Insights into vertebrate development merging bioimaging and computational

Insights into vertebrate development: merging bioimaging and computational modeling Paul Kulesa Stowers Institute for Medical Research

Insights into vertebrate development: merging bioimaging and computational modeling Paul Kulesa Stowers Institute for Medical Research

We have developed culture and imaging techniques to analyze avian development chick alligator duck quail From www. saviorfare. wa & B. S. Arnold et al. , 2001
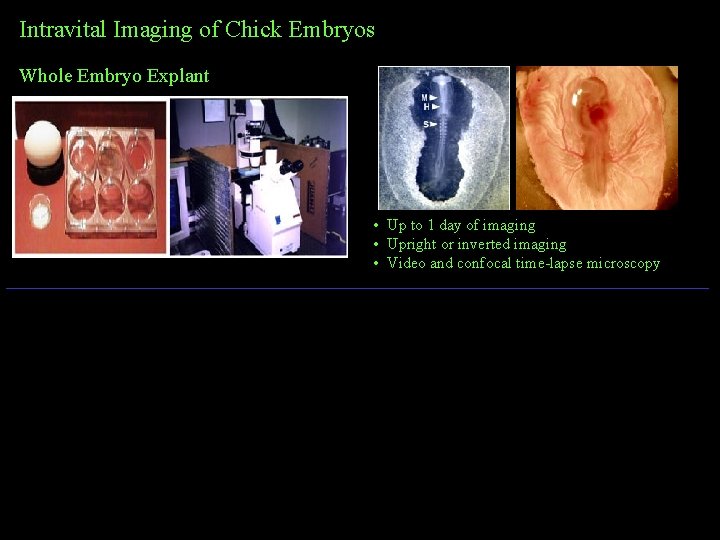
Intravital Imaging of Chick Embryos Whole Embryo Explant • Up to 1 day of imaging • Upright or inverted imaging • Video and confocal time-lapse microscopy
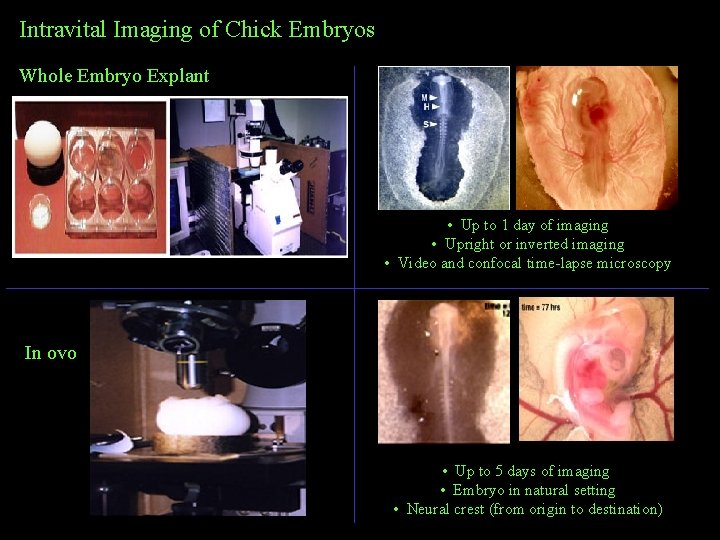
Intravital Imaging of Chick Embryos Whole Embryo Explant • Up to 1 day of imaging • Upright or inverted imaging • Video and confocal time-lapse microscopy In ovo • Up to 5 days of imaging • Embryo in natural setting • Neural crest (from origin to destination)
