Innate natural immune functions in poultry reproductive organs



















- Slides: 38

Innate (natural) immune functions in poultry reproductive organs Drh. Bambang Ariyadi, MP. , Ph. D

INTRODUCTION • The hen ovary and oviduct could be infected by various pathogenic microorganisms: – Salmonella enteritidis – Mycoplasma melagridis – bronchitis viruses – avian influenza viruses • Microorganisms colonising the cloaca may ascend the oviduct through the vagina and uterus. • The immune function of the ovary and oviduct is essential to protect these tissues from infection as well as for the production of hygienic eggs.

PRIMARY BARRIERS TO INFECTIVE AGENTS IN THE OVARY AND OVIDUCT • Many pathogens may enter the ovarian follicles through the blood stream. • Intravenously injected carbons were phagocytised by fibroblast-like cells in theca interna capillary beds. • The mucosal surface of the oviduct is lined with ciliated pseudostratified epithelium. • The movement of the cilia may remove the luminal microorganisms by sending them out of the oviduct.

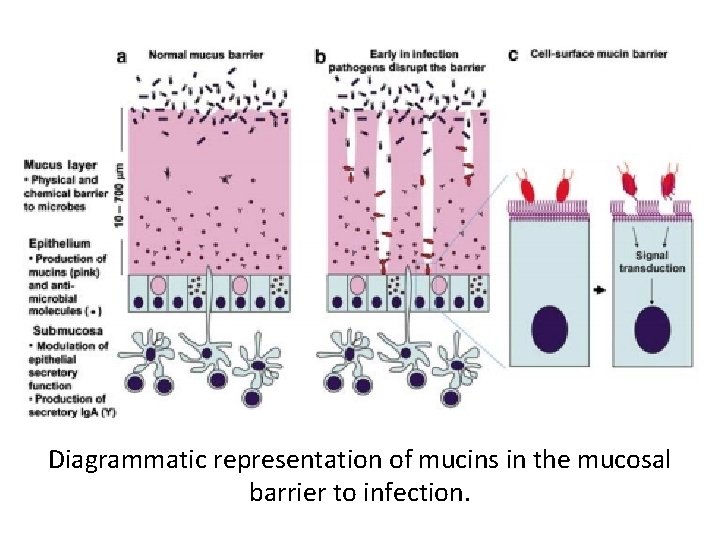
Diagrammatic representation of mucins in the mucosal barrier to infection.

• In each oviductal segment, the apical surface of the epithelial cells is overlaid with mucin substances that may block the attachment of microorganisms to the cells. • Tight junctions of mucosal epithelium are responsible for a paracellular barrier that protects underlying tissue against the most toxic materials or pathogens. • Expression of mucin and claudins in the mucosa of the oviduct is lower in non-laying hens than in laying hens, their expressions are upregulated by estrogen.

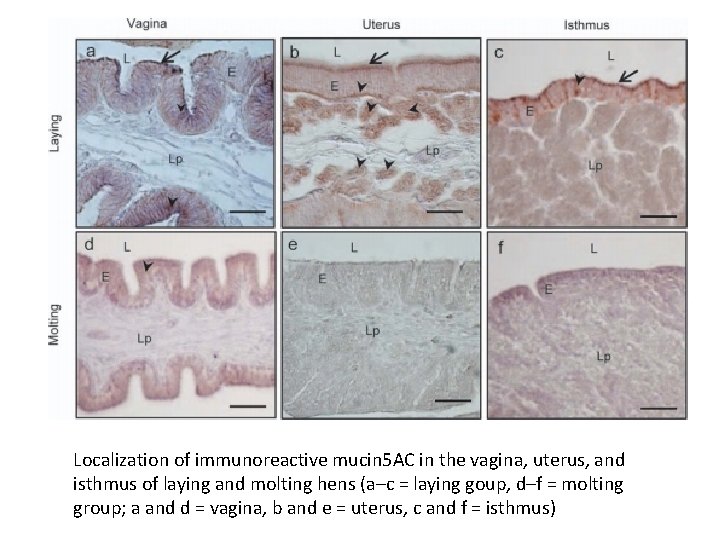
Localization of immunoreactive mucin 5 AC in the vagina, uterus, and isthmus of laying and molting hens (a–c = laying goup, d–f = molting group; a and d = vagina, b and e = uterus, c and f = isthmus)

