Inorganic and Organic Compounds Inorganic not made by


Inorganic and Organic Compounds Inorganic - not made by living things Organic - made by living things - carbon compounds - forms covalent bonds

Properties of Water l Great solvent called a universal solvent l Helps to digest food and remove waste products l Helps with transport in the body
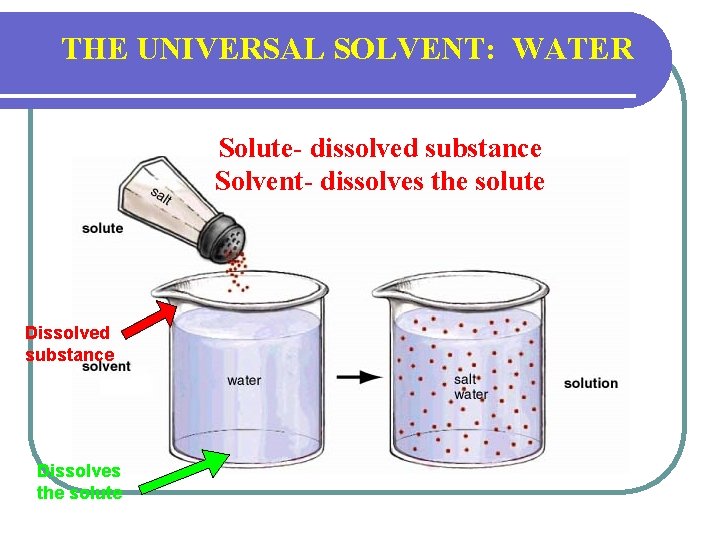
THE UNIVERSAL SOLVENT: WATER Solute- dissolved substance Solvent- dissolves the solute Dissolved substance Dissolves the solute

Water is a Polar Compound l Hydrogen ends holds slightly positive charges l Oxygen end holds a slightly negative charge (+) 1 H p+ p+ (--) 1 H
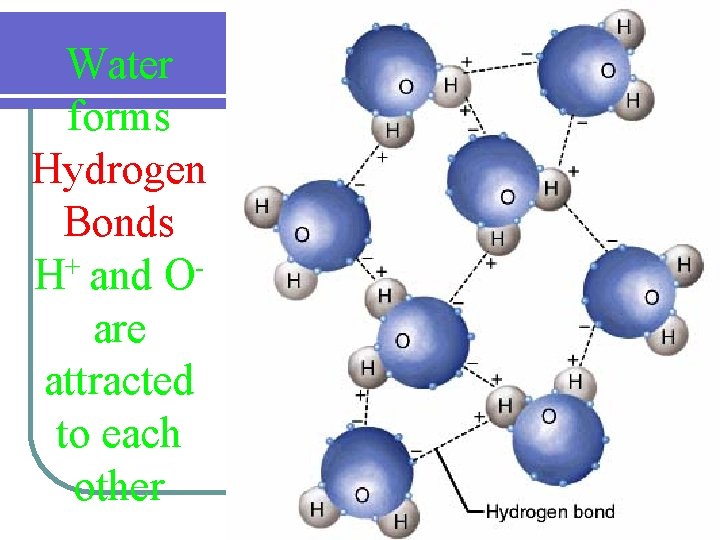
Water forms Hydrogen Bonds + H and O are attracted to each other
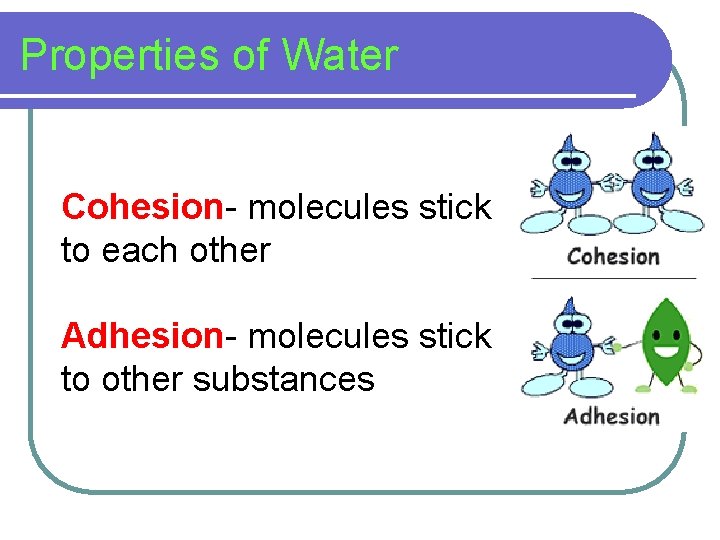
Properties of Water Cohesion- molecules stick to each other Adhesion- molecules stick to other substances
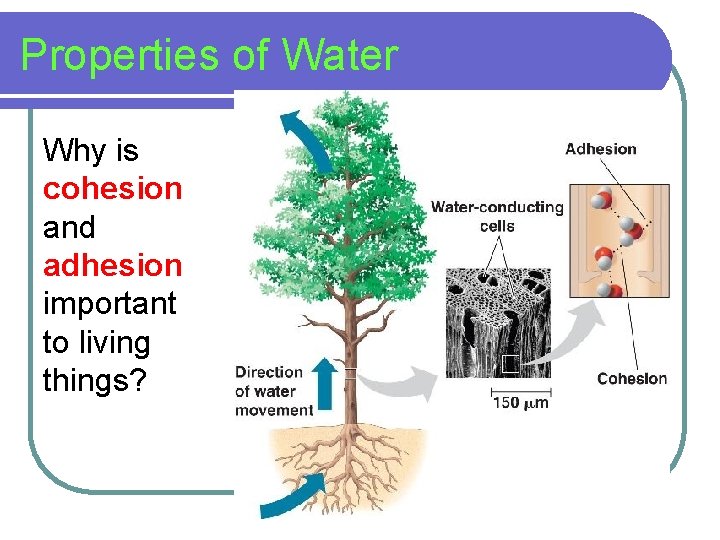
Properties of Water Why is cohesion and adhesion important to living things?

