Inherited Retinal Disease Milestones Reasons to be Excited

Inherited Retinal Disease Milestones: Reasons to be Excited Brian Mansfield, Ph. D Senior Vice President, Research

1971

Affected Families Drive Research Gordon and Llura Gund Eliot Berson, MD, Ben and Beverley Mass. Eye and Ear Berman-Gund Laboratory for the Study of Retinal Degenerations 1968 National Eye Institute 1971 Foundation Fighting Blindness 2018 >55 Organizations in US alone Founded in 1971, the Foundation Fighting Blindness has raised more than $725 MM toward R&D and public health education
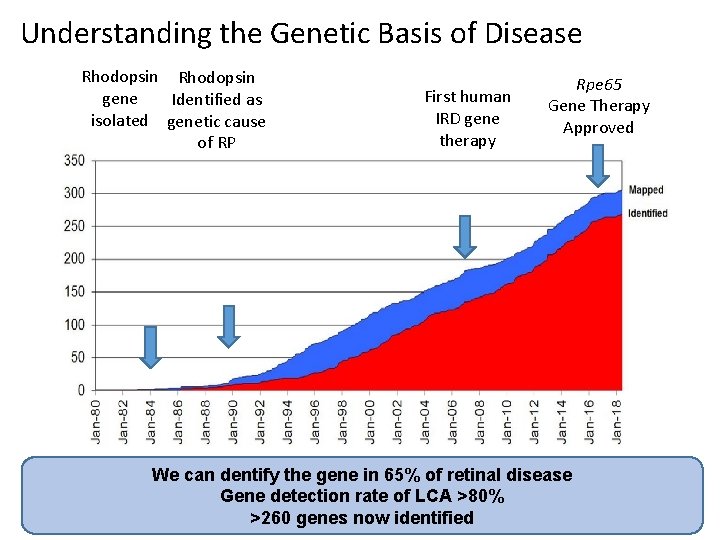
Understanding the Genetic Basis of Disease Rhodopsin gene Identified as isolated genetic cause of RP First human IRD gene therapy Rpe 65 Gene Therapy Approved We can dentify the gene in 65% of retinal disease Gene detection rate of LCA >80% >260 genes now identified

2018 - Approved Products United States: • Argus II Prosthesis 55 electrodes, epiretinal Second Sight • Luxturna RPE 65 Gene Therapy Spark Therapeutics Europe: • IRIS II • Alpha IMS 150 electrodes, subretinal 1, 600 electrodes, subretinal, no camera Compassionate Use – UK • LCA - AIPL 1 - Meira. GTx Pixium Vision Retina Implant
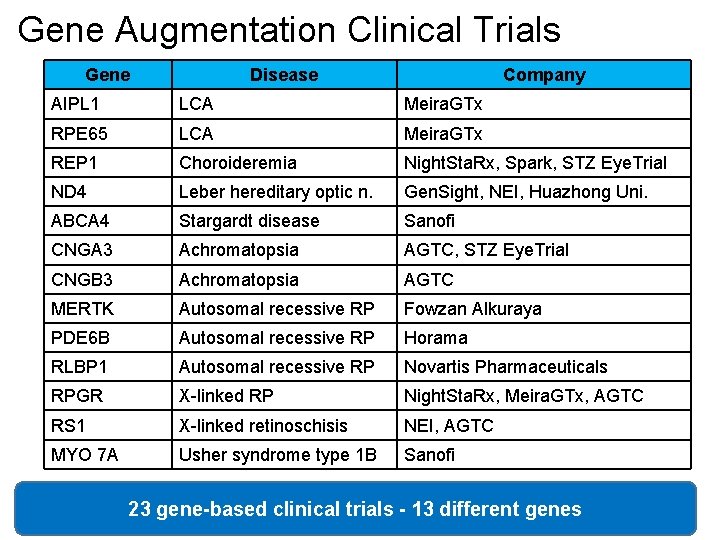
Gene Augmentation Clinical Trials Gene Disease Company AIPL 1 LCA Meira. GTx RPE 65 LCA Meira. GTx REP 1 Choroideremia Night. Sta. Rx, Spark, STZ Eye. Trial ND 4 Leber hereditary optic n. Gen. Sight, NEI, Huazhong Uni. ABCA 4 Stargardt disease Sanofi CNGA 3 Achromatopsia AGTC, STZ Eye. Trial CNGB 3 Achromatopsia AGTC MERTK Autosomal recessive RP Fowzan Alkuraya PDE 6 B Autosomal recessive RP Horama RLBP 1 Autosomal recessive RP Novartis Pharmaceuticals RPGR X-linked RP Night. Sta. Rx, Meira. GTx, AGTC RS 1 X-linked retinoschisis NEI, AGTC MYO 7 A Usher syndrome type 1 B Sanofi 23 gene-based clinical trials - 13 different genes