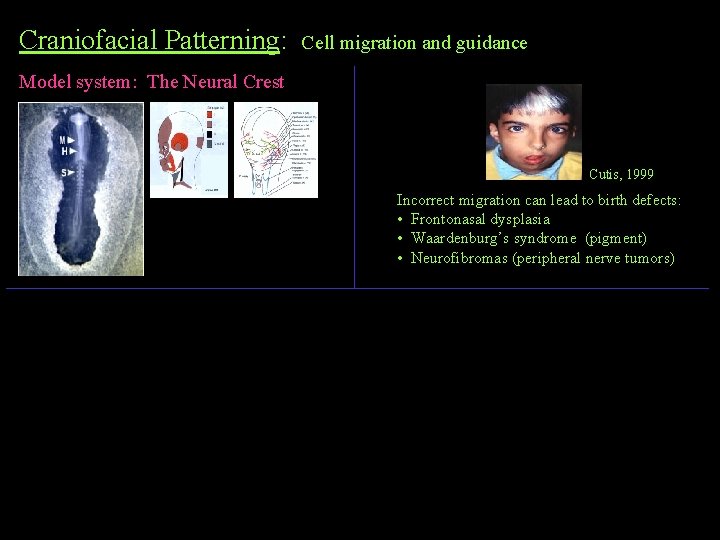
Craniofacial Patterning: Cell migration and guidance Model system: The Neural Crest Cutis, 1999 Incorrect migration can lead to birth defects: • Frontonasal dysplasia • Waardenburg’s syndrome (pigment) • Neurofibromas (peripheral nerve tumors)
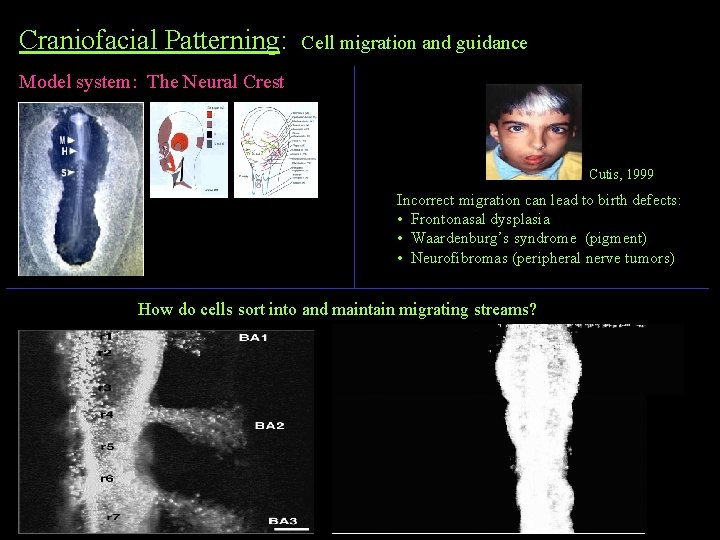
Craniofacial Patterning: Cell migration and guidance Model system: The Neural Crest Cutis, 1999 Incorrect migration can lead to birth defects: • Frontonasal dysplasia • Waardenburg’s syndrome (pigment) • Neurofibromas (peripheral nerve tumors) How do cells sort into and maintain migrating streams?
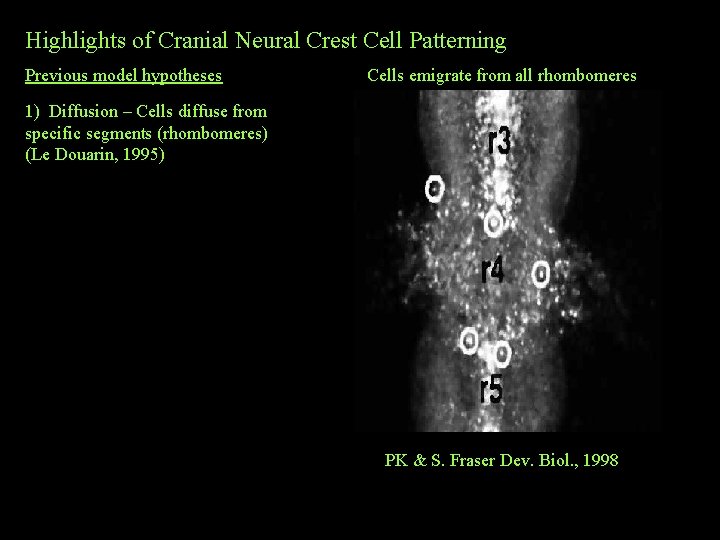
Highlights of Cranial Neural Crest Cell Patterning Previous model hypotheses Cells emigrate from all rhombomeres 1) Diffusion – Cells diffuse from specific segments (rhombomeres) (Le Douarin, 1995) PK & S. Fraser Dev. Biol. , 1998
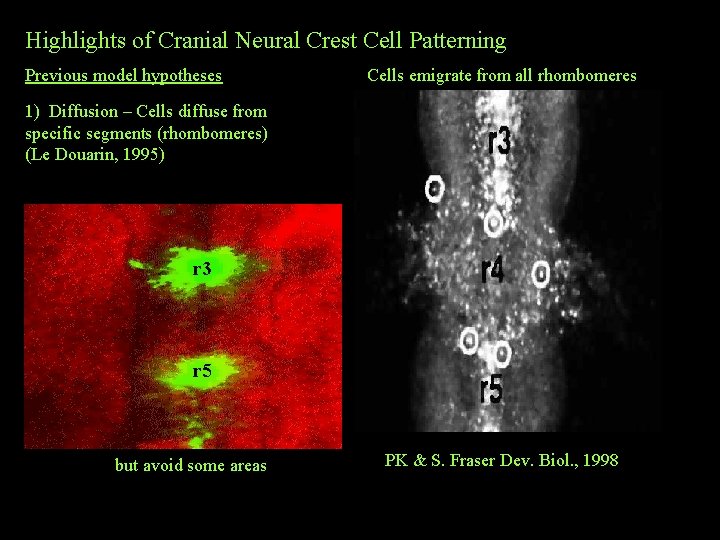