http: //www. youtube. com/watch? f eature=player_embedded&v=D Ail. C 0 sjvy 0#! http: //www. youtube. com/watch? v=HVT 3 Y 3_g. HGg&list=SP 3 EE D 4 C 1 D 684 D 3 ADF&index=2
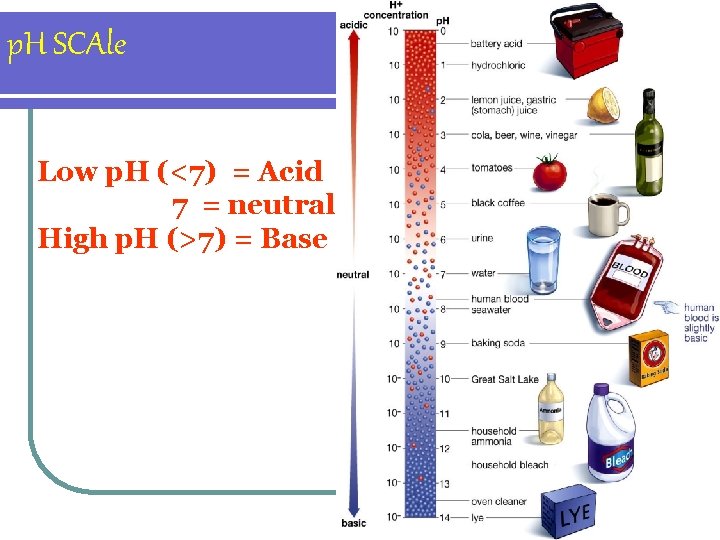
p. H SCAle Low p. H (<7) = Acid 7 = neutral High p. H (>7) = Base

p. H in the body l. Human blood- 7. 4 l. Stomach juice- 2. 0

ORGANIC COMPOUNDS Organic compounds are carbon compounds, made by living things
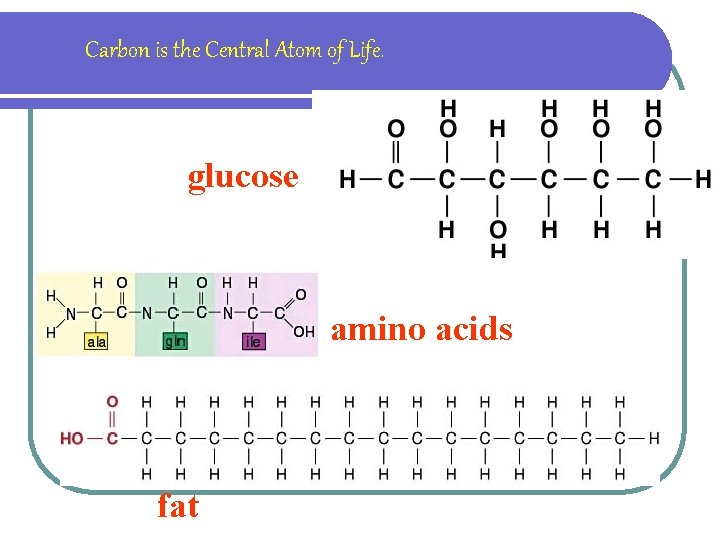
Carbon is the Central Atom of Life. glucose amino acids fat

