Institut fr Physikalische Chemie Elektrochemie Lehrgebiet A STRUCTURE

Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A STRUCTURE OF THE BENZENE DIMER GOVERNED BY DYNAMICS. MELANIE SCHNELL, Center for Free-Electron Laser Science, Hamburg, Germany UNDINE ERLEKAM, GERT V. HELDEN, GERARD MEIJER, Fritz-Haber-Institut, Berlin, Germany PHILIP R. BUNKER, National Research Council of Canada, Ottawa, Canada JENS-UWE GRABOW, Gottfried-Wilhelm-Leibniz-Universtät, Hannover, Germany AD VAN DER AVOIRD, Radboud University, Nijmegen, The Netherlands
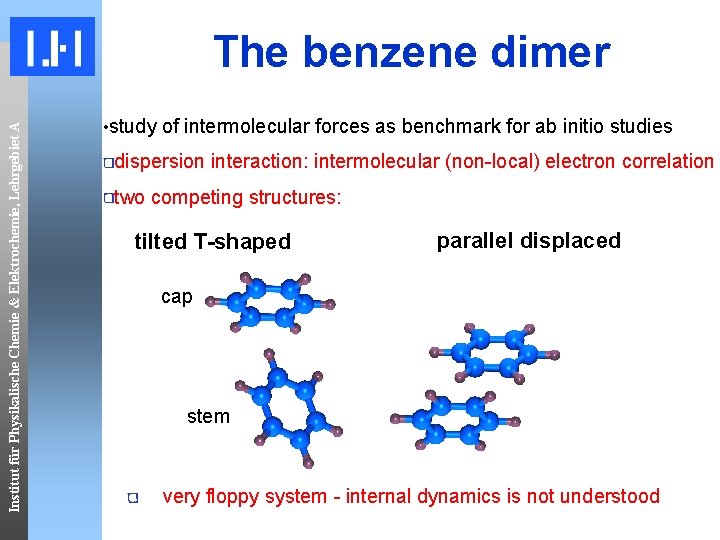
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A The benzene dimer • study of intermolecular forces as benchmark for ab initio studies dispersion interaction: intermolecular (non-local) electron correlation two competing structures: tilted T-shaped parallel displaced cap stem very floppy system - internal dynamics is not understood
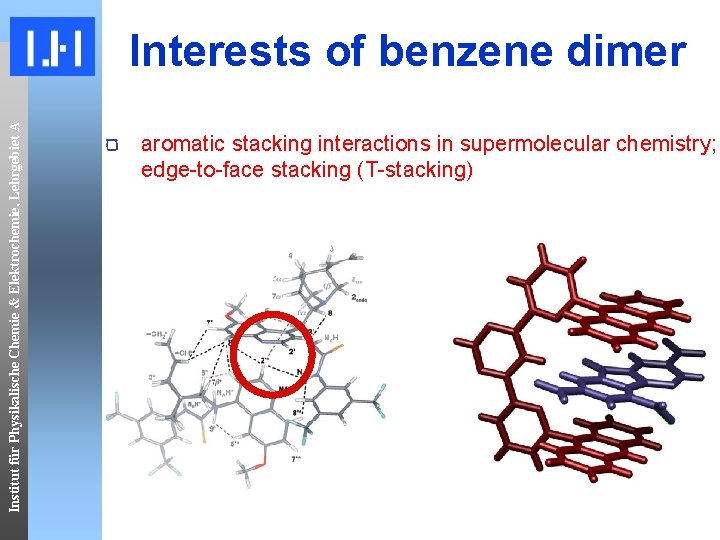
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Interests of benzene dimer aromatic stacking interactions in supermolecular chemistry; edge-to-face stacking (T-stacking)

