Innovations in Clinical Trial Designs Ori BenYehuda MD

Innovations in Clinical Trial Designs Ori Ben-Yehuda, MD Clinical Trials Center Cardiovascular Research Foundation & Columbia University New York, NY

Disclosures: Ori Ben-Yehuda, MD Consultant to Cardiovalve, Inc.

The Traditional Gold Standard • • Placebo or sham control Large Superiority endpoint Hard Endpoints – All cause mortality as single endpoint- ideal – Composite endpoint (eg- cv death/mi/stroke)

Challenges • Expensive • Not real world • Composite Endpoints- endpoints not equal in importance – Mortality plus MI plus Revascularization- but periprocedural MI does not equal death – How does one integrate Qo. L and Functional status into a composite • Time to event analysis- does not account for repeated event • Low event rates- very large sample sizes

Innovations • Hierarchal composite endpoints – Finkelstein-Schoenfeld and Pocock’s Win. Ratio methodology • Adaptive design • Pragmatic trials • Embedding clinical trials into EMR (EHR)

Finkelstein-Schoenfeld Methodology • Compares patients along a hierarchy of endpoints • Winners are declared • Example of hierarchy – Death – Myocardial Infarction – Hospitalization for heart failure – Quality of Life Questionnaire

FS Methodology • Each patient from group A is compared to every patient from group B • If patient from group A is alive and patient from group B has died, patient from group A is declared the winner • If both are alive, or both are dead then go on to the next line in the hierarchy, until a winner is declared or a final tie is declared • Number of wins in each group is counted • Gives greater weight to endpoints that are higher in the hierarchy • Generates a p value, not an effect size – Win Ratio provides insight into effect size

Advantages to FS Methodology • Order of comparison determines importance of endpoints in the hierarchal composite • Allows integration of Qo. L and Functional Status (NYHA, 6 minute walk test, etc) • Increases power
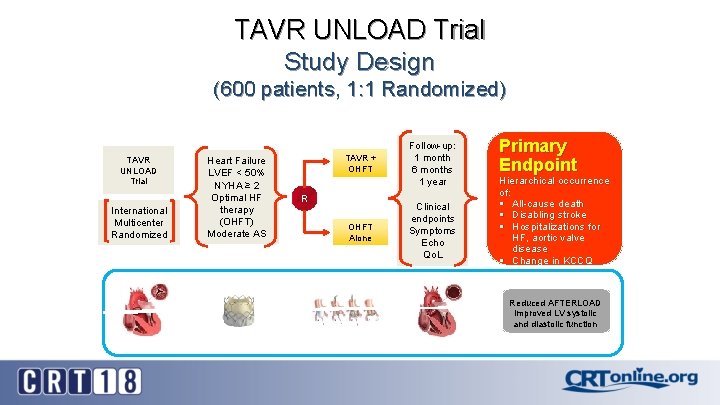
TAVR UNLOAD Trial Study Design (600 patients, 1: 1 Randomized) TAVR UNLOAD Trial International Multicenter Randomized Heart Failure LVEF < 50% NYHA ≥ 2 Optimal HF therapy (OHFT) Moderate AS TAVR + OHFT R OHFT Alone Follow-up: 1 month 6 months 1 year Clinical endpoints Symptoms Echo Qo. L Primary Endpoint Hierarchical occurrence of: § All-cause death § Disabling stroke § Hospitalizations for HF, aortic valve disease § Change in KCCQ KCC Reduced AFTERLOAD Improved LV systolic and diastolic function

TAVR- UNLOAD: FS Methodology Comparison of each potential subject pair (1 from TAVR group and 1 from OHFT) Step 1 If 1 or both subjects die Subject in TAVR group dies first Subject in OHFT group dies first Both subjects die on the same day Step 2 If no ranking yet available Subject in TAVR group suffers a more severe disabling stroke (m. RS 2 to 5) Subject in OHFT group suffers a more severe disabling stroke (m. RS 2 to 5) Both subjects suffer a disabling stroke of the same severity Step 3 If no ranking yet available Subject in TAVR group suffers the stroke first Subject in OHFT group suffers the stroke first Both subjects suffer a disabling stroke of the same severity on the same day ----------------------------------------------------Step 6 If no ranking yet available Subject in TAVR group is in worse KCCQ category at one year** Subject in OHFT group is in worse KCCQ category at one year Both subjects are in same KCCQ category at one year How event is assessed Favors OHFT Favors TAVR Go to step 2 Favors OHFT Favors TAVR Go to step 3 Favors OHFT Favors TAVR Go to step 4 Favors OHFT Favors TAVR Tie

