Inorganic compounds Inorganic compound Any substance in which

Inorganic compounds

Inorganic compound Any substance in which two or more chemical elements (usually other than carbon) are combined, nearly always in definite proportions. Compounds of carbon are usually classified as organic when carbon is bound to hydrogen.

Inorganic compounds • • • Oxides Acids Bases Salts ……. .

Oxides Chemical compounds with one or more oxygen atoms combined with another element

Nomenclature and formulas • Oxides are maned by name of non-oxygen atom + oxide eg. Hydrogen oxide, H 2 O. • Oxidatnion state of non-oxygen element should be given eg. Sulfur (IV) oxide, SO 2. • Sometimes, non-oxygen/oxygen ratios are used to name oxides eg. monooxide, dioxide, trioxide etc. • In the industry, oxides are named by contracting the element name with „a”, eg. magnesia oxide, Mg. O.

Theory of Acids and Bases • There are three major classifications of substances known as acids or bases. • The Arrhenius definition • The Brønsted-Lowry definition • The Lewis definition

Arrhenius definition • An acid produces H+ in solution and a base produces OH-. • This theory was developed in 1883.

Brønsted-Lowry definition • Acids are defined as proton donors; whereas bases are defined as proton acceptors. • A compound that acts as both a Brønsted. Lowry acid and base together is called amphoteric

Lewis definition • Acids act as electron pair acceptors and bases act as electron pair doners.

Acids /Arrhenius/ • Substance that gives H+ ions when dissolved in water. • General Structure HR, where: H is proton (cation) and R in an anion

Nomenclature of oxygenless acids • The most important is anion. After that word „acid”. • If there is no oxygen in acid, the acid is named with the hydro- prefix. • Then is the root. The root in the name of the element. • At the end is suffix –ic. The name of anion with suffix –ide instead of –ic. H 2 S: hydro sulfur ic acid, sulf –ide HCl: hydro chlor ic acid, chlor -ide

Nonmenclature of oxoacids • When there is an oxigen in anion, the name of acid if from the anion, after that word „acid”. • The root is formed from the name of central element, that build anion with the suffix –ic. Oxidation state of central atom should be indicated. The name of anion is build in the same way, but with suffix –ate instead of –ic. H 2 SO 4: sulfur ic (IV) acid, sulf ate (IV) anion

Reactions • oxygenless acids hydrogen + nonmetal → acid H 2 + Cl 2→ 2 HCl + H 2 O → HCl(aq) • oxoacids acid anhydride + water → acid SO 3 + H 2 O → H 2 SO 4 P 4 O 10 + 6 H 2 O → 4 H 3 PO 4 Cl 2 O 7 + H 2 O → HCl. O 4
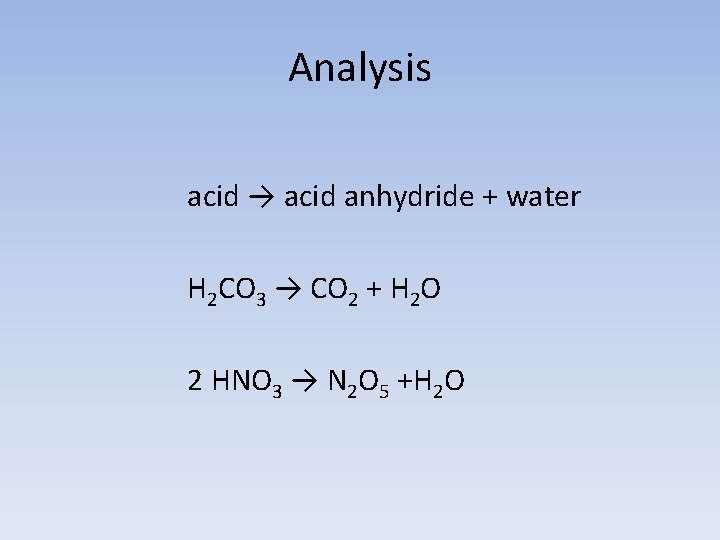
Analysis acid → acid anhydride + water H 2 CO 3 → CO 2 + H 2 O 2 HNO 3 → N 2 O 5 +H 2 O

Neutralization • acid + metal oxide → salt + water Mg. O + H 2 SO 4 → Mg. SO 4 + H 2 O Na 2 O + HCl → Na. Cl + H 2 O 2 Fe. O + 6 H 2 SO 4 → Fe 2(SO 4)3 + 3 SO 2 + 6 H 2 O • acid + base → salt + water KOH + HNO 2 → KNO 2 + H 2 O Ca(OH)2 + HNO 3 → Ca(NO 3)2 + 2 H 2 O