What is known so far. . . Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A J. Chem. Phys. 63 (1975) 1419 J. Chem. Phys. 97 (1992) 2189 J. Chem. Phys. 98 (1993) 4294 symmetric-top spectrum with complex internal rotation
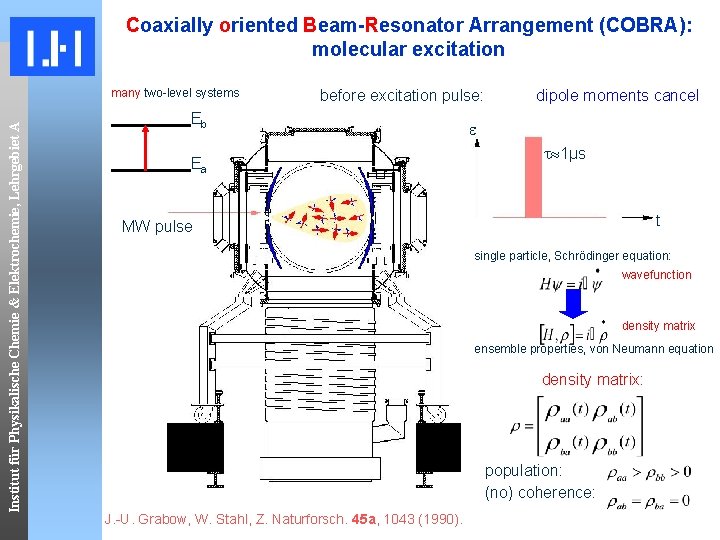
Coaxially oriented Beam-Resonator Arrangement (COBRA): molecular excitation Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A many two-level systems before excitation pulse: Eb Ea dipole moments cancel 1μs t MW pulse single particle, Schrödinger equation: wavefunction density matrix ensemble properties, von Neumann equation density matrix: population: (no) coherence: J. -U. Grabow, W. Stahl, Z. Naturforsch. 45 a, 1043 (1990).
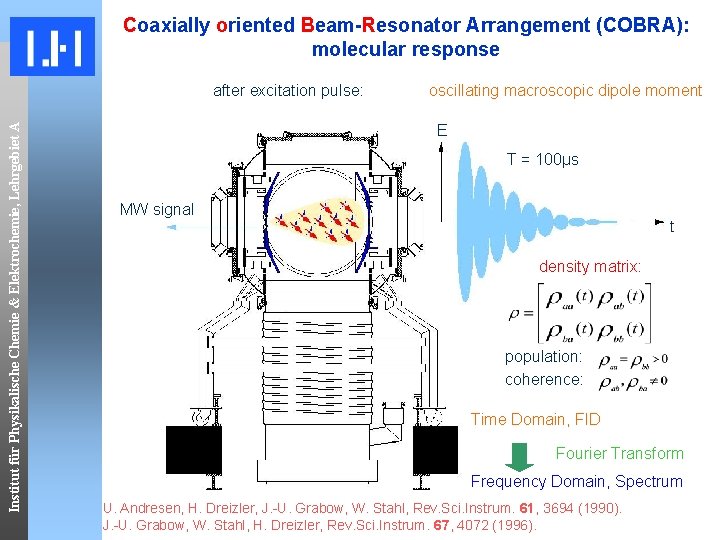
Coaxially oriented Beam-Resonator Arrangement (COBRA): molecular response Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A after excitation pulse: oscillating macroscopic dipole moment E T = 100μs MW signal t density matrix: population: coherence: Time Domain, FID Fourier Transform Frequency Domain, Spectrum U. Andresen, H. Dreizler, J. -U. Grabow, W. Stahl, Rev. Sci. Instrum. 61, 3694 (1990). J. -U. Grabow, W. Stahl, H. Dreizler, Rev. Sci. Instrum. 67, 4072 (1996).
