INNOVATIVE LESSON PLAN PRASANTH M ST JACOBS TRAINING

INNOVATIVE LESSON PLAN PRASANTH M ST JACOB’S TRAINING COLLEGE

Coordination Chemistry ¿ Transition ô Form metals act as Lewis acids complexes/complex ions Fe 3+(aq) + 6 CN-(aq) [Fe(CN)6]3 -(aq) Lewis acid Lewis base Complex ion Ni 2+(aq) + 6 NH 3(aq) [Ni(NH 3)6]2+(aq) Lewis acid Lewis base Complex ion Complex with a net charge = complex ion Complexes have distinct properties

Coordination Chemistry ¿ Coordination compound ©Compound that contains 1 or more complexes ©Example ô [Co(NH 3)6]Cl 3 ô [Cu(NH 3)4][Pt. Cl 4] ô [Pt(NH 3)2 Cl 2]

Coordination Chemistry ¿ Coordination sphere ©Metal and ligands bound to it ¿ Coordination number ©number of donor atoms bonded to the central metal atom or ion in the complex ô Most common = 4, 6 ô Determined by ligands Larger ligands and those that transfer substantial negative charge to metal favor lower coordination numbers

Coordination Chemistry Complex charge = sum of charges on the metal and the ligands [Fe(CN)6]3 -
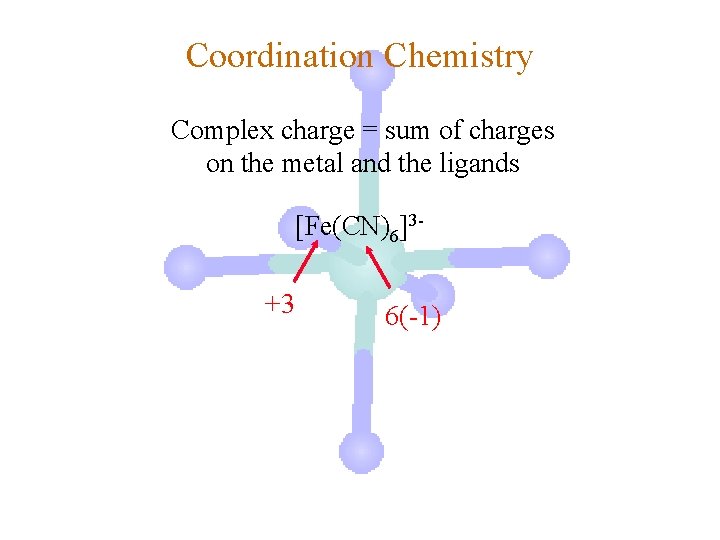
Coordination Chemistry Complex charge = sum of charges on the metal and the ligands [Fe(CN)6]3+3 6(-1)
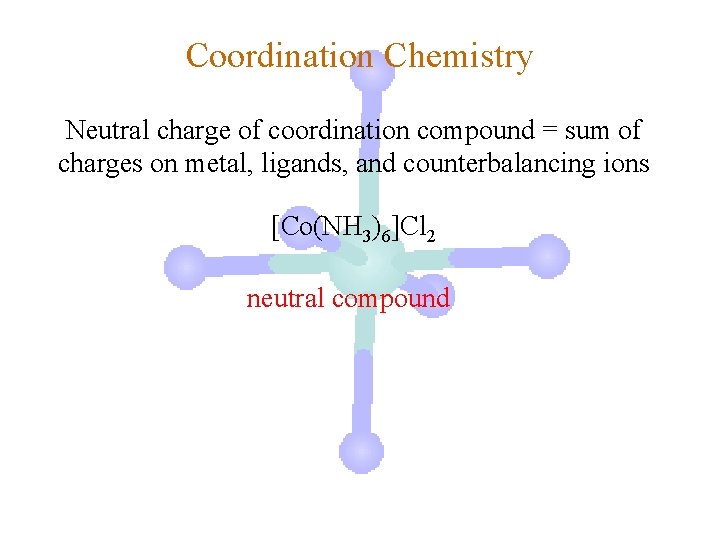
Coordination Chemistry Neutral charge of coordination compound = sum of charges on metal, ligands, and counterbalancing ions [Co(NH 3)6]Cl 2 neutral compound
