Insights from Levant 2 and Global SFA Registry

Insights from Levant 2 and Global SFA Registry (Interim Update) Chris Metzger, M. D. CVA Heart Institute Kingsport, TN, USA

Disclosures • Symposium Honoraria & Course Proctor – Abbott, Medtronic, Tri. Vascular • Symposium Honoraria – Spectranetics, Cordis, Bard, Cardiovascular Research Foundation, Boston Scientific • National PI – CANOPY, SAPPHIRE WW • Stock, Research Grants, etc. – None

Lutonix: The Science

Anatomy of Drug Coated Balloon Drug • All DCBs use Paclitaxel • Paclitaxel can differ from DCB to DCB: • Crystalline • Amorphous Carrier • Important to bind paclitaxel to the balloon • Differs from manufacturer to manufacturer COATING • KEY FEATURES: ü Drug Uniformity ü Drug Retention ü Drug Release 4

LUTONIX® DCB Formulation • Result of extensive development and rigorous testing • 45 pre-clinical studies • >11, 000 histology samples • Resulted in an optimized formulation with a therapeutic dose of 2 µg/mm 2 5

Lutonix: The Technology
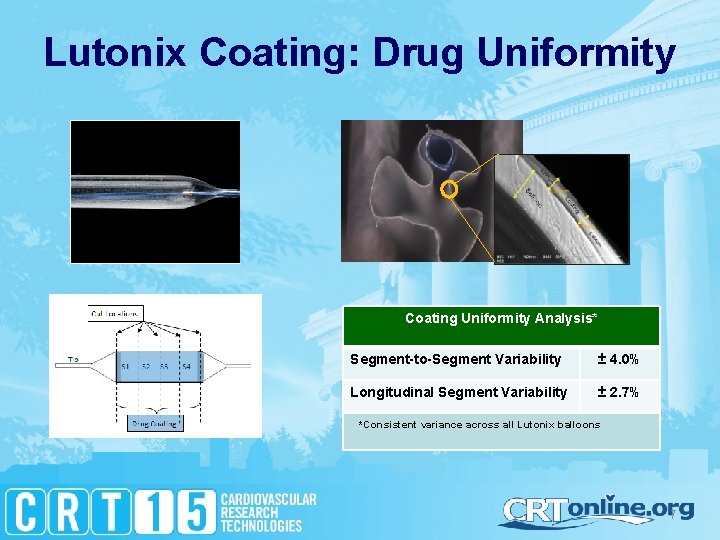
Lutonix Coating: Drug Uniformity Coating Uniformity Analysis* Segment-to-Segment Variability ± 4. 0% Longitudinal Segment Variability ± 2. 7% *Consistent variance across all Lutonix balloons 7

Lutonix Coating: Drug Release In Vivo Administration of Fluorescent-Labeled PTX to Excised Porcine Artery 10% Oregon green labeled paclitaxel incorporated into Lutonix ® DCB coating Uniform Delivery in vivo at 1 hour (Animal vessel cross section after 30 sec. inflation) Lutonix coating uniformity allows uniform drug delivery Preclinical animal data on file. Animal test results may not be indicative of clinical performance. Different test methods may yield different results. 8

Lutonix Coating Durability Dry Inflate “Shake” Test LTX DCB (n=5) Residual Drug on DCB Balloon After Inflation/Shake Drug Loss After DCB Inflation/Shake 99. 4% ± 1. 1% <0. 1%* * Below limits of detection Methods of Kelsch et al. , Invest Radiol 2011; 46(4): 255 -63 Evidence of Durability. 9
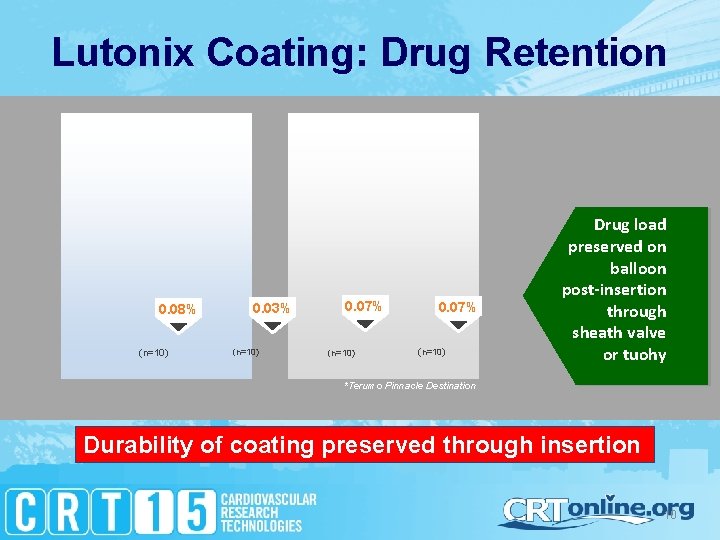
Lutonix Coating: Drug Retention 0. 08% (n=10) 0. 03% (n=10) 0. 07% (n=10) Drug load preserved on balloon post-insertion through sheath valve or tuohy *Terumo Pinnacle Destination Durability of coating preserved through insertion 10

