Inmunoterapia un cambio en el paradigma del tratamiento

Inmunoterapia: un cambio en el paradigma del tratamiento del CNMP avanzado. Madrid, 25 de Octubre de 2017 Delvys Rodríguez Abreu, MD Medical Oncology Dept. Hospital Universitario Insular de Gran Canaria. Spain
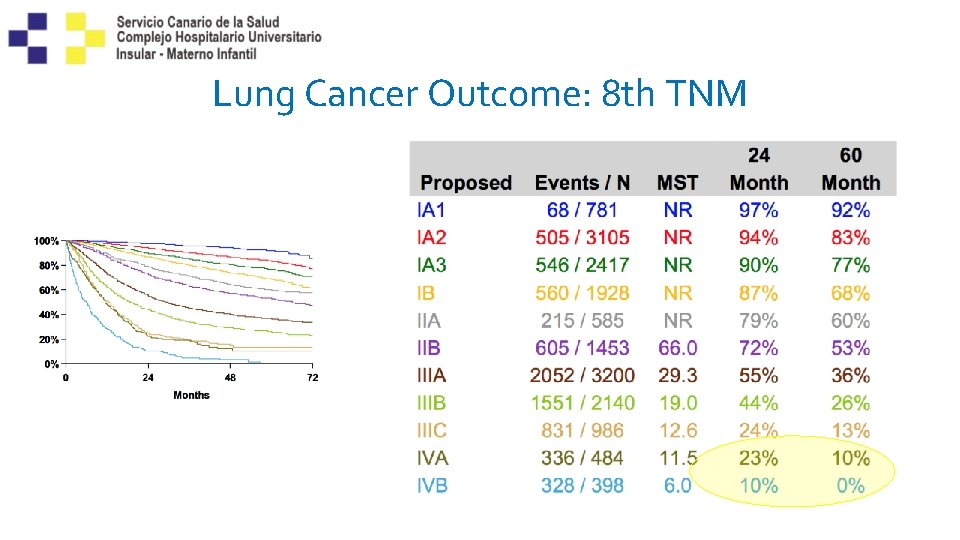
Lung Cancer Outcome: 8 th TNM

Octubre 2017. . . QUÉ TENEMOS DE IO PULMÓN?
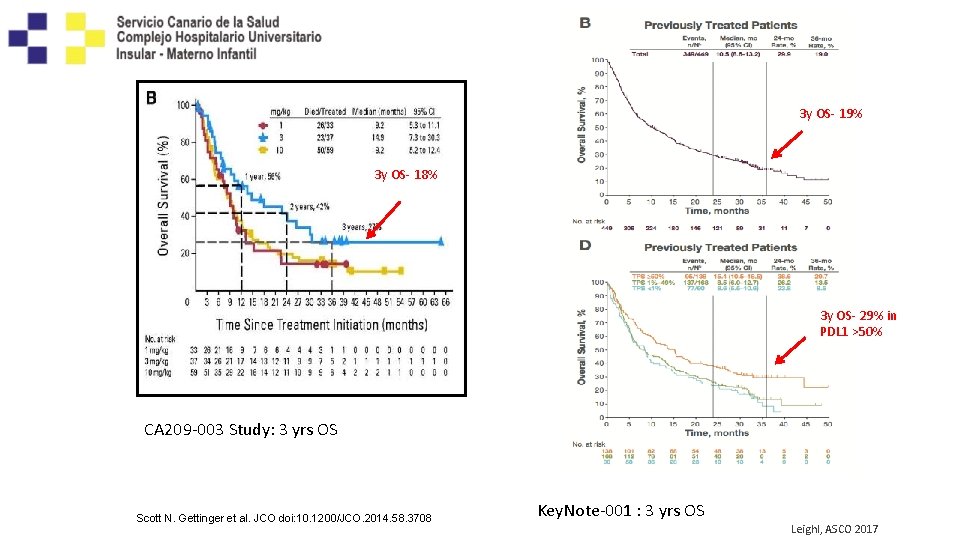
3 y OS- 19% 3 y OS- 18% 3 y OS- 29% in PDL 1 >50% CA 209 -003 Study: 3 yrs OS Scott N. Gettinger et al. JCO doi: 10. 1200/JCO. 2014. 58. 3708 Key. Note-001 : 3 yrs OS Leighl, ASCO 2017
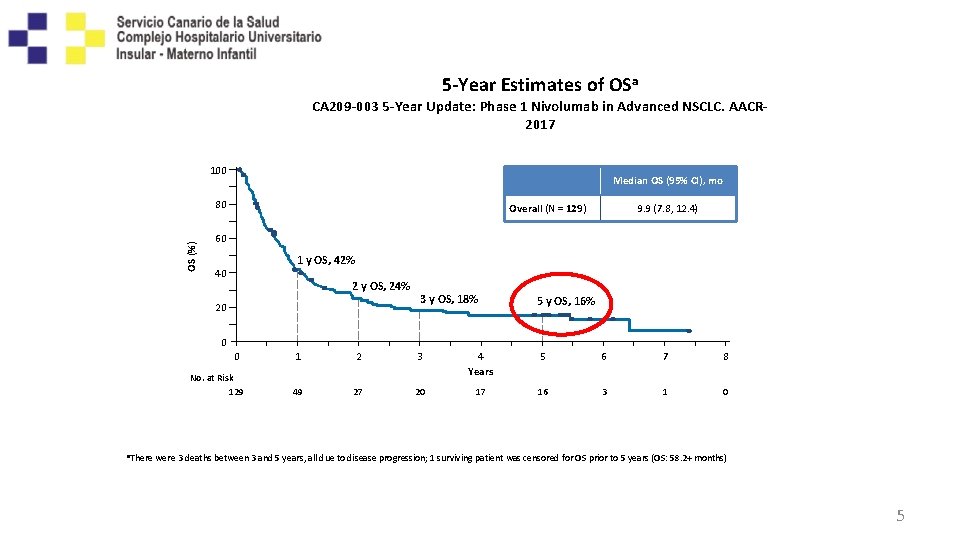
5 -Year Estimates of OSa CA 209 -003 5 -Year Update: Phase 1 Nivolumab in Advanced NSCLC. AACR 2017 100 Median OS (95% CI), mo OS (%) 80 Overall (N = 129) 9. 9 (7. 8, 12. 4) 60 1 y OS, 42% 40 2 y OS, 24% 20 3 y OS, 18% 5 y OS, 16% 0 0 No. at Risk 129 a. There 1 2 3 4 Years 5 6 7 8 49 27 20 17 16 3 1 0 were 3 deaths between 3 and 5 years, all due to disease progression; 1 surviving patient was censored for OS prior to 5 years (OS: 58. 2+ months) 5

Immunotherapy? ? Pretreated patients Chemotherapy? ?
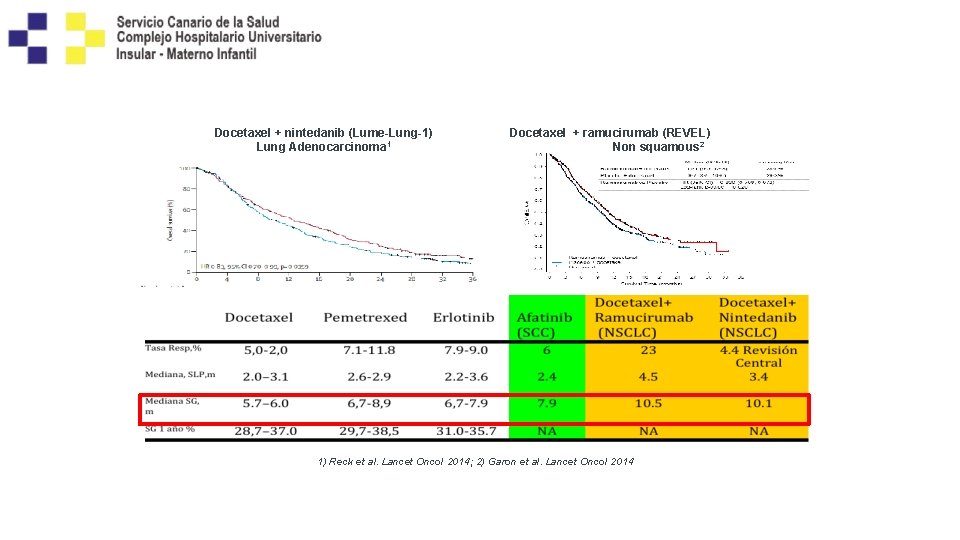
Docetaxel + nintedanib (Lume-Lung-1) Lung Adenocarcinoma 1 Docetaxel + ramucirumab (REVEL) Non squamous 2 1) Reck et al. Lancet Oncol 2014; 2) Garon et al. Lancet Oncol 2014

But lung cancer patients still dying… ‘Insanity: doing the same thing over and over again and expecting different results. ’ Albert Einstein (1879 – 1955)
The Past (2015 – 2016) - Immunotherapy in pretreated patients Nivolumab – Check. Mate 017 (PIII)1 Nivolumab – Check. Mate 057 (PIII)2 GOOD BYE POSITIVE DOCETAXEL!!! TRIALS 2 nd Line, squamous, PD-L 1 All-Comer 2 nd Line, non-squamous, PD-L 1 All-Comer Pembrolizumab - Keynote 010 (PII/III)3 Atezolizumab – OAK (PIII)4 2 nd+ Line, PD-L 1 TPS ≥ 1% 2 nd+ Line, PD-L 1 All-Comer 1. Borghaei H et al. Poster presentation at ASCO 2016. 9025. 2. Brahmer JR et al. Oral presentation at AACR 2017. CT 077. 3. Herbst RS et al. Poster presentation at ASCO 2017. 9090. 4. Rittmeyer A et al. Lancet. 2017; 389(10066): 255 -265.