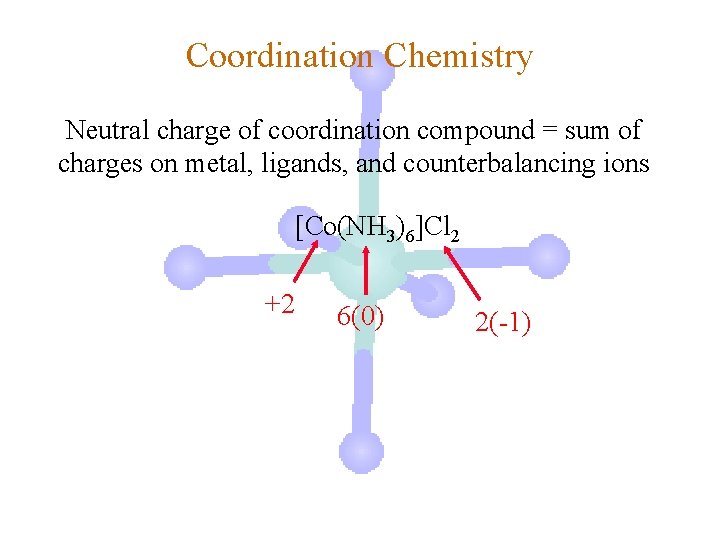
Coordination Chemistry Neutral charge of coordination compound = sum of charges on metal, ligands, and counterbalancing ions [Co(NH 3)6]Cl 2 +2 6(0) 2(-1)

Coordination Chemistry ¿ Ligands ©classified according to the number of donor atoms ©Examples ô monodentate ô bidentate =1 =2 ô tetradentate = 4 ô hexadentate = 6 ô polydentate = 2 or more donor atoms
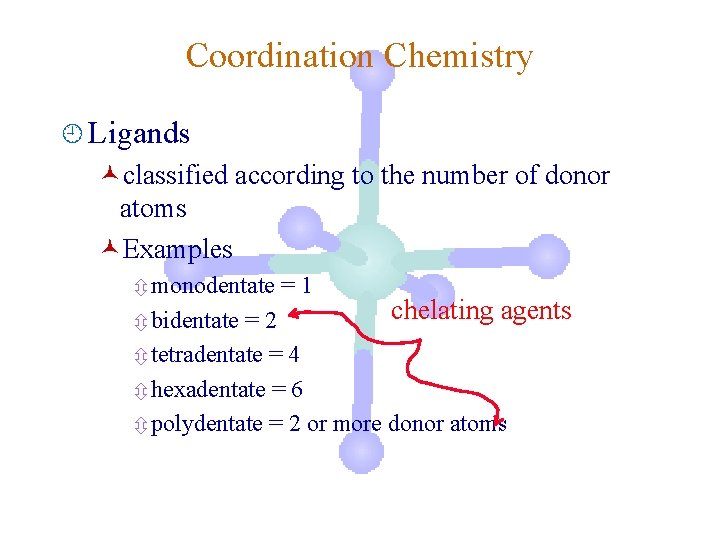
Coordination Chemistry ¿ Ligands ©classified according to the number of donor atoms ©Examples ô monodentate =1 chelating agents =2 ô tetradentate = 4 ô hexadentate = 6 ô polydentate = 2 or more donor atoms ô bidentate