Lutonix Coating Durability* Paclitaxel Surface Conc. (ng/cm 2) Surface Swab Test- Back Table 45. 00 40. 00 35. 00 30. 96 30. 00 25. 00 20. 39 20. 00 15. 00 9. 82 10. 00 5. 00 0. 40 0. 27 0. 34 0. 00 Lutonix 6 x 120 mm Medtronic 6 x 120 mm Lutonix 4 x 120 mm Medtronic 4 x 120 mm Overall Lutonix Overall Medtronic N=9 N=9 N=18 Simulated Clinical Use Test LUTONIX® Amount of drug lost on the back table. (Drug measured- ng/mg) Designed to minimize unnecessary drug exposure to staff and patients 11

Levant 2 Clinical Trial
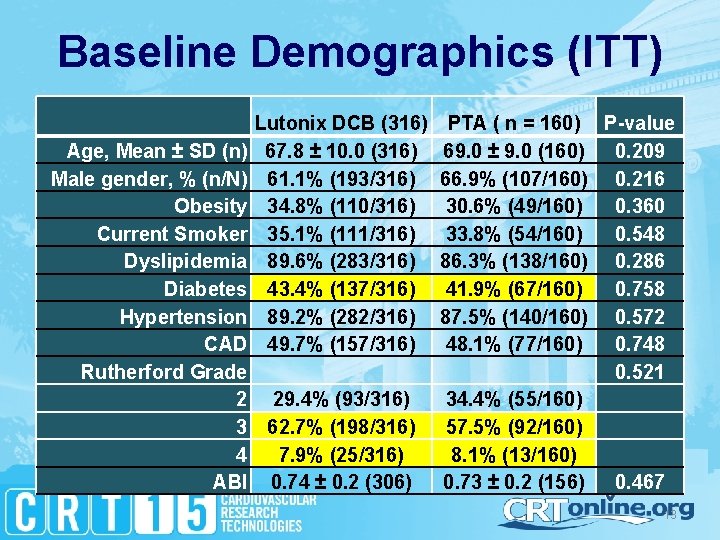
Baseline Demographics (ITT) Lutonix DCB (316) 67. 8 ± 10. 0 (316) 61. 1% (193/316) 34. 8% (110/316) 35. 1% (111/316) 89. 6% (283/316) 43. 4% (137/316) 89. 2% (282/316) 49. 7% (157/316) Age, Mean ± SD (n) Male gender, % (n/N) Obesity Current Smoker Dyslipidemia Diabetes Hypertension CAD Rutherford Grade 2 29. 4% (93/316) 3 62. 7% (198/316) 4 7. 9% (25/316) ABI 0. 74 ± 0. 2 (306) PTA ( n = 160) P-value 69. 0 ± 9. 0 (160) 0. 209 66. 9% (107/160) 0. 216 30. 6% (49/160) 0. 360 33. 8% (54/160) 0. 548 86. 3% (138/160) 0. 286 41. 9% (67/160) 0. 758 87. 5% (140/160) 0. 572 48. 1% (77/160) 0. 748 0. 521 34. 4% (55/160) 57. 5% (92/160) 8. 1% (13/160) 0. 73 ± 0. 2 (156) 0. 467 13