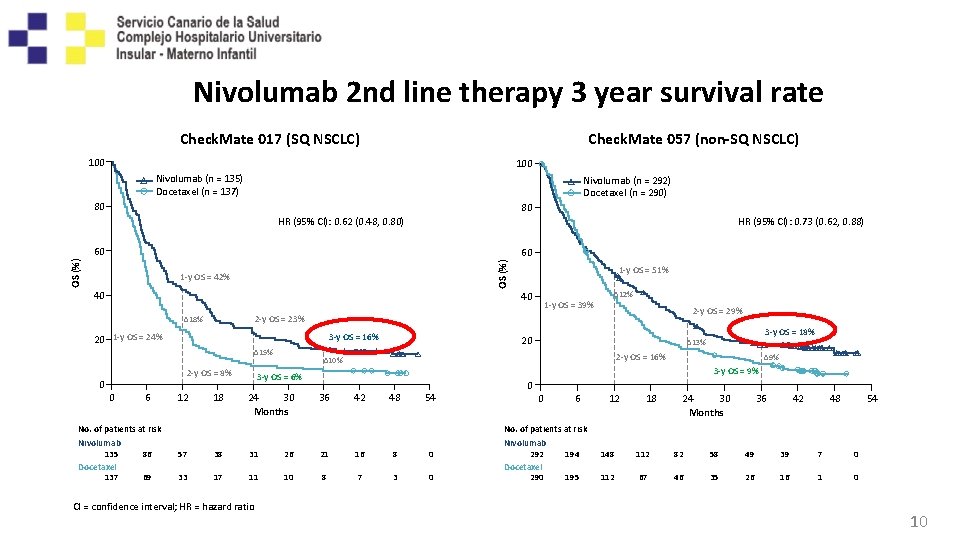
Nivolumab 2 nd line therapy 3 year survival rate Check. Mate 017 (SQ NSCLC) Check. Mate 057 (non-SQ NSCLC) 100 Nivolumab (n = 135) Docetaxel (n = 137) Nivolumab (n = 292) Docetaxel (n = 290) 80 80 HR (95% CI): 0. 62 (0. 48, 0. 80) HR (95% CI): 0. 73 (0. 62, 0. 88) 60 OS (%) 60 1 -y OS = 42% 40 Δ 12% 40 20 1 -y OS = 24% 3 -y OS = 16% Δ 15% 2 -y OS = 8% 0 6 12 2 -y OS = 29% 18 3 -y OS = 18% 20 Δ 13% 2 -y OS = 16% Δ 10% 24 30 Months 36 Δ 9% 3 -y OS = 6% 42 48 54 No. of patients at risk Nivolumab 135 Docetaxel 137 1 -y OS = 39% 2 -y OS = 23% Δ 18% 0 1 -y OS = 51% 0 0 6 12 18 24 30 Months 36 42 48 54 No. of patients at risk 86 57 38 31 26 21 16 8 0 69 33 17 11 10 8 7 3 0 CI = confidence interval; HR = hazard ratio Nivolumab 292 Docetaxel 290 194 148 112 82 58 49 39 7 0 195 112 67 46 35 26 16 1 0 10
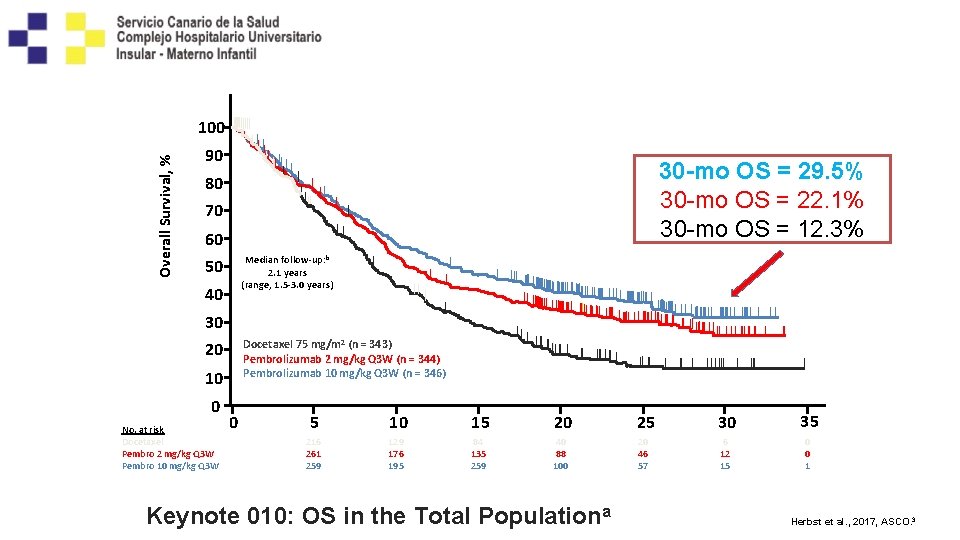
Overall Survival, % 100 90 30 -mo OS = 29. 5% 30 -mo OS = 22. 1% 30 -mo OS = 12. 3% 80 70 60 Median follow-up: b 2. 1 years (range, 1. 5 -3. 0 years) 50 40 30 Docetaxel 75 mg/m 2 (n = 343) Pembrolizumab 2 mg/kg Q 3 W (n = 344) Pembrolizumab 10 mg/kg Q 3 W (n = 346) 20 10 0 No. at risk Docetaxel Pembro 2 mg/kg Q 3 W Pembro 10 mg/kg Q 3 W 0 5 10 15 20 25 30 35 216 261 259 129 176 195 84 135 259 40 88 100 20 46 57 6 12 15 0 0 1 6769 – RS Herbst Keynote 010: OS in the Total Populationa Herbst et al. , 2017, ASCO. 3
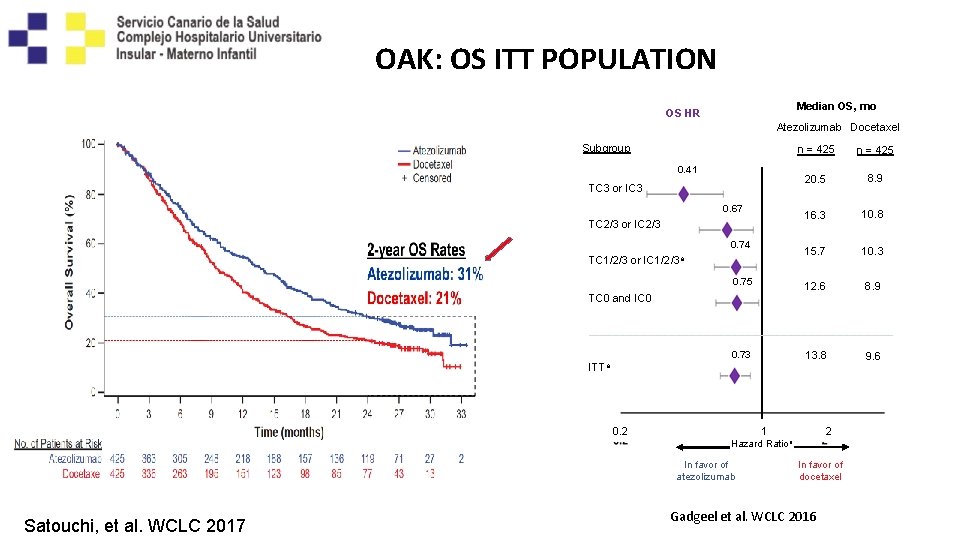
OAK: OS ITT POPULATION Median OS, mo OS HR Atezolizumab Docetaxel Subgroup n = 425 20. 5 8. 9 16. 3 10. 8 15. 7 10. 3 0. 75 12. 6 8. 9 0. 73 13. 8 9. 6 0. 41 TC 3 or IC 3 0. 67 TC 2/3 or IC 2/3 0. 74 TC 1/2/3 or IC 1/2/3 a TC 0 and IC 0 ITTa 0. 2 1 2 Hazard Ratioa In favor of atezolizumab Satouchi, et al. WCLC 2017 In favor of docetaxel Gadgeel et al. WCLC 2016
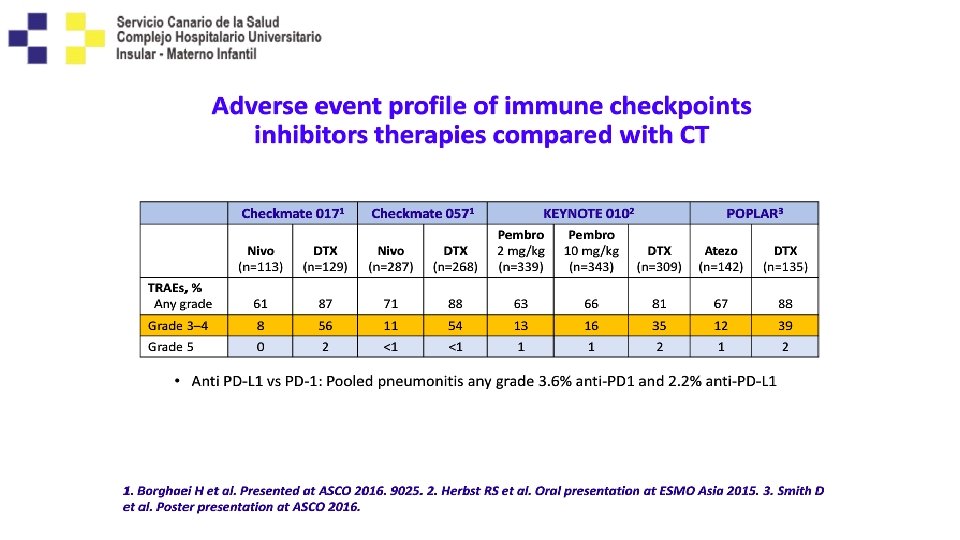

Conclusiones de estos estudios ya antiguos!! • Funciona tanto en escamoso como en no escamoso. • Mayor en fumadores y menos en EGFR++. • Mayor en todos los PD-L 1 positivos. • Respuestas rápidas como la quimio y raramente resp no convencionales. • Mayor efecto en OS y menor en PFS. • Actividad en SNC. • Mejor perfil de toxicidad que el docetaxel. • Plateau en la curva de SG.
