Infrared spectroscopy of the hydrated sulfate dianion Columbus

Infrared spectroscopy of the hydrated sulfate dianion Columbus 2006
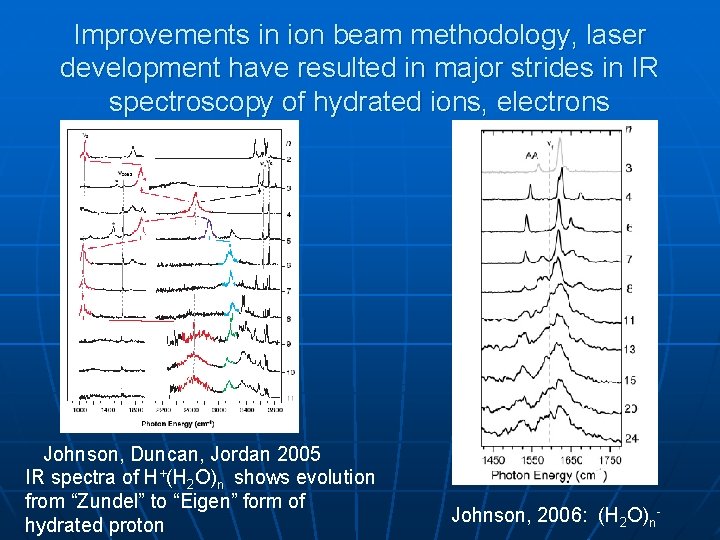
Improvements in ion beam methodology, laser development have resulted in major strides in IR spectroscopy of hydrated ions, electrons Johnson, Duncan, Jordan 2005 IR spectra of H+(H 2 O)n shows evolution from “Zundel” to “Eigen” form of hydrated proton Johnson, 2006: (H 2 O)n-

IR spectra of hydrated multiplycharged anions n n n IR work by Johnson and Okumura on X(H 2 O)n clusters show single hydrogen bonds with solvent molecules What about doublycharged anions? Model system: SO 42 -(H 2 O)n
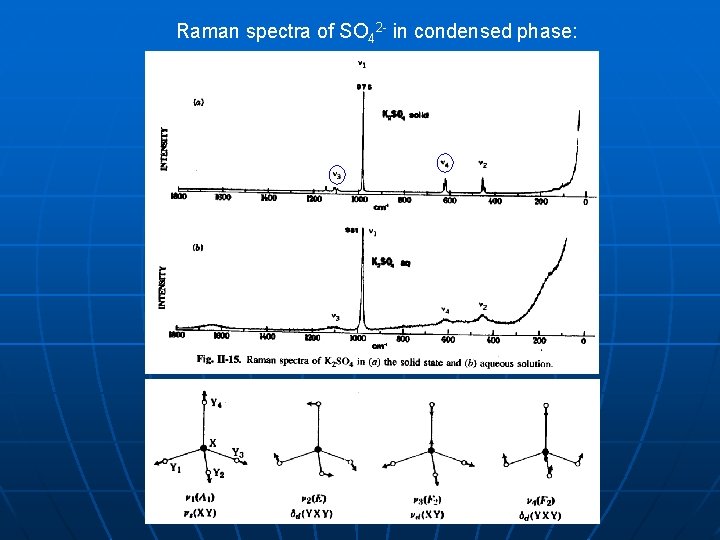
Raman spectra of SO 42 - in condensed phase:
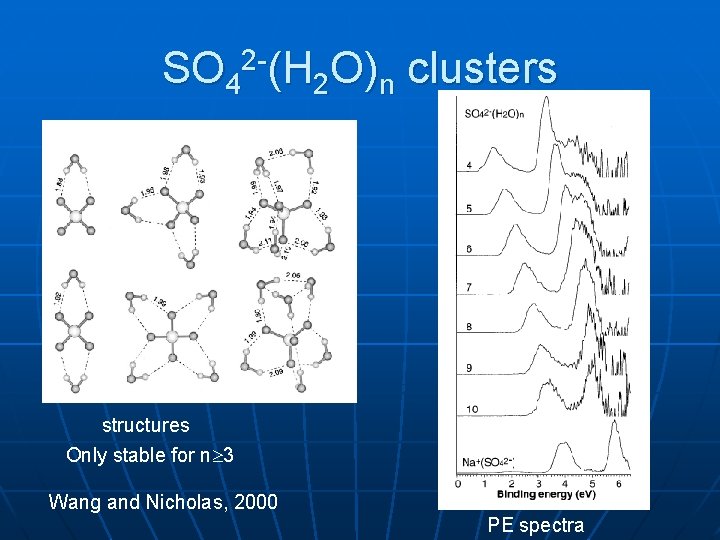
SO 42 -(H 2 O)n clusters structures Only stable for n 3 Wang and Nicholas, 2000 PE spectra
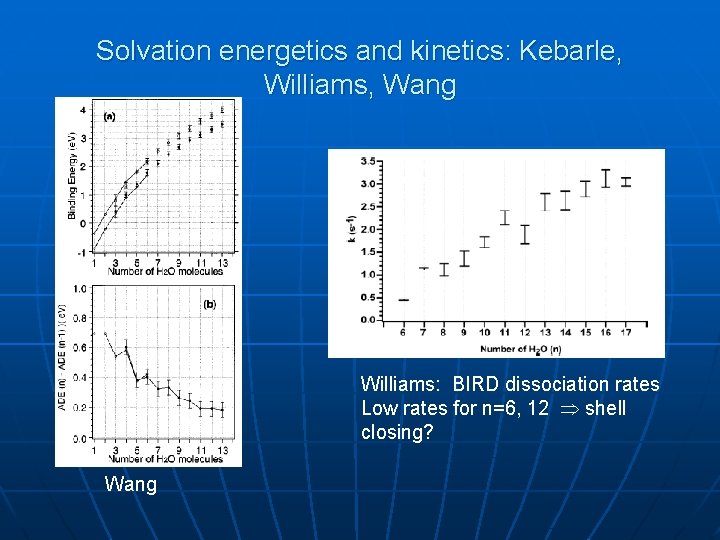
Solvation energetics and kinetics: Kebarle, Williams, Wang Williams: BIRD dissociation rates Low rates for n=6, 12 shell closing? Wang

