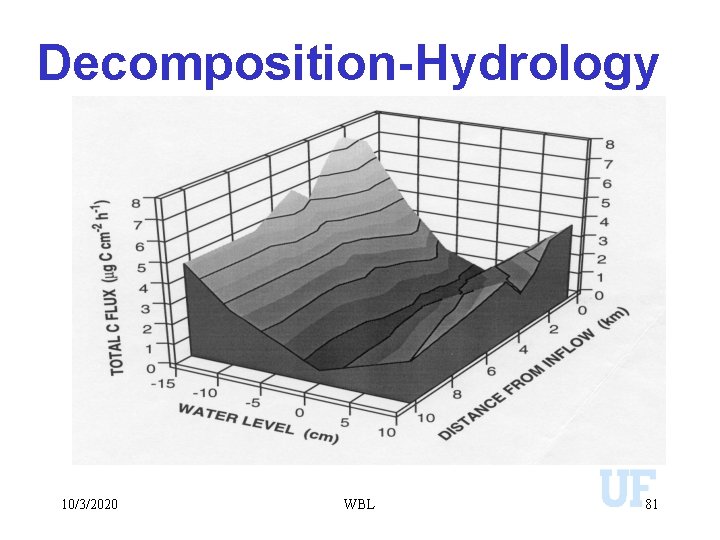
Decomposition-Hydrology 10/3/2020 WBL 81
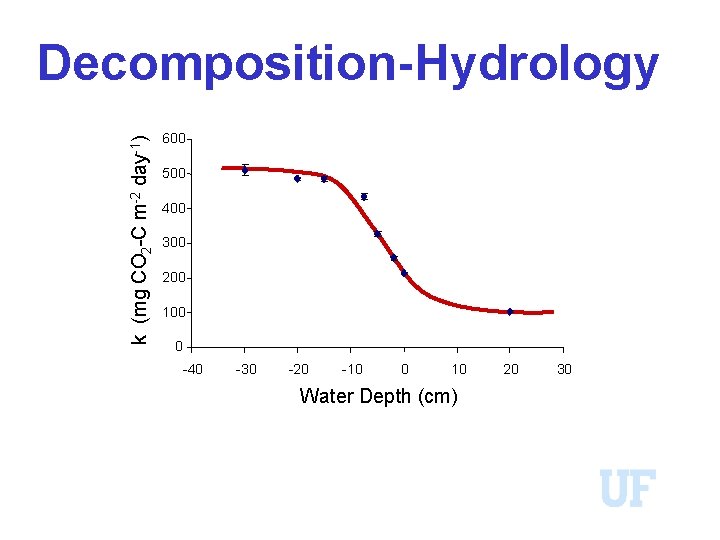
k (mg CO 2 -C m-2 day-1) Decomposition-Hydrology 600 500 400 300 200 100 0 -40 -30 -20 -10 0 10 Water Depth (cm) 20 30
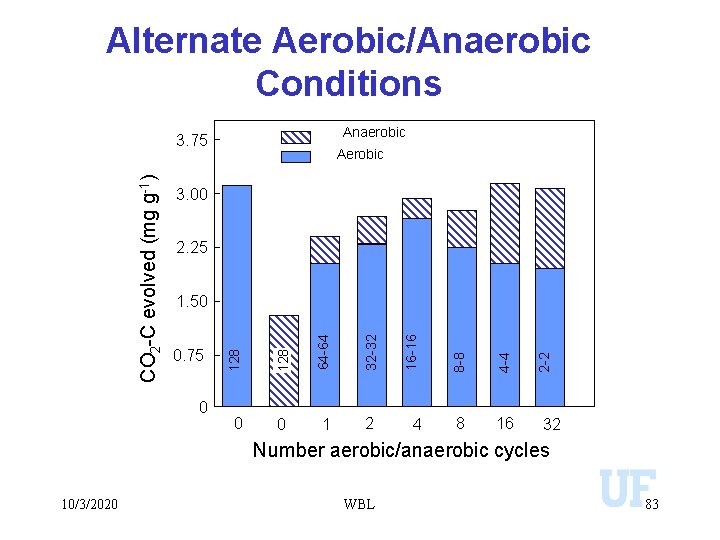
Alternate Aerobic/Anaerobic Conditions Anaerobic Aerobic 3. 00 2. 25 2 4 8 16 2 -2 1 4 -4 0 8 -8 0 16 -16 32 -32 0 64 -64 0. 75 128 1. 50 128 CO 2 -C evolved (mg g-1) 3. 75 32 Number aerobic/anaerobic cycles 10/3/2020 WBL 83
![Decomposition of Detrital Plant Tissue [Lake Apopka Marsh] k/day 0. 16 0. 12 Saggitaria Decomposition of Detrital Plant Tissue [Lake Apopka Marsh] k/day 0. 16 0. 12 Saggitaria](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-84.jpg)
Decomposition of Detrital Plant Tissue [Lake Apopka Marsh] k/day 0. 16 0. 12 Saggitaria 0. 08 0. 04 k/day 0 0. 16 0. 12 Typha 0. 08 Summer Winter 0. 04 0 10/3/2020 Decomposition N-release WBL P-release 84
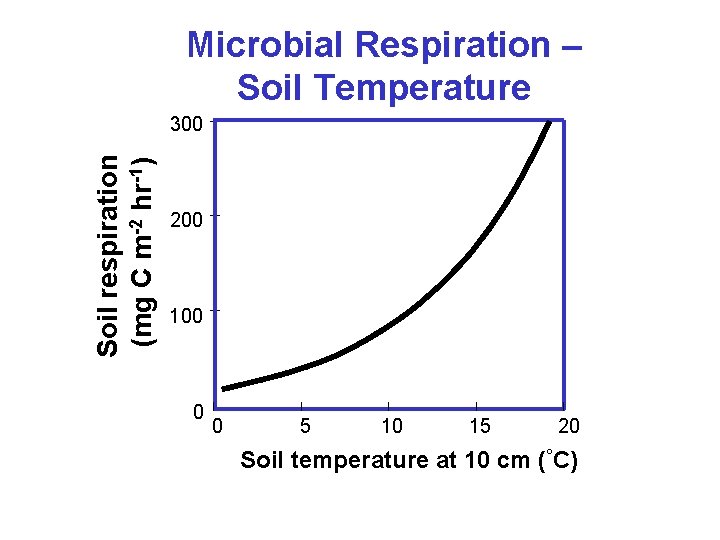
Microbial Respiration – Soil Temperature Soil respiration (mg C m-2 hr-1) 300 200 100 0 0 5 10 15 20 Soil temperature at 10 cm (°C)