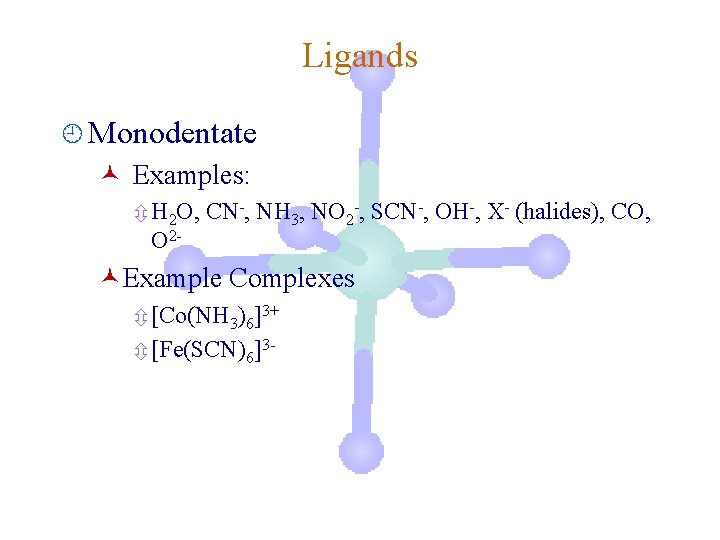
Ligands ¿ Monodentate © Examples: ô H 2 O, O 2 - CN-, NH 3, NO 2 -, SCN-, OH-, X- (halides), CO, ©Example Complexes ô [Co(NH 3)6]3+ ô [Fe(SCN)6]3 -
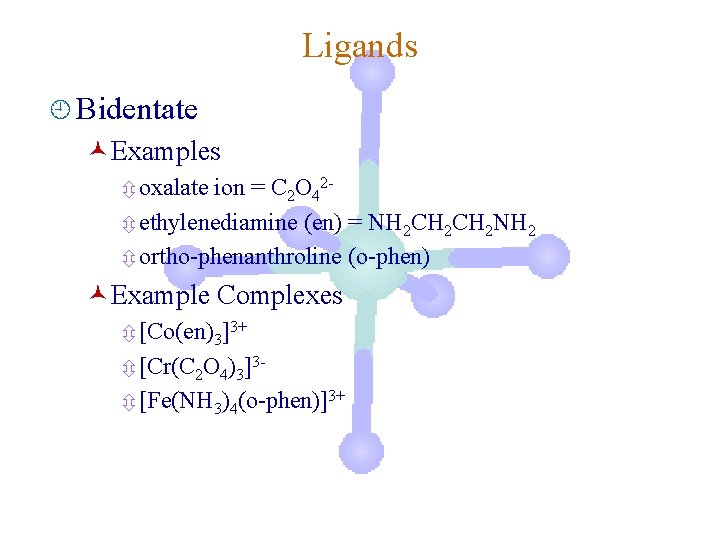
Ligands ¿ Bidentate ©Examples ô oxalate ion = C 2 O 42ô ethylenediamine (en) = NH 2 CH 2 NH 2 ô ortho-phenanthroline (o-phen) ©Example Complexes ô [Co(en)3]3+ ô [Cr(C 2 O 4)3]3ô [Fe(NH 3)4(o-phen)]3+
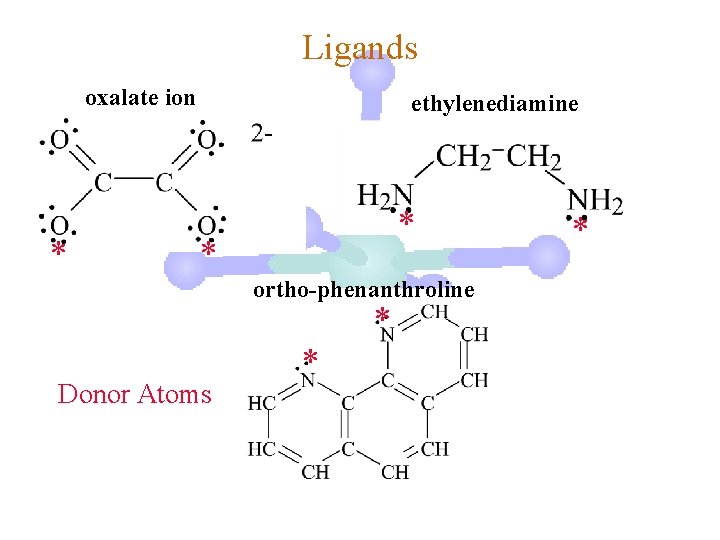
Ligands oxalate ion * ethylenediamine * * ortho-phenanthroline * Donor Atoms * *
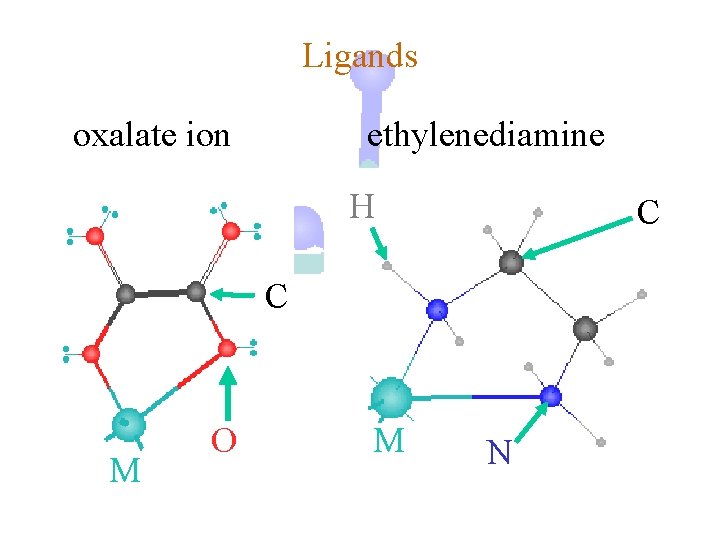
Ligands oxalate ion ethylenediamine H C C M O M N
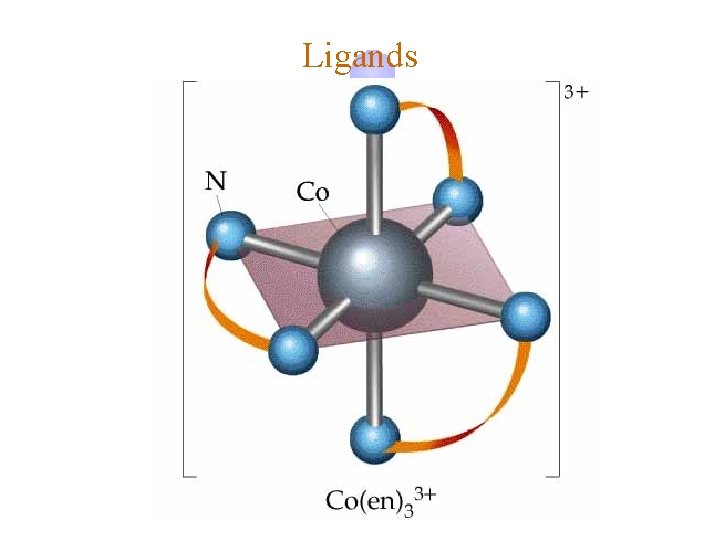
