INFRAREDACTIVE VIBRON BANDS ASSOCIATED WITH RARE GAS SUBSTITUTIONAL
INFRARED-ACTIVE VIBRON BANDS ASSOCIATED WITH RARE GAS SUBSTITUTIONAL IMPURITIES IN SOLID HYDROGEN PAUL L. RASTON and DAVID T. ANDERSON, Department of Chemistry, University of Wyoming, Laramie, WY 82071 Ne H 2 Ar Kr Xe

H 2 -Rare gas (Rg) Systems * Important for understanding basic van der Waals interactions. * Model systems with regard to determination of accurate potential energy surfaces from experimental data. * Attractive to study because of the relative simplicity and fundamental nature of these systems. H 2 -Rg: R. J. Le Roy and J. M. Hutson, J. Chem. Phys. 86, 837 (1987). H 2 -H 2: P. Diep, and J. K. Johnson, J. Chem. Phys. 112, 4465 (2000).
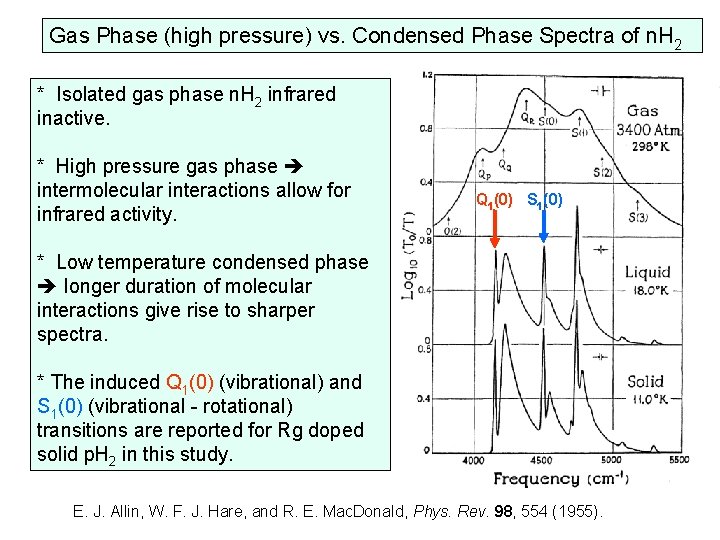
Gas Phase (high pressure) vs. Condensed Phase Spectra of n. H 2 * Isolated gas phase n. H 2 infrared inactive. * High pressure gas phase intermolecular interactions allow for infrared activity. Q 1(0) S 1(0) * Low temperature condensed phase longer duration of molecular interactions give rise to sharper spectra. * The induced Q 1(0) (vibrational) and S 1(0) (vibrational - rotational) transitions are reported for Rg doped solid p. H 2 in this study. E. J. Allin, W. F. J. Hare, and R. E. Mac. Donald, Phys. Rev. 98, 554 (1955).
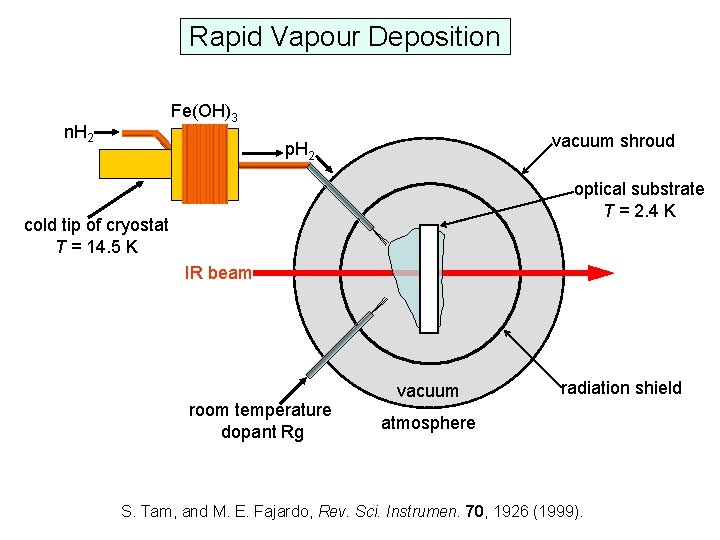
Rapid Vapour Deposition Fe(OH)3 n. H 2 vacuum shroud p. H 2 optical substrate T = 2. 4 K cold tip of cryostat T = 14. 5 K IR beam room temperature dopant Rg vacuum radiation shield atmosphere S. Tam, and M. E. Fajardo, Rev. Sci. Instrumen. 70, 1926 (1999).