Highlights of Cranial Neural Crest Cell Patterning Previous model hypotheses Cells emigrate from all rhombomeres 1) Diffusion – Cells diffuse from specific segments (rhombomeres) (Le Douarin, 1995) r 3 r 5 but avoid some areas PK & S. Fraser Dev. Biol. , 1998
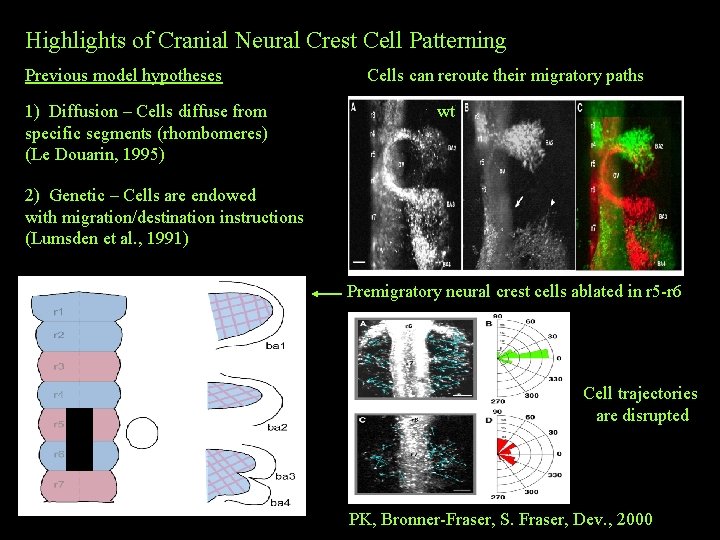
Highlights of Cranial Neural Crest Cell Patterning Previous model hypotheses 1) Diffusion – Cells diffuse from specific segments (rhombomeres) (Le Douarin, 1995) Cells can reroute their migratory paths wt 2) Genetic – Cells are endowed with migration/destination instructions (Lumsden et al. , 1991) Premigratory neural crest cells ablated in r 5 -r 6 Cell trajectories are disrupted PK, Bronner-Fraser, S. Fraser, Dev. , 2000
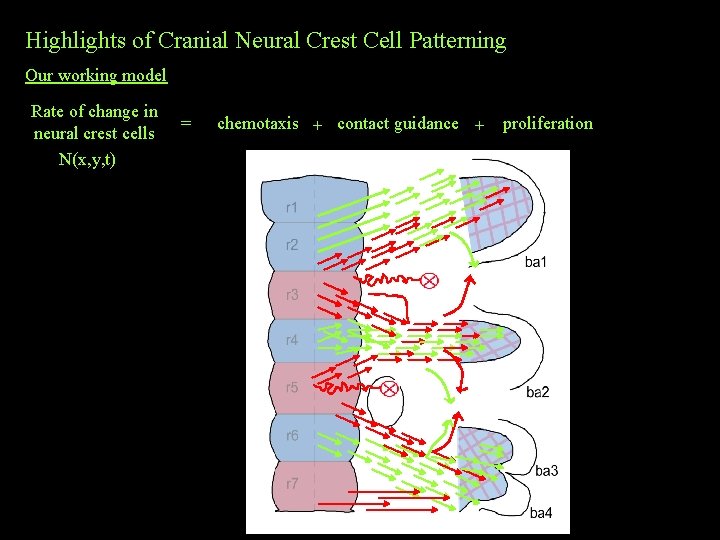
Highlights of Cranial Neural Crest Cell Patterning Our working model Rate of change in neural crest cells N(x, y, t) = chemotaxis + contact guidance + proliferation ? ? ?