Ligands

Ligands ¿ Hexadentate © ethylenediaminetetraacetate (EDTA) = (O 2 CCH 2)2 N(CH 2 CO 2)24©Example Complexes ô [Fe(EDTA)]-1 ô [Co(EDTA)]-1
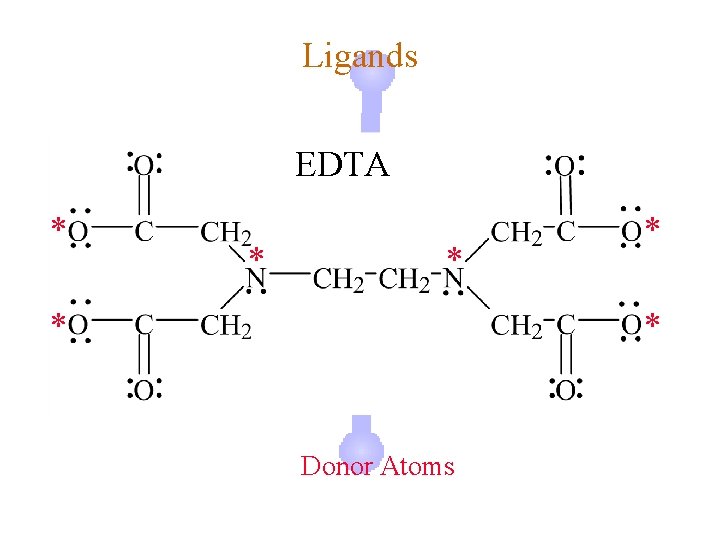
Ligands EDTA * * * Donor Atoms
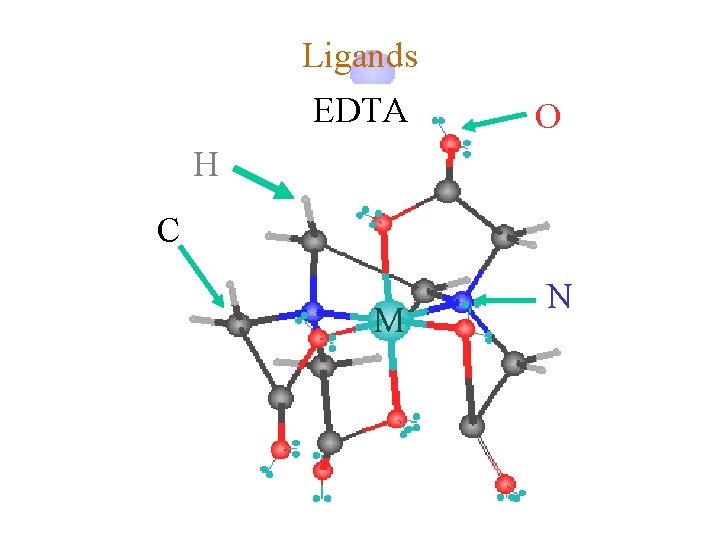
Ligands EDTA O H C M N
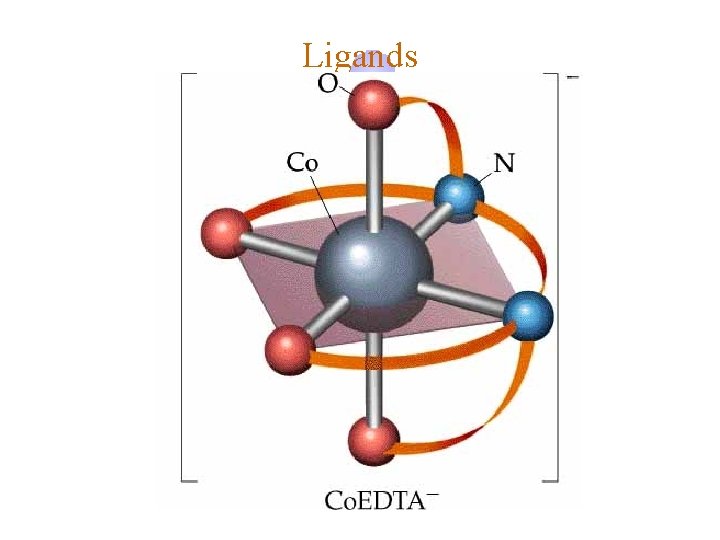
Ligands EDTA
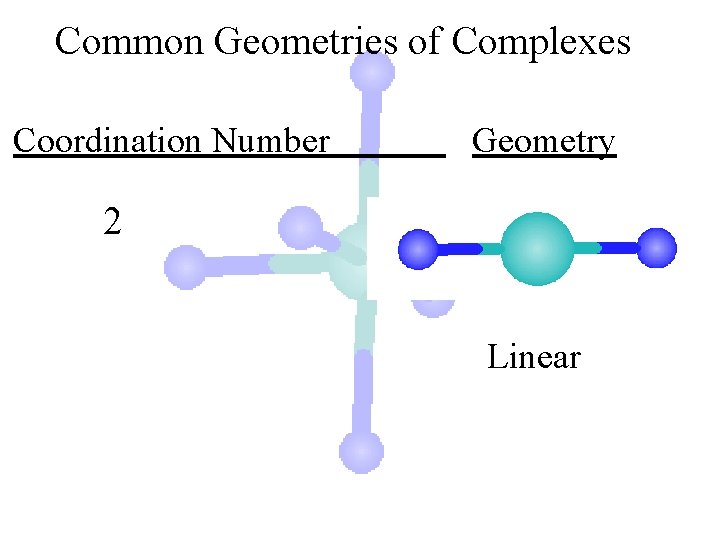
Common Geometries of Complexes Coordination Number Geometry 2 Linear
![Common Geometries of Complexes Coordination Number Geometry 2 Linear Example: [Ag(NH 3)2]+ Common Geometries of Complexes Coordination Number Geometry 2 Linear Example: [Ag(NH 3)2]+](http://slidetodoc.com/presentation_image_h/de01a45ea2d868aca11d7d697aa6dc79/image-21.jpg)