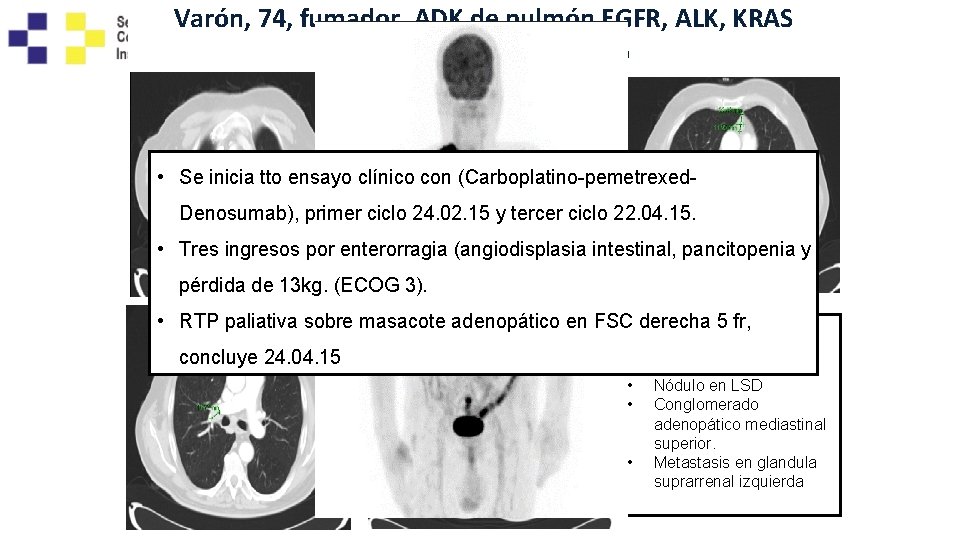
Varón, 74, fumador, ADK de pulmón EGFR, ALK, KRAS Positivo. PD-L 1 --negativo • Se inicia tto ensayo clínico con (Carboplatino-pemetrexed. Denosumab), primer ciclo 24. 02. 15 y tercer ciclo 22. 04. 15. • Tres ingresos por enterorragia (angiodisplasia intestinal, pancitopenia y pérdida de 13 kg. (ECOG 3). • RTP paliativa sobre masacote adenopático en FSC derecha 5 fr, TAC cráneo-tóraxconcluye 24. 04. 15 • • • abdomen superior (05. 01. 15) Nódulo en LSD Conglomerado adenopático mediastinal superior. Metastasis en glandula suprarrenal izquierda
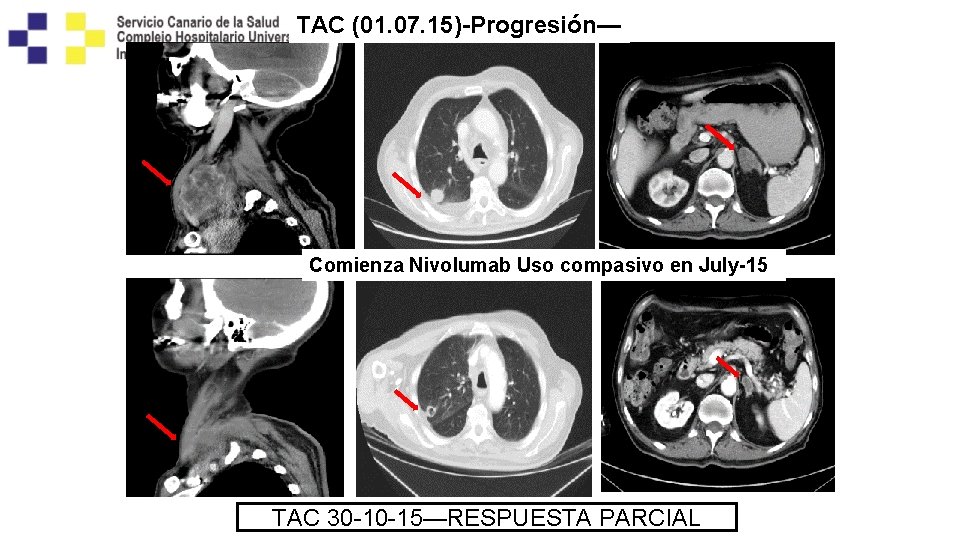
TAC (01. 07. 15)-Progresión— Comienza Nivolumab Uso compasivo en July-15 TAC 30 -10 -15—RESPUESTA PARCIAL
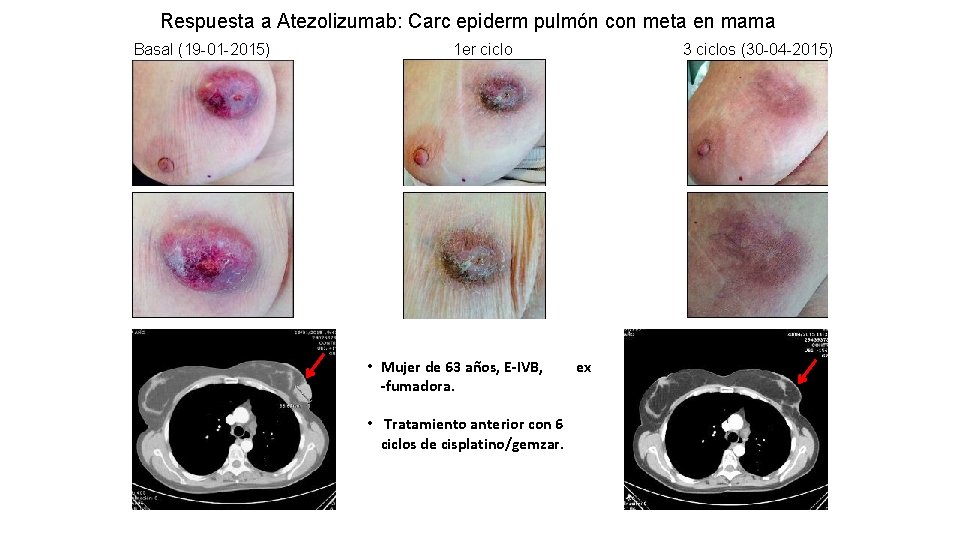
Respuesta a Atezolizumab: Carc epiderm pulmón con meta en mama Basal (19 -01 -2015) 1 er ciclo • Mujer de 63 años, E-IVB, -fumadora. • Tratamiento anterior con 6 ciclos de cisplatino/gemzar. 3 ciclos (30 -04 -2015) ex
ESMO and NCCN guideline 2017 • NCCN guidelines 2 nd

Inmunoterapia en 1 era línea? 2016 -2017 Monoterapia y Combinaciones. 19
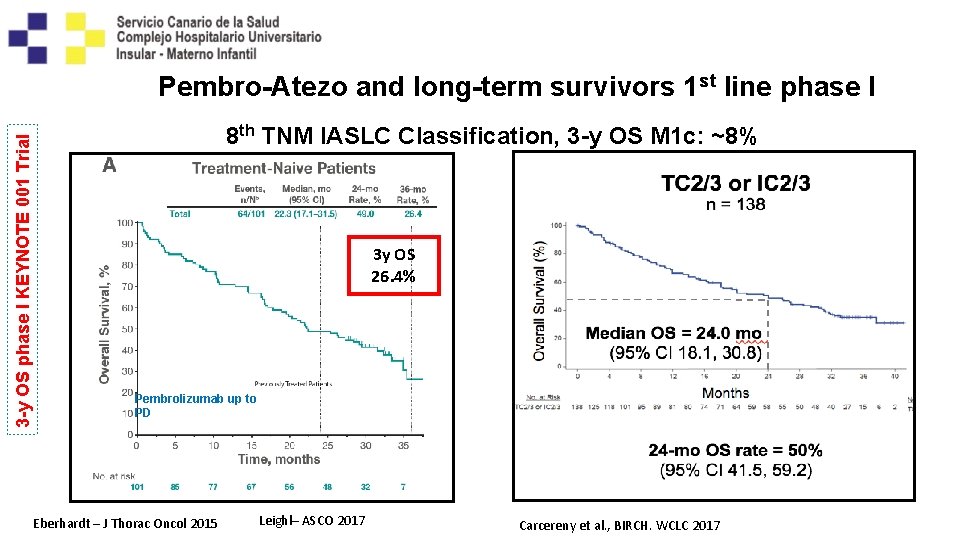
3 -y OS phase I KEYNOTE 001 Trial Pembro-Atezo and long-term survivors 1 st line phase I 8 th TNM IASLC Classification, 3 -y OS M 1 c: ~8% 3 y OS 26. 4% PD-L 1 > 50% (N=138) 3 y. OS: 29. 7% Pembrolizumab up to PD Eberhardt – J Thorac Oncol 2015 Leighl– ASCO 2017 Carcereny et al. , BIRCH. WCLC 2017
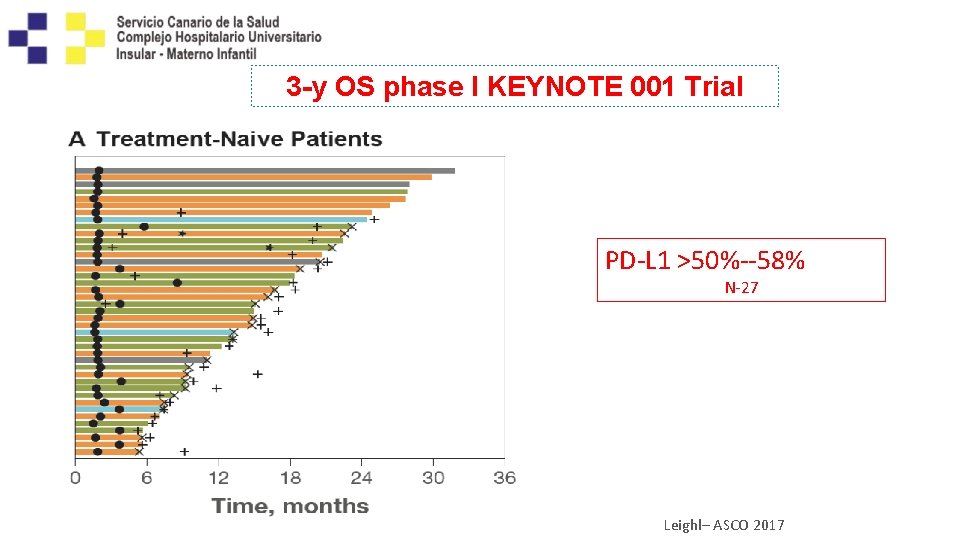
3 -y OS phase I KEYNOTE 001 Trial PD-L 1 >50%--58% N-27 Leighl– ASCO 2017
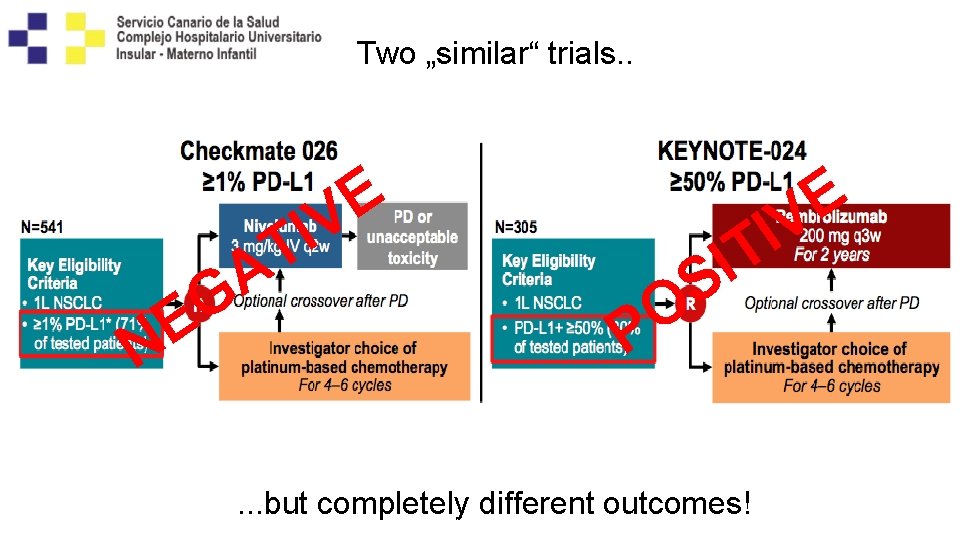
Two „similar“ trials. . E V I T E N A G O P E V I T I S . . . but completely different outcomes! Carbone D et al, NEJM 2017; 376 (25): 2415 -2426; Reck M et al, NEJM 2016; 375 (19):

NCCN guidelines 1 st line- version 9. 2017 Ø b m Pe m u z i l o r e n o y l n. o e e t lin h t s s i r i f ab g u r d IO o r p ap in d ve
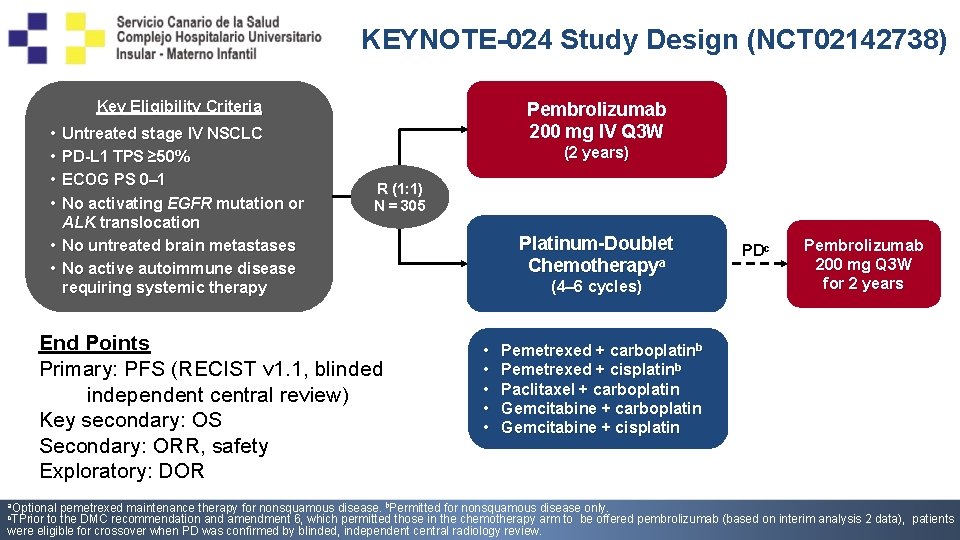
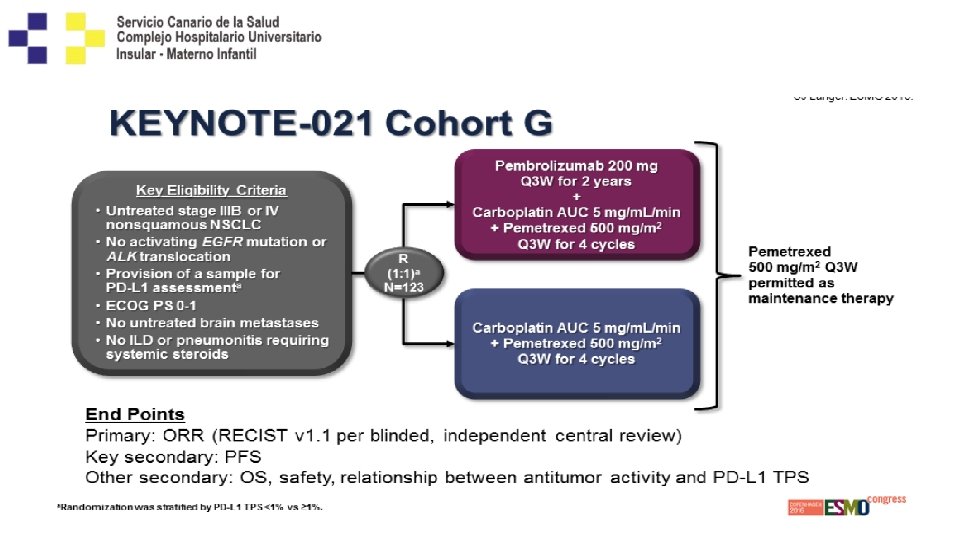