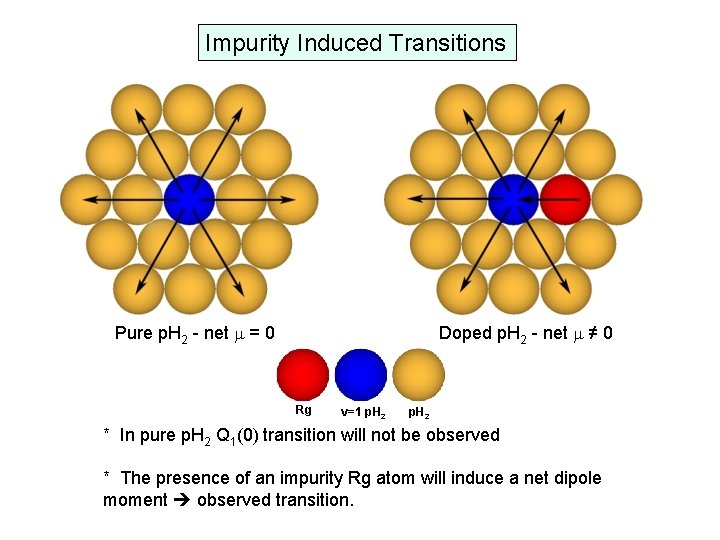
Impurity Induced Transitions Pure p. H 2 - net m = 0 Doped p. H 2 - net m ≠ 0 Rg v=1 p. H 2 * In pure p. H 2 Q 1(0) transition will not be observed * The presence of an impurity Rg atom will induce a net dipole moment observed transition.
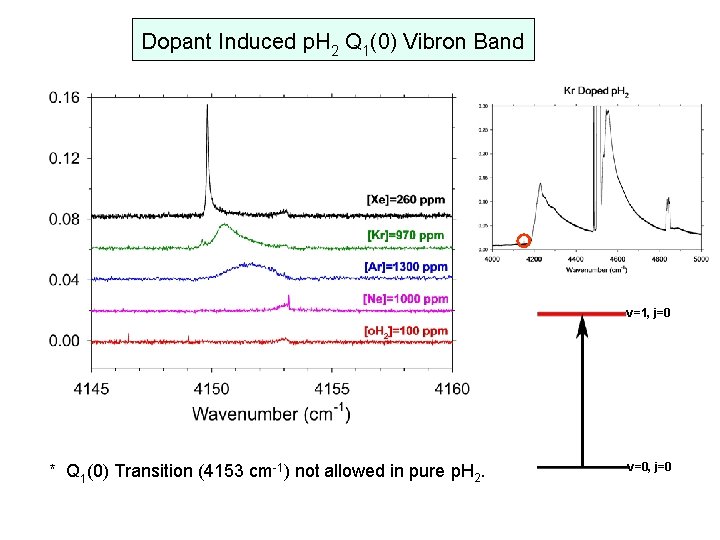
Dopant Induced p. H 2 Q 1(0) Vibron Band v=1, j=0 * Q 1(0) Transition (4153 cm-1) not allowed in pure p. H 2. v=0, j=0
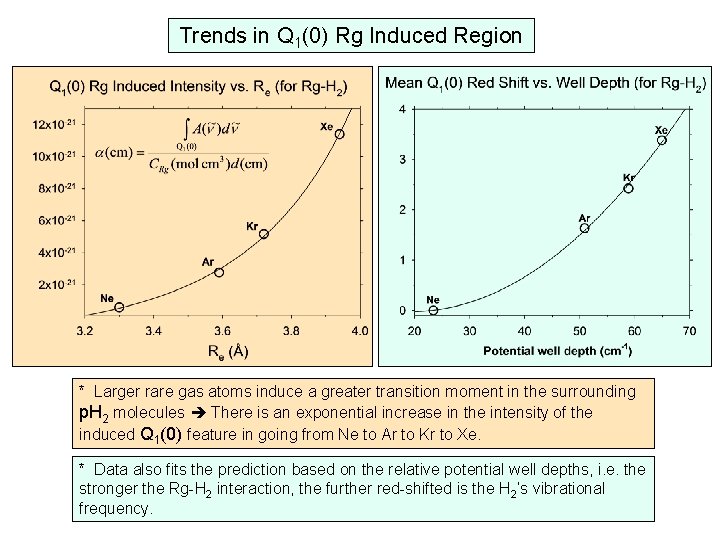
Trends in Q 1(0) Rg Induced Region * Larger rare gas atoms induce a greater transition moment in the surrounding p. H 2 molecules There is an exponential increase in the intensity of the induced Q 1(0) feature in going from Ne to Ar to Kr to Xe. * Data also