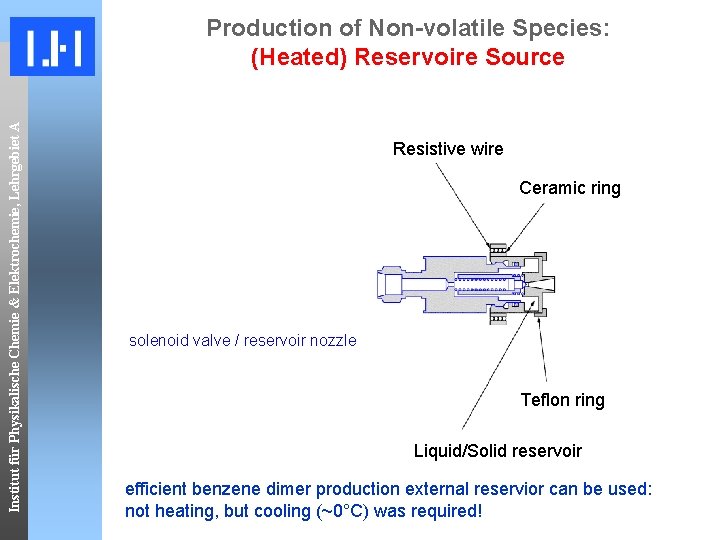
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Production of Non-volatile Species: (Heated) Reservoire Source Resistive wire Ceramic ring solenoid valve / reservoir nozzle Teflon ring Liquid/Solid reservoir efficient benzene dimer production external reservior can be used: not heating, but cooling (~0°C) was required!
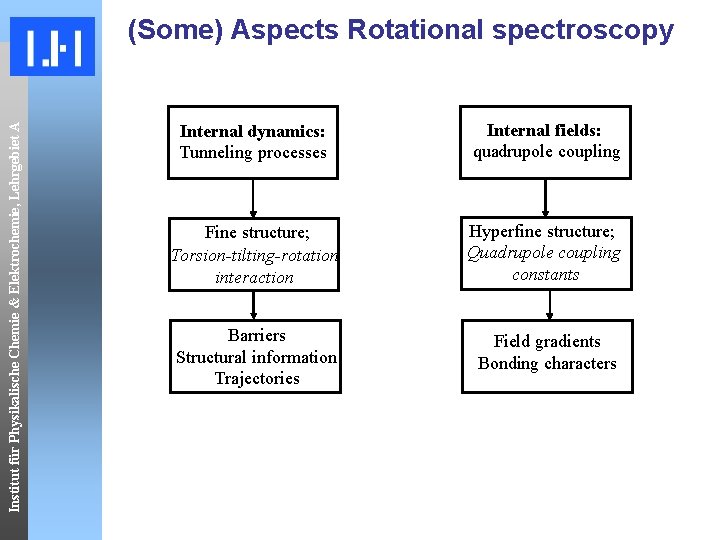
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A (Some) Aspects Rotational spectroscopy Internal dynamics: Tunneling processes Internal fields: quadrupole coupling Fine structure; Torsion-tilting-rotation interaction Hyperfine structure; Quadrupole coupling constants Barriers Structural information Trajectories Field gradients Bonding characters
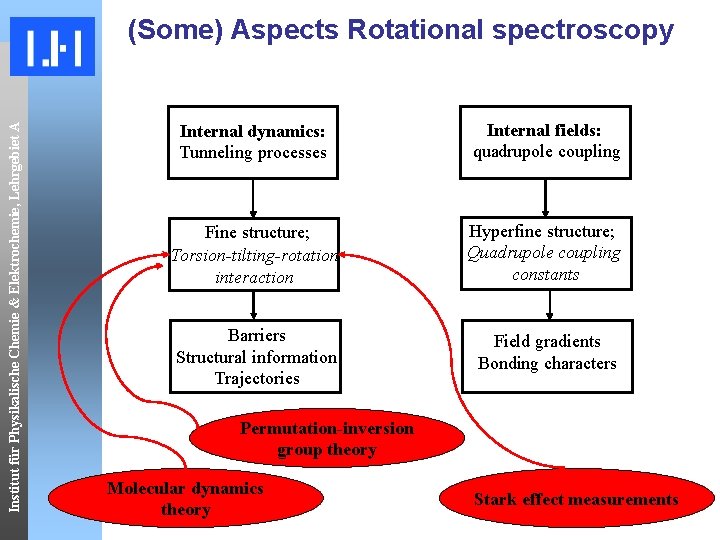
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A (Some) Aspects Rotational spectroscopy Internal dynamics: Tunneling processes Internal fields: quadrupole coupling Fine structure; Torsion-tilting-rotation interaction Hyperfine structure; Quadrupole coupling constants Barriers Structural information Trajectories Field gradients Bonding characters Permutation-inversion group theory Molecular dynamics theory Stark effect measurements