Arrhenius Equation k = Ae - E / RT k = Reaction Rate Constant ; A = Arrhenius coefficient ; E = Activation Energy ; R = Gas constant ; and T = Temperature (K) k 1 = k 2 10/3/2020 WBL T 1 T 2 86
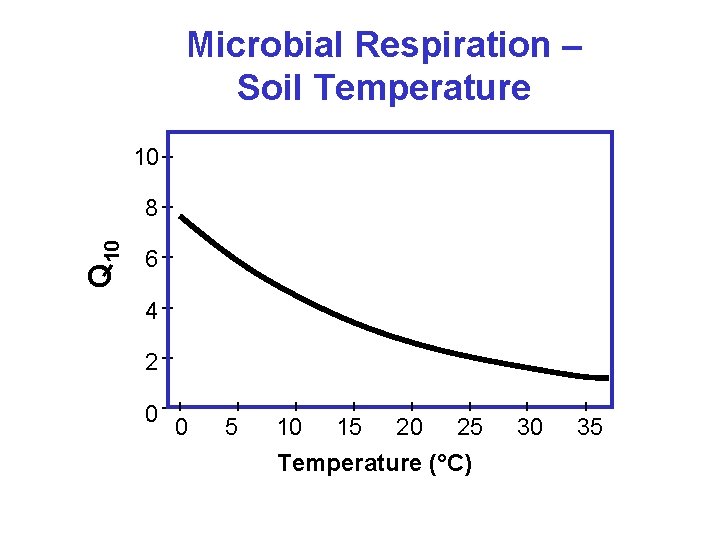
Microbial Respiration – Soil Temperature 10 Q 10 8 6 4 2 0 0 5 10 15 20 25 Temperature (°C) 30 35
![Microbial Activity CO 2 Production (mg C kg-1 h-1) [Site: Water Conservation 2 A] Microbial Activity CO 2 Production (mg C kg-1 h-1) [Site: Water Conservation 2 A]](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-88.jpg)
Microbial Activity CO 2 Production (mg C kg-1 h-1) [Site: Water Conservation 2 A] 250 Drained conditions y = 0. 07 x + 52 200 R 2 = 0. 58 150 100 Flooded conditions 50 y = 0. 06 x + 26 R 2 = 0. 72 0 0 500 1000 1500 2000 Total Phosphorus (mg P kg-1) 10/3/2020 WBL 88
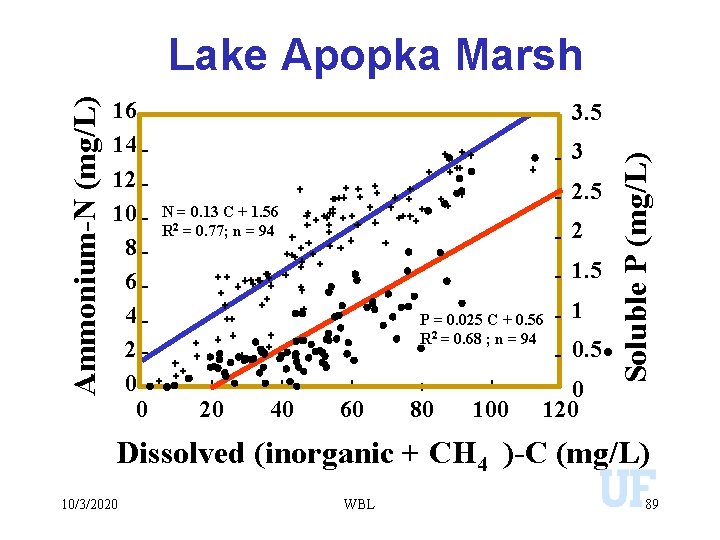
16 14 12 10 8 6 4 2 0 0 3. 5 3 2. 5 N = 0. 13 C + 1. 56 R 2 = 0. 77; n = 94 2 1. 5 P = 0. 025 C + 0. 56 R 2 = 0. 68 ; n = 94 20 40 60 80 100 1 0. 5 0 120 Soluble P (mg/L) Ammonium-N (mg/L) Lake Apopka Marsh Dissolved (inorganic + CH 4 )-C (mg/L) 10/3/2020 WBL 89