fits the prediction based on the relative potential well depths, i. e. the stronger the Rg-H 2 interaction, the further red-shifted is the H 2’s vibrational frequency.
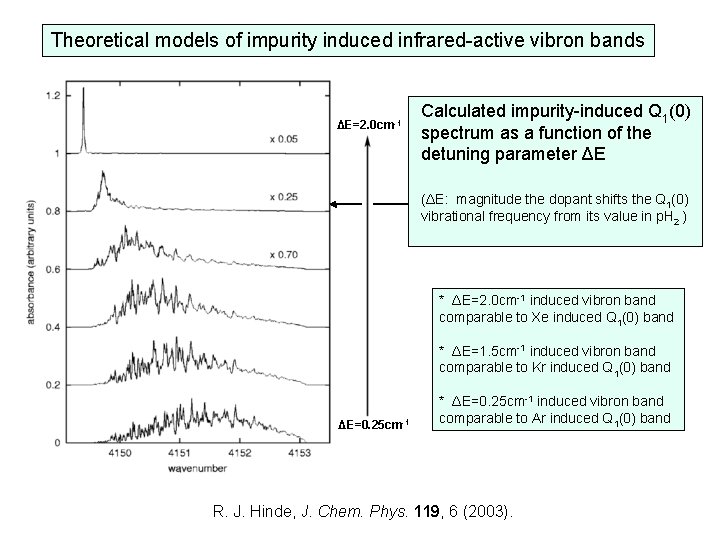
Theoretical models of impurity induced infrared-active vibron bands ΔE=2. 0 cm-1 Calculated impurity-induced Q 1(0) spectrum as a function of the detuning parameter ΔE (ΔE: magnitude the dopant shifts the Q 1(0) vibrational frequency from its value in p. H 2 ) * ΔE=2. 0 cm-1 induced vibron band comparable to Xe induced Q 1(0) band * ΔE=1. 5 cm-1 induced vibron band comparable to Kr induced Q 1(0) band ΔE=0. 25 cm-1 * ΔE=0. 25 cm-1 induced vibron band comparable to Ar induced Q 1(0) band R. J. Hinde, J. Chem. Phys. 119, 6 (2003).