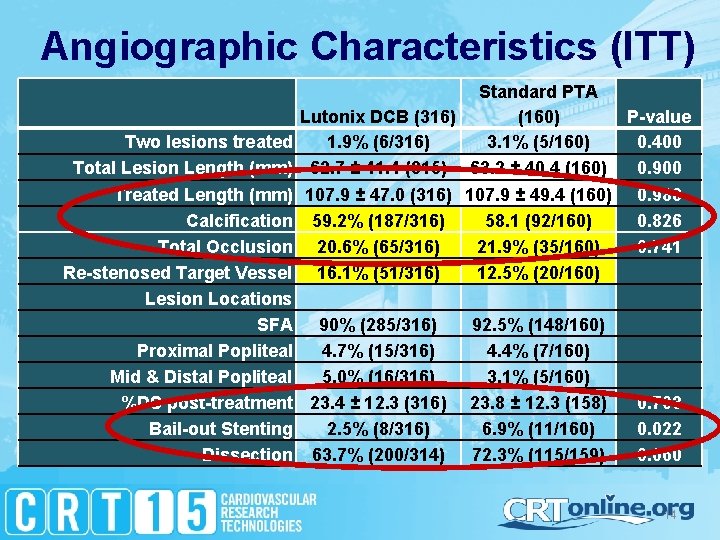
Angiographic Characteristics (ITT) Standard PTA Lutonix DCB (316) (160) P-value Two lesions treated 1. 9% (6/316) 3. 1% (5/160) 0. 400 Total Lesion Length (mm) 62. 7 ± 41. 4 (315) 63. 2 ± 40. 4 (160) 0. 900 Treated Length (mm) 107. 9 ± 47. 0 (316) 107. 9 ± 49. 4 (160) 0. 988 Calcification 59. 2% (187/316) 58. 1 (92/160) 0. 826 Total Occlusion 20. 6% (65/316) 21. 9% (35/160) 0. 741 Re-stenosed Target Vessel 16. 1% (51/316) 12. 5% (20/160) Lesion Locations SFA 90% (285/316) 92. 5% (148/160) Proximal Popliteal 4. 7% (15/316) 4. 4% (7/160) Mid & Distal Popliteal 5. 0% (16/316) 3. 1% (5/160) %DS post-treatment 23. 4 ± 12. 3 (316) 23. 8 ± 12. 3 (158) 0. 703 Bail-out Stenting 2. 5% (8/316) 6. 9% (11/160) 0. 022 Dissection 63. 7% (200/314) 72. 3% (115/159) 0. 060 14
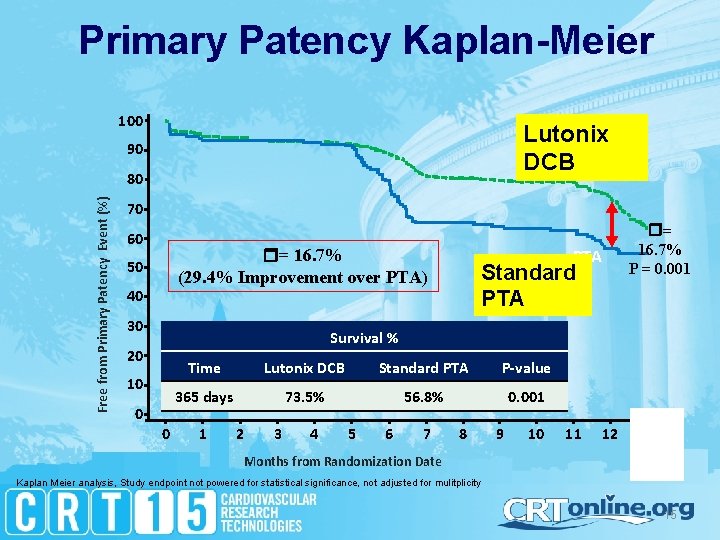