To Understand the BIG, You’d Better Know the LITTLE (and how small and big are linked)
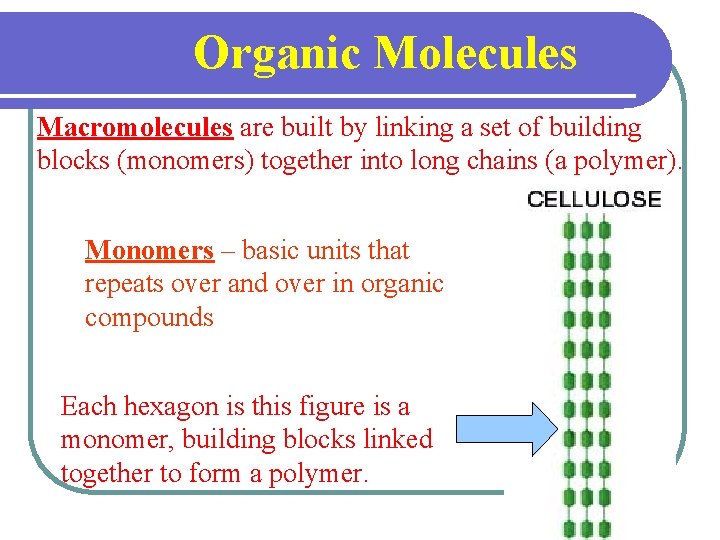
Organic Molecules Macromolecules are built by linking a set of building blocks (monomers) together into long chains (a polymer). Monomers – basic units that repeats over and over in organic compounds Each hexagon is this figure is a monomer, building blocks linked together to form a polymer.
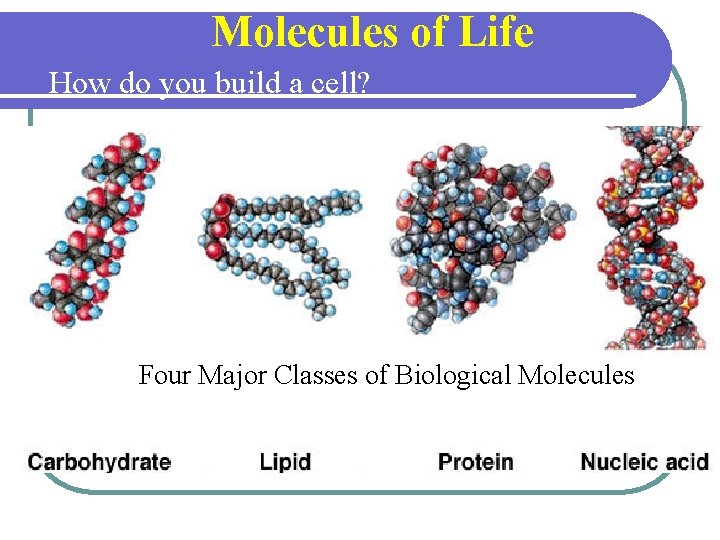
Molecules of Life How do you build a cell? Start with water, add lots of small carboncontaining molecules and ……. Four Major Classes of Biological Molecules

FOUR TYPES OF ORGANIC COMPOUNDS Common names l CARBOHYDRATES l l SUGARS AND STARCHES LIPIDS l FATS, OILS, AND WAXES l PROTEINS l NUCLEIC ACIDS l DNA AND RNA (GENETIC MATERIAL)
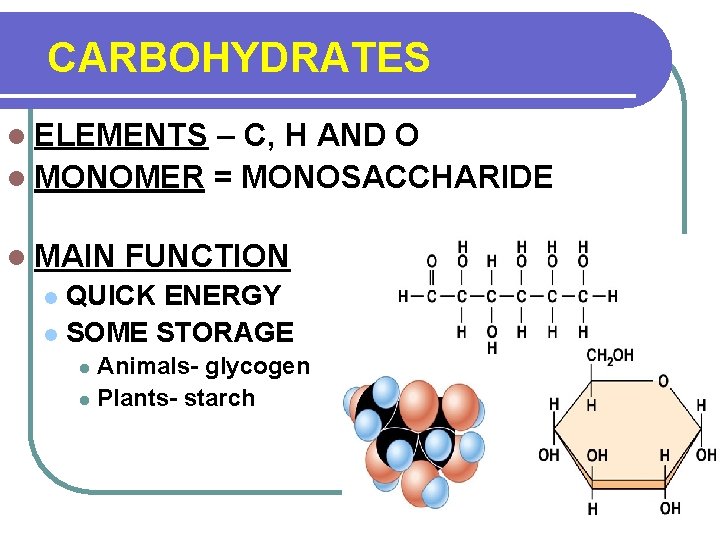
CARBOHYDRATES l ELEMENTS – C, H AND O l MONOMER = MONOSACCHARIDE l MAIN FUNCTION QUICK ENERGY l SOME STORAGE l Animals- glycogen l Plants- starch l