KEYNOTE-024 Study Design (NCT 02142738) Key Eligibility Criteria • • Untreated stage IV NSCLC PD-L 1 TPS ≥ 50% ECOG PS 0– 1 No activating EGFR mutation or ALK translocation • No untreated brain metastases • No active autoimmune disease requiring systemic therapy Pembrolizumab 200 mg IV Q 3 W (2 years) R (1: 1) N = 305 End Points Primary: PFS (RECIST v 1. 1, blinded independent central review) Key secondary: OS Secondary: ORR, safety Exploratory: DOR a. Optional c. TPrior to Platinum-Doublet Chemotherapya (4– 6 cycles) • • • PDc Pembrolizumab 200 mg Q 3 W for 2 years Pemetrexed + carboplatinb Pemetrexed + cisplatinb Paclitaxel + carboplatin Gemcitabine + cisplatin pemetrexed maintenance therapy for nonsquamous disease. b. Permitted for nonsquamous disease only. the DMC recommendation and amendment 6, which permitted those in the chemotherapy arm to be offered pembrolizumab (based on interim analysis 2 data), patients were eligible for crossover when PD was confirmed by blinded, independent central radiology review.
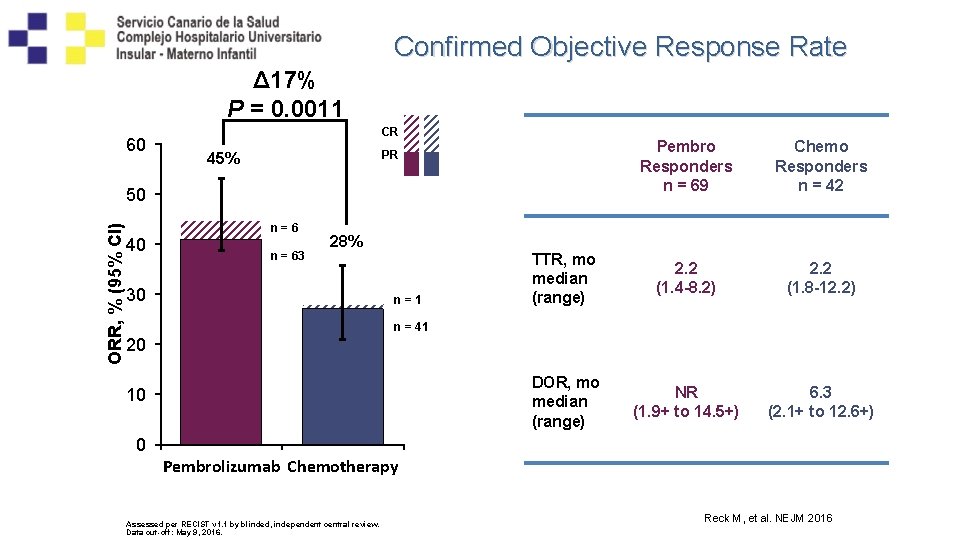
Confirmed Objective Response Rate Δ 17% P = 0. 0011 60 CR Pembro Responders n = 69 Chemo Responders n = 42 TTR, mo median (range) 2. 2 (1. 4 -8. 2) 2. 2 (1. 8 -12. 2) DOR, mo median (range) NR (1. 9+ to 14. 5+) 6. 3 (2. 1+ to 12. 6+) PR 45% ORR, % (95% CI) 50 40 n=6 n = 63 28% 30 n=1 n = 41 20 10 0 Pembrolizumab Chemotherapy Assessed per RECIST v 1. 1 by blinded, independent central review. Data cut-off: May 9, 2016. Reck M, et al. NEJM 2016
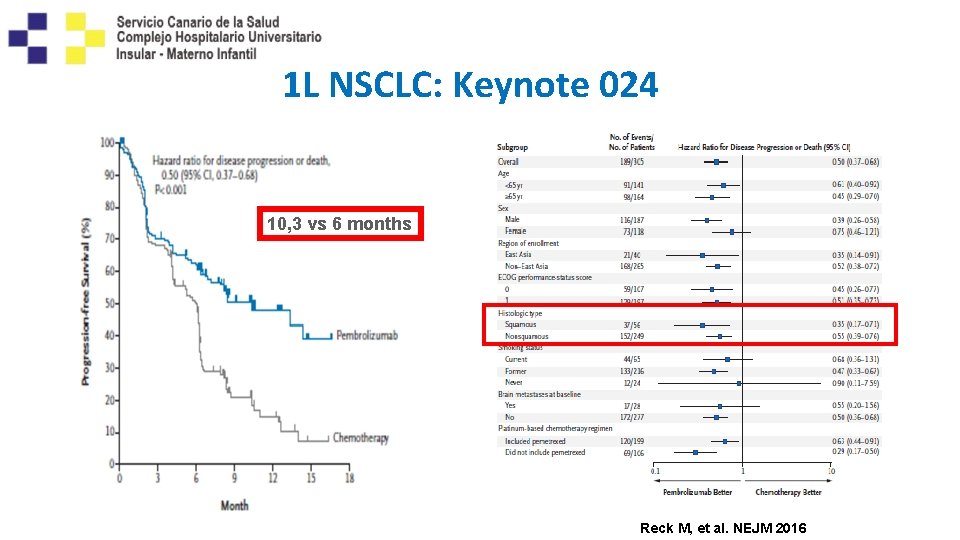
1 L NSCLC: Keynote 024 10, 3 vs 6 months Reck M, et al. NEJM 2016
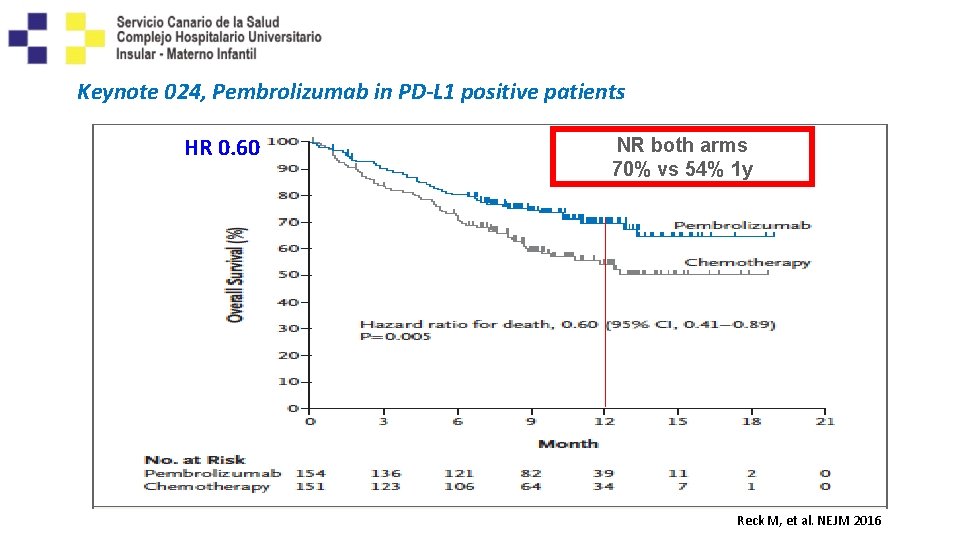
Keynote 024, Pembrolizumab in PD-L 1 positive patients HR 0. 60 NR both arms 70% vs 54% 1 y Reck M, et al. NEJM 2016
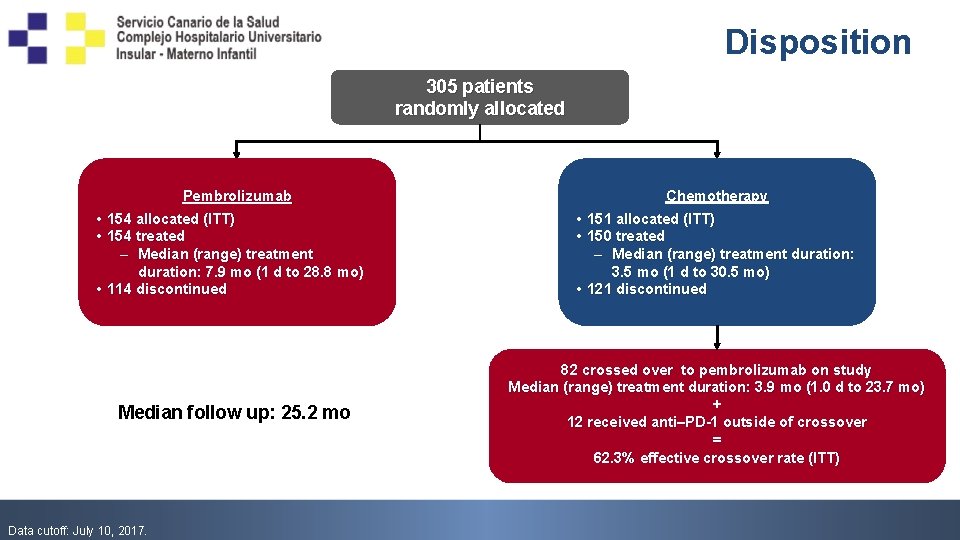
Disposition 305 patients randomly allocated Pembrolizumab Chemotherapy • 154 allocated (ITT) • 154 treated – Median (range) treatment duration: 7. 9 mo (1 d to 28. 8 mo) • 114 discontinued • 151 allocated (ITT) • 150 treated – Median (range) treatment duration: 3. 5 mo (1 d to 30. 5 mo) • 121 discontinued Median follow up: 25. 2 mo 82 crossed over to pembrolizumab on study Median (range) treatment duration: 3. 9 mo (1. 0 d to 23. 7 mo) + 12 received anti–PD-1 outside of crossover = 62. 3% effective crossover rate (ITT) Data cutoff: July 10, 2017.