Primary Patency Kaplan-Meier 100 Lutonix DCBDCB 90 Free from Primary Patency Event (%) 80 70 60 = 16. 7% (29. 4% Improvement over PTA) 50 40 30 = 16. 7% P = 0. 001 PTA Standard PTA Survival % 20 10 0 0 Time Lutonix DCB Standard PTA P-value 365 days 73. 5% 56. 8% 0. 001 1 2 3 4 5 6 7 8 9 10 11 12 13 Months from Randomization Date Kaplan Meier analysis, Study endpoint not powered for statistical significance, not adjusted for mulitplicity 15
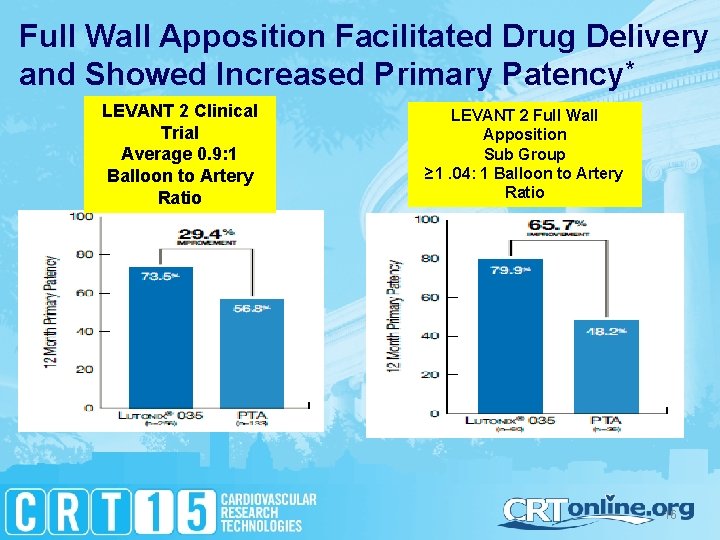
Full Wall Apposition Facilitated Drug Delivery and Showed Increased Primary Patency* LEVANT 2 Clinical Trial Average 0. 9: 1 Balloon to Artery Ratio LEVANT 2 Full Wall Apposition Sub Group ≥ 1. 04: 1 Balloon to Artery Ratio 16
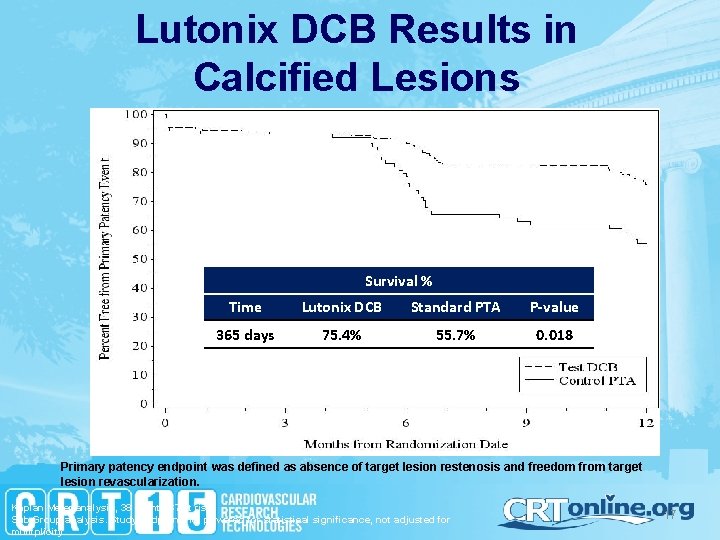
Lutonix DCB Results in Calcified Lesions Survival % Time Lutonix DCB Standard PTA P-value 365 days 75. 4% 55. 7% 0. 018 Primary patency endpoint was defined as absence of target lesion restenosis and freedom from target lesion revascularization. Kaplan Meier analysis, 38 events 67 at risk Sub Group analysis. Study endpoint not powered for statistical significance, not adjusted for mulitplicity 17
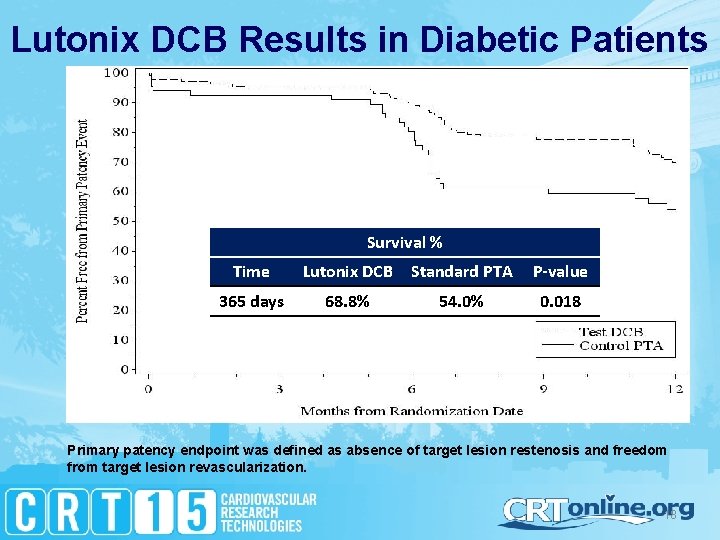