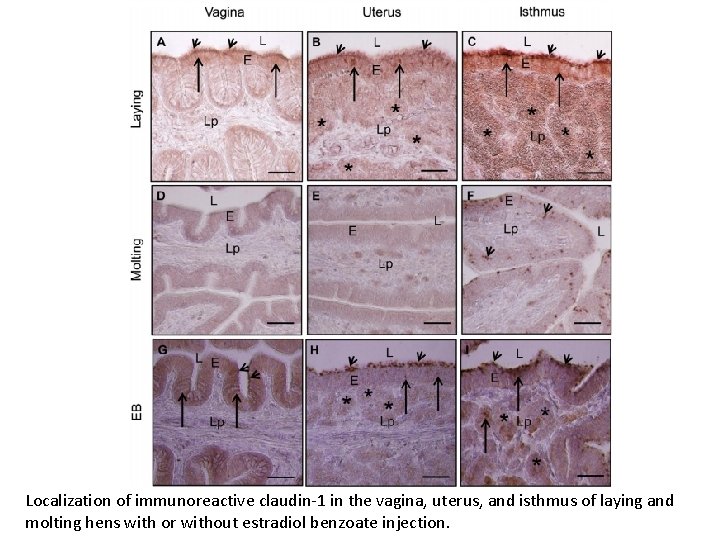
Localization of immunoreactive claudin-1 in the vagina, uterus, and isthmus of laying and molting hens with or without estradiol benzoate injection.

IMMUNOCOMPETENT CELLS IN FEMALE REPRODUCTIVE ORGANS • Cellular and humoral immuno-response play roles in local host defence. • Antigen presenting cells expressing MHC class. II such as dendritic cells and macrophages present antigens to CD 4+T cells. • The activated CD 4+ T cells activate B cells to differentiate and synthesise antibodies. • The other CD 4+T cells may stimulate phagocytic activity of macrophages. • The Ig. Y secreted in the follicles may be incorporated into the yolk as maternal immunoglobulins.

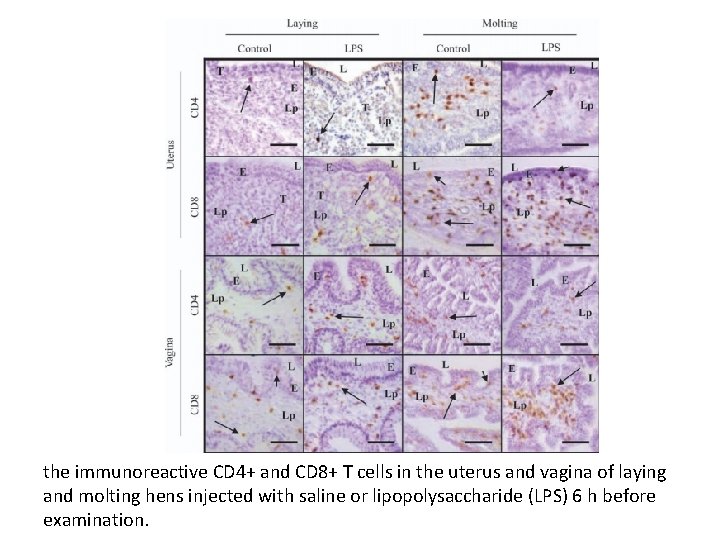
the immunoreactive CD 4+ and CD 8+ T cells in the uterus and vagina of laying and molting hens injected with saline or lipopolysaccharide (LPS) 6 h before examination.

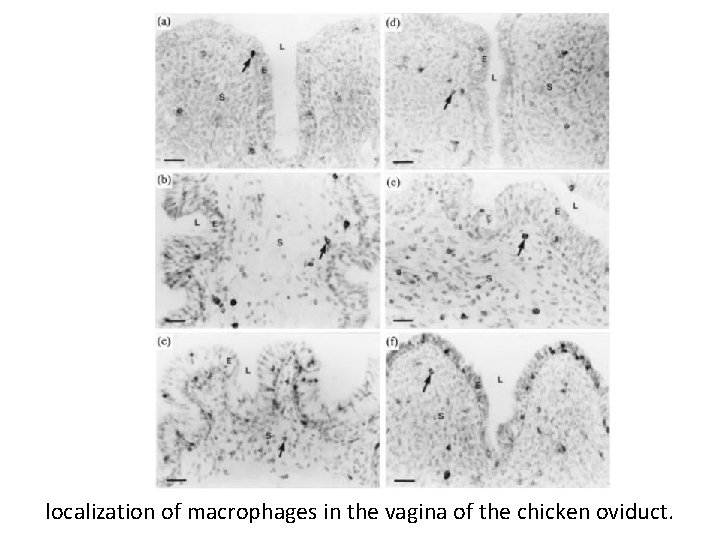
localization of macrophages in the vagina of the chicken oviduct.