Optogenetic Gene Therapy Clinical Trials • Photoreceptors lost • Bacterial light-sensitive gene • Insert into healthy retinal cells – bipolar, ganglion cells Gene Disease Company CHR 2 Optogenetics -RP Allergan Chrimson Optogenetics - RP Gensight Biologics There is a pipeline of >16 similar therapies in preclinical development

Gene Patch Clinical Trials • Gene itself does not encode a bad protein • The instructions that drive the gene fool it to act wrongly • “Deep Intronic” mutations • A small DNA molecule is injected to cover up the faulty instruction • Not virus – repeat injections every few months Gene CEP 290 Disease LCA 10 - single mutation - p. Cys 998 X Company Pro. QR • 3 month interim data – well tolerated, improved VA, mobility course • Planning a pivotal Phase 2/3 trial A similar approach for a group of mutations in USH 2 A is near clinic
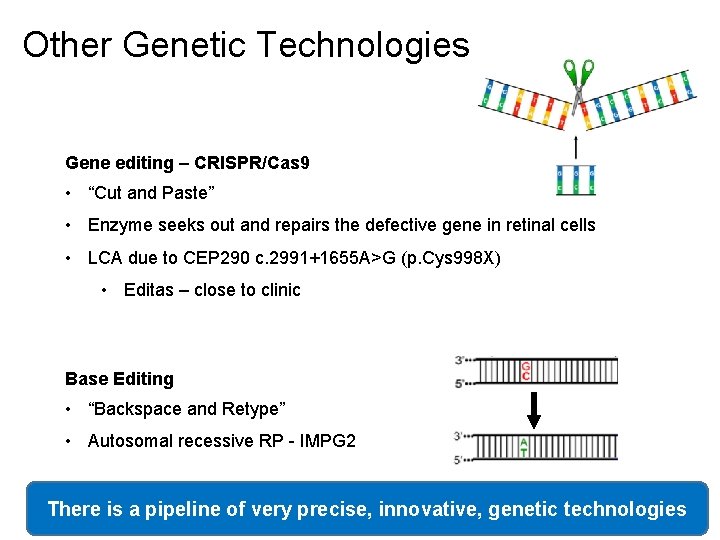
Other Genetic Technologies Gene editing – CRISPR/Cas 9 • “Cut and Paste” • Enzyme seeks out and repairs the defective gene in retinal cells • LCA due to CEP 290 c. 2991+1655 A>G (p. Cys 998 X) • Editas – close to clinic Base Editing • “Backspace and Retype” • Autosomal recessive RP - IMPG 2 There is a pipeline of very precise, innovative, genetic technologies
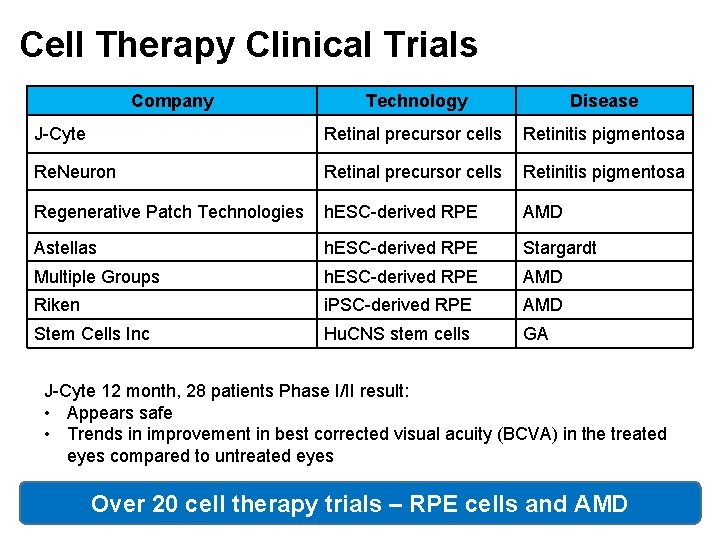
Cell Therapy Clinical Trials Company Technology Disease J-Cyte Retinal precursor cells Retinitis pigmentosa Re. Neuron Retinal precursor cells Retinitis pigmentosa Regenerative Patch Technologies h. ESC-derived RPE AMD Astellas h. ESC-derived RPE Stargardt Multiple Groups h. ESC-derived RPE AMD Riken i. PSC-derived RPE AMD Stem Cells Inc Hu. CNS stem cells GA J-Cyte 12 month, 28 patients Phase I/II result: • Appears safe • Trends in improvement in best corrected visual acuity (BCVA) in the treated eyes compared to untreated eyes Over 20 cell therapy trials – RPE cells and AMD
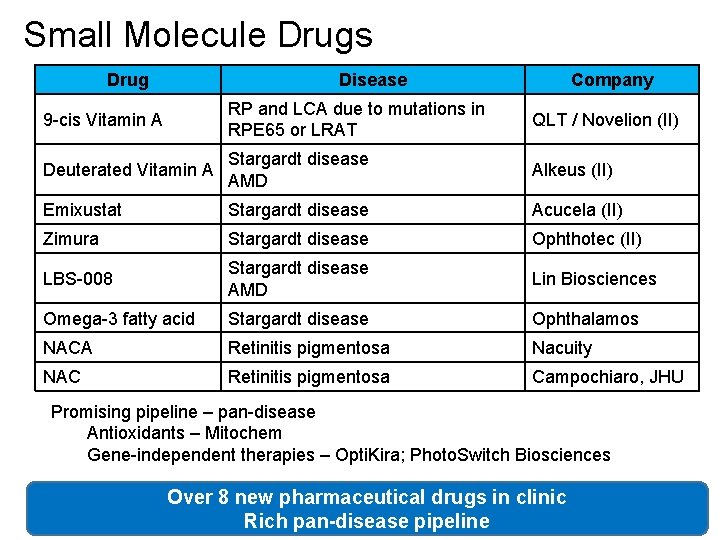
Small Molecule Drugs Drug Disease Company 9 -cis Vitamin A RP and LCA due to mutations in RPE 65 or LRAT QLT / Novelion (II) Deuterated Vitamin A Stargardt disease AMD Alkeus (II) Emixustat Stargardt disease Acucela (II) Zimura Stargardt disease Ophthotec (II) LBS-008 Stargardt disease AMD Lin Biosciences Omega-3 fatty acid Stargardt disease Ophthalamos NACA Retinitis pigmentosa Nacuity NAC Retinitis pigmentosa Campochiaro, JHU Promising pipeline – pan-disease Antioxidants – Mitochem Gene-independent therapies – Opti. Kira; Photo. Switch Biosciences Over 8 new pharmaceutical drugs in clinic Rich pan-disease pipeline