Lutonix DCB Results in Diabetic Patients Survival % Time Lutonix DCB Standard PTA P-value 365 days 68. 8% 54. 0% 0. 018 Primary patency endpoint was defined as absence of target lesion restenosis and freedom from target lesion revascularization. 18
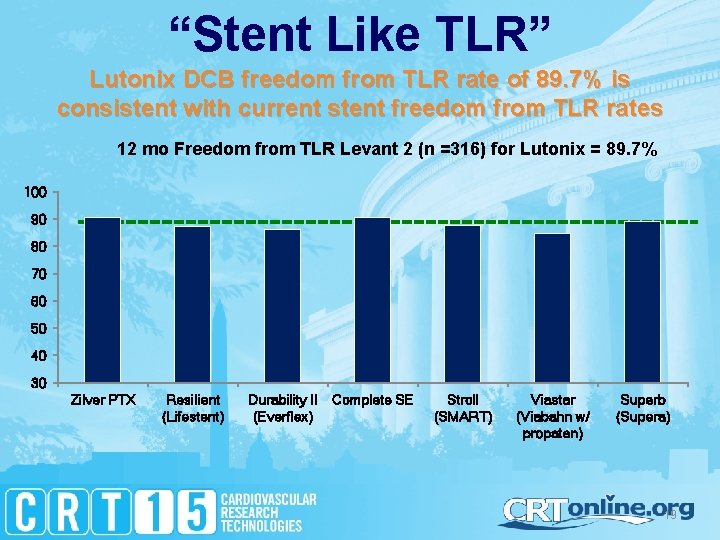
“Stent Like TLR” Lutonix DCB freedom from TLR rate of 89. 7% is consistent with current stent freedom from TLR rates 12 mo Freedom from TLR Levant 2 (n =316) for Lutonix = 89. 7% 100 90 80 70 60 50 40 30 Zilver PTX Resilient (Lifestent) Durability II Complete SE (Everflex) Stroll (SMART) Viastar (Viabahn w/ propaten) Superb (Supera) 19
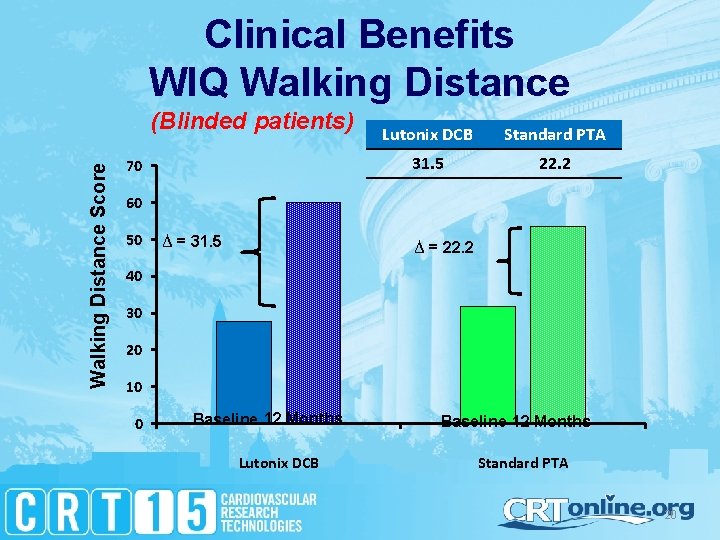
Clinical Benefits WIQ Walking Distance Score (Blinded patients) 70 Lutonix DCB Standard PTA 31. 5 22. 2 60 50 ∆ = 31. 5 ∆ = 22. 2 40 30 20 10 0 Baseline 12 Months Lutonix DCB Baseline 12 Months Standard PTA 20
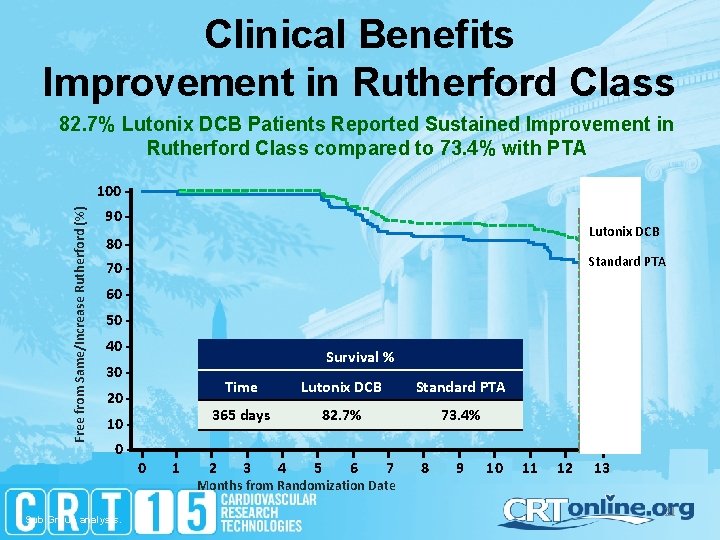
Clinical Benefits Improvement in Rutherford Class 82. 7% Lutonix DCB Patients Reported Sustained Improvement in Rutherford Class compared to 73. 4% with PTA Free from Same/Increase Rutherford (%) 100 90 - Lutonix DCB 80 - Standard PTA 70 60 50 40 - Survival % 30 20 10 0 - Sub Group analysis. 0 1 Time Lutonix DCB Standard PTA 365 days 82. 7% 73. 4% 2 3 4 5 6 7 Months from Randomization Date 8 9 10 11 12 13 21