HUMORAL FACTORS FOR IMMUNE DEFENCE SYSTEM IN FEMALE REPRODUCTIVE ORGANS • Humoral antimicrobial factors also play an important role in the defence of the mucosa or incorporated into the eggs. • Lysozyme, an enzyme that kills Gram-positive bacteria by breaking down their cell walls, is present in saliva, tears, and egg white. • This enzyme is synthesised in the oviduct and incorporated into the albumen, and possibly in the eggshell membrane and eggshell matrix.

• Avidin, a host defence protein involved in antimicrobial action, is also synthesised in the oviduct and incorporated in the albumen. • The mucosal epithelial cells that were localised at the bottom region of the secondary folds of the hen vagina show peroxidase activity. • Avian β-defensins (Av. BD) are expressed in hen ovary and oviduct and the expression may be regulated by the interaction of Toll-like receptors (TLRs) and their ligands.

TOLL-LIKE RECEPTORS IN FEMALE REPRODUCTIVE ORGANS • Toll-like receptors (TLRs) are elements that recognise microbial agents in the innate immune system. • In hen ovary, m. RNAs of TLR 1 -2, 2 -1, 3 – 5, 7, 15 and 21 are expressed. • In the oviduct of sexually mature birds all types of TLRs apart from TLR 1 -1 are expressed. • The expressions of TLR 2 -1, 2 -2 and 4 are abundant and differ with respect to sexual maturation in the chicken vagina.

PROINFLAMMATORY CYTOKINES IN FEMALE REPRODUCTIVE ORGANS • Proinflammatory cytokines and chemokines are critical in initiating innate and adaptive immunoresponses as well as local inflammatory responses. • IL-1β may induce T cell proliferation, fever, and acute phase response. • IL-6 may regulate macrophage differentiation, T cell activation and immunoglobulin production by B cells.

AVIAN β-DEFENSINS • Defensins are very common cationic antimicrobial peptides among vertebrates. • They actively kill Gram-positive and Gramnegative bacteria, protozoa as well as some fungi and enveloped viruses. • Av. BD 11 detected in the vitelline membrane has antimicrobial activity against both Gram-positive and Gram-negative bacteria.

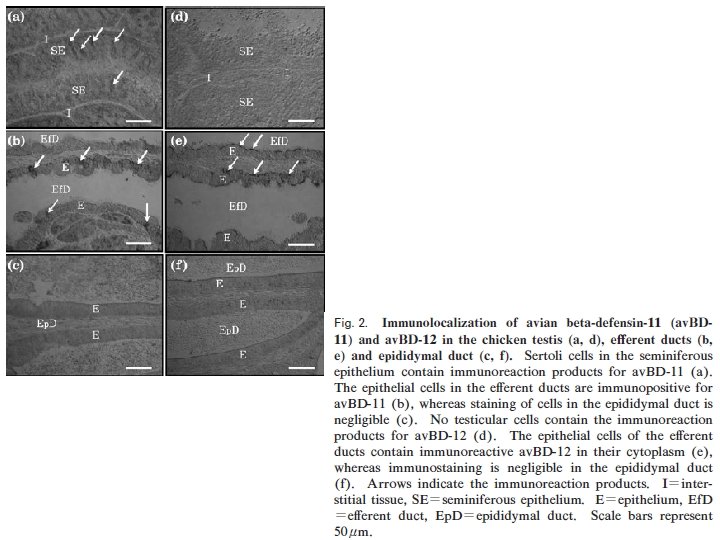
SYNTHESIS OF AVIAN ß-DEFENSINS IN THE OVARY AND OVIDUCT • Av. BDs genes are expressed in the hen ovary. • Av. BDs are identified in each oviductal segment spanning the infundibulum to the vagina. • Gallin (ovodefensin), a member of the avian defensin family with antimicrobial activity is synthesised in the tubular glands of the magnum. • The expression of Av. BD 11 in the oviduct may be enhanced with sexual maturation by the effects of estrogen.