ACTIVE Trial- Cardioband in MR Co-primary endpoint I • Prevalence of MR ≤ 2+ at one year. Co-primary endpoint II • Hierarchical comparison between the Device and Control groups using Finkelstein-Schoenfeld methodology: • Time to cardiovascular death • Number of heart failure hospitalizations • Improvement in 6 Minute Walk Test vs. baseline (≥ 40 meters improvement) • Improvement in Kansas City Cardiomyopathy Questionnaire at one year vs. baseline (KCCQ; ≥ 10 points improvement)

Adaptive Design (Mehta and Pockock) • Addresses sample size re-estimation – Initial sample size based on assumptions • Event rates in control arm • Effect size (is it clinically relevant) • Measurement standard deviation • What if initial sample size is wrong? – – • Too big – waste of resources Too small- risk of type II error (false negative trial) Traditional interim look – alpha penalty (0. 05 threshold adjusted)

Adaptive Methodology • Interim look without alpha penalty • Identify a predefined “promising zone” based on conditional power of the trial at interim analysis – Favorable- no change in sample size – Promising- sample size re-estimation – Unfavorable – no change in sample size • Because trial may still be negative, no alpha penalty • Can be done on blinded data as well as unblinded • Best predefined and handled by the DSMB

Pragmatic vs. Explanatory Trials Terms coined by Schwartz & Lellough in 1967 Dialogues Clin Neurosci. 2011 Jun; 13(2): 217– 224.

Explanatory vs. Pragmatic Trials • • Explanatory Highly Controlled Setting Strict I & E If and how an intervention works? High internal validity • • • Pragmatic Broad range of patients Looser I & E Does intervention work in real life? External validity Patient centric outcomes

PRECIS Tool: Pragmatic-Explanatory Continuum Indicator Summary Example in Pragmatic Trial Domains 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Eligibility criteria Flexibility of the experimental intervention Practitioner expertise (experimental) Flexibility of the comparison Practitioner expertise (comparison) Follow-up intensity Outcomes Participant compliance Practitioner adherence Primary outcomes. 1. 2. There are no I &E criteria Practitioners are not constricted by guidelines on how to apply the intervention 3. The experimental intervention is applied by all 4. The best alternative treatments are used 5. The comparative treatment is applied by all 6. No formal follow-up sections 7. The outcome is a simple but meaningful one 8. There are no plans to improve or alter compliance 9. No strategy to motivate practitioner's adherence. 10. Intention to treat J Clin Epidemiol. 2009 May; 62(5): 464 -75.

Limitations of Pragmatic Trials • Dilution of effect- may require very large sample size to avoid type II error, especially for medium efficacy Rx • Long follow up may be required (ALLHAT took 8 yrs!) • Geographic variation • Less applicable to evaluation of devices/techniques that require high expertise • Who pays?

TAPAS: Thrombus Aspiration during Primary Percutaneous Coronary Intervention • Single center • 1071 patients enrolled • Primary end point: a myocardial blush grade of 0 (absent reperfusion) or 1 (minimal myocardial reperfusion) – 17. 1% in the thrombus-aspiration group – 26. 3% in the conventional-PCI group – risk ratio, 0. 65; 95% confidence interval [CI], 0. 51 to 0. 83; P<0. 001) • Death in 11 of 529 (2. 1%) and 21 of 531 (4. 0%) (risk ratio, 0. 52; 95% CI, 0. 26 to 1. 07; P=0. 07) N Engl J Med 2008; 358: 557 -567
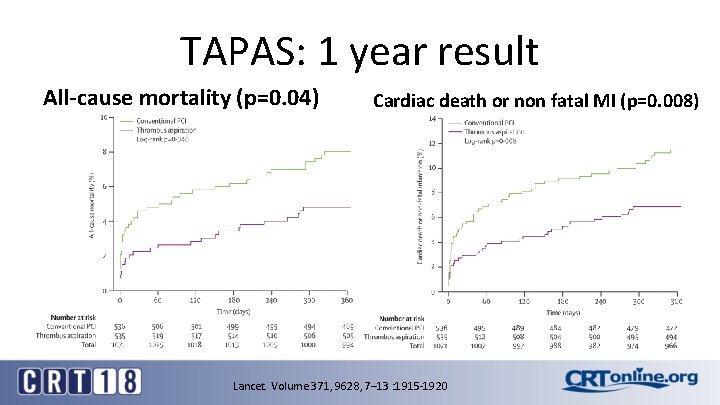
TAPAS: 1 year result All-cause mortality (p=0. 04) Cardiac death or non fatal MI (p=0. 008) Lancet. Volume 371, 9628, 7– 13 : 1915 -1920

