Inputs to a casebased HIV surveillance system Objectives

Inputs to a case-based HIV surveillance system

Objectives � Review HIV case definitions � Understand � Identify clinical and immunologic staging the sentinel events in a case reporting system � Identify events existing data sources to report sentinel

WHO Surveillance Case Definitions

Background � 2004 – 2005 � WHO � April regional meetings to revise guidelines 2006 � WHO global consensus meeting to finalize the revision of clinical stage and case definitions

Case Definition: HIV Infection � Adults and children 18 months or older: Positive HIV antibody testing (rapid or laboratory-based enzyme immunoassay) confirmed by a second HIV antibody test (rapid or laboratory-based enzyme immunoassay) relying on different antigens or of different operating characteristics. and /or; � Positive virological test for HIV or its components (HIV-RNA or HIVDNA or ultrasensitive HIV p 24 antigen) confirmed by a second virological test obtained from a separate determination. � � Children � younger than 18 months: Positive virological test for HIV or its components (HIV-RNA or HIVDNA or ultrasensitive HIV p 24 antigen) confirmed by a second virological test obtained from a separate determination taken more than four weeks after birth. � Note: Positive antibody testing is not recommended for definitive or confirmatory diagnosis of HIV infection in children until 18 months of age.

Case Definition: Advanced HIV Infection (including AIDS) �Adults and children with confirmed HIV infection: � Clinical criteria: � Presumptive condition or definitive diagnosis of any stage 3 or stage 4 � Immunological � CD 4 criteria: count less than 350/mm of blood �Children younger than five years of age with confirmed HIV infection: � %CD 4+ <30 among those younger than 12 months � %CD 4+ <25 among those aged 12– 35 months � %CD 4+ <20 among those aged 36– 59 months

Primary HIV Infection �Definition: The time of HIV infection. Some clinical symptoms, but no (or few) antibodies present in blood � There is no standard case definition � This can be identified by recent appearance of HIV or by identifying viral products (HIV-RNA or HIV-DNA and/or ultrasensitive HIV p 24 antigen) with negative (or weakly reactive) HIV antibody � If identified, this should be reported � Primary HIV Infection should not be confused with clinical staging conditions developed with established HIV infection