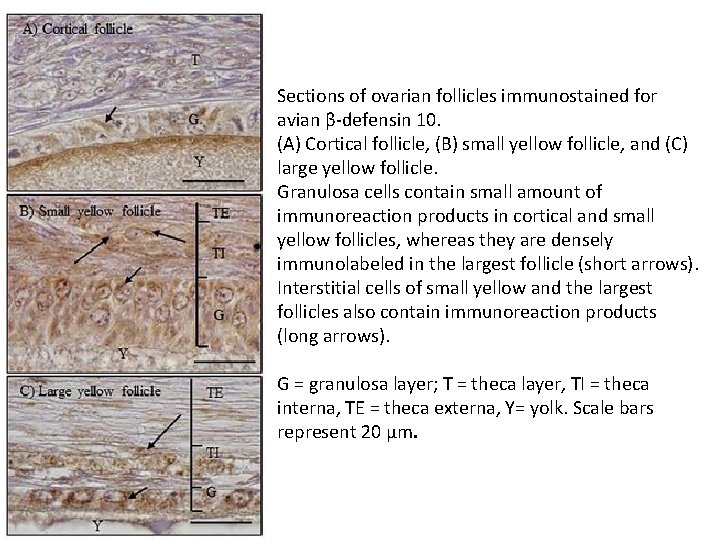
Sections of ovarian follicles immunostained for avian β-defensin 10. (A) Cortical follicle, (B) small yellow follicle, and (C) large yellow follicle. Granulosa cells contain small amount of immunoreaction products in cortical and small yellow follicles, whereas they are densely immunolabeled in the largest follicle (short arrows). Interstitial cells of small yellow and the largest follicles also contain immunoreaction products (long arrows). G = granulosa layer; T = theca layer, TI = theca interna, TE = theca externa, Y= yolk. Scale bars represent 20 μm.

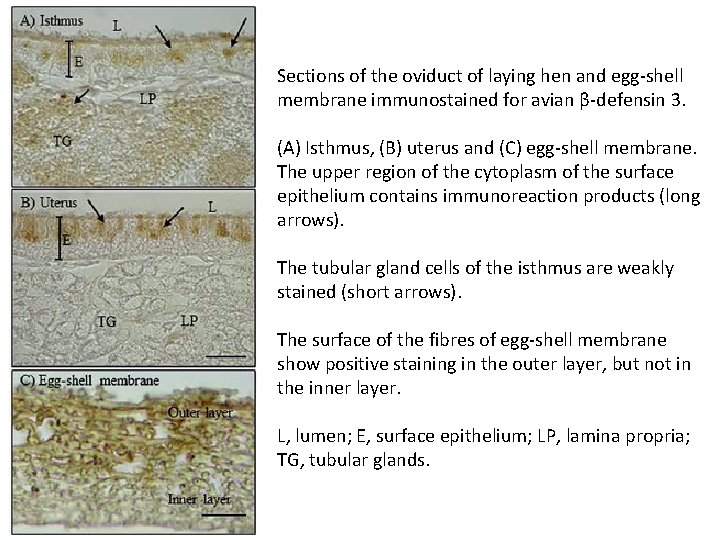
Sections of the oviduct of laying hen and egg-shell membrane immunostained for avian β-defensin 3. (A) Isthmus, (B) uterus and (C) egg-shell membrane. The upper region of the cytoplasm of the surface epithelium contains immunoreaction products (long arrows). The tubular gland cells of the isthmus are weakly stained (short arrows). The surface of the fibres of egg-shell membrane show positive staining in the outer layer, but not in the inner layer. L, lumen; E, surface epithelium; LP, lamina propria; TG, tubular glands.

SECRETION OF AVIAN ß-DEFENSINS TO EGGS • The perivitelline layer contains immunopositive Av. BD in the growing follicles. • Av. BDs may be secreted by the granulosa cells. • Gallin, a member of the defensin family, is synthesised in the magnum glandular cells and secreted into the egg albumin. • Av. BD 3 synthesised in the uterus may be secreted to coat eggshell membrane, while Av. BD 3, 10 and 12 may be incorporated into the eggshell matrix.

• The eggshell and its membrane exhibit antimicrobial activities. • Antimicrobial proteins including β-N-acetylglucosaminidase, lysozyme and ovo-transferrin have been identified in the eggshell membrane. • Presence of antimicrobial proteins such as L-type lysozyme, ovotransferrin, ovocalyxin-proteins has also been reported in the eggshell matrix. • Av. BDs in the albumen, eggshell membrane and eggshell matrix may form the defence system of the egg in combination with other antimicrobial proteins.