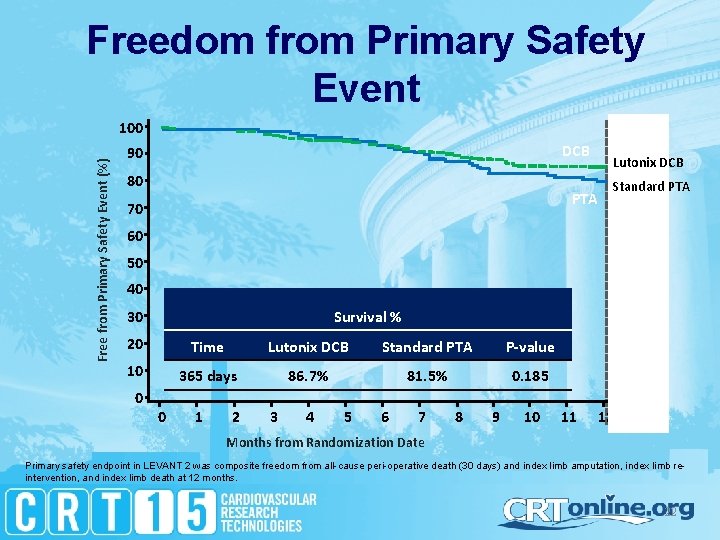
Freedom from Primary Safety Event Free from Primary Safety Event (%) 100 DCB 90 80 Lutonix DCB PTA 70 Standard PTA 60 50 40 30 Survival % 20 Time Lutonix DCB Standard PTA P-value 10 365 days 86. 7% 81. 5% 0. 185 0 0 1 2 3 4 5 6 7 8 9 10 11 12 13 Months from Randomization Date Primary safety endpoint in LEVANT 2 was composite freedom from all-cause peri-operative death (30 days) and index limb amputation, index limb reintervention, and index limb death at 12 months. 22
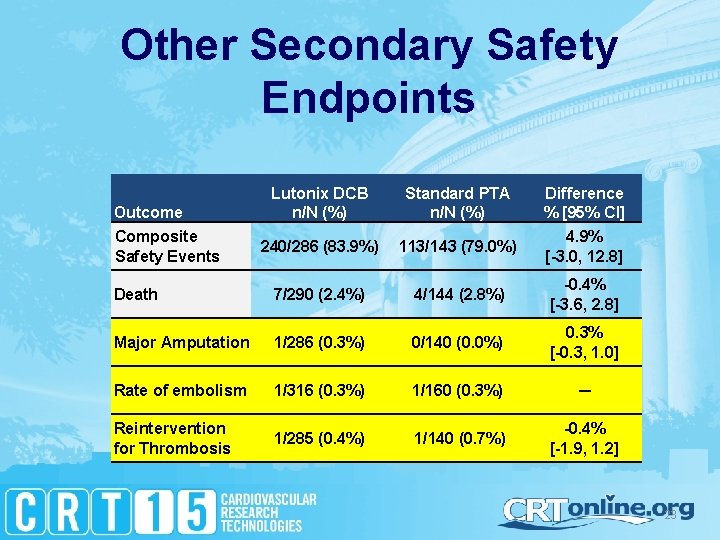
Other Secondary Safety Endpoints Lutonix DCB n/N (%) Standard PTA n/N (%) Difference % [95% CI] 240/286 (83. 9%) 113/143 (79. 0%) 4. 9% [-3. 0, 12. 8] Death 7/290 (2. 4%) 4/144 (2. 8%) -0. 4% [-3. 6, 2. 8] Major Amputation 1/286 (0. 3%) 0/140 (0. 0%) 0. 3% [-0. 3, 1. 0] Rate of embolism 1/316 (0. 3%) 1/160 (0. 3%) ─ Reintervention for Thrombosis 1/285 (0. 4%) 1/140 (0. 7%) -0. 4% [-1. 9, 1. 2] Outcome Composite Safety Events 23