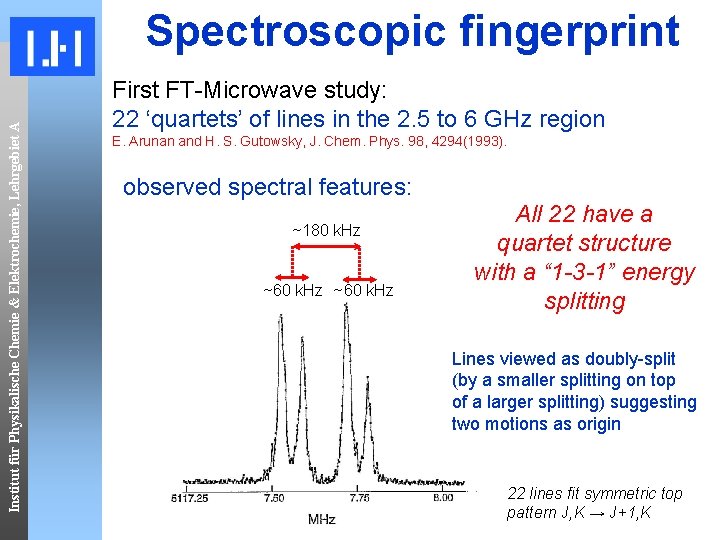
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Spectroscopic fingerprint First FT-Microwave study: 22 ‘quartets’ of lines in the 2. 5 to 6 GHz region E. Arunan and H. S. Gutowsky, J. Chem. Phys. 98, 4294(1993). observed spectral features: ~180 k. Hz ~60 k. Hz All 22 have a quartet structure with a “ 1 -3 -1” energy splitting Lines viewed as doubly-split (by a smaller splitting on top of a larger splitting) suggesting two motions as origin 22 lines fit symmetric top pattern J, K → J+1, K
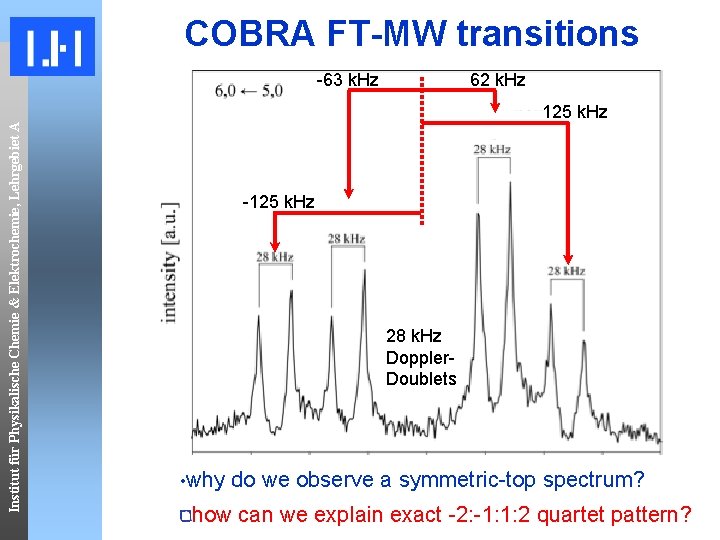
COBRA FT-MW transitions Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A -63 k. Hz 62 k. Hz 125 k. Hz -125 k. Hz 28 k. Hz Doppler. Doublets • why do we observe a symmetric-top spectrum? 1 exact -2: -1: 1: 2 2 1 pattern? how can we explain quartet
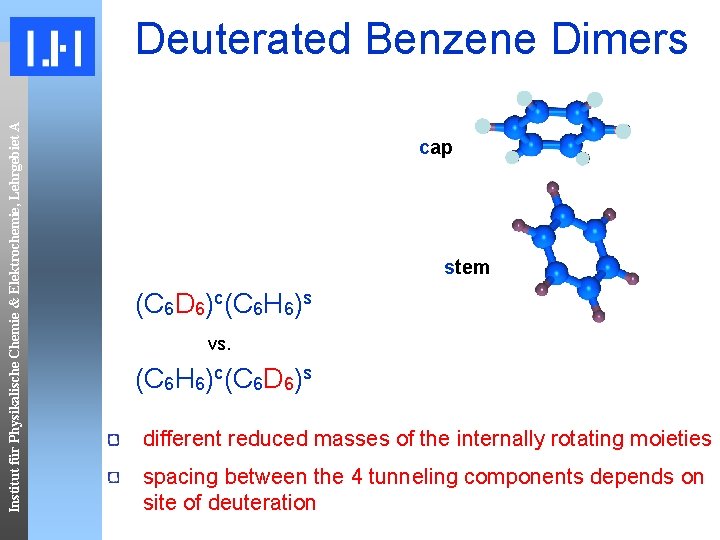
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Deuterated Benzene Dimers cap stem (C 6 D 6)c(C 6 H 6)s vs. (C 6 H 6)c(C 6 D 6)s different reduced masses of the internally rotating moieties spacing between the 4 tunneling components depends on site of deuteration
