Infrared Studies of Ammonia Borane Kevin Meaux Background

Infrared Studies of Ammonia Borane Kevin Meaux

Background Alternatives to fossil fuels to power transportation are sought To use hydrogen for fuel, a more effective way of storing H 2 is needed High volumetric density, hydrogen mass %, stability, and reversibility are desired Liquid and gaseous H 2 are not dense enough Metal hydrides and similar compounds offer potential solutions

Ammonia Borane (H 3 N: BH 3) description Melting point Nonhazardous stable white powder 1 104 ⁰C (377 K) Density 0. 78 g/cm 3 Mass % H 19. 6% Hydrogen Density 0. 153 g H/cm 3 H 3 N: BH 3 1 For Comparison… Mass % H of Li. Al. H 4 10. 6% Mass % H of Na. BH 4 10. 7% Density of L H 2 0. 068 g/cm 3 [1] Marder, T. , Angewandte Chemie International Edition, 2007, 46, 8116– 8118.

Goal For effective use, more info is needed on the dehydrogenation process of ammonia borane (AB) Important first steps involve developing a working experimental setup to study this change
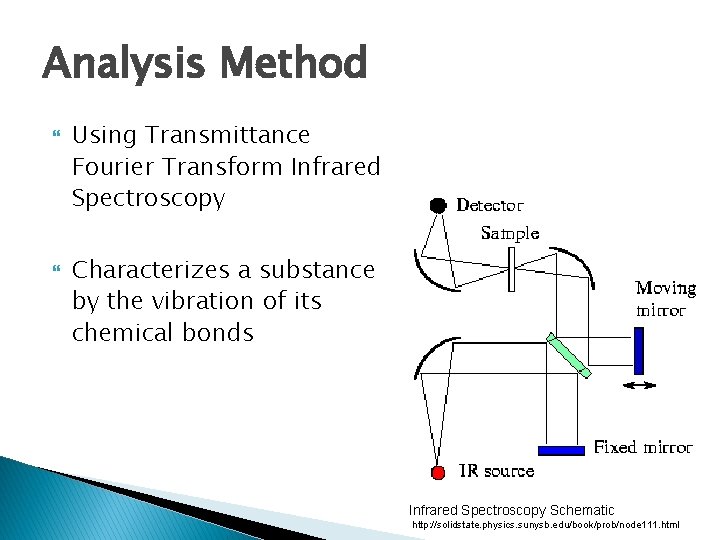
Analysis Method Using Transmittance Fourier Transform Infrared Spectroscopy Characterizes a substance by the vibration of its chemical bonds Infrared Spectroscopy Schematic http: //solidstate. physics. sunysb. edu/book/prob/node 111. html