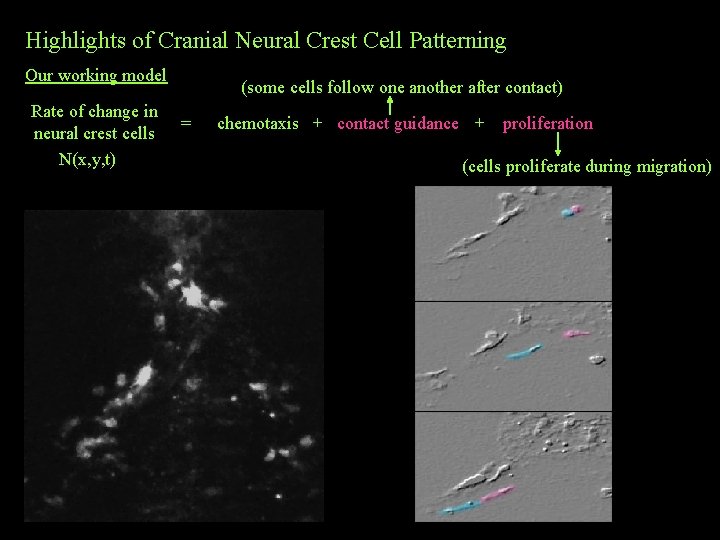
Highlights of Cranial Neural Crest Cell Patterning Our working model Rate of change in neural crest cells N(x, y, t) (some cells follow one another after contact) = chemotaxis + contact guidance + proliferation (cells proliferate during migration)
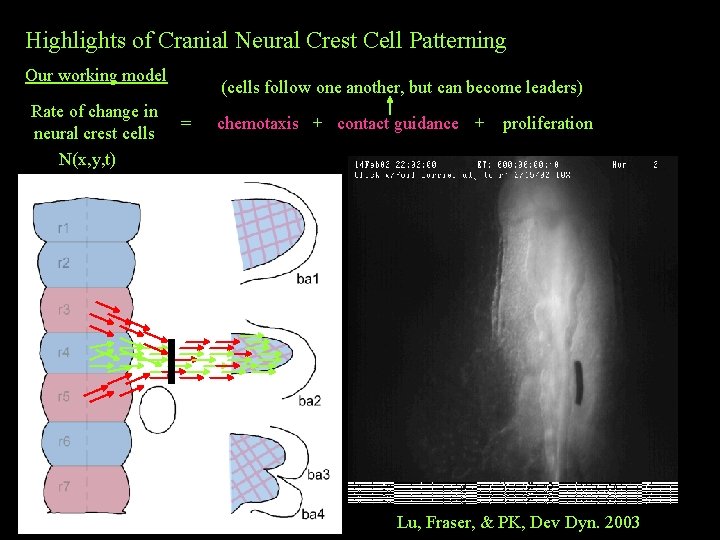
Highlights of Cranial Neural Crest Cell Patterning Our working model Rate of change in neural crest cells N(x, y, t) (cells follow one another, but can become leaders) = ? ? ? chemotaxis + contact guidance + proliferation ? ? Lu, Fraser, & PK, Dev Dyn. 2003
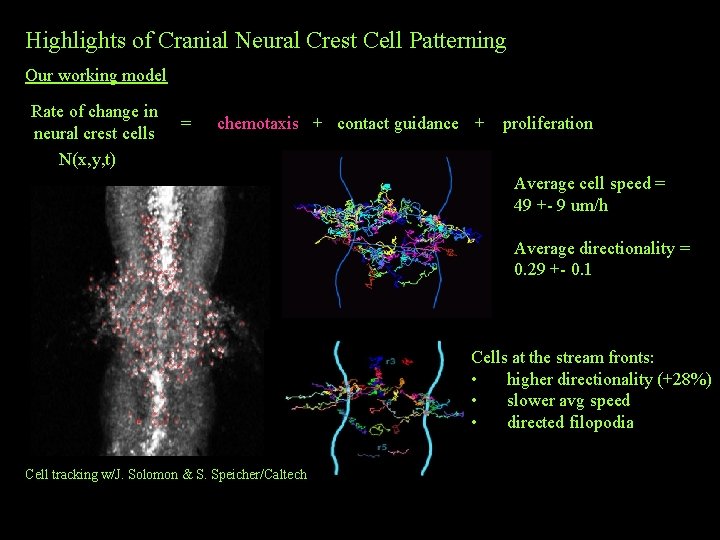
Highlights of Cranial Neural Crest Cell Patterning Our working model Rate of change in neural crest cells N(x, y, t) = chemotaxis + contact guidance + proliferation Average cell speed = 49 +- 9 um/h Average directionality = 0. 29 +- 0. 1 Cells at the stream fronts: • higher directionality (+28%) • slower avg speed • directed filopodia Cell tracking w/J. Solomon & S. Speicher/Caltech
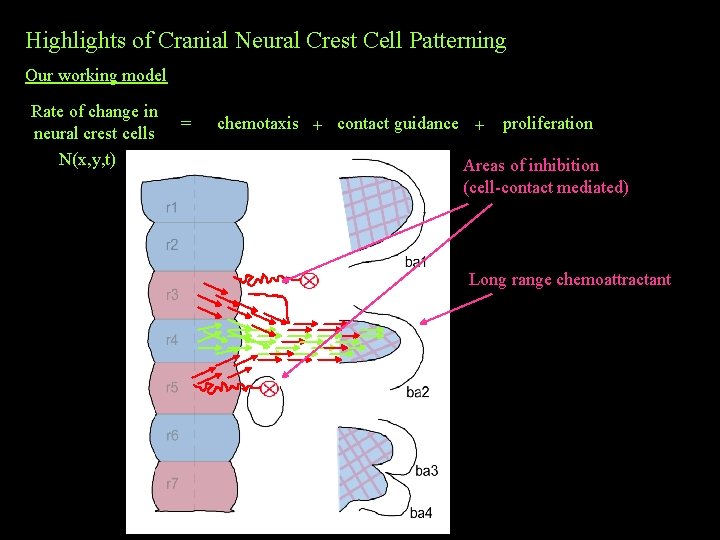
Highlights of Cranial Neural Crest Cell Patterning Our working model Rate of change in neural crest cells N(x, y, t) = chemotaxis + contact guidance + proliferation Areas of inhibition (cell-contact mediated) ? ? ? Long range chemoattractant
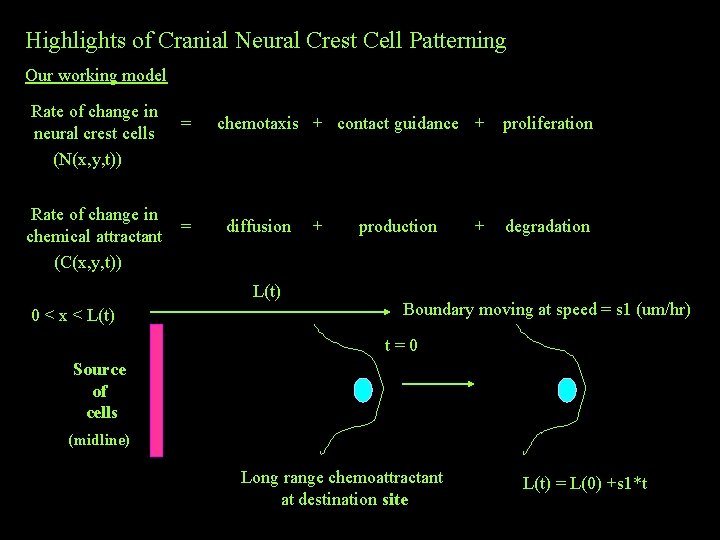