Demonstrated Safety of Lutonix DCB Low rate of embolic events, similar to PTA Low rates of re-intervention due to thrombosis, similar to PTA Every adverse event (including serious and nonserious) was adjudicated in Levant 2 by an independent panel of medical experts. 24

Insights from Levant 2 and Real World Global SFA Registry (Interim Update)

Baseline DCB Demographics Levant 2 vs Global SFA Registry* Levant 2 DCB (316) Global SFA Registry (691) Age, Mean ± SD (n) 67. 8 ± 10. 0 (316) 68. 3 ± 9. 8 (691) Male gender, % (n/N) Obesity Current Smoker Dyslipidemia Diabetes Hypertension Rutherford Grade 2 3 4 5 and 6 61. 1% (193/316) 34. 8% (110/316) 35. 1% (111/316) 89. 6% (283/316) 43. 4% (137/316) 89. 2% (282/316) 67. 6% (467/691) 29. 4% (93/316) 62. 7% (198/316) 7. 9% (25/316) 0. 0% (0/316) 17. 6% 62. 5% 7. 3% 1. 6% * Interim Results N = 289 36. 5% (252/690) 69. 9% (483/691) 39. 5% (273/691) 85. 2% (589/691) 26

DCB Angiographic Demographics Levant 2 vs Global SFA Registry* Levant 2 DCB (316) Global SFA Registry (691) Calcification 59. 2% (187/316) Total Occlusion 20. 6% (65/316) 14. 2% (98/688) 100. 9 ± 84. 2 (684) 197. 3 ± 133. 2 (202) 34. 2% (235/687) 30. 9% (212/686) SFA 90% (285/316) 70% (477/678) Proximal Popliteal 4. 7% (15/316) 16. 8% (114/678) Mid & Distal Popliteal 5. 0% (16/316) 12. 8% (87/678) 23. 4 ± 12. 3 (316) 14. 3 ± 18. 3 (680) 27. 6% (191/691) 38. 1% (263/691) Two lesions treated 1. 9% (6/316) Total Lesion Length (mm) 62. 7 ± 41. 4 (315) Treated Length (mm) 107. 9 ± 47. 0 (316) Lesion Locations %DS post-treatment Bail-out Stenting Dissection * Interim Results N = 289 2. 5% (8/316) 63. 7% (200/314) 27
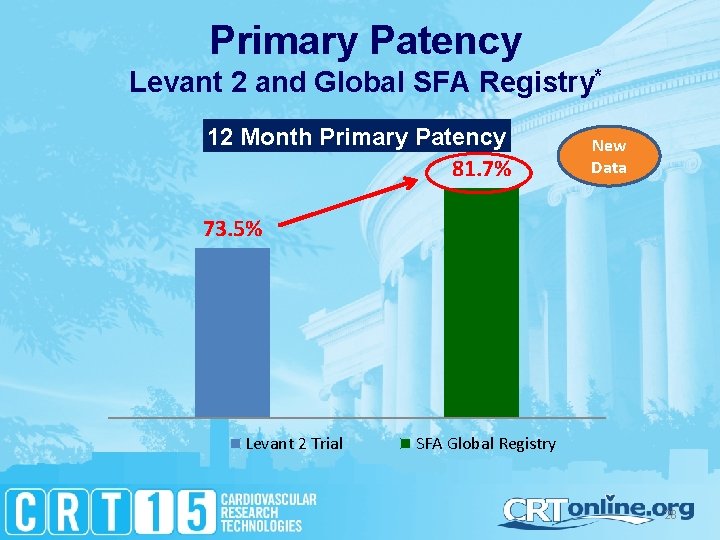
Primary Patency Levant 2 and Global SFA Registry* 12 Month Primary Patency 81. 7% New Data 73. 5% Levant 2 Trial SFA Global Registry 28
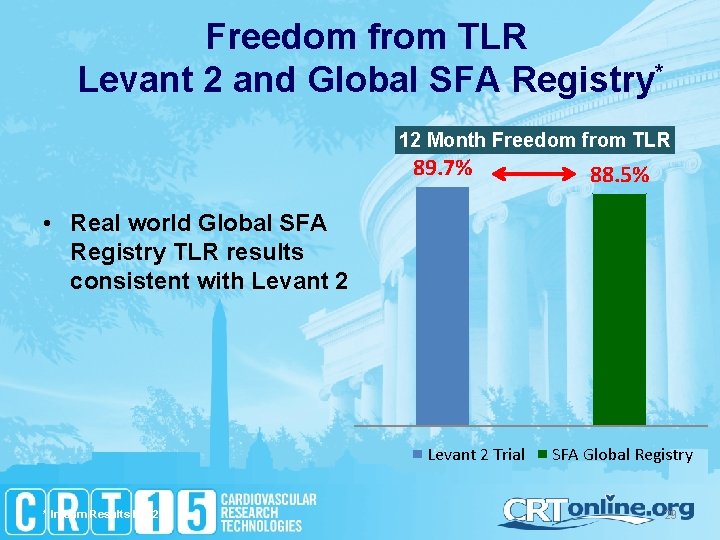
Freedom from TLR Levant 2 and Global SFA Registry* 12 Month Freedom from TLR 89. 7% 88. 5% • Real world Global SFA Registry TLR results consistent with Levant 2 Trial * Interim Results N = 279 SFA Global Registry 29