CARBOHYDRATES Other facts 2: 1 ratio of Hydrogen to Oxygen One sugar C 6 H 12 O 6 monosaccharide Two sugars C 12 H 22 O 11 disaccharide Many sugars- polysaccharide
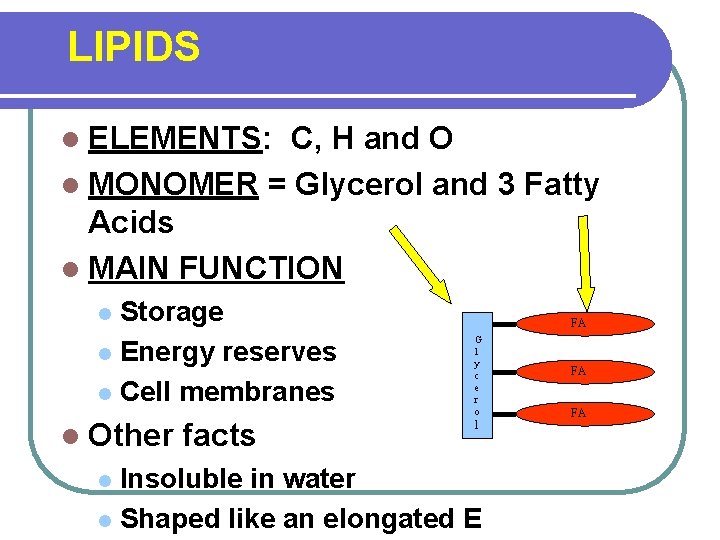
LIPIDS l ELEMENTS: C, H and O l MONOMER = Glycerol and 3 Fatty Acids l MAIN FUNCTION Storage l Energy reserves l Cell membranes l l Other facts FA G l y c e r o l Insoluble in water l Shaped like an elongated E l FA FA
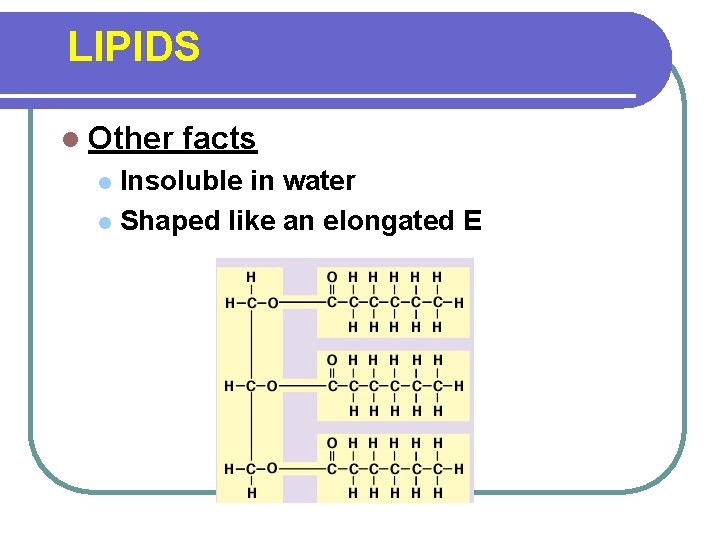
LIPIDS l Other facts Insoluble in water l Shaped like an elongated E l

PROTEINS l ELEMENTS: C, H, O and N l MONOMER: Amino Acids l FUNCTIONS Basic building blocks of living material l Hair, Muscles, Fingernails l Enzymes – speed up chemical reactions l Transport in cell l
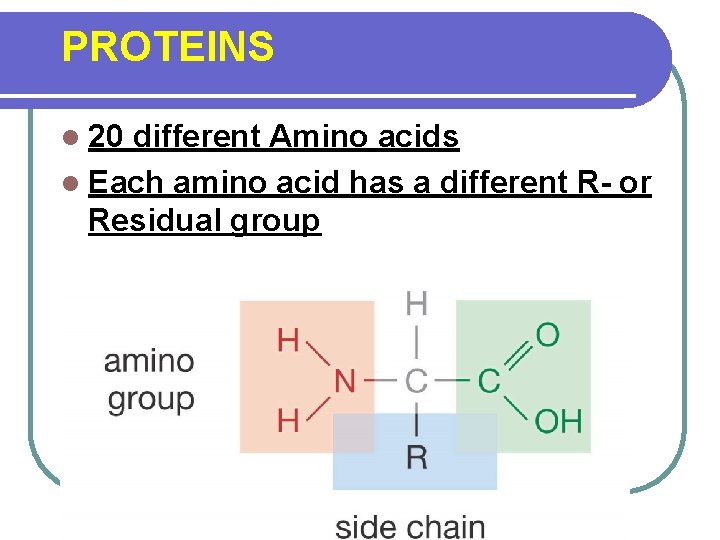
PROTEINS l 20 different Amino acids l Each amino acid has a different R- or Residual group