Highlights of Cranial Neural Crest Cell Patterning Our working model Rate of change in neural crest cells (N(x, y, t)) Rate of change in chemical attractant (C(x, y, t)) = = chemotaxis + contact guidance + proliferation diffusion L(t) 0 < x < L(t) + production + degradation Boundary moving at speed = s 1 (um/hr) t=0 Source of cells (midline) Long range chemoattractant at destination site L(t) = L(0) +s 1*t
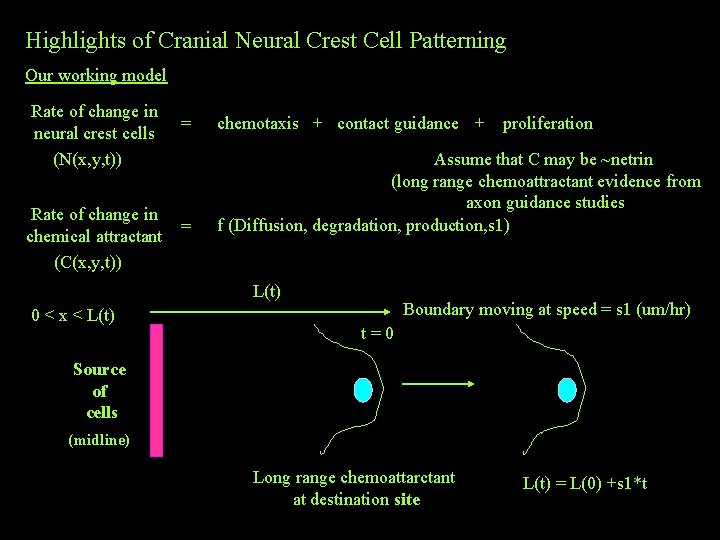
Highlights of Cranial Neural Crest Cell Patterning Our working model Rate of change in neural crest cells (N(x, y, t)) Rate of change in chemical attractant (C(x, y, t)) = chemotaxis + contact guidance + proliferation = Assume that C may be ~netrin (long range chemoattractant evidence from axon guidance studies f (Diffusion, degradation, production, s 1) L(t) 0 < x < L(t) Boundary moving at speed = s 1 (um/hr) t=0 Source of cells (midline) Long range chemoattarctant at destination site L(t) = L(0) +s 1*t
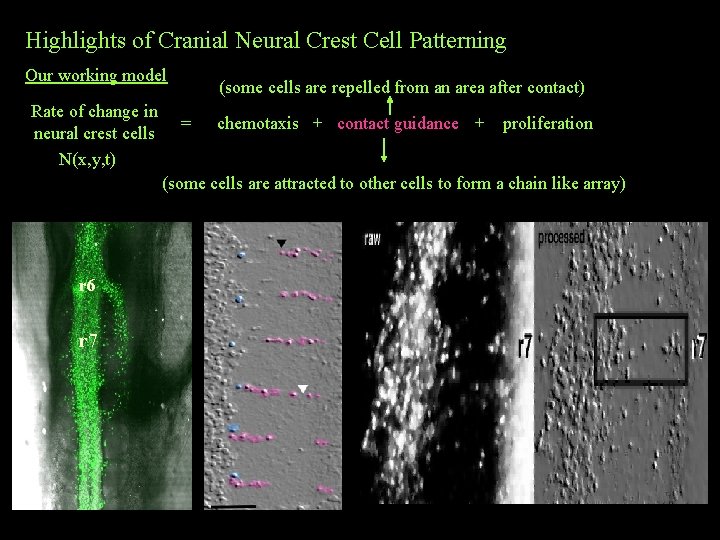
Highlights of Cranial Neural Crest Cell Patterning Our working model Rate of change in neural crest cells N(x, y, t) (some cells are repelled from an area after contact) = chemotaxis + contact guidance + proliferation (some cells are attracted to other cells to form a chain like array) r 6 r 7
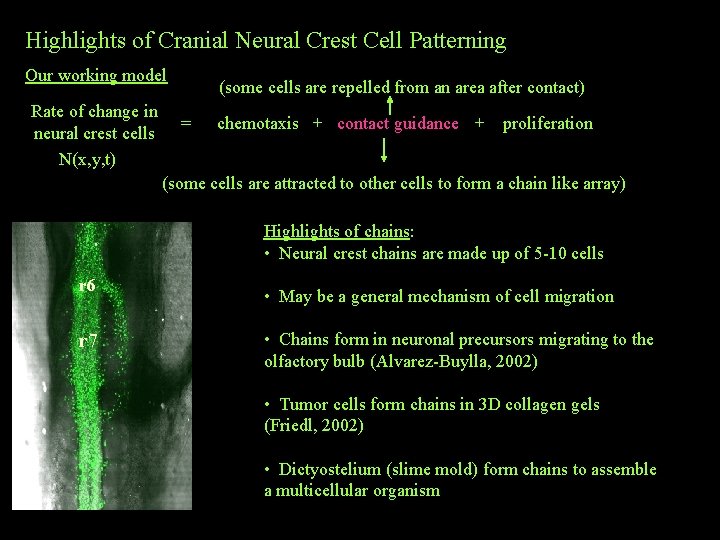
Highlights of Cranial Neural Crest Cell Patterning Our working model Rate of change in neural crest cells N(x, y, t) (some cells are repelled from an area after contact) = chemotaxis + contact guidance + proliferation (some cells are attracted to other cells to form a chain like array) Highlights of chains: • Neural crest chains are made up of 5 -10 