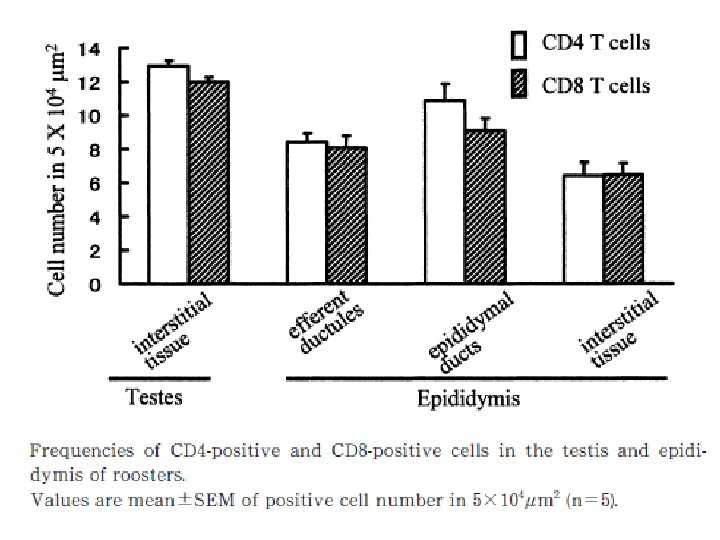
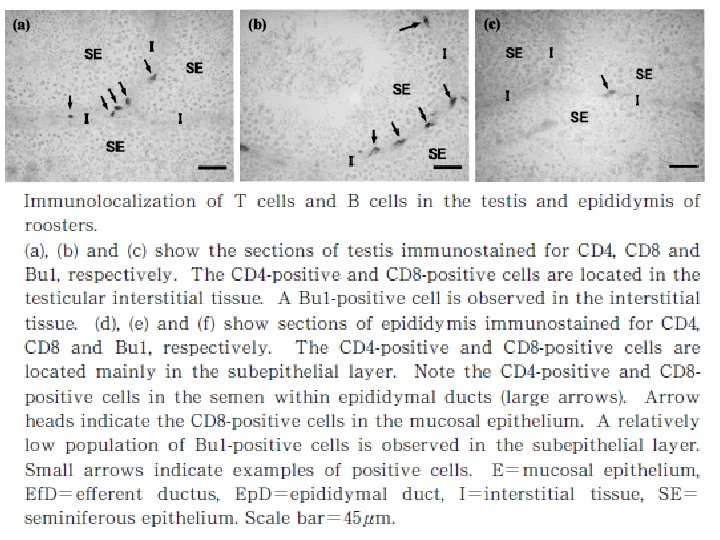
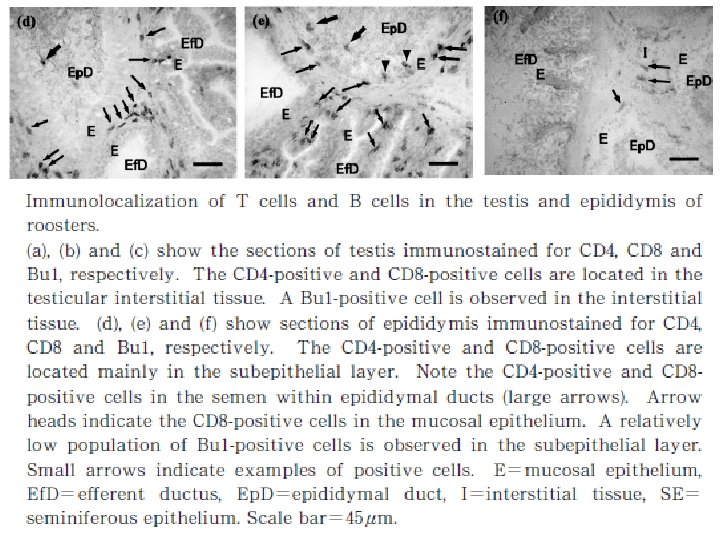
Natural immune function in male • The testis, epididymis and ductus deference are located in the abdominal cavity in birds. • The testis consists of numerous tubules seminiferous containing germ cells and Sertoli cells that form blood-testis barrier, narrow interstitial tissue and tunica albuginea covering the surface of testis. • The tubules seminiferous connect with the rete testis, which drains to the ductus system in epididymis, namely the ductules eferent and ductus epididymal.

• The male reproductive organs are the site that maybe infected by pathogens such as avian leukosis virus, herpesvirus, mycoplasma, and bronchitis virus. • Normal immune function in male reproductive organs is important to protect them from such pathogenic infection. • In the epididymis, sperm and germ cells detached from the seminiferous epithelium were phagocytosed by mucosal epithelial cells and macrophages, suggesting that degraded germ cells and un-ejaculated sperm in male genital tract are absorbed by them.

• if the immune functions are imbalanced, the immune response to germ cells and spermatozoa may cause reproductive disorder to decrease fertility. • Thus the immune system plays significant roles in defense to pathogens and also may affect the reproductive functions. • Although the population of B cells is relatively low, probably to avoid the induction of antisperm antibody.