Thrombus Aspiration in ST Elevation Myocardial Infarction in Scandinavia (TASTE) • Randomized within the SCAAR/SWEDEHEART • Within the registry all patients with suspected STEMI and planned PCI after angiography • Recruitment: oral consent followed by written consent • 60% participation • 1: 1 randomization to thrombus aspiration followed by PCI or to PCI only • 7244 patients underwent randomization (Sweden, Iceland, Denmark)
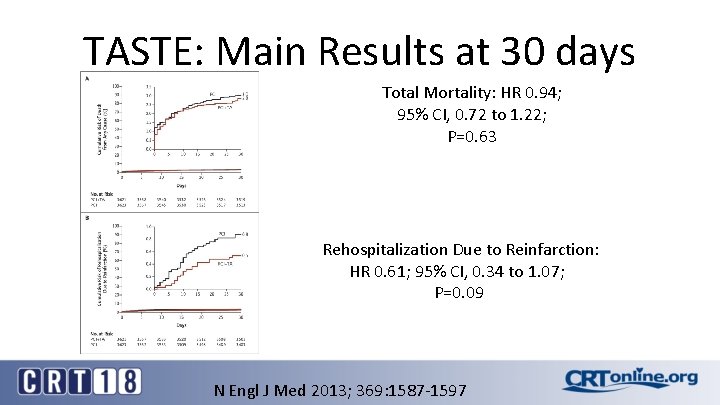
TASTE: Main Results at 30 days Total Mortality: HR 0. 94; 95% CI, 0. 72 to 1. 22; P=0. 63 Rehospitalization Due to Reinfarction: HR 0. 61; 95% CI, 0. 34 to 1. 07; P=0. 09 N Engl J Med 2013; 369: 1587 -1597
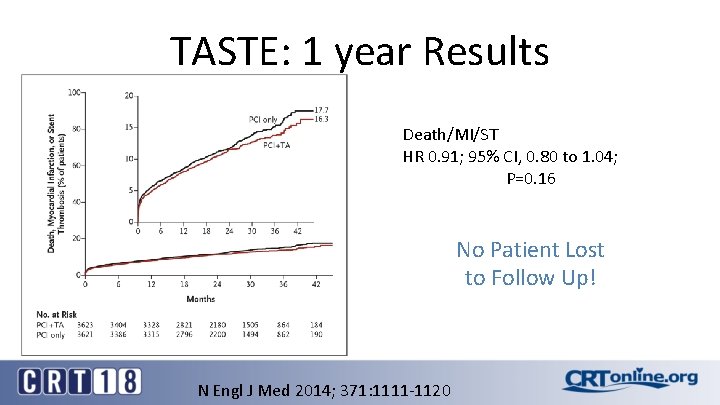
TASTE: 1 year Results Death/MI/ST HR 0. 91; 95% CI, 0. 80 to 1. 04; P=0. 16 No Patient Lost to Follow Up! N Engl J Med 2014; 371: 1111 -1120
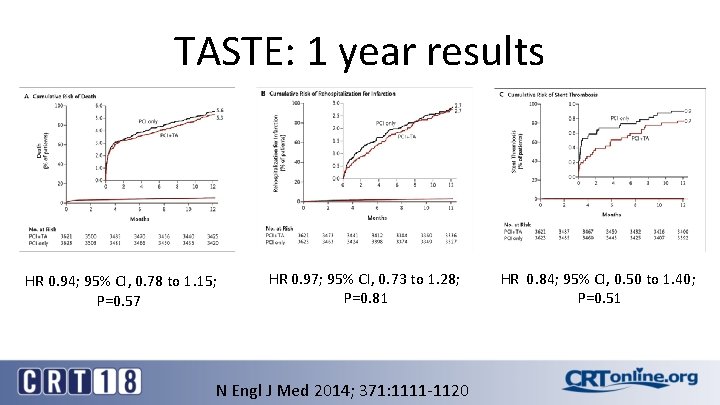
TASTE: 1 year results HR 0. 94; 95% CI, 0. 78 to 1. 15; P=0. 57 HR 0. 97; 95% CI, 0. 73 to 1. 28; P=0. 81 N Engl J Med 2014; 371: 1111 -1120 HR 0. 84; 95% CI, 0. 50 to 1. 40; P=0. 51

Cost Typical Registration Device Trial • $20, 000 per patient Registry Trial such as TASTE • $50 per patient 400 Fold Difference! >99% Discount


Limitations and Challenges of TASTE Approach • No CEC • Limited data elements • Worked well in SWEDEHEART – Easy to enroll- common language – Well organized and tested database – Single payer system • Adoption is slow

EHR in Clinical Trials Embedding trials into routine clinical care EHR will identify patients eligible by I & E Electronic enrollment F/up by collecting data in EHR Advantages: real world, therapy incorporated into routine care • Good for large pragmatic trials: eg, asa for primary prevention • • •

Thank You
- Slides: 28