WHO Clinical and Immunological Classification for HIV

Purpose of Classification System � Classification is used to assess patients before they are put on treatment (presumptive and definitive diagnosis) � Proposed to use clinical staging events and immunologic classification to assess individuals once they are receiving ART
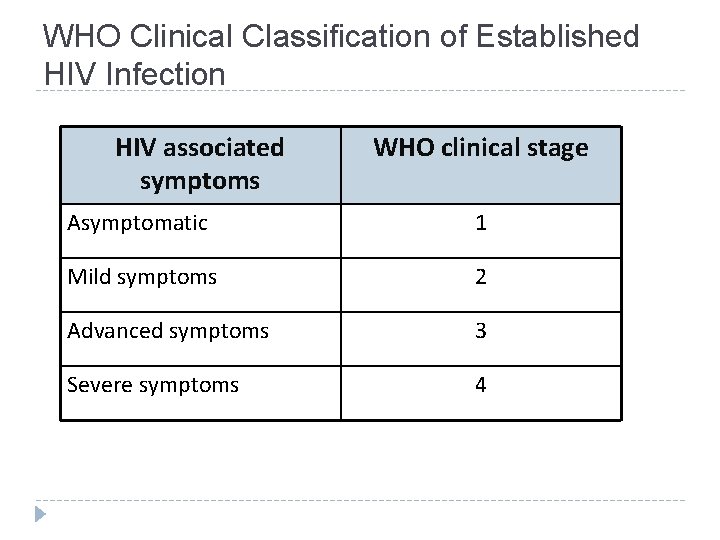
WHO Clinical Classification of Established HIV Infection HIV associated symptoms WHO clinical stage Asymptomatic 1 Mild symptoms 2 Advanced symptoms 3 Severe symptoms 4
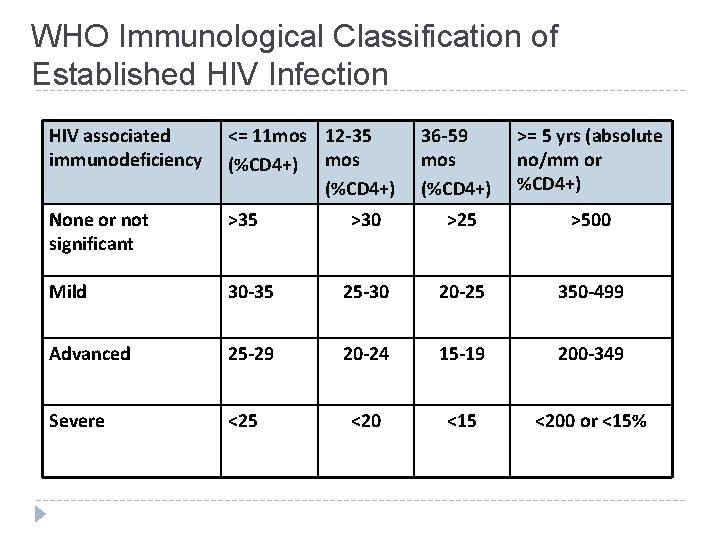
WHO Immunological Classification of Established HIV Infection HIV associated immunodeficiency <= 11 mos 12 -35 (%CD 4+) mos (%CD 4+) 36 -59 mos (%CD 4+) >= 5 yrs (absolute no/mm or %CD 4+) None or not significant >35 >30 >25 >500 Mild 30 -35 25 -30 20 -25 350 -499 Advanced 25 -29 20 -24 15 -19 200 -349 Severe <25 <20 <15 <200 or <15%
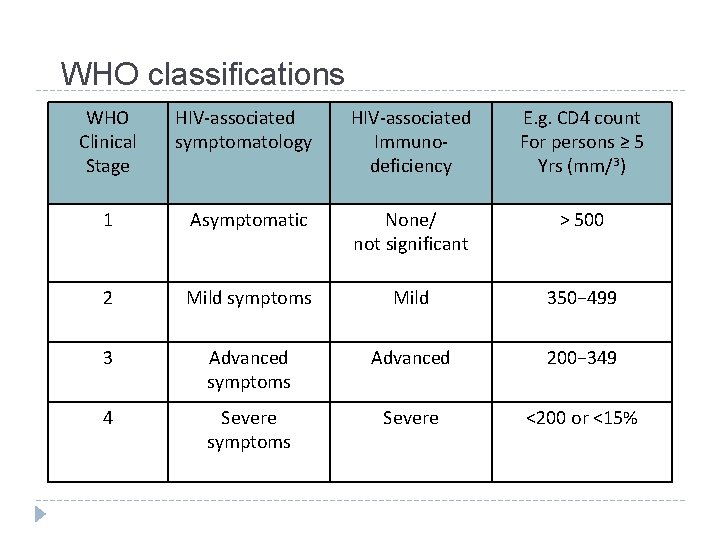
WHO classifications WHO Clinical Stage HIV-associated symptomatology HIV-associated Immunodeficiency E. g. CD 4 count For persons ≥ 5 Yrs (mm/3) 1 Asymptomatic None/ not significant > 500 2 Mild symptoms Mild 350− 499 3 Advanced symptoms Advanced 200− 349 4 Severe symptoms Severe <200 or <15%

HIV Case-Based Surveillance

Review: WHO Surveillance System Recommendations � WHO now recommends that reporting be expanded to cover the entire spectrum of HIV disease. � Previous case definitions focused only on AIDS cases � WHO recommends that countries standardise their reporting practices. � Countries � Report may: all HIV cases (clinical stages 1 through 4) � Report advanced HIV disease (clinical stages 3 and 4) � Report AIDS cases (clinical stage 4)

Pro and Cons of WHO Options �Report all HIV cases (clinical stages 1 through 4) � Pros: � Full picture of epidemic � Allows tracking of disease progression � Follow disease trends for both new infections and later stages � Identifies priority populations that may need additional surveillance activities and prevention services � Can be generalized to entire population � Cons: � Can be difficult to implement � Can be expensive

Pro and Cons of WHO Options �Report advanced HIV disease (clinical stages 3 and 4) � Pros: � Measure of clinical burden Those how are diagnosed with HIV and who need ART � Some disease trends � Cons: � Under-reporting � May increase even as prevention decreases new cases

Pro and Cons of WHO Options Report AIDS cases (clinical stage 4) Pros: Clinical burden Easier to manage Cons: Under estimation of epidemic Trends for AIDS cases may differ from new HIV infection
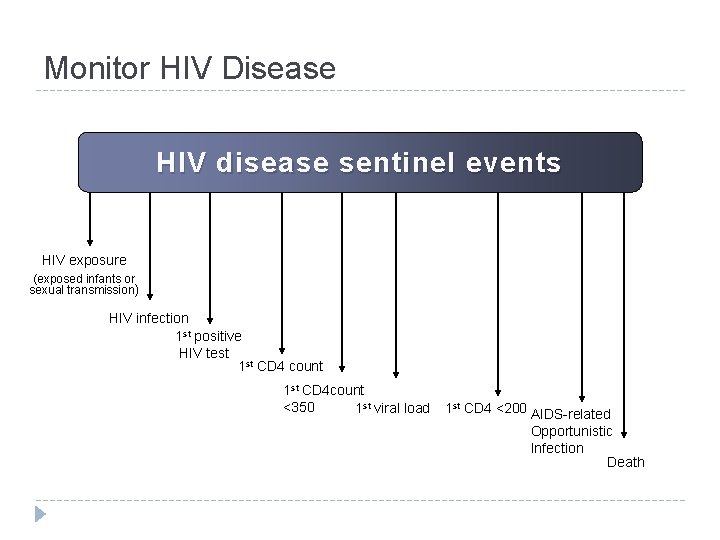
Monitor HIV Disease HIV disease sentinel events HIV exposure (exposed infants or sexual transmission) HIV infection 1 st positive HIV test 1 st CD 4 count 1 st CD 4 count <350 1 st viral load 1 st CD 4 <200 AIDS-related Opportunistic Infection Death