Original Experimental Setup KBr and NH 3 BH 3 are mixed in a 25: 1 ratio KBr alone and the KBr/AB mixture are pressed into a tungsten mesh in different spots Have varied the reference salt, absolute amounts, and ratios
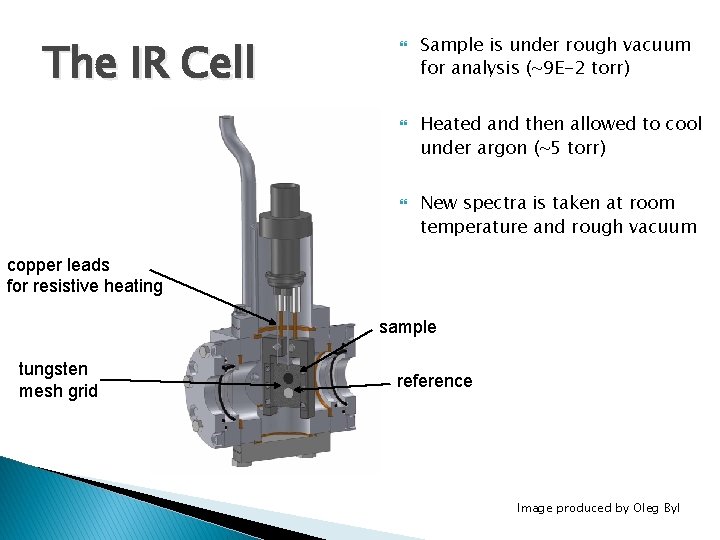
The IR Cell Sample is under rough vacuum for analysis (~9 E-2 torr) Heated and then allowed to cool under argon (~5 torr) New spectra is taken at room temperature and rough vacuum copper leads for resistive heating sample tungsten mesh grid reference Image produced by Oleg Byl
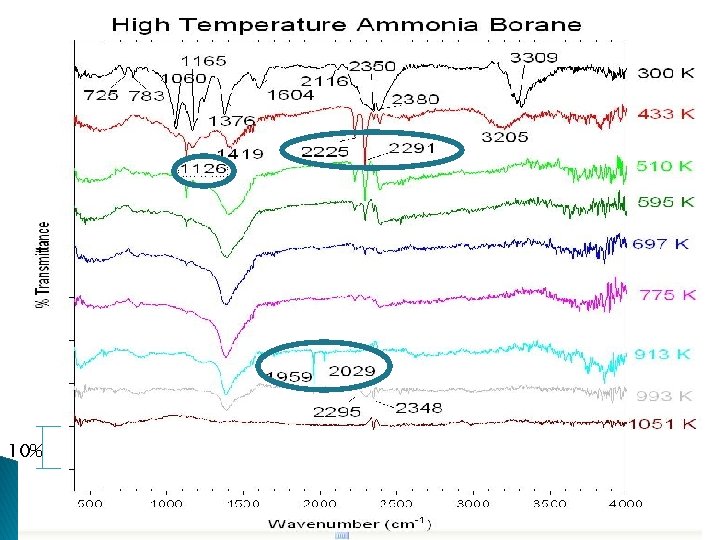
10%
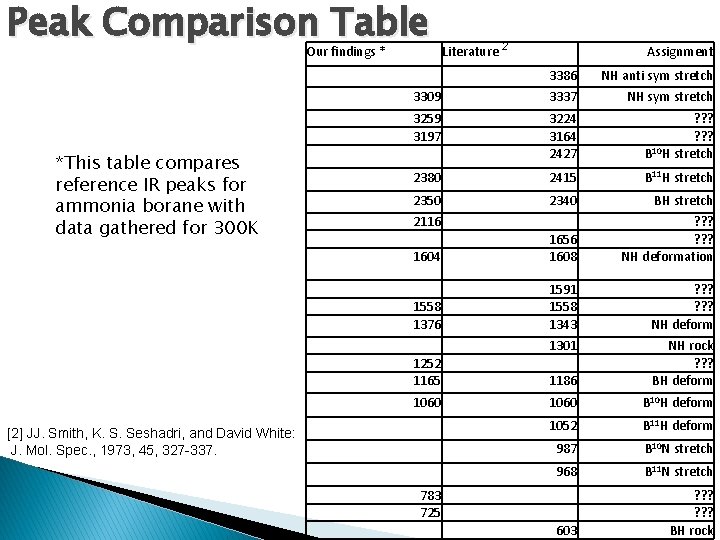
Peak Comparison Table Our findings * *This table compares reference IR peaks for ammonia borane with data gathered for 300 K Literature 2 Assignment 3386 NH anti sym stretch 3309 3337 NH sym stretch 3259 3197 3224 3164 2427 ? ? ? 10 B H stretch 2380 2415 B 11 H stretch 2350 2340 BH stretch 1604 1656 1608 ? ? ? NH deformation 1558 1376 1591 1558 1343 ? ? ? NH deform 1301 2116 1252 1165 1186 NH rock ? ? ? BH deform 1060 B 10 H deform 1052 B 11 H deform 987 B 10 N stretch 968 B 11 N stretch 603 ? ? ? BH rock [2] JJ. Smith, K. S. Seshadri, and David White: J. Mol. Spec. , 1973, 45, 327 -337. 783 725

Analysis The 1126, 2225, and 2291 peaks shown forming at 433 K correspond to KBH 4 3 The 1959/2029 peaks formed at 913 K are likely the formation of B 11 O 2/B 10 O 2 anions 4 The other peaks observed at 433 K do not fit with borazine, boron oxides, or boron nitride [3] K. B. Harvey and N. R. Mc. Quaker: Canadian Journal of Chemistry, 1971, 49, 3272 -3281. [4] I. C. Hisatsune and Noelia Haddock Suarez: Inorganic Chemistry, 1964, 3, 168 -174.