Localized vs. Delocalized Vibron Localized vibron Delocalized vibron
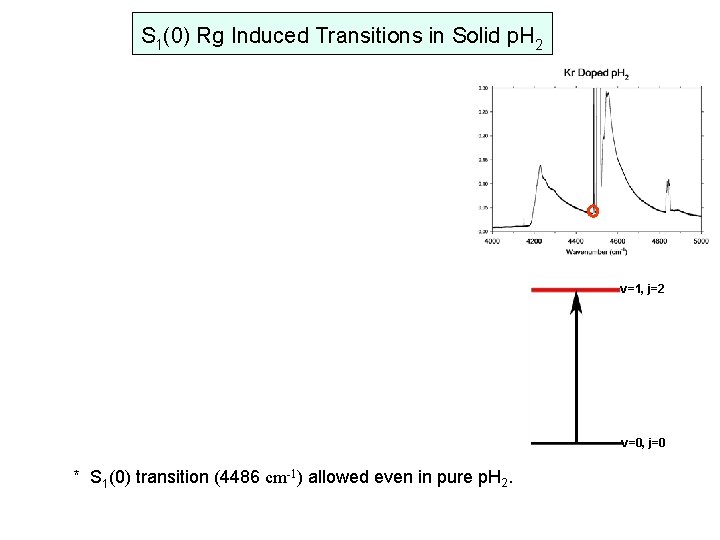
S 1(0) Rg Induced Transitions in Solid p. H 2 v=1, j=2 v=0, j=0 * S 1(0) transition (4486 cm-1) allowed even in pure p. H 2.

S 1(0) Xe Induced Transitions in Solid p. H 2 * Complicated spectrum – why a minimum of 6 satellite peaks…
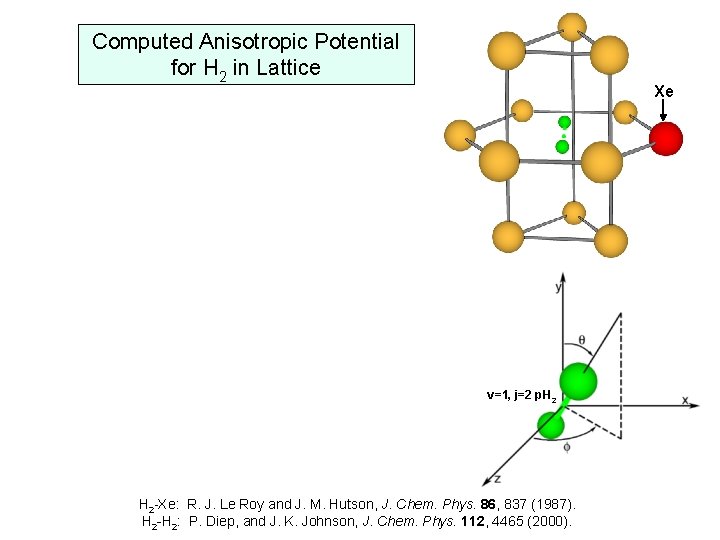
Computed Anisotropic Potential for H 2 in Lattice Xe v=1, j=2 p. H 2 -Xe: R. J. Le Roy and J. M. Hutson, J. Chem. Phys. 86, 837 (1987). H 2 -H 2: P. Diep, and J. K. Johnson, J. Chem. Phys. 112, 4465 (2000).