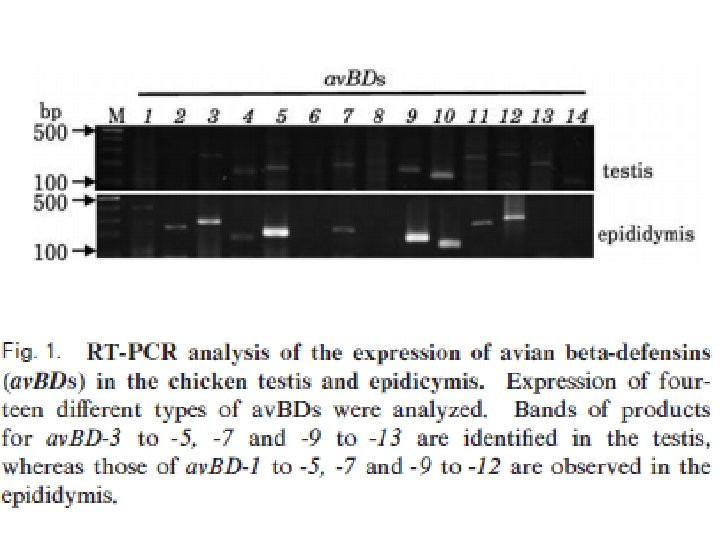
• There are the expression of nine Avian βdefensins (Av. BDs) in the testis, and 10 Av. BDs in the epididymis. • Sertoli cells, sperm cells, and epithelial cells in the epididymis show Av. BD immunopositive staining. • Sperm cells stained immunopositive for Av. BD 3, and the immunopositive Av. BD are retained even after the sperm enter the sperm storage tubules in the oviduct.

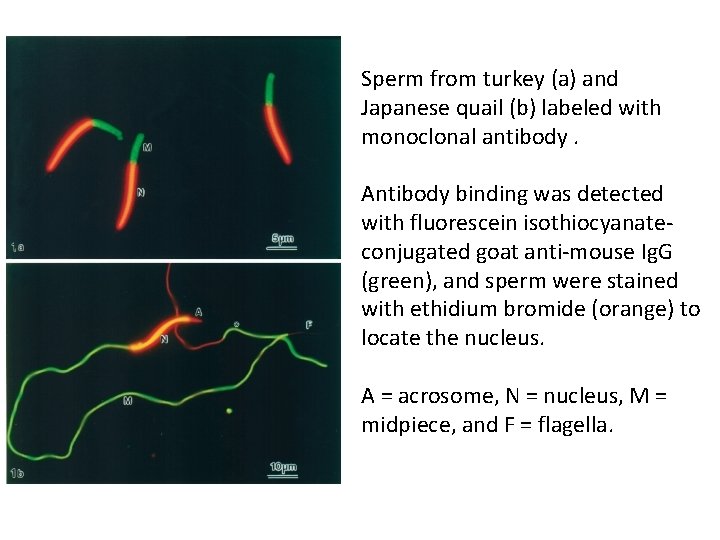
Sperm from turkey (a) and Japanese quail (b) labeled with monoclonal antibody. Antibody binding was detected with fluorescein isothiocyanateconjugated goat anti-mouse Ig. G (green), and sperm were stained with ethidium bromide (orange) to locate the nucleus. A = acrosome, N = nucleus, M = midpiece, and F = flagella.




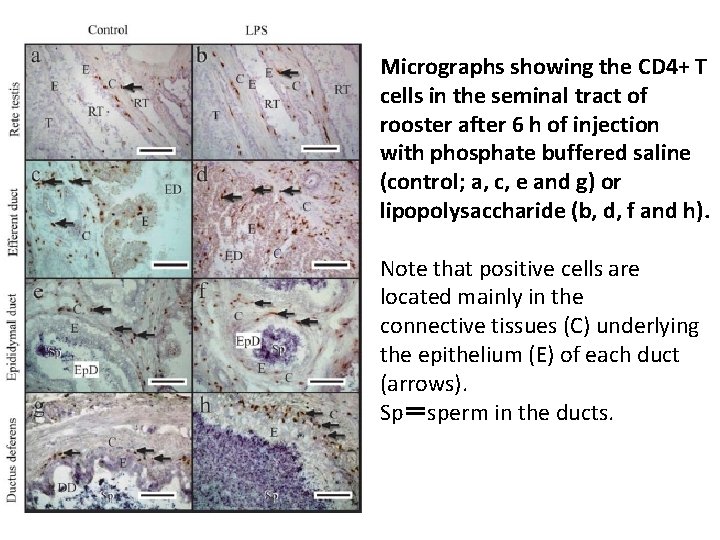
Micrographs showing the CD 4+ T cells in the seminal tract of rooster after 6 h of injection with phosphate buffered saline (control; a, c, e and g) or lipopolysaccharide (b, d, f and h). Note that positive cells are located mainly in the connective tissues (C) underlying the epithelium (E) of each duct (arrows). Sp=sperm in the ducts.