Secondary Safety Endpoints – Levant 2 and Global SFA Registry* Outcome Composite Safety Events Death Major Amputation Rate of embolism Reintervention for Thrombosis Levant 2 Global SFA Registry 240/286 (83. 9%) 7/290 (2. 4%) 5/267 (1. 9%) 1/286 (0. 3%) 0/262 (0. 0%) 1/316 (0. 3%) 2/264 (0. 8%) 1/285 (0. 4%) 0/262 (0. 0%) 30

Effectiveness Without Compromising Safety • Levant 2 demonstrated superior patency for Lutonix DCB against PTA – 29. 4% Improved patency over PTA – Lutonix DCB effective in challenging patient population – TLR rates of 89% consistent in both Clinical Trial and Real. World Registry • Levant 2 demonstrated safety of Lutonix DCB – Low rates of re-intervention for thrombosis and embolism – No unanticipated safety events in over 1000 patients** • Patients who received Lutonix DCB reported Clinical Benefits in Levant 2 – Sustained improvement in Rutherford Class – Improvement in self-reported Walking Distance scores 31

Lessons Learned from Levant 2 • Levant 2 - rigorous trial w/ rapid enrollment • Good PTA technique improved PTA results • DCB can be done with LOW bailout stent rates with “stent-like” freedom from TLR • DCB’s should be sized ≥ 1: 1 for ↑ results • Good DCB technique (incl. no geographic miss) is important for optimal results • DCB’s: > results, = safety c/w PTA- all pts

Thank You for Your Attention!!

Lutonix Coating Durability Lutonix 035 IN. PACT Simulated dry inflate test. Not intended to represent actual handling or preparation and not representative of actual clinical use. 34
- Slides: 34