Career Development Awards Founded 1968 Attract and retain young researchers into our field Establish a career – enable them to attract funding – NIH and beyond

Bright Minds - Cutting Edge Innovation David Gamm, Ph. D University of Wisconsin Photoreceptor transplantation i. PSC and microengineering Shannon Boye, Ph. D University of Florida Dual AAV vector-mediated therapy for Myosin 7 a Usher syndrome (USH 1 B) The Foundation continues to fund ground breaking translational research in the area of cell and gene therapy

Focus on Translational Research Providing 16 years of preclinical research support ($10 M) FFB helped, with others, to catalyze the first commercial gene therapy

Industry Expertise Guiding Development Konstantin Petrukhin, Ph. D Columbia University Optimization of Small molecule RBP 4 antagonists for treatment of Stargardt’s disease The Foundation collaborates with the Harrington Discovery Institute to speed translation of new therapies

Strategic Funding of Companies Clinical Gene Therapy Pipeline • • X-linked retinoschisis – RS 1 Achromatopsia – CNGA 3 Achromatopsia – CNGB 3 X-linked retinitis pigmentosa RPGR AGTC leveraged an early FFB investment to garner $265 M to Develop Gene Therapies

Co-Funding Commercialization Start up company founded in 2017 • Based on FFB supported research in Dr. Campochiaro’s lab • NACA – N-acetyl cysteine amide • Independent of the genetic cause of disease • Reduces oxidative stress on cells • Novel aqueous humor clinical biomarkers Oral NACA has just completed Phase 1 clinical testing in Australia