Common Geometries of Complexes Coordination Number Geometry 2 Linear Example: [Ag(NH 3)2]+
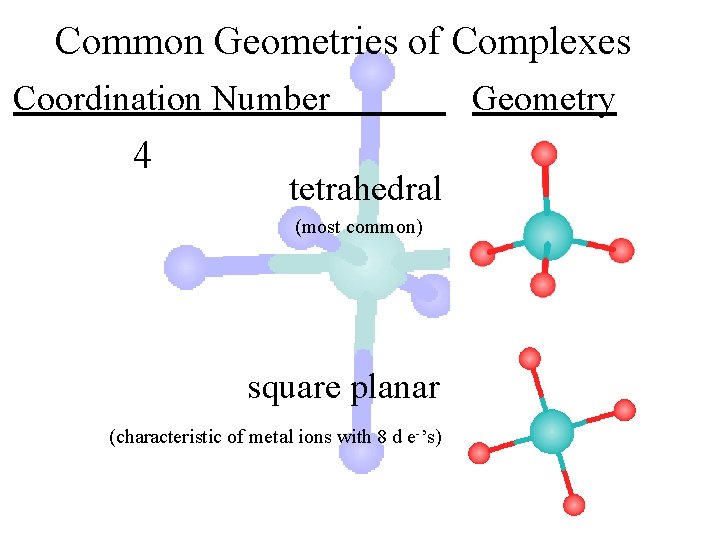
Common Geometries of Complexes Coordination Number 4 tetrahedral (most common) square planar (characteristic of metal ions with 8 d e-’s) Geometry
![Common Geometries of Complexes Coordination Number 4 tetrahedral Examples: [Zn(NH 3)4]2+, [Fe. Cl 4]- Common Geometries of Complexes Coordination Number 4 tetrahedral Examples: [Zn(NH 3)4]2+, [Fe. Cl 4]-](http://slidetodoc.com/presentation_image_h/de01a45ea2d868aca11d7d697aa6dc79/image-23.jpg)