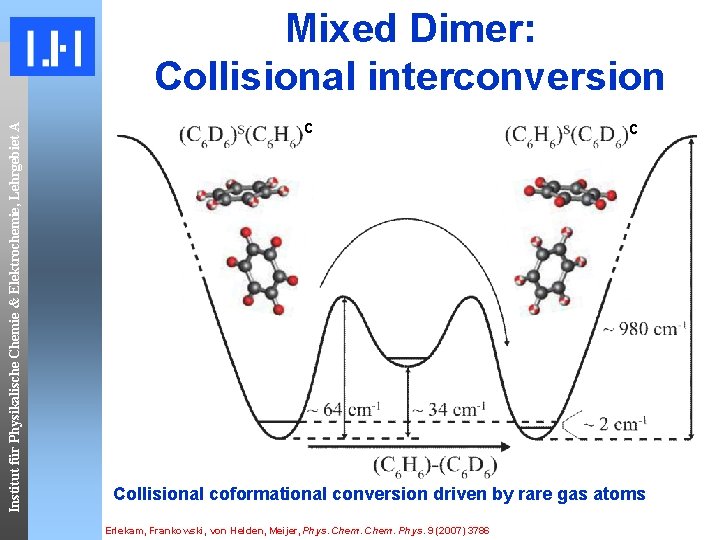
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Mixed Dimer: Collisional interconversion c c Collisional coformational conversion driven by rare gas atoms Erlekam, Frankowski, von Helden, Meijer, Phys. Chem. Phys. 9 (2007) 3786
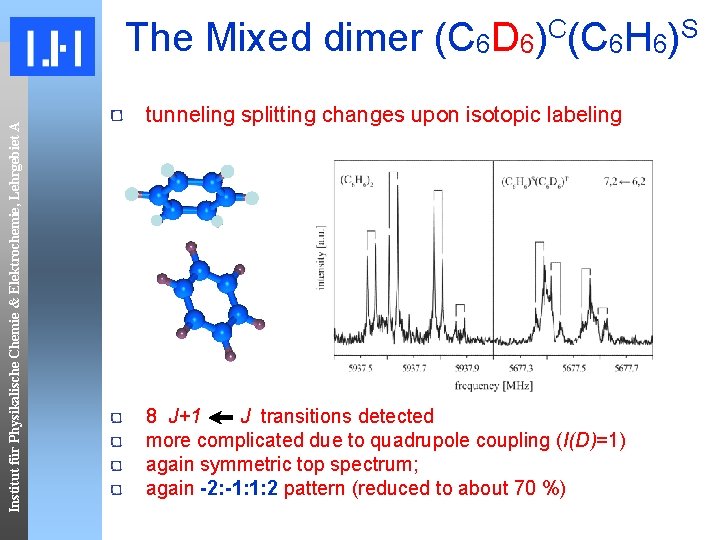
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A The Mixed dimer (C 6 D 6)C(C 6 H 6)S tunneling splitting changes upon isotopic labeling 8 J+1 J transitions detected more complicated due to quadrupole coupling (I(D)=1) again symmetric top spectrum; again -2: -1: 1: 2 pattern (reduced to about 70 %)
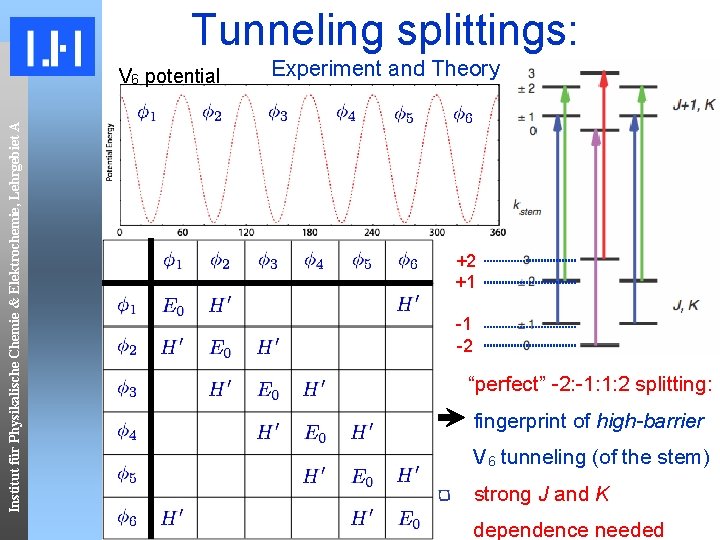
Tunneling splittings: Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A V 6 potential Experiment and Theory +2 +1 -1 -2 “perfect” -2: -1: 1: 2 splitting: fingerprint of high-barrier V 6 tunneling (of the stem) strong J and K dependence needed

Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Symmetric-top character 6 D calculations show: cap is nearly freely rotating about its C 6 axis group theory (G 576): all motions, but insignificant cap turnover and cap/stem exchange: accidental degeneracy for Kcap=3 n 1 K++K-, G 7++G 7 -, G 1++G 1 - states behave as symmetric top similar findings for other benzene complexes with low barriers, such as benzene-CO Reduced-dimensionality approach and group theory: M. Schnell, U. Erlekam, P. R. Bunker, G. von Helden, J. -U. Grabow, G Meijer, A. van der Avoird, Phys. Chem. Phys. 15 (2013) 10207. 6 D calculations: A. van der Avoird, R. Podeszwa, K. Szalewicz, C. Leforestier, R. van Harrevelt, P. R. Bunker, M. Schnell, G. von Helden, G. Meijer, Phys. Chem. Phys. 12 (2010) 8219. Benzene-CO: T. Brupbacher, A. Bauder, J. Chem. Phys. 99 (1993) 9394
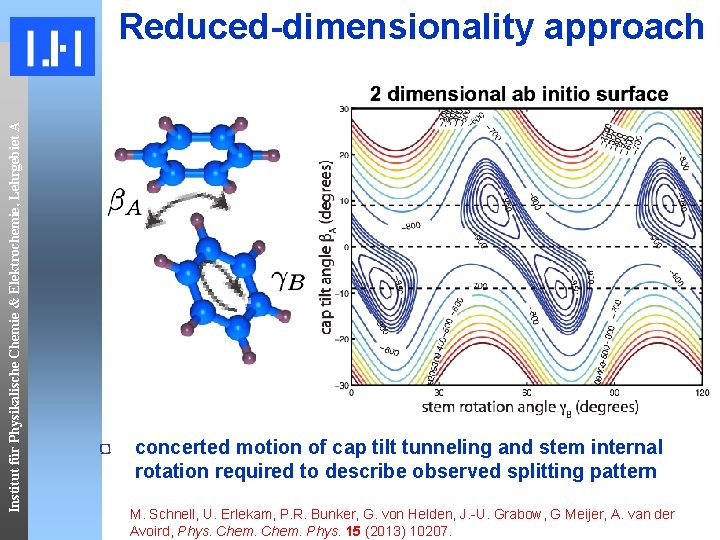
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Reduced-dimensionality approach concerted motion of cap tilt tunneling and stem internal rotation required to describe observed splitting pattern M. Schnell, U. Erlekam, P. R. Bunker, G. von Helden, J. -U. Grabow, G Meijer, A. van der Avoird, Phys. Chem. Phys. 15 (2013) 10207.