Equity Investments - Startups Start up founded in 2016 • Rod-derived cone viability factor (Rd. CVF) • Pan-disease gene therapy • Technology seeded by FFB grants at several universities – international collaboration • Scientific founders: • Jose Sahel, MD • Thierry Leveillard, Ph. D Sparing Vision is dedicated to developing Rd. CVF as a novel therapeutic target for treating IRDs
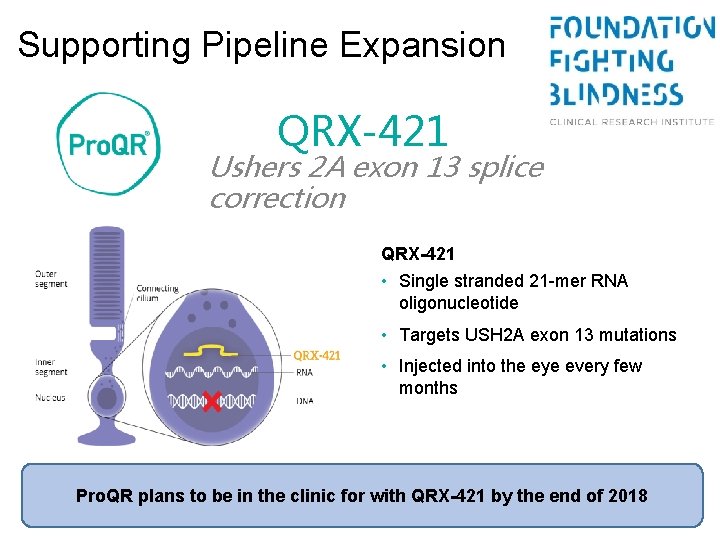
Supporting Pipeline Expansion QRX-421 Ushers 2 A exon 13 splice correction QRX-421 • Single stranded 21 -mer RNA oligonucleotide • Targets USH 2 A exon 13 mutations • Injected into the eye every few months Pro. QR plans to be in the clinic for with QRX-421 by the end of 2018

Understanding Disease Progression Natural History Study of Stargardt disease • How variable is disease? • How does the disease progress? • How do we know if a therapy has changed the course of disease? 11 publications to date Identified and endpoint that is acceptable to the FDA
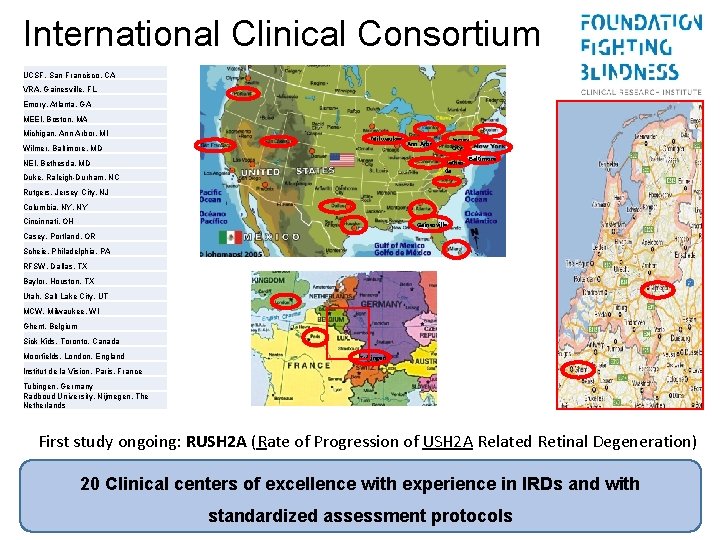
International Clinical Consortium UCSF, San Francisco, CA VRA, Gainesville, FL Emory, Atlanta, GA MEEI, Boston, MA Michigan, Ann Arbor, MI Milwaukee Wilmer, Baltimore, MD NEI, Bethesda, MD Duke, Raleigh-Durham, NC Ann Arbor Jersey City Baltimore Bethes da Rutgers, Jersey City, NJ Columbia, NY Cincinnati, OH Gainesville Casey, Portland, OR Scheie, Philadelphia, PA RFSW, Dallas, TX Baylor, Houston, TX Utah, Salt Lake City, UT MCW, Milwaukee, WI Ghent, Belgium Sick Kids, Toronto, Canada Moorfields, London, England Tubingen Institut de la Vision, Paris, France Tubingen, Germany Radboud University, Nijmegen, The Netherlands First study ongoing: RUSH 2 A (Rate of Progression of USH 2 A Related Retinal Degeneration) 20 Clinical centers of excellence with experience in IRDs and with standardized assessment protocols