Monitor HIV Disease HIV disease sentinel events HIV exposure (exposed infants or sexual transmission) HIV infection 1 st positive HIV test 1 st CD 4 count AIDS reporting 1 st CD 4 count <350 1 st viral load 1 st CD 4 <200 AIDS-related Opportunistic Infection Death
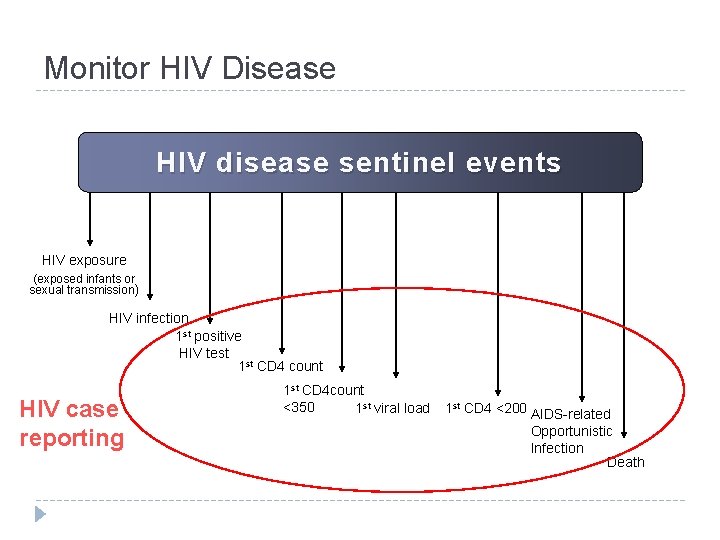
Monitor HIV Disease HIV disease sentinel events HIV exposure (exposed infants or sexual transmission) HIV infection 1 st positive HIV test 1 st CD 4 count HIV case reporting 1 st CD 4 count <350 1 st viral load 1 st CD 4 <200 AIDS-related Opportunistic Infection Death

Where Will the Data Come From? � HIV antibody test (positives only) – probably already exists in most countries � Laboratories, rapid testing from facilities � Clinical stages? Which ones? � HIV facility identifies clinical stage at first visit and reports it � All CD 4 tests? � Labs � All Viral load tests? � Labs
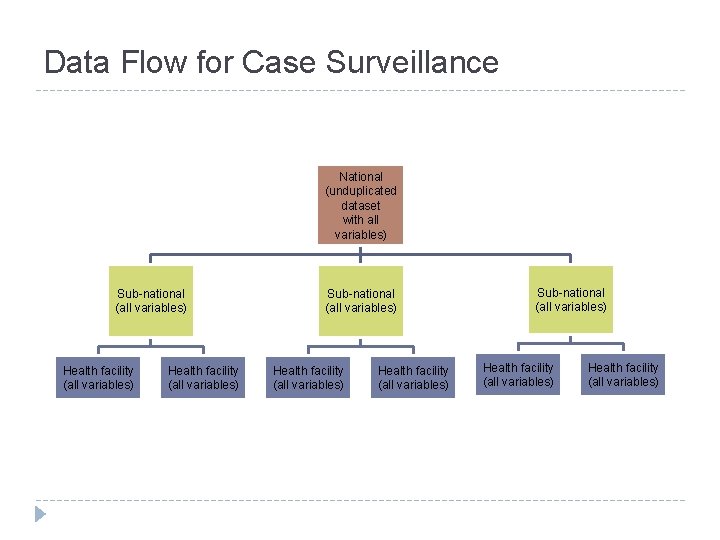
Data Flow for Case Surveillance National (unduplicated dataset with all variables) Sub-national (all variables) Health facility (all variables) Sub-national (all variables) Health facility (all variables)

How Will Cases be Identified? Potential sources of HIV case reports: All laboratories Health centres ART treatment clinics TB clinics VCT sites Private physicians Hospitals PMTCT programme Blood banks Hospice Vital statistics registries

Thank You Working Together to Plan, Implement, and Use HIV Surveillance Systems
- Slides: 24