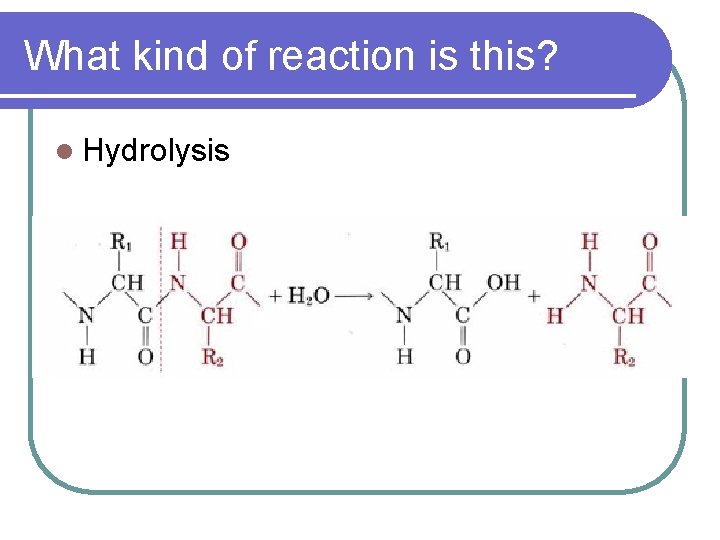
PROTEINS l Forms peptide bonds between amino acids l 1 amino acid- peptide l 2 amino acids- dipeptide l Many amino acids- polypeptide
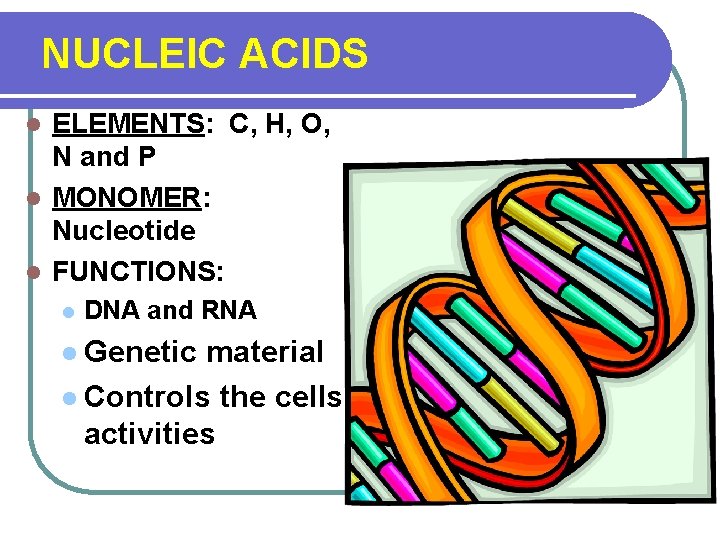
NUCLEIC ACIDS ELEMENTS: C, H, O, N and P l MONOMER: Nucleotide l FUNCTIONS: l l DNA and RNA l Genetic material l Controls the cells activities

DNA- carries the genetic code RNA- carries out the instructions
Organic molecules Video 14 minutes http: //www. youtube. com/watch? v=H 8 WJ 2 KENl K 0&list=SP 3 EED 4 C 1 D 684 D 3 ADF
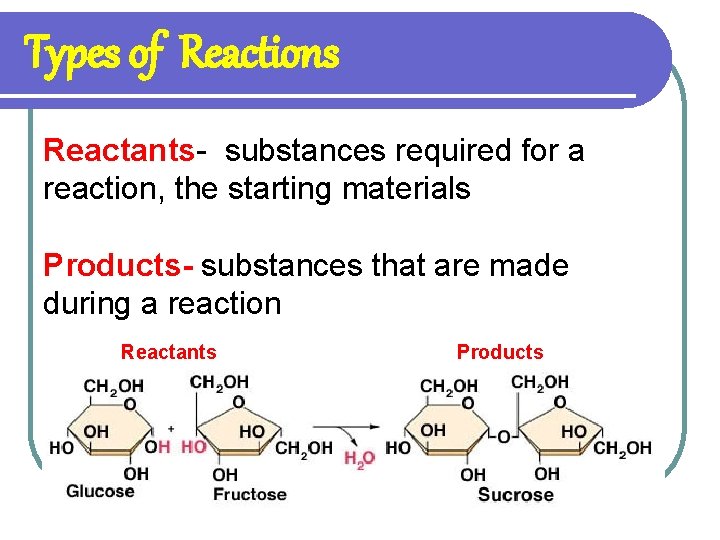
Types of Reactions Reactants- substances required for a reaction, the starting materials Products- substances that are made during a reaction Reactants Products