Neutralization • acid + metal → salt + hydrogen H 2 CO 3 + 2 Na → Na 2 CO 3 +H 2↑ 2 H 3 PO 4 + 3 Mg → Mg 3(PO 4)2 + 6 H 2↑ HNO 3 + Fe → Fe(NO 3)2 + H 2↑
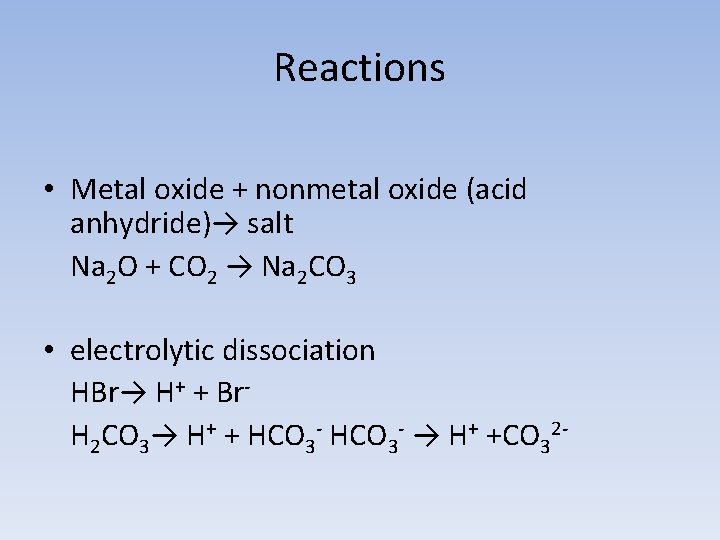
Reactions • Metal oxide + nonmetal oxide (acid anhydride)→ salt Na 2 O + CO 2 → Na 2 CO 3 • electrolytic dissociation HBr→ H+ + Br. H 2 CO 3→ H+ + HCO 3 - → H+ +CO 32 -

Bases /Arrhenius/ • Substance that gives OH- ions when dissolved in water. • General Structure M(OH)x, where: M is usually metal (cation) and OH is a hydroxylic group • Nomenclature: cation (metal) (oxidation state) + hydroxide Co(OH)3 cobalt (III) hydroxide

Creation • metal + water → base + H 2↑ 2 Na + 2 H 2 O --> 2 Na. OH + H 2 Ca + 2 H 2 O --> Ca(OH)2 + H 2 • metal oxide + water → hydroxide + salt Fe. Cl 3 + 3 Na. OH ---> Fe(OH)3 + 3 Na Cl Cu. SO 4 + 2 Na. OH ---> Cu(OH)2 + Na 2 SO 4

Creation • metal hydride + water → base + H 2↑ • metal peroxide + water → base + H 2 O 2 • metal carbide + water → base + unsaturated hydrocarbon↑

Reactions • non metal oxide + base → salt + water SO 2 + 2 KOH → K 2 SO 3 + H 2 O • base + non metal oxide → salt + water 2 Na. OH + CO 2→ Na 2 CO 3 + H 2 O • electrolytic dissociation Na. OH → Na+ + OH-

Salts • An ionic compound that results from the neutralization reaction of an acid and a base. • Salts are composed of related numbers of cations and anions so that the product is electrically neutral.

Nomenclature • Name of the cation, then name of the anion, same rules like for acids and bases. • Fe 2(SO 4)3: Iron (III) sulfate (VI)

Acid salts • Salts formed by the partial neutralization of diprotic or polyprotic acids. Acid is only partially neutralized, one or more hydrogen atoms still remain in the structure. • To indicate hydrogen presence word hydro- is given between cation and anion. Amount of hydrogen atoms should be indicated: Na. H 2 PO 4: sodium di hydro phosphate (V)

Basic salts • Salts formed by the partial neutralization of dihydroxylic or polyhydroxylic basess. Base is only partially neutralized, one or more hydroxylic group still remain in the structure. • To indicate OH- group presence word hydro- is given between cation and anion. Amount of hydrogen atoms should be indicated: Cu 2(OH)3 Cl: dicopper (II) trihydroxyl chloride

Reactions with salts • salt 1 + acid 1(stronger) → salt 2 + acid 2 (weaker) Mg. CO 3 + 2 HCl→ Mg. Cl 2 + CO 2 + H 2 O • salt 1 + salt 2→ salt 3 + salt 4↓ Ca. Cl 2 + Na 2 CO 3 → Ca. CO 3↓ + 2 Na. Cl Ca 2++2 Cl- + 2 Na+ + CO 32 - → Ca. CO 3↓ +2 Na+ + 2 Cl. Ag. Cl. O 4 + NH 4 Cl → Ag. Cl↓ + NH 4 Cl. O 4 Ag+ + Cl. O 4 - + NH 4+ + Cl-→ Ag. Cl↓ + Cl. O 4 - + NH 4+

Sources • http: //chemwiki. ucdavis. edu/Core/Inorganic_ Chemistry/Descriptive_Chemistry/Main_Grou p_Reactions/Compounds/Oxides • http: //chemwiki. ucdavis. edu/Core/Physical_C hemistry/Acids_and_Bases/Acid
- Slides: 27