Calculated magnitude of splitting of J=2 level by Xe in hcp lattice 357. 56 cm-1 m. J=± 2 356. 02 cm-1 3 cm-1 (total observed splitting is ~1. 5 cm-1) 355. 28 cm-1 m. J=± 1 354. 54 cm-1 m. J=0 * Inherent axial symmetry to each substitutional crystal site in hcp lattice lifting of the J=2 upper state giving three rotational states with m. J = ± 2, ± 1, and 0. * Two different substitutional sites exist for a Xenon impurity in a p. H 2 hcp lattice, in-plane (IP) and out-of-plane (OP) Potential explanation for the 3 x 2 = 6 excited state levels…
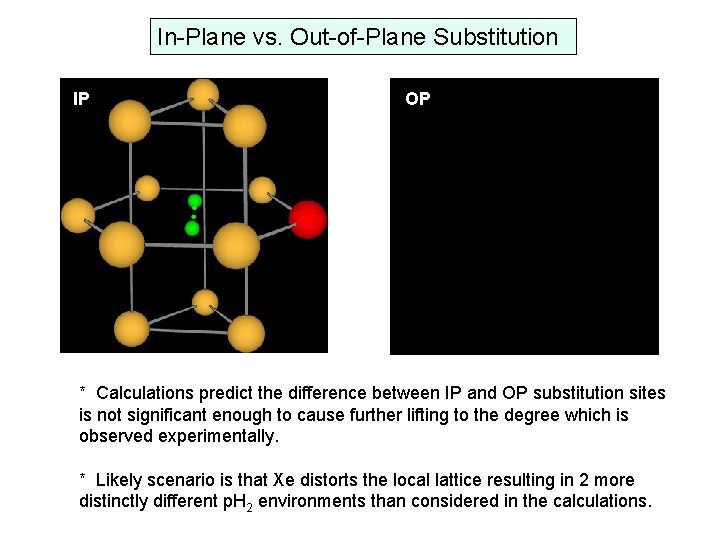
In-Plane vs. Out-of-Plane Substitution IP OP * Calculations predict the difference between IP and OP substitution sites is not significant enough to cause further lifting to the degree which is observed experimentally. * Likely scenario is that Xe distorts the local lattice resulting in 2 more distinctly different p. H 2 environments than considered in the calculations.
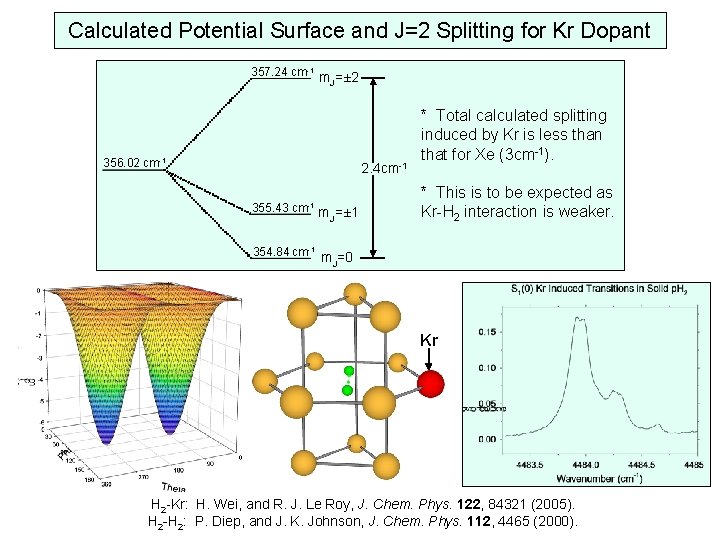
Calculated Potential Surface and J=2 Splitting for Kr Dopant 357. 24 cm-1 m =± 2 J 356. 02 cm-1 2. 4 cm-1 355. 43 cm-1 m =± 1 J 354. 84 cm-1 * Total calculated splitting induced by Kr is less than that for Xe (3 cm-1). * This is to be expected as Kr-H 2 interaction is weaker. m. J=0 Kr H 2 -Kr: H. Wei, and R. J. Le Roy, J. Chem. Phys. 122, 84321 (2005). H 2 -H 2: P. Diep, and J. K. Johnson, J. Chem. Phys. 112, 4465 (2000).