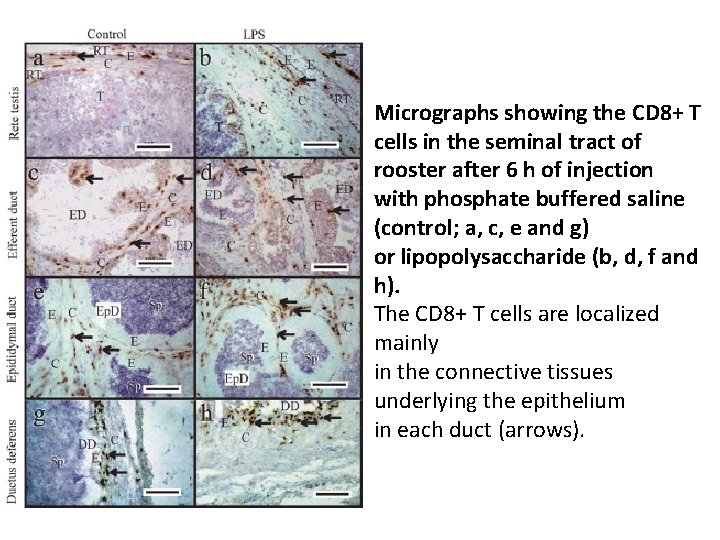
Micrographs showing the CD 8+ T cells in the seminal tract of rooster after 6 h of injection with phosphate buffered saline (control; a, c, e and g) or lipopolysaccharide (b, d, f and h). The CD 8+ T cells are localized mainly in the connective tissues underlying the epithelium in each duct (arrows).



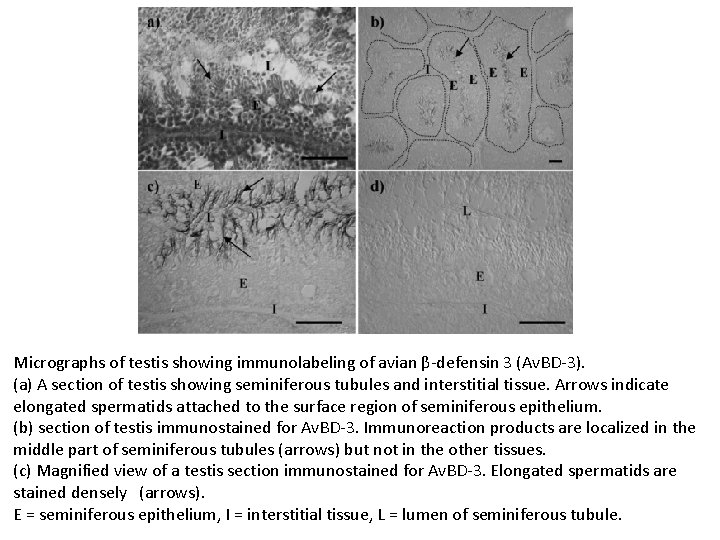
Micrographs of testis showing immunolabeling of avian β-defensin 3 (Av. BD-3). (a) A section of testis showing seminiferous tubules and interstitial tissue. Arrows indicate elongated spermatids attached to the surface region of seminiferous epithelium. (b) section of testis immunostained for Av. BD-3. Immunoreaction products are localized in the middle part of seminiferous tubules (arrows) but not in the other tissues. (c) Magnified view of a testis section immunostained for Av. BD-3. Elongated spermatids are stained densely (arrows). E = seminiferous epithelium, I = interstitial tissue, L = lumen of seminiferous tubule.

Micrographs of epididymis and ductus deferens showing immunolabeling of avian β-defensin 3 (Av. BD-3). (a) A section of epididymis. The epithelial cells of the epididymal ducts are not stained. The staining of the epithelial cells of efferent ductules is nonspecific because they were stained occasionally in control. (b) Section of the epididymal duct immunostained for Av. BD-3. Note the positive staining of sperm (arrow). (c) Section of luminal contents in the ductus deferens immunostained for Av. BD-3. Sperm are stained positive (arrow). E = epithelium; Ef = efferent ductile; Ep = epididymal duct.