Common Geometries of Complexes Coordination Number 4 tetrahedral Examples: [Zn(NH 3)4]2+, [Fe. Cl 4]- square planar Example: [Ni(CN)4]2 - Geometry
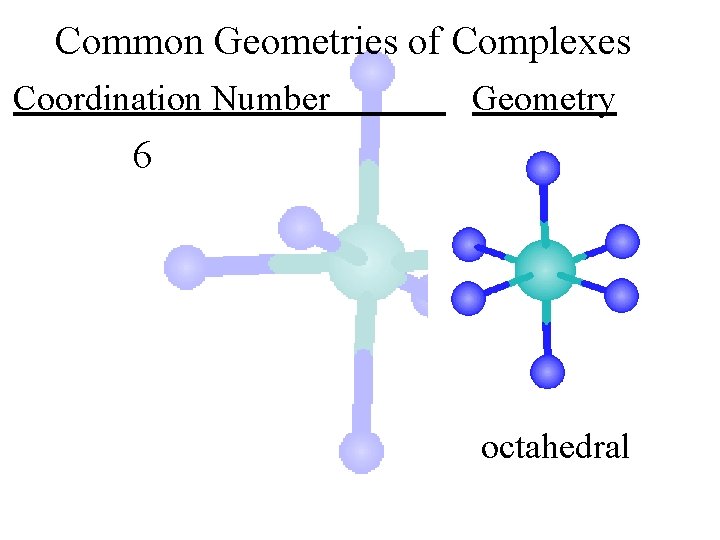
Common Geometries of Complexes Coordination Number Geometry 6 octahedral
![Common Geometries of Complexes Coordination Number Geometry 6 Examples: [Co(CN)6]3 -, [Fe(en)3]3+ octahedral Common Geometries of Complexes Coordination Number Geometry 6 Examples: [Co(CN)6]3 -, [Fe(en)3]3+ octahedral](http://slidetodoc.com/presentation_image_h/de01a45ea2d868aca11d7d697aa6dc79/image-25.jpg)
Common Geometries of Complexes Coordination Number Geometry 6 Examples: [Co(CN)6]3 -, [Fe(en)3]3+ octahedral
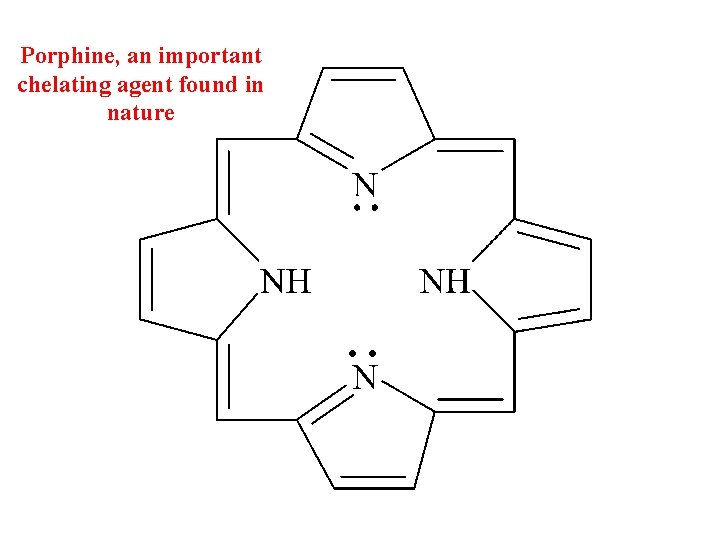