Soil Organic Matter 10/3/2020 WBL 90
![Plant Detritus Decomposition Detrital plant tissue or Carbon loading Residue [lignin] CO 2 Microbial Plant Detritus Decomposition Detrital plant tissue or Carbon loading Residue [lignin] CO 2 Microbial](http://slidetodoc.com/presentation_image/c24a0a1c6266e9b411848dcd6b85ed8f/image-91.jpg)
Plant Detritus Decomposition Detrital plant tissue or Carbon loading Residue [lignin] CO 2 Microbial biomass HUMUS Humus: Total of the organic compounds in soil exclusive of undecayed plant and animal tissues, their “partial decomposition” products and the soil microbial biomass 10/3/2020 WBL 91
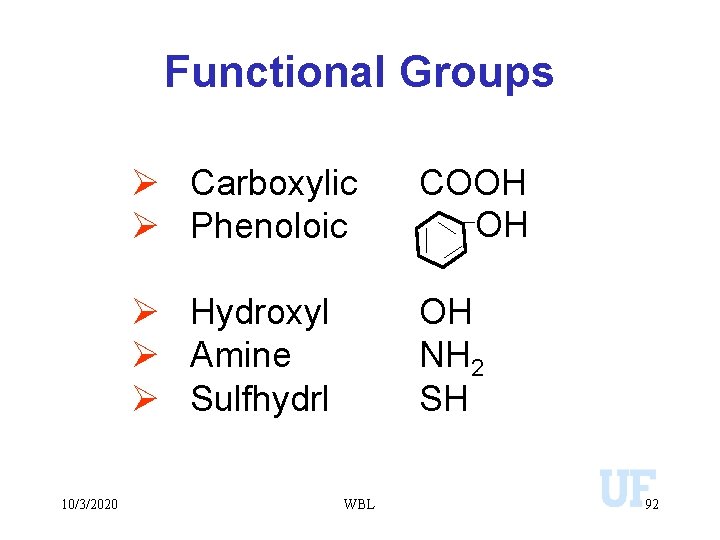
Functional Groups 10/3/2020 Ø Carboxylic Ø Phenoloic COOH OH Ø Hydroxyl Ø Amine Ø Sulfhydrl OH NH 2 SH WBL 92
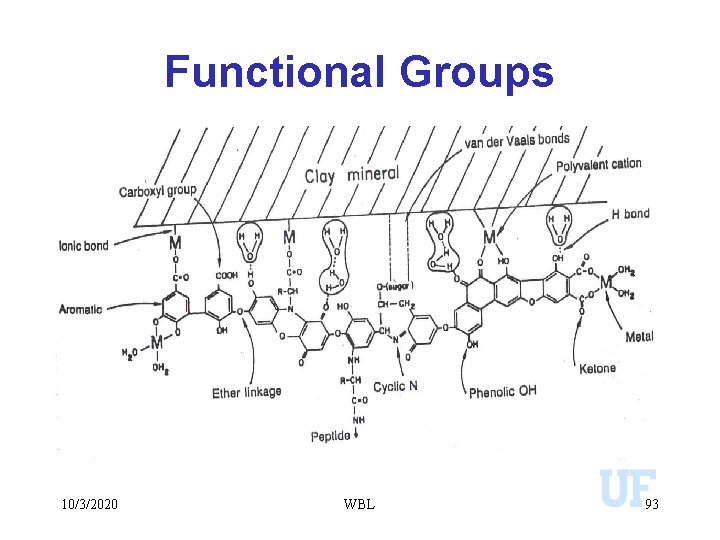
Functional Groups 10/3/2020 WBL 93

Functions of Organic Matter • • • Source of nutrients for plant growth. Source of energy for soil microorganisms. Source of exchange capacity for cations. Provides long-term storage for nutrients. Strong adsorbing agent for toxic organic compounds. • Complexation of metals. 10/3/2020 WBL 94