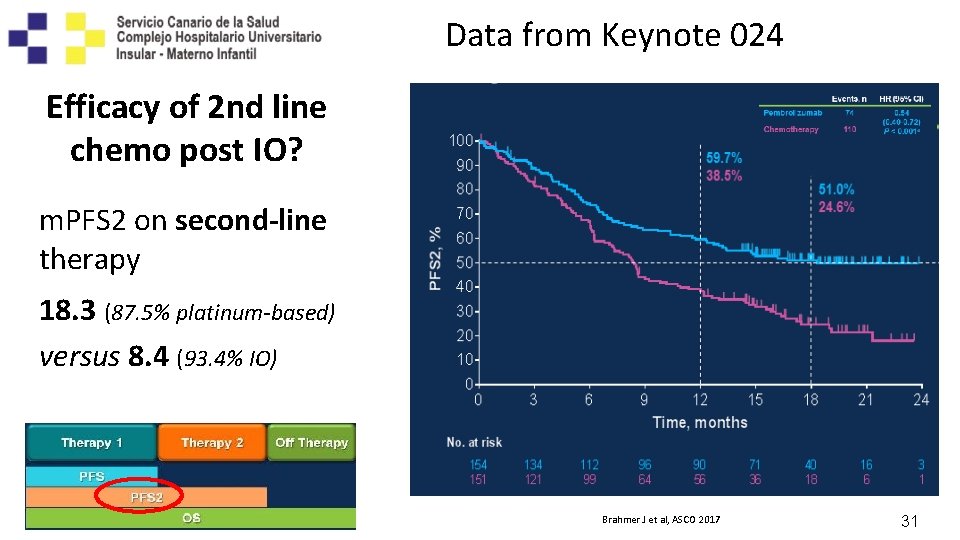
Data from Keynote 024 Efficacy of 2 nd line chemo post IO? m. PFS 2 on second-line therapy 18. 3 (87. 5% platinum-based) versus 8. 4 (93. 4% IO) Brahmer J et al, ASCO 2017 31
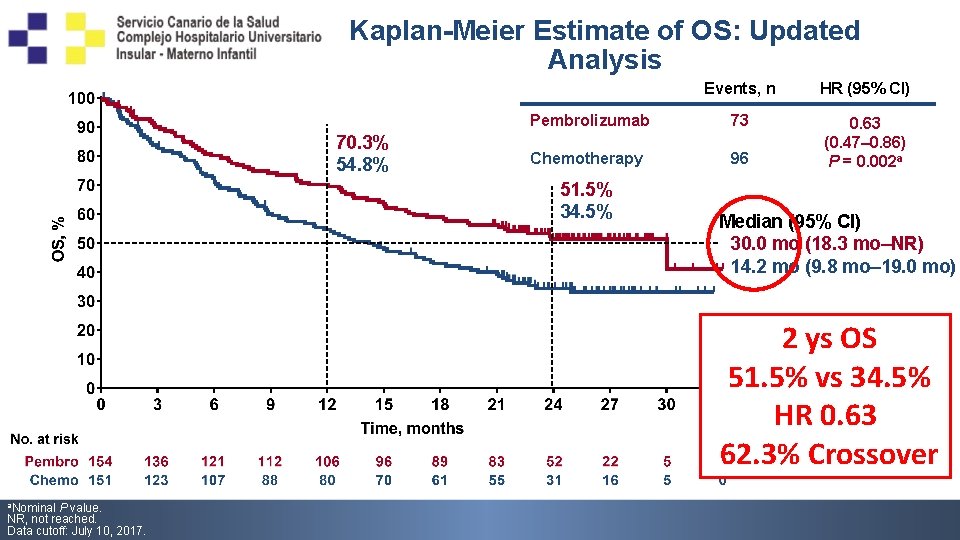
Kaplan-Meier Estimate of OS: Updated Analysis 70. 3% 54. 8% Events, n HR (95% CI) Pembrolizumab 73 Chemotherapy 96 0. 63 (0. 47– 0. 86) P = 0. 002 a 51. 5% 34. 5% Median (95% CI) 30. 0 mo (18. 3 mo–NR) 14. 2 mo (9. 8 mo– 19. 0 mo) 2 ys OS 51. 5% vs 34. 5% HR 0. 63 62. 3% Crossover a. Nominal P value. NR, not reached. Data cutoff: July 10, 2017.
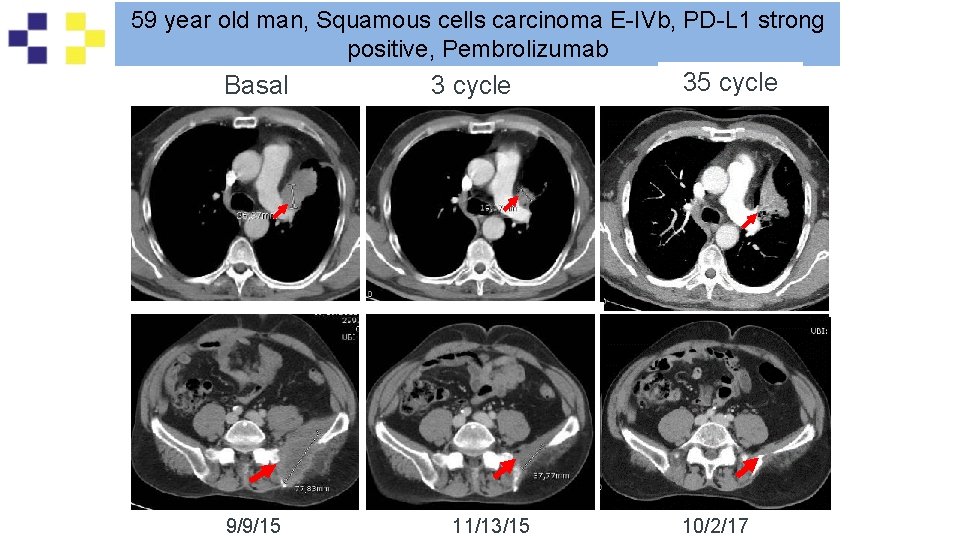
59 year old man, Squamous cells carcinoma E-IVb, PD-L 1 strong positive, Pembrolizumab Basal 9/9/15 3 cycle 11/13/15 cycle 6 35 cycle 10/2/17 01/14/16
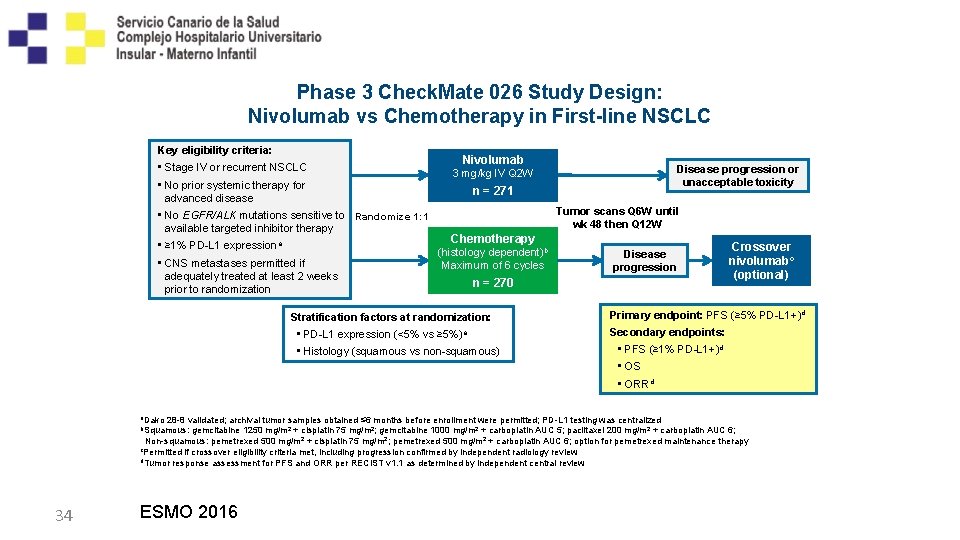
Phase 3 Check. Mate 026 Study Design: Nivolumab vs Chemotherapy in First-line NSCLC Key eligibility criteria: Nivolumab • Stage IV or recurrent NSCLC Disease progression or 3 mg/kg IV Q 2 W unacceptable toxicity • No prior systemic therapy for n = 271 advanced disease Tumor scans Q 6 W until • No EGFR/ALK mutations sensitive to Randomize 1: 1 wk 48 then Q 12 W available targeted inhibitor therapy Chemotherapy • ≥ 1% PD-L 1 expressiona Crossover (histology dependent)b Disease nivolumabc • CNS metastases permitted if Maximum of 6 cycles progression (optional) adequately treated at least 2 weeks n = 270 prior to randomization Stratification factors at randomization: • PD-L 1 expression (<5% vs ≥ 5%)a • Histology (squamous vs non-squamous) a. Dako Primary endpoint: PFS (≥ 5% PD-L 1+)d Secondary endpoints: • PFS (≥ 1% PD-L 1+)d • OS • ORRd 28 -8 validated; archival tumor samples obtained ≤ 6 months before enrollment were permitted; PD-L 1 testing was centralized gemcitabine 1250 mg/m 2 + cisplatin 75 mg/m 2; gemcitabine 1000 mg/m 2 + carboplatin AUC 5; paclitaxel 200 mg/m 2 + carboplatin AUC 6; Non-squamous: pemetrexed 500 mg/m 2 + cisplatin 75 mg/m 2; pemetrexed 500 mg/m 2 + carboplatin AUC 6; option for pemetrexed maintenance therapy c. Permitted if crossover eligibility criteria met, including progression confirmed by independent radiology review d. Tumor response assessment for PFS and ORR per RECIST v 1. 1 as determined by independent central review b. Squamous: 34 ESMO 2016
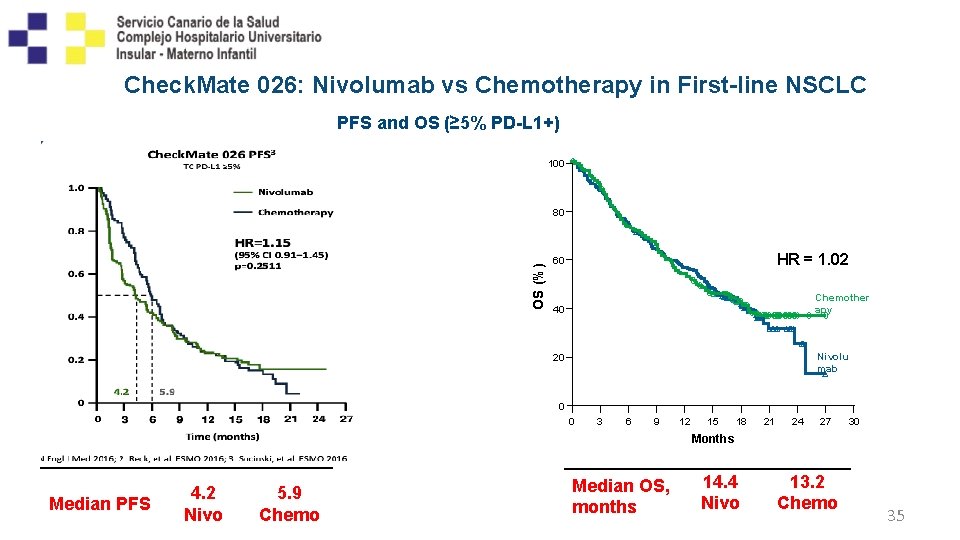