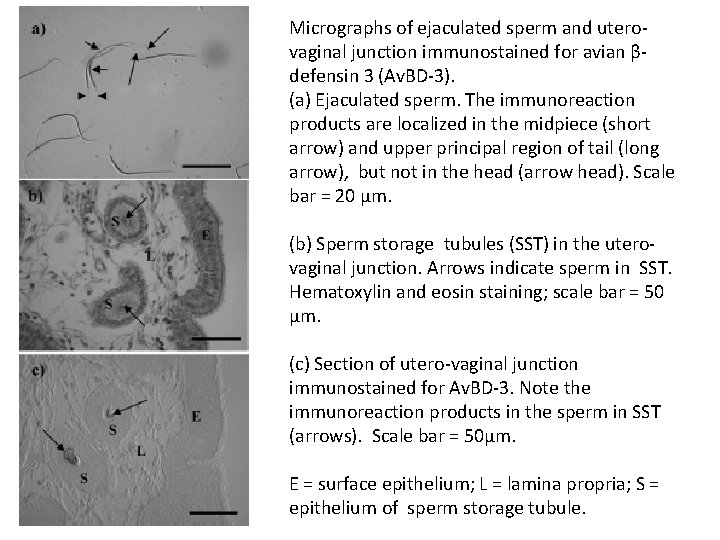
Micrographs of ejaculated sperm and uterovaginal junction immunostained for avian βdefensin 3 (Av. BD-3). (a) Ejaculated sperm. The immunoreaction products are localized in the midpiece (short arrow) and upper principal region of tail (long arrow), but not in the head (arrow head). Scale bar = 20 μm. (b) Sperm storage tubules (SST) in the uterovaginal junction. Arrows indicate sperm in SST. Hematoxylin and eosin staining; scale bar = 50 μm. (c) Section of utero-vaginal junction immunostained for Av. BD-3. Note the immunoreaction products in the sperm in SST (arrows). Scale bar = 50μm. E = surface epithelium; L = lamina propria; S = epithelium of sperm storage tubule.

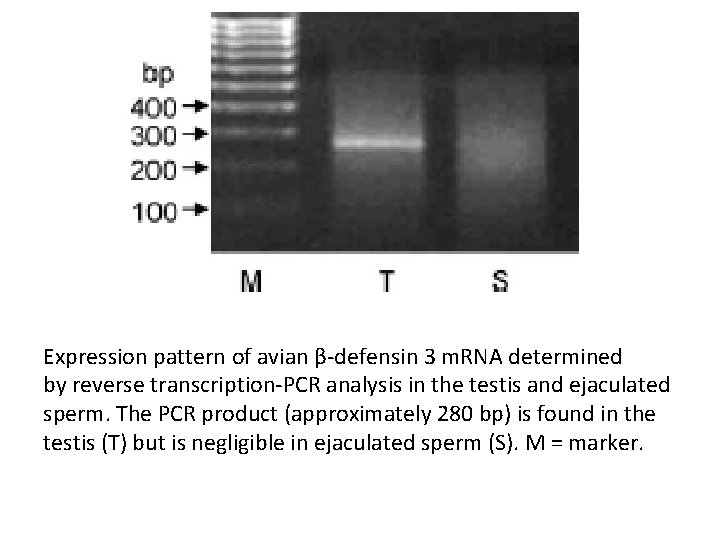
Expression pattern of avian β-defensin 3 m. RNA determined by reverse transcription-PCR analysis in the testis and ejaculated sperm. The PCR product (approximately 280 bp) is found in the testis (T) but is negligible in ejaculated sperm (S). M = marker.

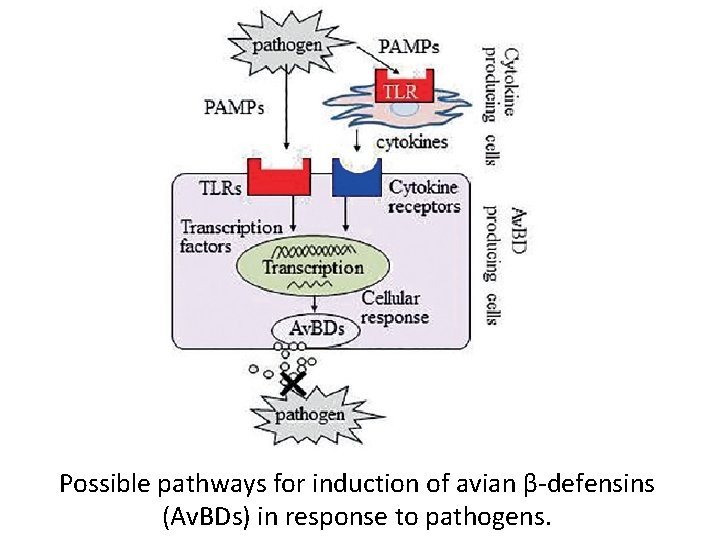
Possible pathways for induction of avian β-defensins (Av. BDs) in response to pathogens.

CONCLUSION • TLRs and Av. BDs are expressed in the chicken reproductive organs. • The expression of some Av. BDs is likely to be upregulated by TLR 4 stimulation. • Proinflammatory cytokines may be involved in this process to induce Av. BDs. • The synthesised Av. BDs may play roles in the local defence against pathogens expressed in the chicken reproductive organs. • These factors are also secreted into eggs to enhance the defence against microbes in eggs laid by healthy birds.