Attracting Industry - Registries • What is the best disease to test my technology or drug on? • How many people are affected? • Where are they? • How do we contact them? • What is the age of onset? • How does that disease progress? • What impacts does the disease have on patient lives? • What is a meaningful treatment for patients? i. e. is there an accessible, comprehensive patient registry? A comprehensive registry is a key resource for rare disease drug development 22

Your Knowledge is a Key Resource • Sharing what you know is critical • Your understanding of the disease • How it impacts you and your family in daily life • What would you do for a treatment – injection every week? • What would mean the most to you in a treatment or cure • What would not be acceptable to you in a therapy – your fears • What don’t clinicians, researchers, regulatory agencies really understand • Focus groups • Patient journey • Testing new endpoints - maze Patient input is a critical part of the drug development pathway
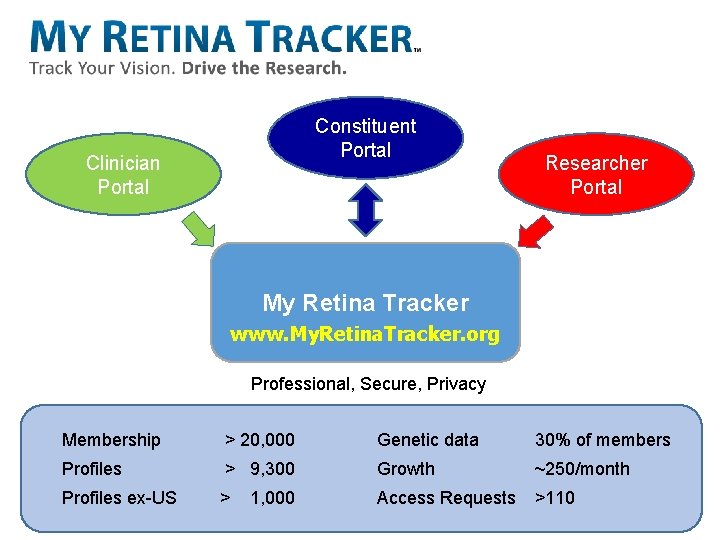
Constituent Portal Clinician Portal Researcher Portal My Retina Tracker www. My. Retina. Tracker. org Professional, Secure, Privacy 24 Membership > 20, 000 Genetic data 30% of members Profiles > 9, 300 Growth ~250/month Profiles ex-US > Access Requests >110 1, 000
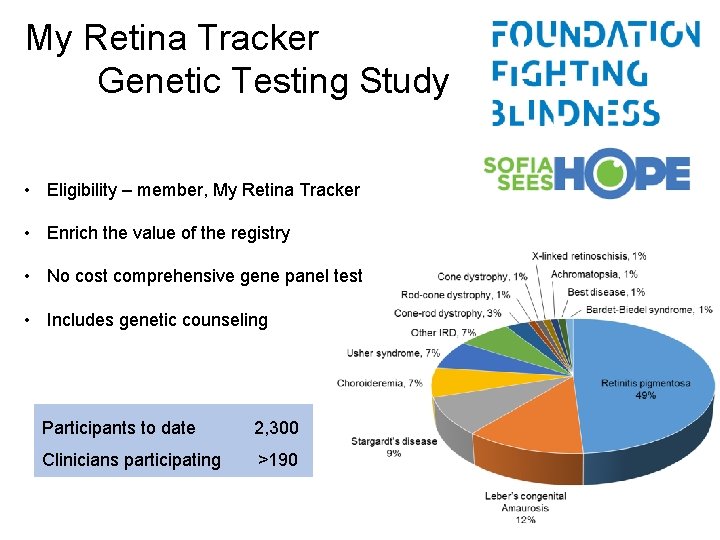
My Retina Tracker Genetic Testing Study • Eligibility – member, My Retina Tracker • Enrich the value of the registry • No cost comprehensive gene panel test • Includes genetic counseling Participants to date 2, 300 Clinicians participating >190

BEAM Mito. Chem Therapeutics, Inc. Over 40 biotechs and companies developing novel therapies Most in contact with the Foundation

Collaboration Sofia Sees Hope – Foundation Fighting Blindness: • Jointly promote awareness of: • Retinal disease research and resources – quarterly newsletters • Importance of patient registries • Importance of genetic testing • Jointly authored LCA Information and Resource page • National Organization for Rare Disorders • Co-fund: • My Retina Tracker Genetic Testing Study • Key research especially related to LCA • Communicate and collaborate closely to accelerate treatments and cures

- Slides: 28