Check. Mate 026: Nivolumab vs Chemotherapy in First-line NSCLC PFS and OS (≥ 5% PD-L 1+) 100 OS (%) 80 HR = 1. 02 60 Chemother apy 40 Nivolu mab 20 0 0 3 6 9 12 15 18 21 24 27 30 Months Median PFS 4. 2 Nivo 5. 9 Chemo Median OS, months 14. 4 Nivo 13. 2 Chemo 35
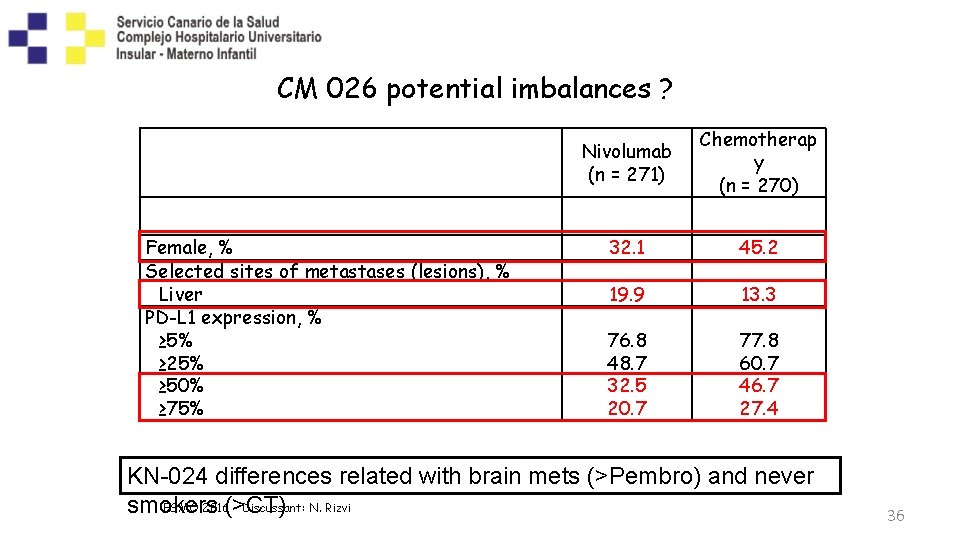
CM 026 potential imbalances ? Female, % Selected sites of metastases (lesions), % Liver PD-L 1 expression, % ≥ 5% ≥ 25% ≥ 50% ≥ 75% Nivolumab (n = 271) Chemotherap y (n = 270) 32. 1 45. 2 19. 9 13. 3 76. 8 48. 7 32. 5 20. 7 77. 8 60. 7 46. 7 27. 4 KN-024 differences related with brain mets (>Pembro) and Socinski et al, ESMO 2016 never ESMO 2016(>CT) - Discussant: N. Rizvi smokers 36
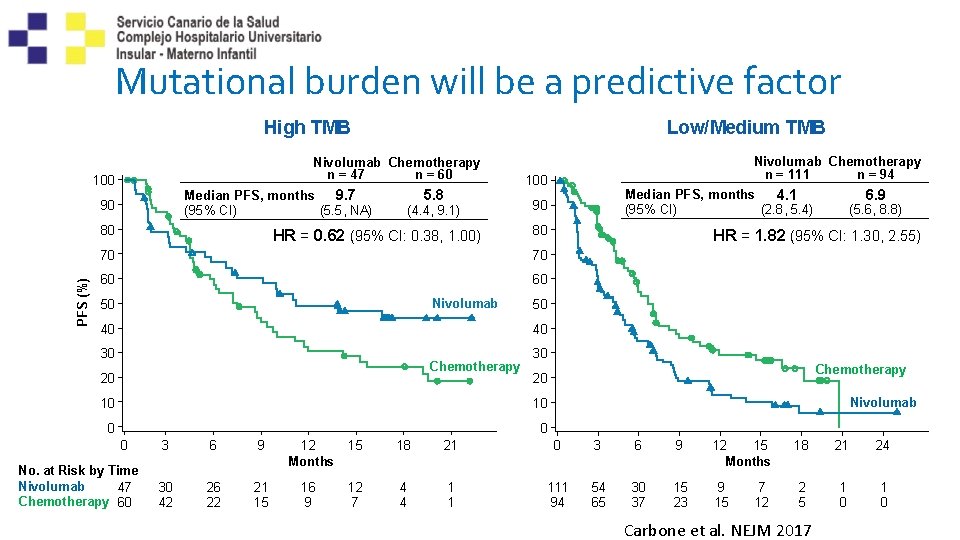
Mutational burden will be a predictive factor High TMB Nivolumab Chemotherapy n = 47 n = 60 100 Median PFS, months 9. 7 (95% CI) (5. 5, NA) 90 80 PFS (%) Low/Medium TMB 5. 8 (4. 4, 9. 1) HR = 0. 62 (95% CI: 0. 38, 1. 00) 100 80 70 60 60 Nivolumab Median PFS, months 4. 1 (95% CI) (2. 8, 5. 4) 90 70 50 Nivolumab Chemotherapy n = 111 n = 94 6. 9 (5. 6, 8. 8) HR = 1. 82 (95% CI: 1. 30, 2. 55) 50 40 40 30 Chemotherapy 20 30 10 10 0 No. at Risk by Time Nivolumab 47 Chemotherapy 60 Chemotherapy 20 Nivolumab 3 6 9 12 Months 15 18 21 0 3 6 9 12 15 Months 30 42 26 22 21 15 16 9 12 7 4 4 1 1 111 94 54 65 30 37 15 23 9 15 7 12 18 21 24 2 5 1 0 Carbone et al. NEJM 2017
More trials to come. . .
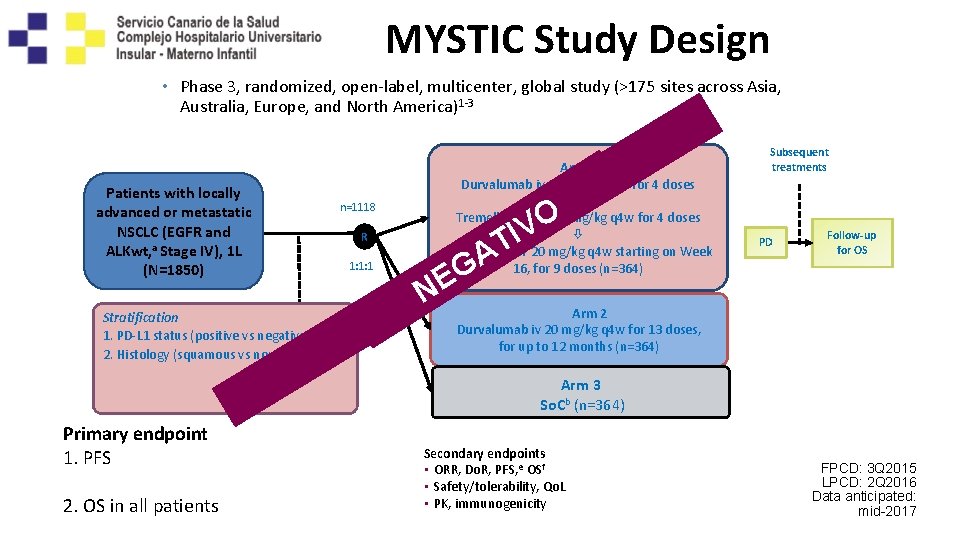
MYSTIC Study Design • Phase 3, randomized, open-label, multicenter, global study (>175 sites across Asia, Australia, Europe, and North America)1 -3 Patients with locally advanced or metastatic NSCLC (EGFR and ALKwt, a Stage IV), 1 L (N=1850) Stratification 1. PD-L 1 status (positive vs negative) 2. Histology (squamous vs nonsquamous) Arm 1 Durvalumab iv 20 mg/kg q 4 w for 4 doses + Tremelimumab iv 1 mg/kg q 4 w for 4 doses Durvalumab iv 20 mg/kg q 4 w starting on Week 16, for 9 doses (n=364) n=1118 C O L C V NTSI R : A O I RG 1: 1: 1 Subsequent treatments PD Follow-up for OS M EE T N IS Arm 2 Durvalumab iv 20 mg/kg q 4 w for 13 doses, for up to 12 months (n=364) Arm 3 So. Cb (n=364) Primary endpoint 1. PFS in all patients and in PD-L 1(+) patients 2. OS in all patients Secondary endpoints • ORR, Do. R, PFS, e OSf • Safety/tolerability, Qo. L • PK, immunogenicity FPCD: 3 Q 2015 LPCD: 2 Q 2016 Data anticipated: mid-2017

Nuevos Retos en 1 era Línea…. . • PD-L 1 0 -50% • Cómo manejamos estos pacientes? • Combinaciones?