cells r 6 r 7 • May be a general mechanism of cell migration • Chains form in neuronal precursors migrating to the olfactory bulb (Alvarez-Buylla, 2002) • Tumor cells form chains in 3 D collagen gels (Friedl, 2002) • Dictyostelium (slime mold) form chains to assemble a multicellular organism
Highlights of Cranial Neural Crest Cell Patterning Our working model hypotheses (Discrete model for contact guidance term) 1) Cells in the chain are linked together by filopodia 2) A cell within a chain emits a chemoattractant at its posterior end (evidence from dictyostelium (c. AMP)) 3) A cell links with another cell after contacting posterior end
Highlights of Cranial Neural Crest Cell Patterning Our working model hypotheses (Discrete model for contact guidance term) 1) Cells in the chain are linked together by filopodia 2) A cell within a chain emits a chemoattractant at its posterior end (evidence from dictyostelium (c. AMP)) 3) A cell links with another cell after contacting posterior end Main assumption for all 3 hypotheses: Either lead cell chews a hole in the extracellular matrix (ECM) or ECM is permissive and lead cell lays down a trail for others to follow. Simple model (cellular automata) • Define a lead cell • Lead cell moves mostly in lateral direction • Leaves open spaces behind which other cells may move into • Gives clues as to how close the lead cell must stay to attract followers • Can leave behind clues instead of open spaces, such as chemoattractant • short or long range interactions?