Porphine, an important chelating agent found in nature
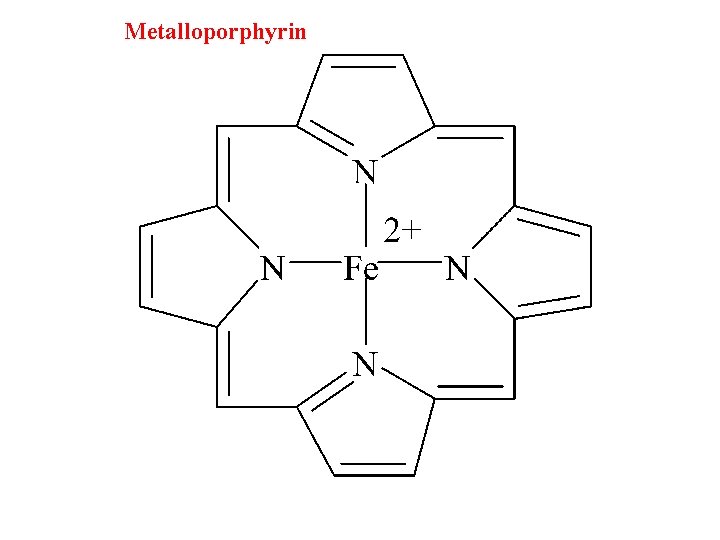
Metalloporphyrin
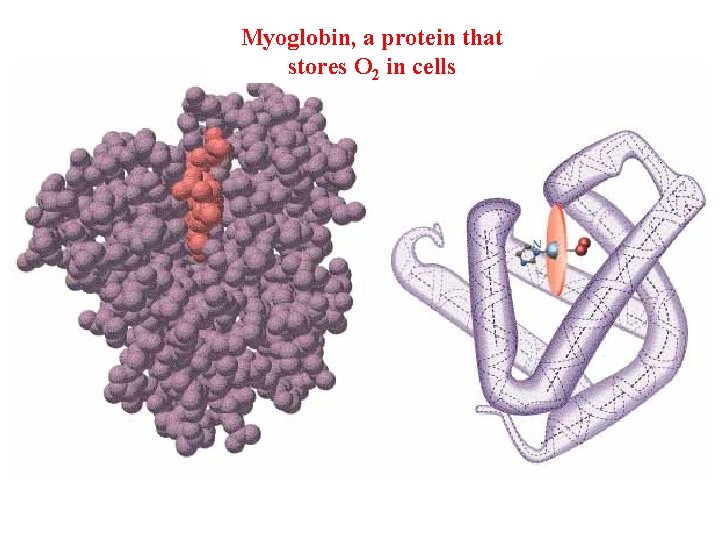
Myoglobin, a protein that stores O 2 in cells
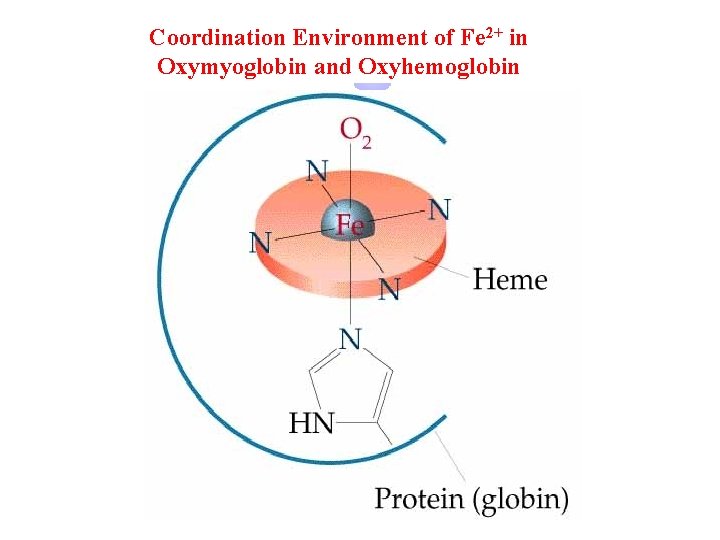
Coordination Environment of Fe 2+ in Oxymyoglobin and Oxyhemoglobin
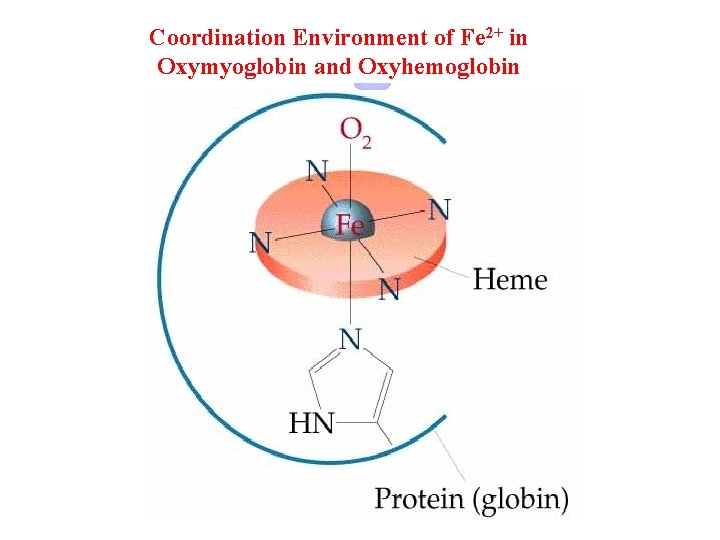
Coordination Environment of Fe 2+ in Oxymyoglobin and Oxyhemoglobin
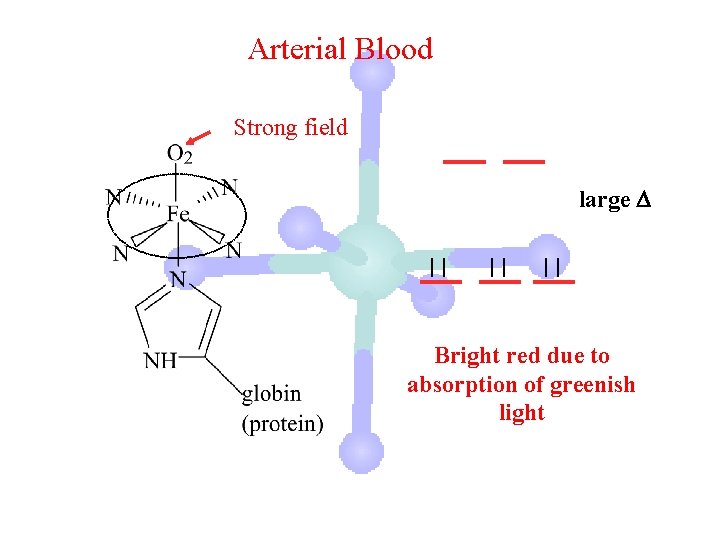
Arterial Blood Strong field large Bright red due to absorption of greenish light
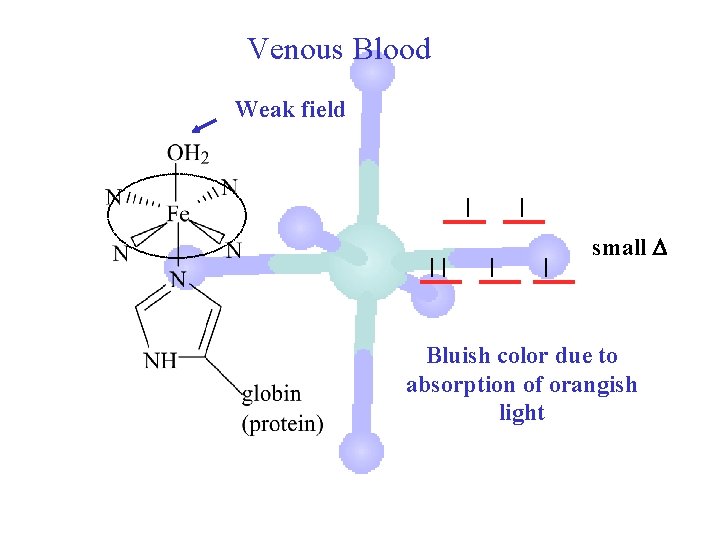
Venous Blood Weak field small Bluish color due to absorption of orangish light


End of Presentation
- Slides: 34