Combinations IO-IO IO-CT
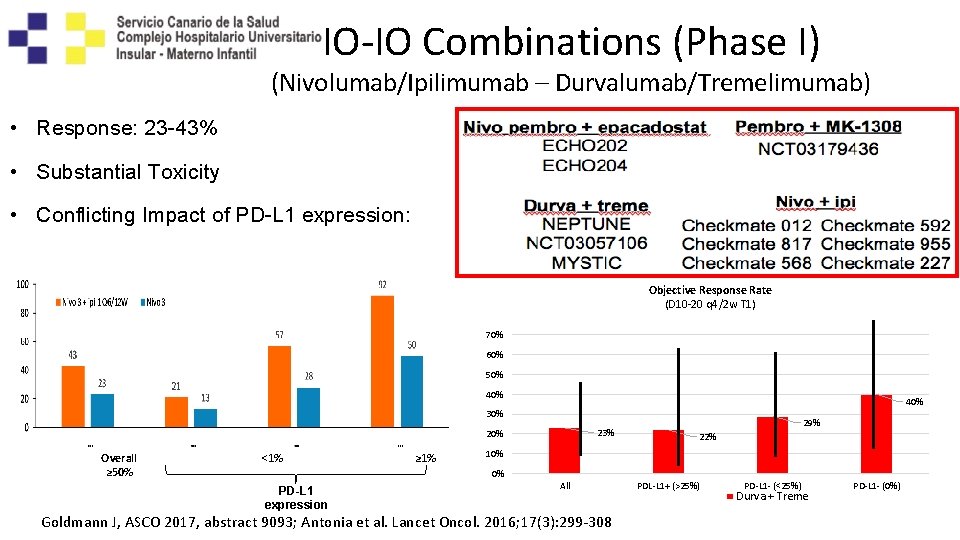
IO-IO Combinations (Phase I) (Nivolumab/Ipilimumab – Durvalumab/Tremelimumab) • Response: 23 -43% • Substantial Toxicity • Conflicting Impact of PD-L 1 expression: Objective Response Rate (D 10 -20 q 4/2 w T 1) 70% 60% 50% 40% 30% 23% 20% Overall ≥ 50% <1% ≥ 1% 29% 22% 10% 0% PD-L 1 expression All Goldmann J, ASCO 2017, abstract 9093; Antonia et al. Lancet Oncol. 2016; 17(3): 299 -308 PDL-L 1+ (>25%) PD-L 1 - (<25%) Durva + Treme PD-L 1 - (0%)
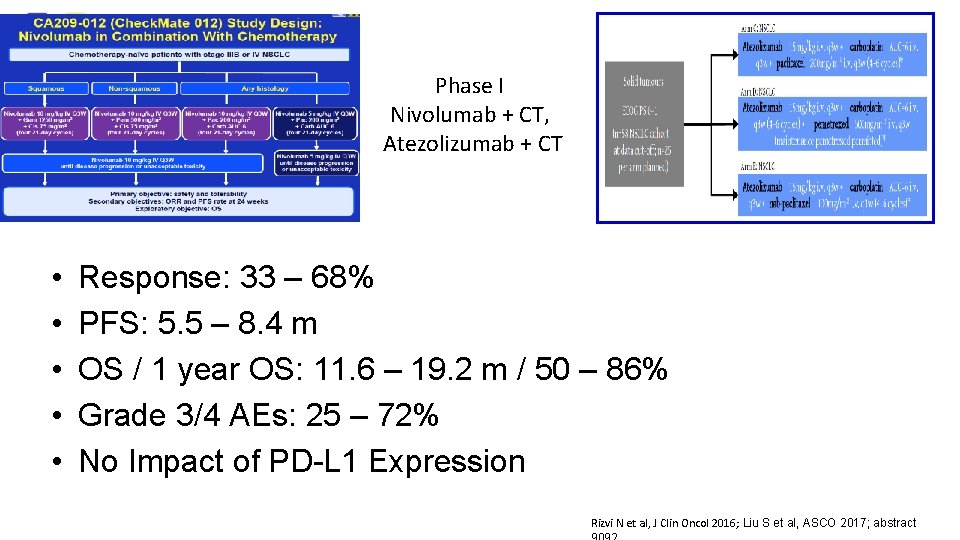
Phase I Nivolumab + CT, Atezolizumab + CT • • • Response: 33 – 68% PFS: 5. 5 – 8. 4 m OS / 1 year OS: 11. 6 – 19. 2 m / 50 – 86% Grade 3/4 AEs: 25 – 72% No Impact of PD-L 1 Expression Rizvi N et al, J Clin Oncol 2016; Liu S et al, ASCO 2017; abstract
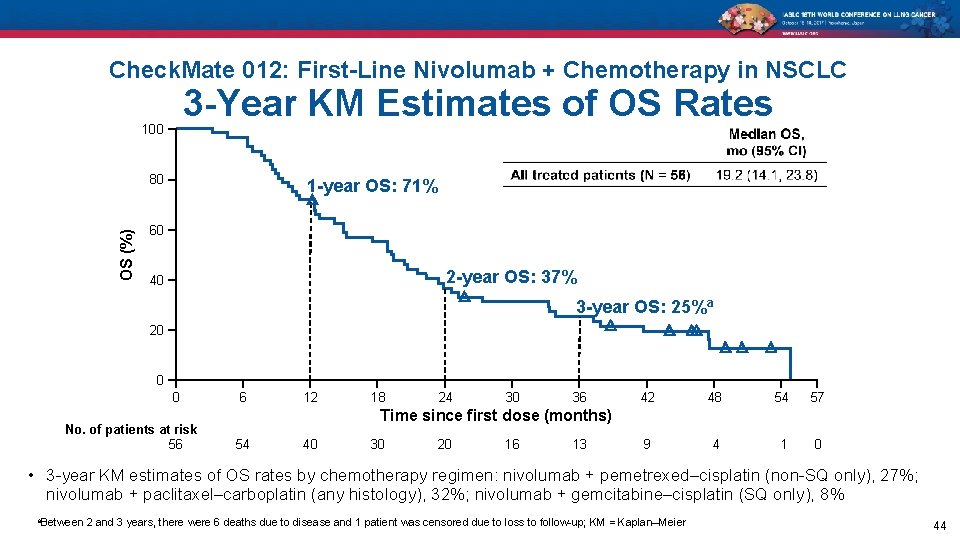
Check. Mate 012: First-Line Nivolumab + Chemotherapy in NSCLC 3 -Year KM Estimates of OS Rates 100 OS (%) 80 1 -year OS: 71% 60 2 -year OS: 37% 40 3 -year OS: 25%a 20 0 0 No. of patients at risk 56 6 12 18 24 30 36 42 48 54 57 9 4 1 0 Time since first dose (months) 54 40 30 20 16 13 • 3 -year KM estimates of OS rates by chemotherapy regimen: nivolumab + pemetrexed–cisplatin (non-SQ only), 27%; nivolumab + paclitaxel–carboplatin (any histology), 32%; nivolumab + gemcitabine–cisplatin (SQ only), 8% a. Between 2 and 3 years, there were 6 deaths due to disease and 1 patient was censored due to loss to follow-up; KM = Kaplan–Meier 44
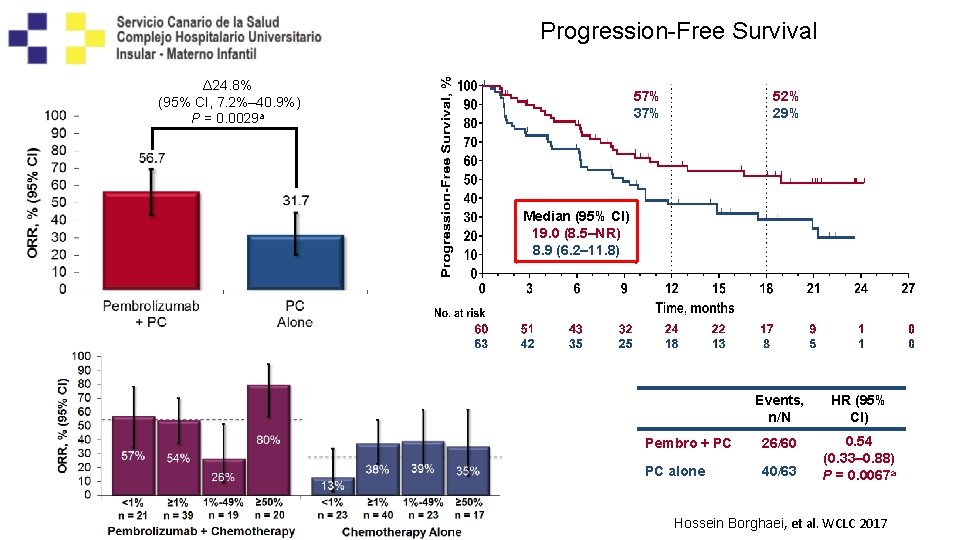
Progression-Free Survival Δ 24. 8% (95% CI, 7. 2%‒ 40. 9%) P = 0. 0029 a 57% 37% 52% 29% Median (95% CI) 19. 0 (8. 5–NR) 8. 9 (6. 2– 11. 8) Events, n/N HR (95% CI) Pembro + PC 26/60 PC alone 40/63 0. 54 (0. 33– 0. 88) P = 0. 0067 a Hossein Borghaei, et al. WCLC 2017
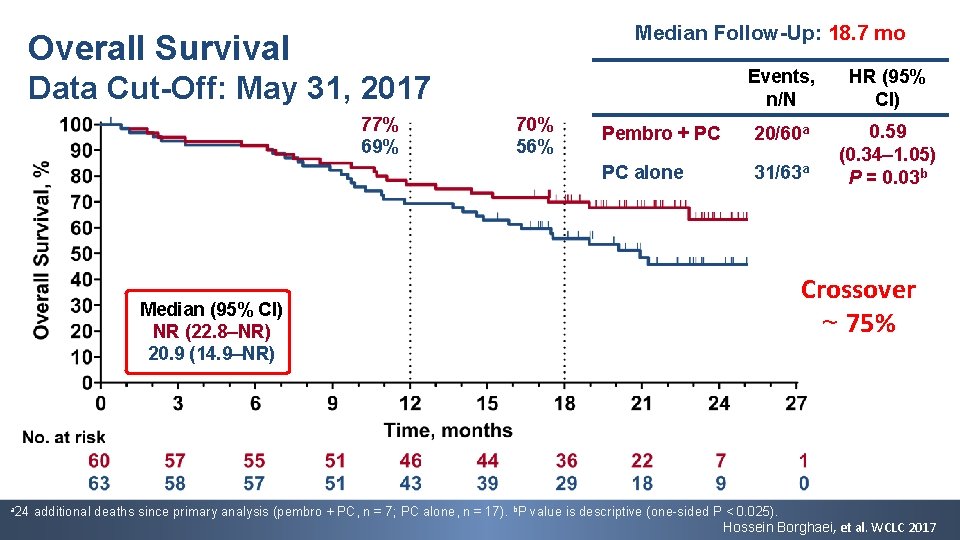
Median Follow-Up: 18. 7 mo Overall Survival Events, n/N HR (95% CI) Pembro + PC 20/60 a PC alone 31/63 a 0. 59 (0. 34– 1. 05) P = 0. 03 b Data Cut-Off: May 31, 2017 77% 69% 70% 56% Crossover ~ 75% Median (95% CI) NR (22. 8–NR) 20. 9 (14. 9–NR) a 24 additional deaths since primary analysis (pembro + PC, n = 7; PC alone, n = 17). b. P value is descriptive (one-sided P < 0. 025). Hossein Borghaei, et al. WCLC 2017
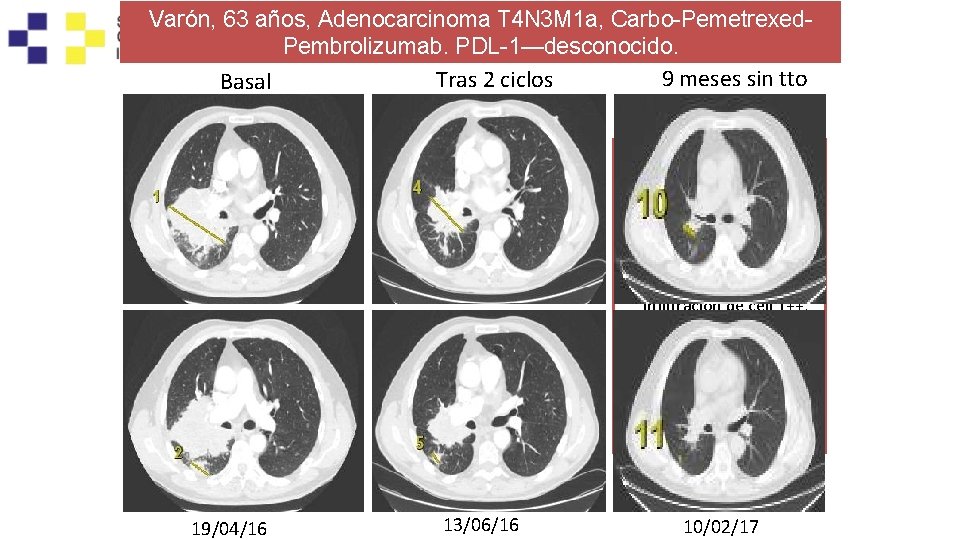
Varón, 63 años, Adenocarcinoma T 4 N 3 M 1 a, Carbo-Pemetrexed. Pembrolizumab. PDL-1—desconocido. 9 meses sin tto Tras 2 ciclos Basal Última dosis: 6 -mayo-2016 • 15/06/16—Creat-4. 6. • Biopsia Renal: Nefritis intersticial aguda medicamentosa con infiltración de cell T++, con granulomas epiteloides no necrotizantes. • Metilpredniosolona 1. 5 mg/kg. 19/04/16 13/06/16 10/02/17
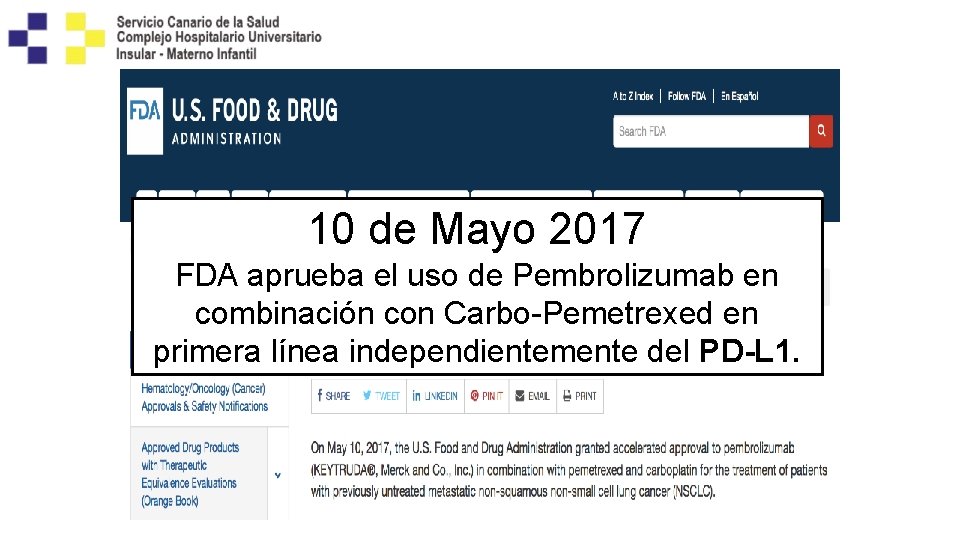
10 de Mayo 2017 FDA aprueba el uso de Pembrolizumab en combinación con Carbo-Pemetrexed en primera línea independientemente del PD-L 1.