Cellular structure of the chains Our working model hypotheses (Discrete model for contact guidance term) 1) Cells in the chain are linked together by filopodia Di. I
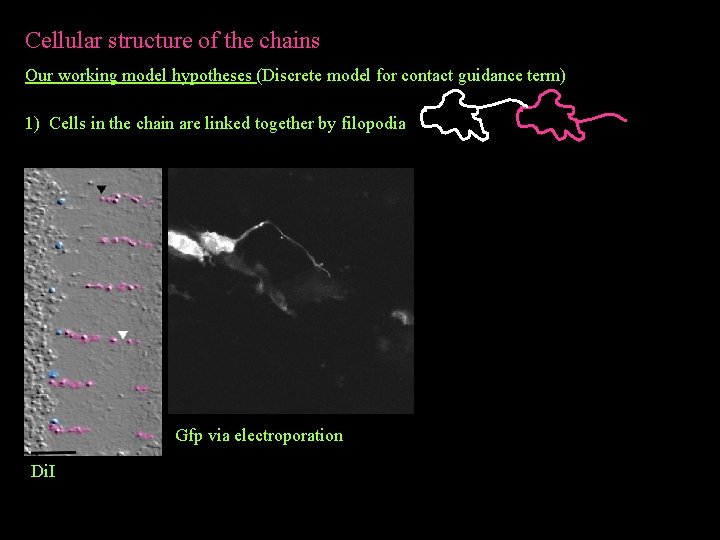
Cellular structure of the chains Our working model hypotheses (Discrete model for contact guidance term) 1) Cells in the chain are linked together by filopodia Gfp via electroporation Di. I
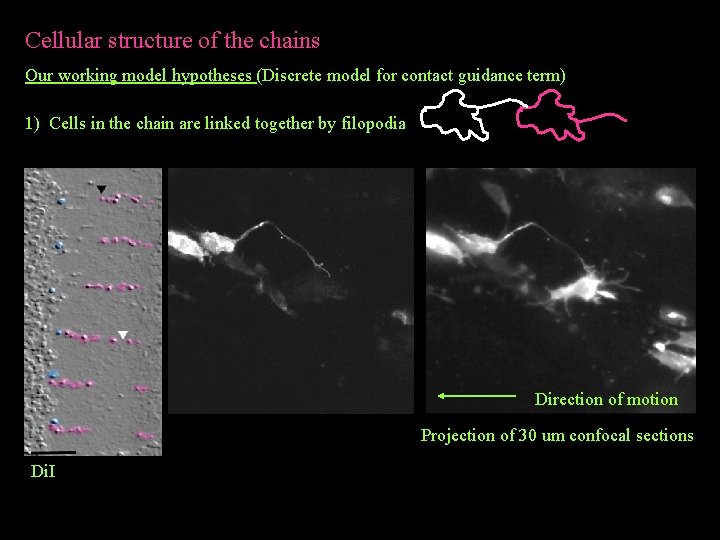
Cellular structure of the chains Our working model hypotheses (Discrete model for contact guidance term) 1) Cells in the chain are linked together by filopodia Direction of motion Projection of 30 um confocal sections Di. I

r 4 r 5
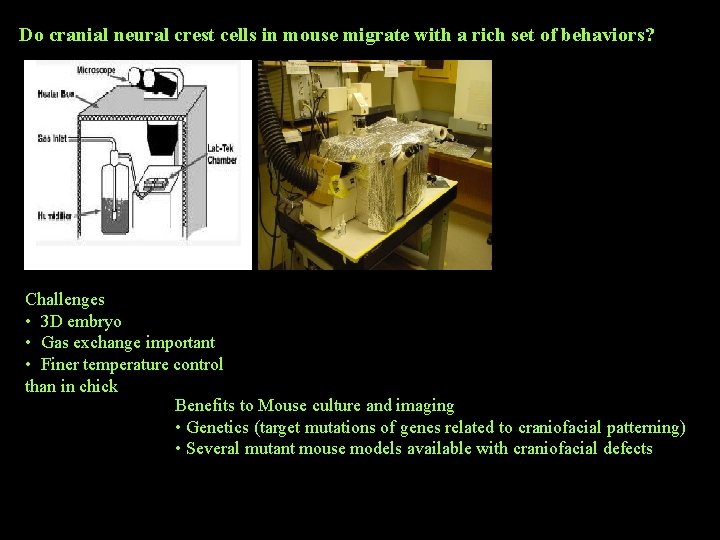
Do cranial neural crest cells in mouse migrate with a rich set of behaviors? Challenges • 3 D embryo • Gas exchange important • Finer temperature control than in