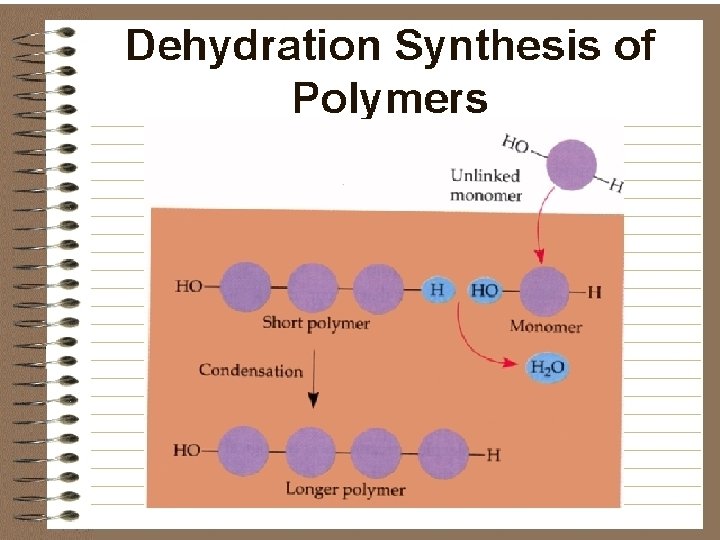
CONDENSATION / DEHYDRATION REACTIONS l CONDENSATION REACTIONS (DEHYDRATION SYNTHESIS REACTIONS) l Joining molecules together by removing water (-H and –OH are removed to make a water molecule) l C 6 H 12 O 6 + C 6 H 12 O 6 GLUCOSE C 12 H 22 O 11 + H 2 O MALTOSE WATER

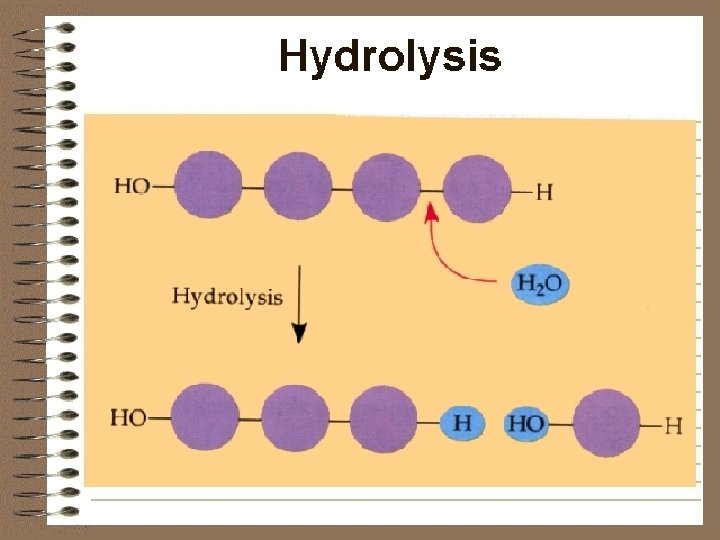
REACTIONS OF BIOLOGICAL COMPOUNDS l HYDROLYSIS l l REACTIONS Water is added to break apart molecules C 12 H 22 O 11 + H 2 O MALTOSE WATER C 6 H 12 O 6 + C 6 H 12 O 6 GLUCOSE

What kind of reaction is this? l Condensation
What kind of reaction is this? l Hydrolysis
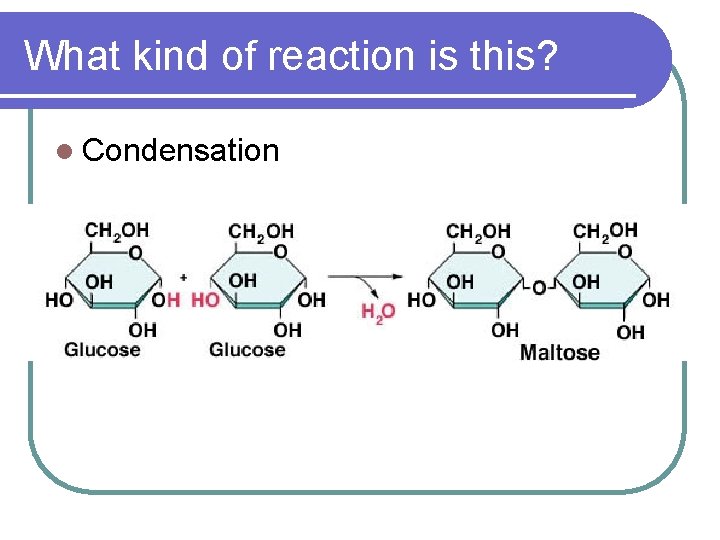
What kind of reaction is this? l Condensation