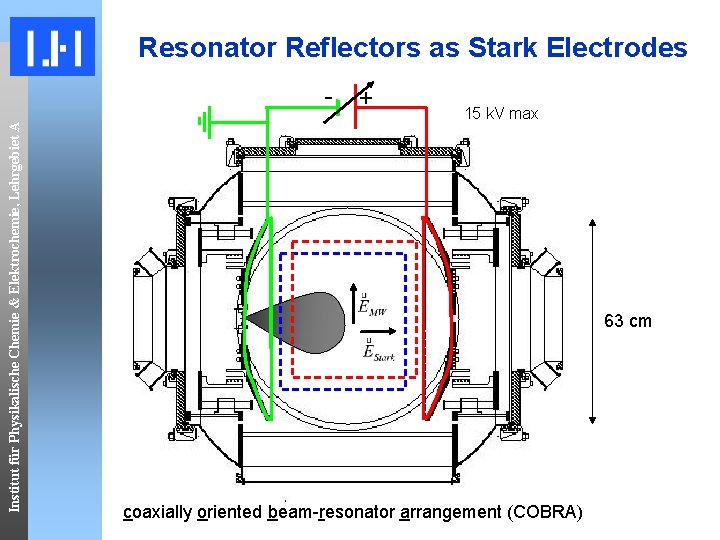
Resonator Reflectors as Stark Electrodes Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A - + 15 k. V max 63 cm coaxially oriented beam-resonator arrangement (COBRA)

Circular Electrodes for Field Homogenization coaxially aligned electrodes for Stark-effect applied in resonators (CAESAR) Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A decreasing potential ground HV M-selection rule: M = 1 M. Schnell, D. Banser, J. -U. Grabow, Rev. Sci. Instrum. 75, 2111(2004).
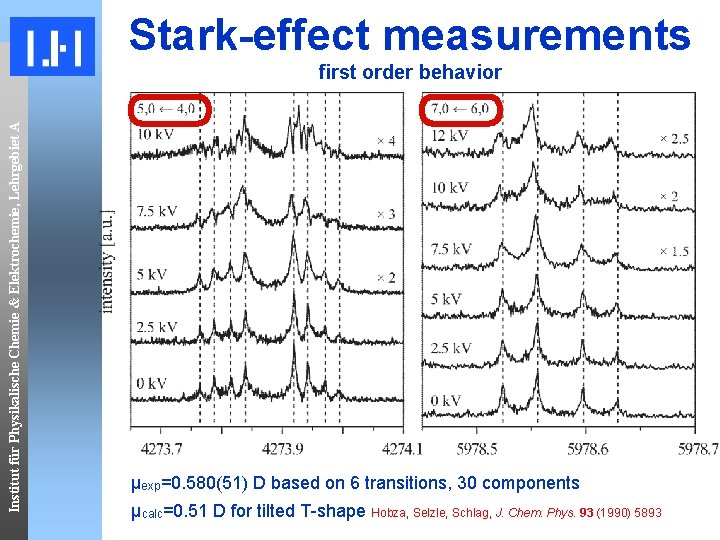
Stark-effect measurements Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A first order behavior µexp=0. 580(51) D based on 6 transitions, 30 components µcalc=0. 51 D for tilted T-shape Hobza, Selzle, Schlag, J. Chem. Phys. 93 (1990) 5893

Stark effect of torsional species Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Condition for linear Stark effect: J. K. G. Watson, J. Mol. Spectr. 50, 281 (1974). The anti-symmetric square [ 2]antisym of the torsional symmetry species needs to contain the symmetry species ( f) of the space fixed components of the dipole moment operator f Experimental results show: Cap torsion, cap tilting, and stem torsion are feasible: MS group G 144 = Gcap Gstem = C 6 v(M) D 6(M) (G 144): cap stem Symmetry of Kcap=3 n 1 levels in C 6 v(M) is: (Kcap=3 n 1) = E 1 or E 2 Symmetry of Kcap=1, 2, . . levels in C 6 v(M) is: (Kcap=1, 2, . . ) = A 1+A 2 or B 1+B 2 [ 2(E 