chick Benefits to Mouse culture and imaging • Genetics (target mutations of genes related to craniofacial patterning) • Several mutant mouse models available with craniofacial defects
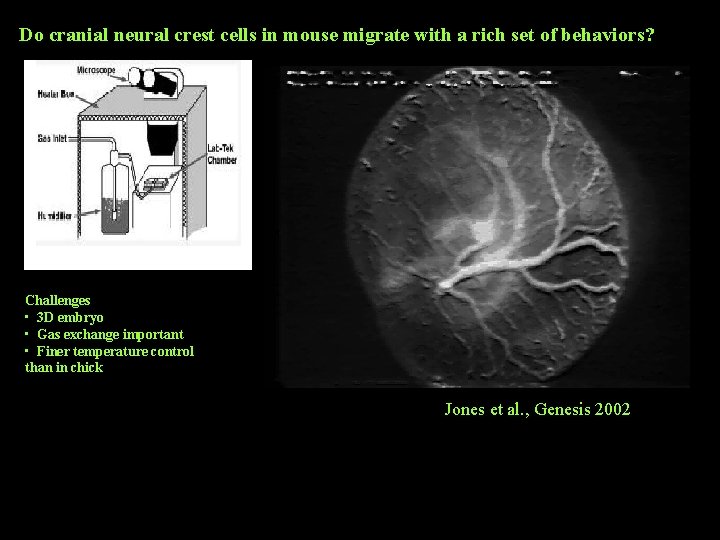
Do cranial neural crest cells in mouse migrate with a rich set of behaviors? Challenges • 3 D embryo • Gas exchange important • Finer temperature control than in chick Jones et al. , Genesis 2002
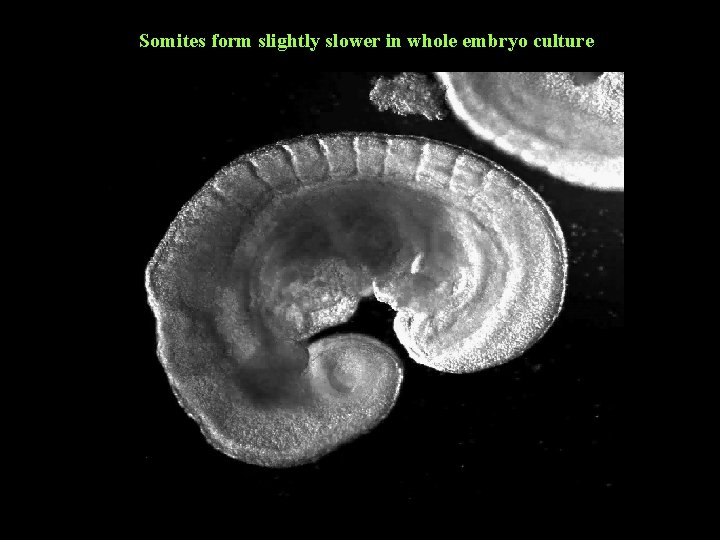
Somites form slightly slower in whole embryo culture
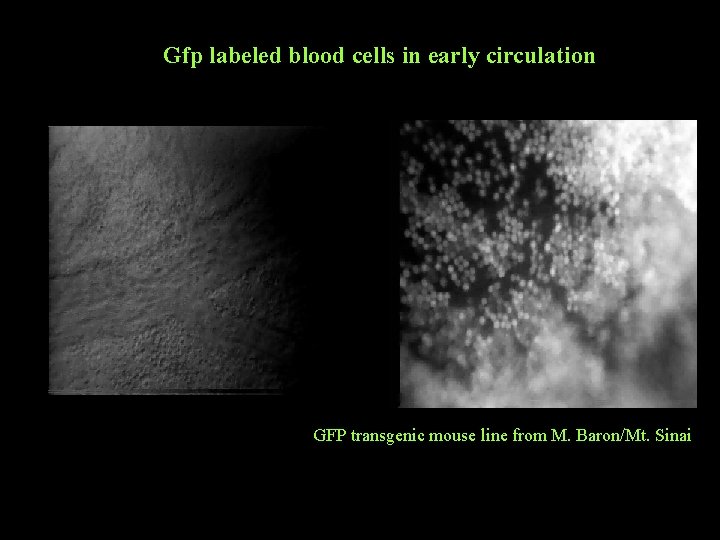
Gfp labeled blood cells in early circulation GFP transgenic mouse line from M. Baron/Mt. Sinai
It is important to maintain the embryo in one place P. Trainor

Acknowledgements Caltech • Scott Fraser • Marianne Bronner-Fraser • Mary Dickinson • Dave Crotty Stowers Institute for Medical Research • Paul Trainor

- Slides: 32