Current Sample Prep Idea Had problems with KBr reactivity in pellet preparation Instead it is prepared on a KBr window Dissolved AB in THF Deposited solution on KBr disk Boiled off THF (67 C) Mounted disk onto mesh Thermocouple KBr Window
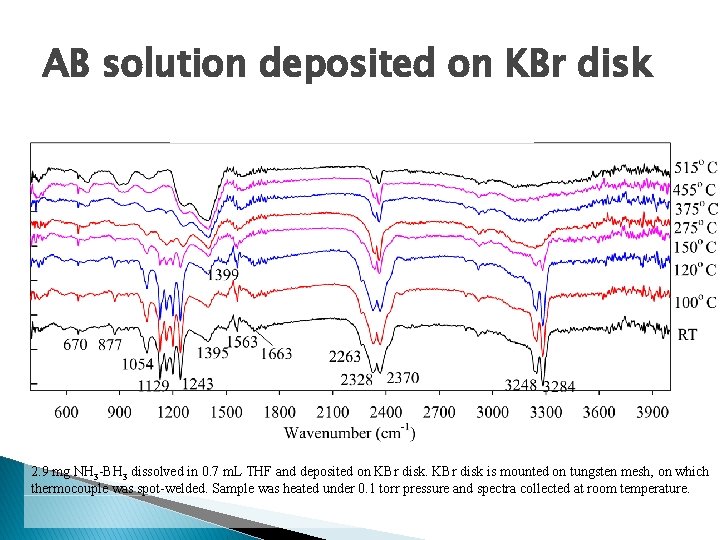
AB solution deposited on KBr disk 2. 9 mg NH 3 -BH 3 dissolved in 0. 7 m. L THF and deposited on KBr disk is mounted on tungsten mesh, on which thermocouple was spot-welded. Sample was heated under 0. 1 torr pressure and spectra collected at room temperature.
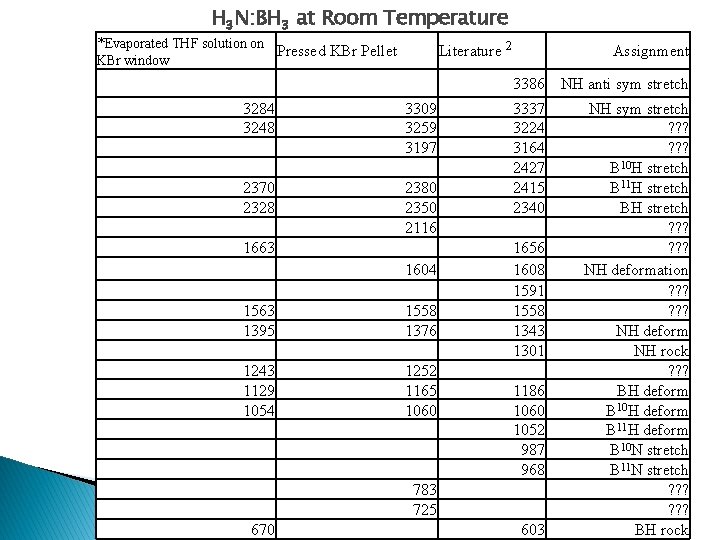
H 3 N: BH 3 at Room Temperature *Evaporated THF solution on KBr window Literature 2 Pressed KBr Pellet Assignment 3386 NH anti sym stretch 3284 3248 3309 3259 3197 2370 2328 2380 2350 2116 1663 1604 1563 1395 1558 1376 1243 1129 1054 1252 1165 1060 3337 3224 3164 2427 2415 2340 1656 1608 1591 1558 1343 1301 1186 1060 1052 987 968 783 725 670 603 NH sym stretch ? ? ? B 10 H stretch B 11 H stretch BH stretch ? ? ? NH deformation ? ? ? NH deform NH rock ? ? ? BH deform B 10 H deform B 11 H deform B 10 N stretch B 11 N stretch ? ? ? BH rock

Analysis The new sample prep method shows more clearly resolved peaks The accuracy of temperatures needs to be verified The peaks are shifted somewhat from literature values The spectra demonstrates dehydrogenation with heating Neither the KBH 4 nor BO 2 - peaks are seen

Summary AB can be useful as a hydrogen storage media More work needs to be done in characterizing its dehydrogenation Current method yields noisy spectra but shows potential Will be dealing with the noise issues in the near future

Acknowledgements This work was supported by the National Science Foundation under EEC-NSF Grant # 1062943 I would like to thank Dr. Trenary and Dr. Jash for their guidance, as well as Dr. Takoudis and Dr. Jursich for administrating the UIC REU program
- Slides: 16