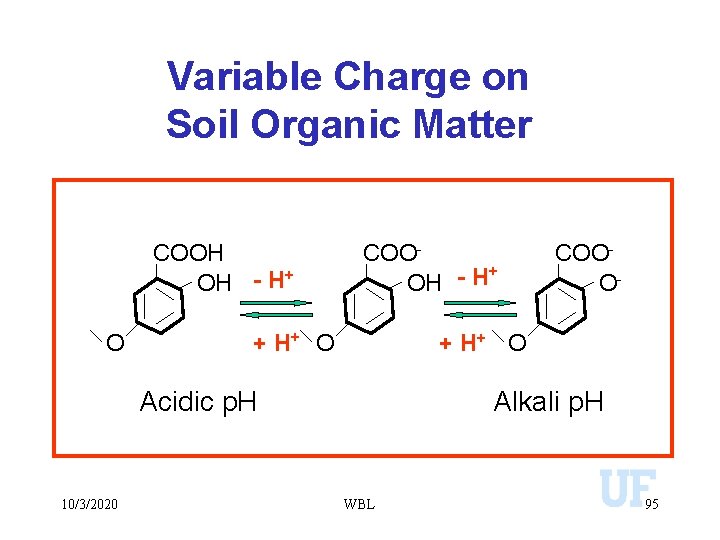
Variable Charge on Soil Organic Matter COOH OH - H+ O COO+ OH - H + H+ O Acidic p. H 10/3/2020 COOO- Alkali p. H WBL 95

Complexation with Metals • Metal ions that would convert to insoluble precipitates are maintained in solution. • Influences the bioavailability of metals. • Some organic complexes with metals may low solubility. . complexation with humic acids. • Inhibits enzyme activity. • Plays a significant role in transporting metals from one ecosystem to another. 10/3/2020 WBL 96
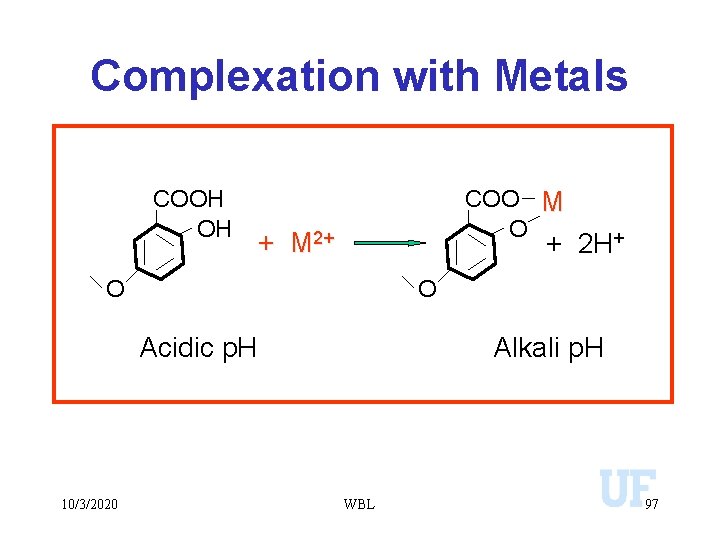
Complexation with Metals COOH OH COO M O + M 2+ + 2 H+ O O Acidic p. H 10/3/2020 Alkali p. H WBL 97

Greenhouse Gases 10/3/2020 WBL 98
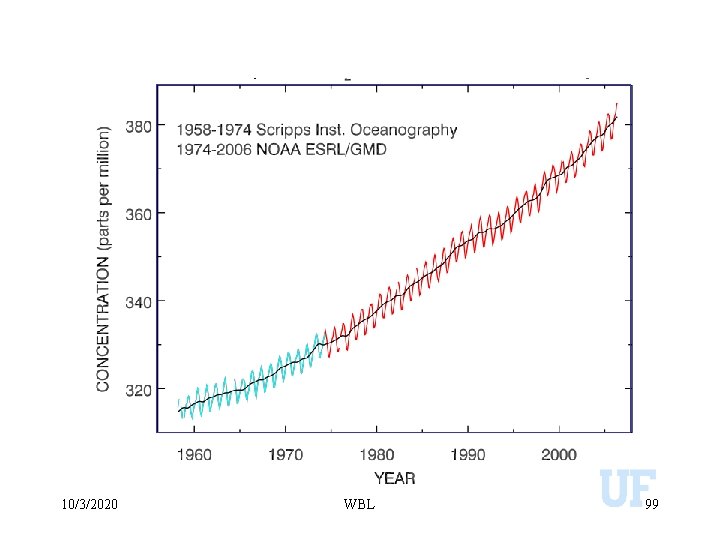
10/3/2020 WBL 99
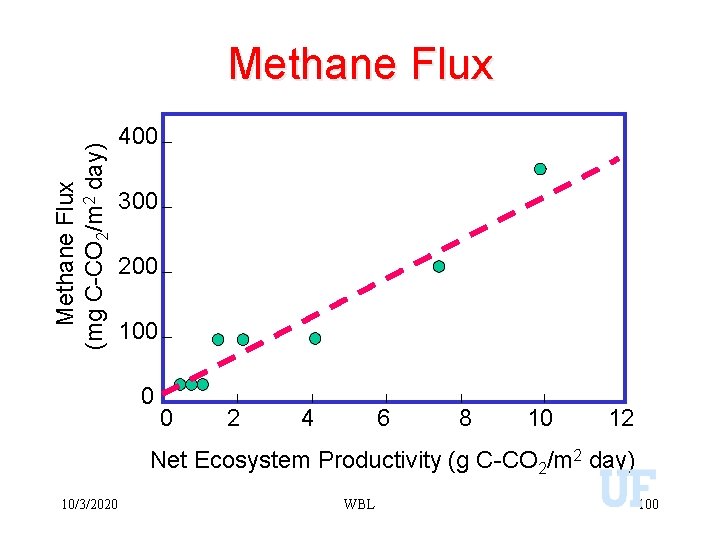
Methane Flux (mg C-CO 2/m 2 day) Methane Flux 400 300 200 100 0 0 2 4 6 8 10 12 Net Ecosystem Productivity (g C-CO 2/m 2 day) 10/3/2020 WBL 100
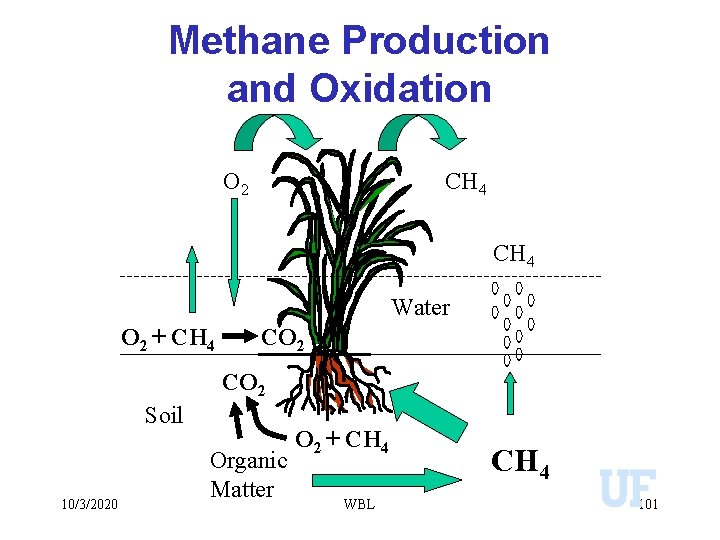