1)]antisym = [ 2(E 2)]antisym = [ 2(A 1+A 2)]antisym =[ 2(B 1+B 2)]antisym = A 2 Symmetry of f in G 144 is: ( f) = A 2 A 1 first order Stark effect in levels having symmetry: E 1 stem or E 2 stem levels with Kcap=3 n 1 but not Kcap=0 will have a first order Stark effect M. Schnell, P. R. Bunker, G. von Helden, J. -U. Grabow, G Meijer, A. van der Avoird, to be published.
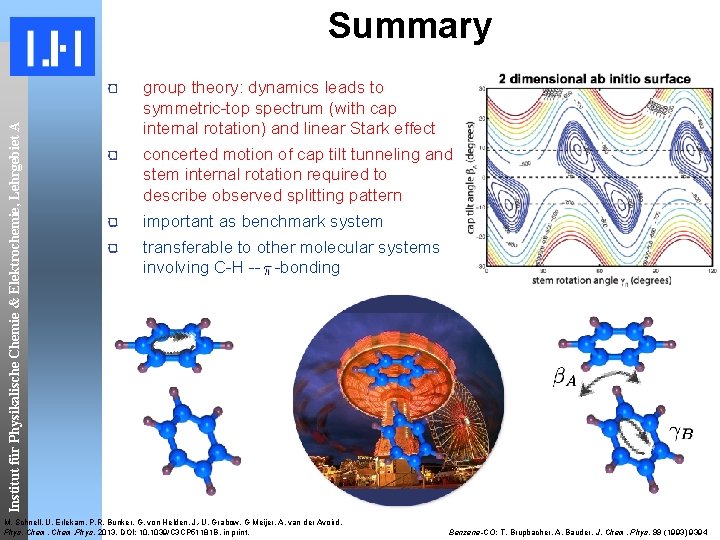
Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Summary group theory: dynamics leads to symmetric-top spectrum (with cap internal rotation) and linear Stark effect concerted motion of cap tilt tunneling and stem internal rotation required to describe observed splitting pattern important as benchmark system transferable to other molecular systems involving C-H -- -bonding M. Schnell, U. Erlekam, P. R. Bunker, G. von Helden, J. -U. Grabow, G Meijer, A. van der Avoird, Phys. Chem. Phys. 2013, DOI: 10. 1039/C 3 CP 51181 B, in print. Benzene-CO: T. Brupbacher, A. Bauder, J. Chem. Phys. 99 (1993) 9394

Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Intramolecular dynamics The benzene dimer: Structure governed by dynamics Angew. Chem. Int. Ed. 52 (2013) 5180 -5183.

Institut für Physikalische Chemie & Elektrochemie, Lehrgebiet A Thanks! Wolfgang Rogge, electronic shop of the PCI Mechanical shop of the PCI Fonds der Chemischen Industrie Deutsche Forschungsgemeinschaft Land Niedersachsen Deutscher Akademischer Austauschdienst
- Slides: 24