Conclusions * The line shapes of the Q 1(0) features induced by Rg atoms in solid p. H 2 provide information on the extent of localization of the vibron. * The intensity of the Rg atom induced Q 1(0) feature provides information on the induction mechanism and on the Rg-p. H 2 intermolecular potential. * Satellite lines in the S 1(0) region induced by the presence of Rg atoms provide information on the anisotropy of the Rg-p. H 2 potential, and on the anisotropy of the crystal structure when the p. H 2 molecule is in a J=2 rotational state. Future Work * Calculations that take into account distortions in the hcp lattice induced by Xenon…

Acknowledgements * M. E. Fajardo * R. J. Hinde * R. J. Le Roy * B. D. Lorenz * G. V. Subrahmanyam * Funding Sources: *Petroleum Research Fund *The Research Corporation *National Science Foundation

End


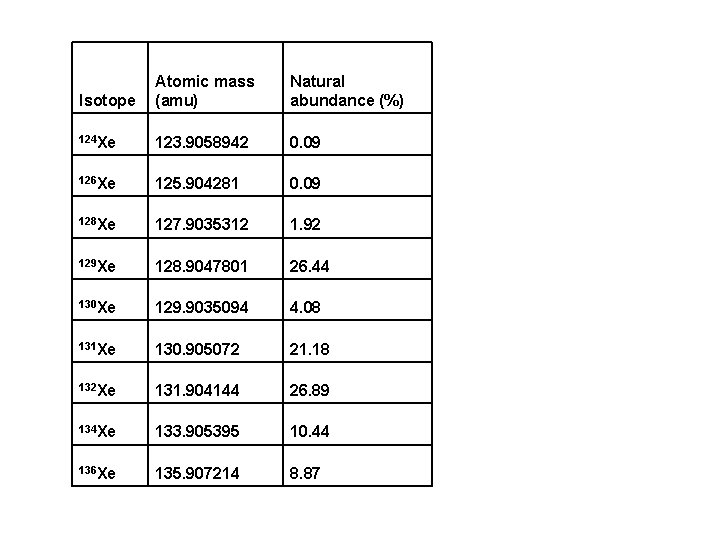
Isotope Atomic mass (amu) Natural abundance (%) 124 Xe 123. 9058942 0. 09 126 Xe 125. 904281 0. 09 128 Xe 127. 9035312 1. 92 129 Xe 128. 9047801 26. 44 130 Xe 129. 9035094 4. 08 131 Xe 130. 905072 21. 18 132 Xe 131. 904144 26. 89 134 Xe 133. 905395 10. 44 136 Xe 135. 907214 8. 87
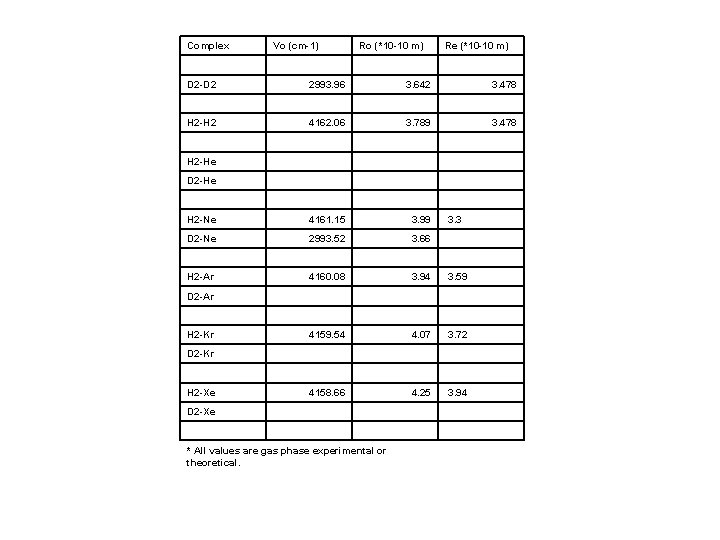
Complex Vo (cm-1) Ro (*10 -10 m) Re (*10 -10 m) D 2 -D 2 2993. 96 H 2 -H 2 3. 642 4162. 06 3. 478 3. 789 3. 478 H 2 -He D 2 -He H 2 -Ne 4161. 15 3. 99 3. 3 D 2 -Ne 2993. 52 3. 66 H 2 -Ar 4160. 08 3. 94 3. 59 D 2 -Ar H 2 -Kr 4159. 54 4. 07 3. 72 D 2 -Kr H 2 -Xe 4158. 66 4. 25 3. 94 D 2 -Xe * All values are gas phase experimental or theoretical.
- Slides: 22