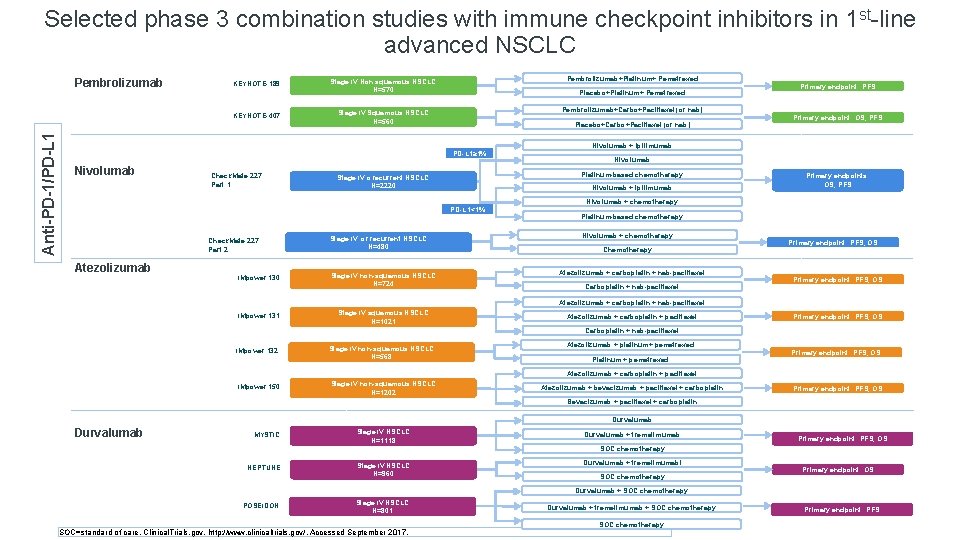
Selected phase 3 combination studies with immune checkpoint inhibitors in 1 st-line advanced NSCLC Pembrolizumab KEYNOTE-189 Anti-PD-1/PD-L 1 KEYNOTE-407 Stage IV Non squamous NSCLC N=570 Pembrolizumab+Platinum+ Pemetrexed Stage IV Squamous NSCLC N=560 Pembrolizumab+Carbo+Paclitaxel (or nab) Placebo+Platinum+ Pemetrexed Placebo+Carbo+Paclitaxel (or nab) PD-L 1≥ 1% Nivolumab Check. Mate 227 Part 1 Check. Mate 227 Part 2 Atezolizumab IMpower 130 Stage IV or recurrent NSCLC N=480 Stage IV non-squamous NSCLC N=724 Nivolumab + ipilimumab PD-L 1<1% Primary endpoint: OS, PFS Nivolumab + ipilimumab Platinum-based chemotherapy Stage IV o recurrent NSCLC N=2220 Primary endpoint: PFS Primary endpoints: OS, PFS Nivolumab + chemotherapy Platinum-based chemotherapy Nivolumab + chemotherapy Chemotherapy Atezolizumab + carboplatin + nab-paclitaxel Carboplatin + nab-paclitaxel Primary endpoint: PFS, OS Atezolizumab + carboplatin + nab-paclitaxel IMpower 131 Stage IV squamous NSCLC N=1021 Atezolizumab + carboplatin + paclitaxel Stage IV non-squamous NSCLC N=568 Atezolizumab + platinum+ pemetrexed Primary endpoint: PFS, OS Carboplatin + nab-paclitaxel IMpower 132 Platinum + pemetrexed Primary endpoint: PFS, OS Atezolizumab + carboplatin + paclitaxel IMpower 150 Stage IV non-squamous NSCLC N=1202 Atezolizumab + bevacizumab + paclitaxel + carboplatin Primary endpoint: PFS, OS Bevacizumab + paclitaxel + carboplatin Durvalumab MYSTIC Stage IV NSCLC N=1118 Durvalumab + tremelimumab Primary endpoint: PFS, OS SOC chemotherapy NEPTUNE Stage IV NSCLC N=960 Durvalumab + tremelimumabl SOC chemotherapy Primary endpoint: OS Durvalumab + SOC chemotherapy POSEIDON Stage IV NSCLC N=801 SOC=standard of care. Clinical. Trials. gov. http: //www. clinicaltrials. gov/. Accessed September 2017. Durvalumab + tremelimumab + SOC chemotherapy Primary endpoint: PFS

Planned and ongoing trials Da kommt einiges auf uns zu. . .
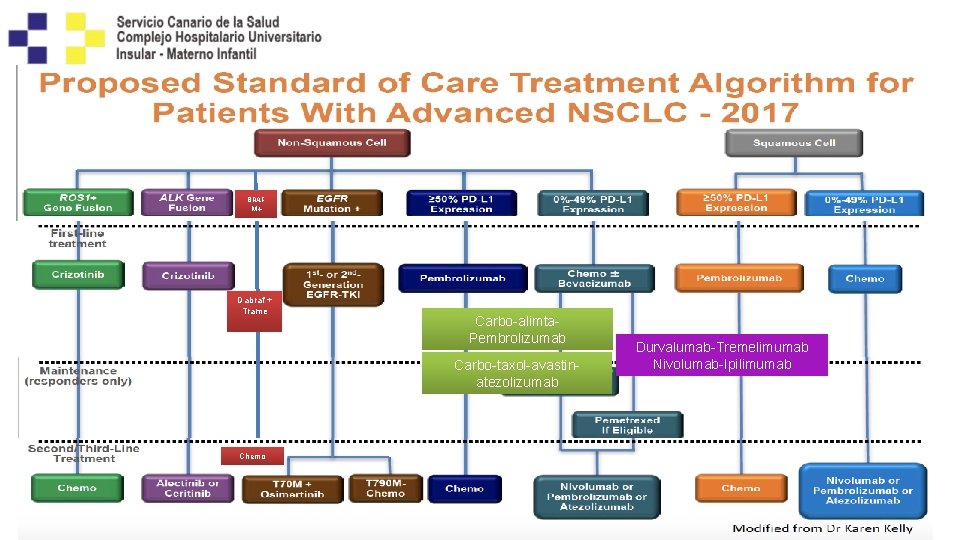
BRAF M+ Dabraf + Trame Carbo-alimta. Pembrolizumab Carbo-taxol-avastinatezolizumab Chemo Durvalumab-Tremelimumab Nivolumab-Ipilimumab

Mensajes • La Inmunoterapia funciona en cáncer de pulmón. • Confirmada superioridad de Inmunoterapia frente a Docetaxel. • En primera línia PD-L 1>50%--PEMBROLIZUMAB nuevo estándar. • Alta tasa de respuestas y duraderas, menor toxicidad, aumento de Supervivencia Global. • Las Combinaciones ya están cerca!! • Necesidad de mejores biomarcadores. • No olvidar la toxicidad!! Es diferente.

Ver enfermos sin leer libros es como navegar sin tener carta de navegación, pero leer libros sin ver enfermos es como nunca haber navegado. William Osler (1849 -1919) Padre de la Medicina Moderna.

GRACIAS !! Dr. Delvys Rodríguez Abreu Servicio Oncología Médica Hospital Universitario Insular de Gran Canaria drodabr@gobiernodecanarias. org
- Slides: 55