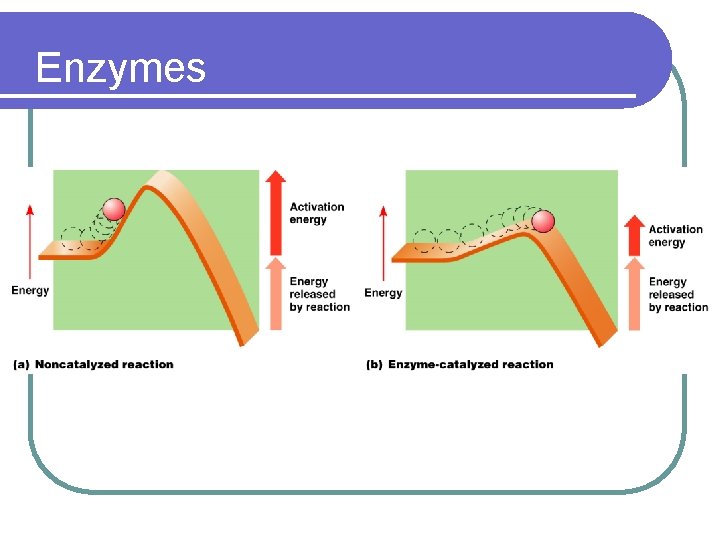
ENZYMES l CATALYSTS FOR BIOLOGICAL REACTIONS- speeds up a reaction l MOST ARE PROTEINS l LOWER THE ACTIVATION ENERGY NEEDED TO CARRY OUT A REACTION l INCREASES THE RATE OF REACTIONS
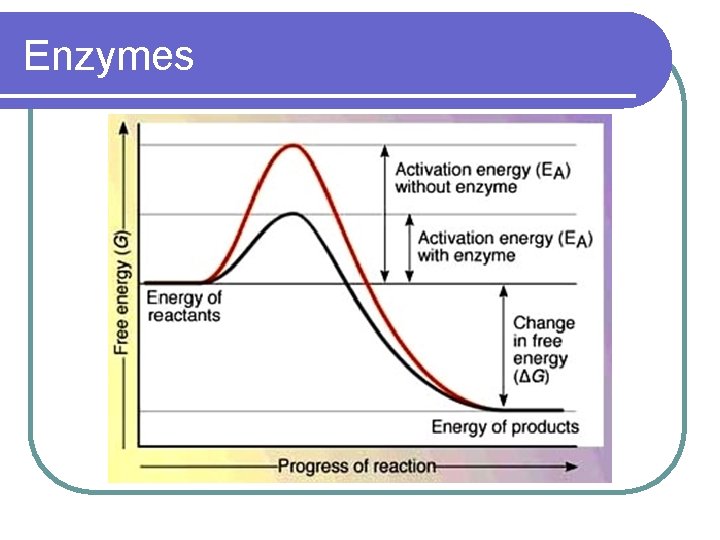
Enzymes
Enzymes
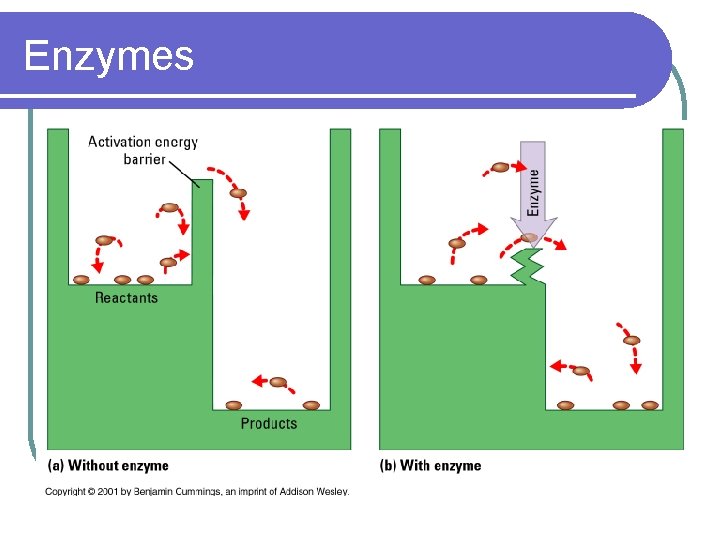
Enzymes

Activation energy l http: //www. ryancshaw. com/Files/micro/A nimations/Enzyme. Substrate/micro_enzyme-substrate. swf