Methane Production and Oxidation O 2 CH 4 Water O 2 + CH 4 CO 2 Soil 10/3/2020 Organic Matter O 2 + CH 4 WBL CH 4 101
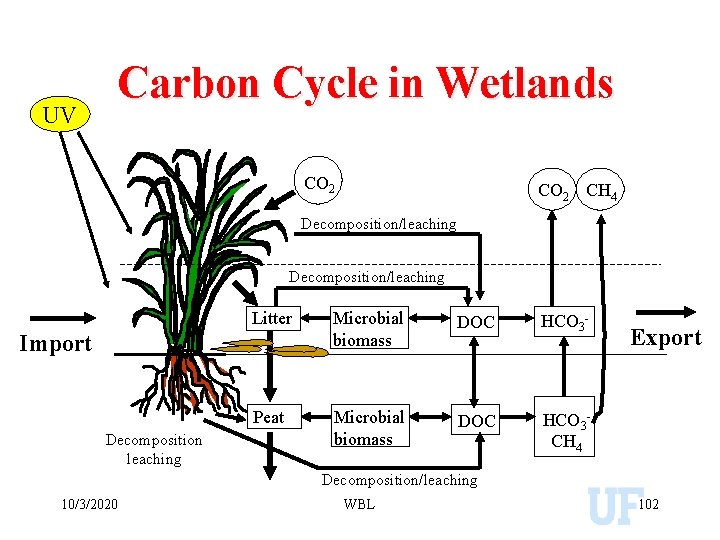
UV Carbon Cycle in Wetlands CO 2 CH 4 Decomposition/leaching Litter Microbial biomass DOC HCO 3 - Peat Microbial biomass DOC HCO 3 CH 4 Import Decomposition leaching Export Decomposition/leaching 10/3/2020 WBL 102

Carbon Cycling Processes Summary Carbon is important for living systems because it can exist in a variety of oxidation states (-4, 0, +4) and serves as a source of electrons for microbial processes. v Most decomposition of organic matter is driven by oxygen, but less efficient electron acceptors are used in anaerobic processes v Humic substances are divided into three major groups: Fulvic acid (acid and base soluble); Humic acid (acid insoluble and base soluble); Humin (acid and base insoluble) v Detrital matter is broken down into complex polymers (cellulose, proteins, lipids, lignin). Enzymes break these polymers into simple monomers (sugars, amino acids, fatty acids) v Organic mater is a source (short term and long term storage) of nutrients for plants and soil microbes v Enzymatic hydrolysis is the rate limiting step in SOM decomposition v 10/3/2020 WBL 103

Carbon Cycling Processes Summary Decomposition is regulated by substrate quality, electron acceptors (who, how many), limiting nutrients, and temperature v Functions of Organic Matter: Source of nutrients for plant growth; source of energy for soil microorganisms; provides long-term storage for nutrients; strong adsorbing agent for toxic organic compounds; complexation of metals v Aerobic decomposition results in the production of oxidized species (CO 2. H 2 O, NO 3 -, SO 42 -, and Mn 4+ and Fe 3+ oxides), while the anaerobic decomposition results in the production of reduced species (H 2, fatty acids, NH 4+, N 2 O, sulfides, CH 4, Fe 2+ and Mn 2+) v Wetlands contain approximately 15 to 22% of the terrestrial carbon and one of the major contributor to the global methane flux , which accounts for approximately 20 to 25% of global methane to atmosphere v 10/3/2020 WBL 104

Dissolved Organic Matter 10/3/2020 WBL 105

10/3/2020 http: //wetlands. ifas. ufl. edu http: //soils. ifas. ufl. edu WBL 106
- Slides: 106