ENZYME ACTION: LOCK & KEY MODEL l SUBSTRATE- SUBSTANCE AN ENZYME ACTS UPON l ACTIVE SITE- REGION WHERE SUBSTRATE AND ENZYME BONDS TOGETHER l PRODUCT IS RELEASED l ENZYME IS UNCHANGED
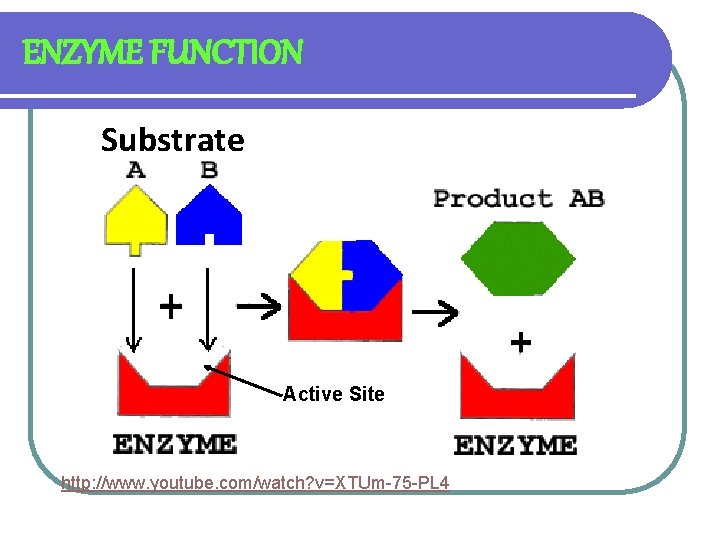
ENZYME FUNCTION Substrate Active Site http: //www. youtube. com/watch? v=XTUm-75 -PL 4
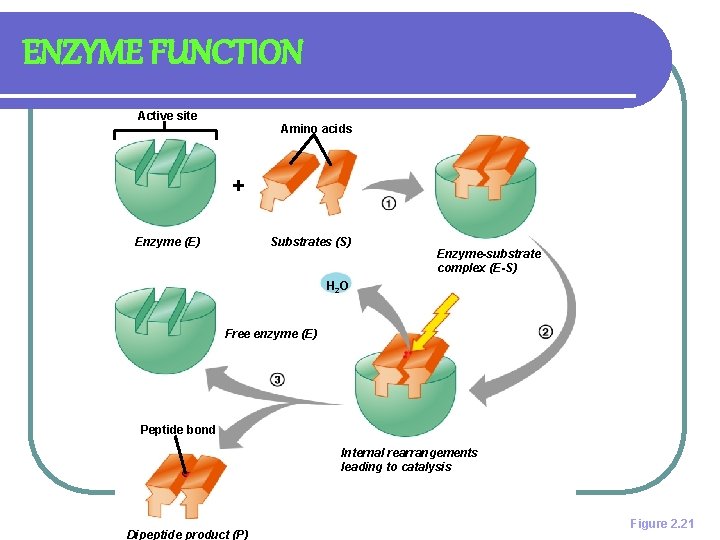
ENZYME FUNCTION Active site Amino acids + Enzyme (E) Substrates (S) Enzyme-substrate complex (E-S) H 2 O Free enzyme (E) Peptide bond Internal rearrangements leading to catalysis Dipeptide product (P) Figure 2. 21

Enzyme Activity l http: //www. lpscience. fatcow. com/jwanam aker/animations/Enzyme%20 activity. html l http: //highered. mcgrawhill. com/sites/0072495855/student_view 0/chapter 2/animation__how_enzymes_w ork. html l http: //www. biotopics. co. uk/other/enzyme. html

FACTORS AFFECTING ENZYME ACTION l TEMPERATURE LITTLE ACTIVITY AT LOW TEMPS l RATE INCREASES WITH TEMPERATURE l MOST ACTIVE TEMP IN HUMANS (37ºC) l ACTIVITY LOST WITH DENATURATION AT HIGH TEMPERATURE l l p. H l Concentration of substrate/enzyme
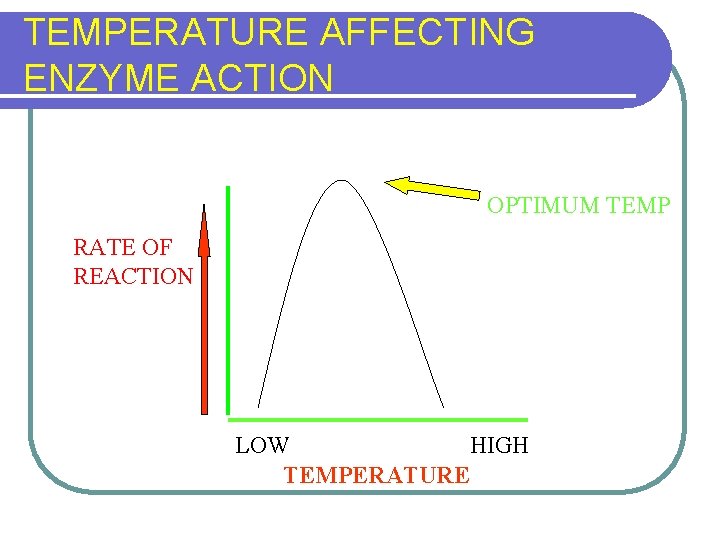
TEMPERATURE AFFECTING ENZYME ACTION OPTIMUM TEMP RATE OF REACTION LOW HIGH TEMPERATURE
- Slides: 46