How to measure IR spectra? n n Need to perform “action spectroscopy” in which photon absorption coupled to ion depletion or production Binding energy (BE) of H 2 O ligands vary from 0. 6 -0. 3 e. V (Wang PE spectra) Expect BE>h for most vibrational transitions Method: Resonant multiphoton absorption
FELIX: Free Electron Laser for Infrared e. Xperiments 100 -2000 cm-1 50 m. J/macropulse 10 Hz, 2% bandwidth

Guided Ion Beam Tandem Mass Spectrometer Ion detection 90° deflector guiding decapole (Ar buffer gas) quadrupole mass filter 1 ← ion sources (parent ions) - laser ablation - ion spray - crossed e‾-beam quadrupole mass filter 2 (product ions) high vacuum 10 -8 mbar cooled RF ion trap (He buffer gas, 16 K) IX L FE
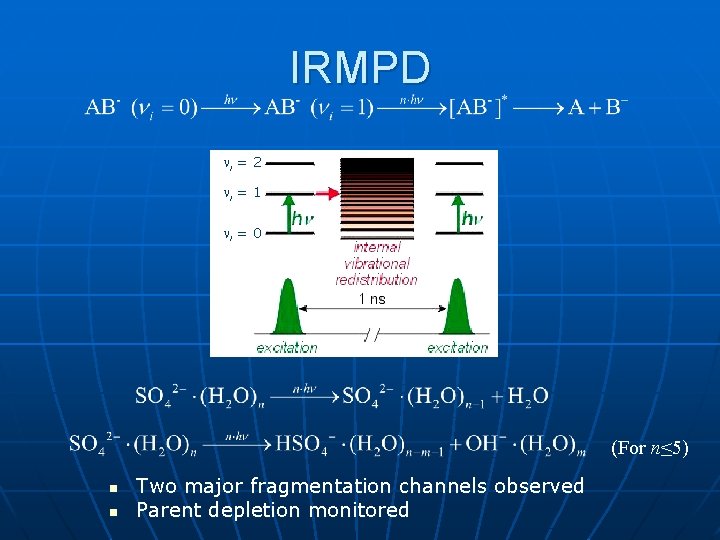
IRMPD i = 2 i = 1 i = 0 (For n≤ 5) n n Two major fragmentation channels observed Parent depletion monitored
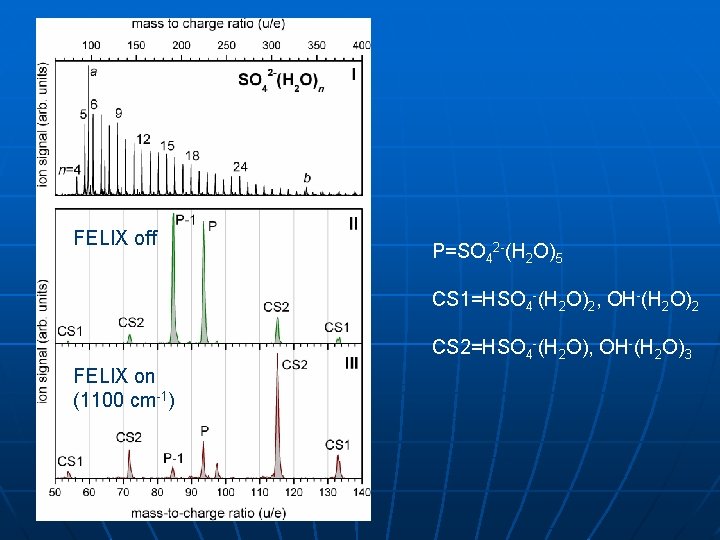
FELIX off P=SO 42 -(H 2 O)5 CS 1=HSO 4 -(H 2 O)2, OH-(H 2 O)2 CS 2=HSO 4 -(H 2 O), OH-(H 2 O)3 FELIX on (1100 cm-1)
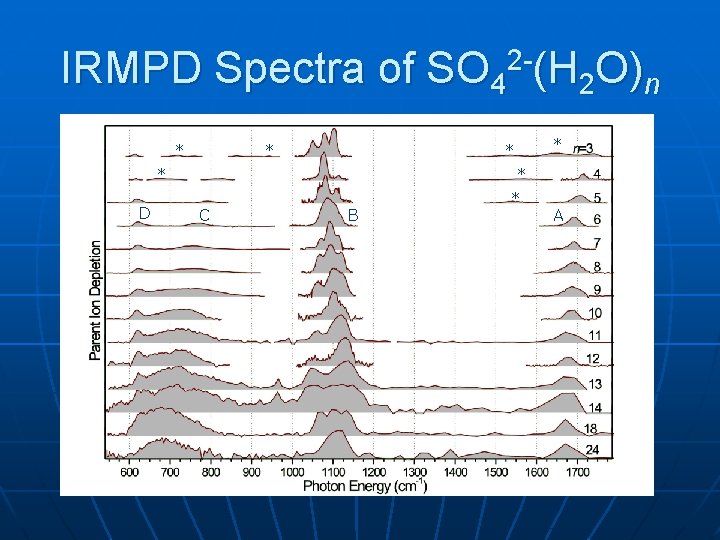
IRMPD Spectra of SO 42 -(H 2 O)n * * * D * * * C B * A
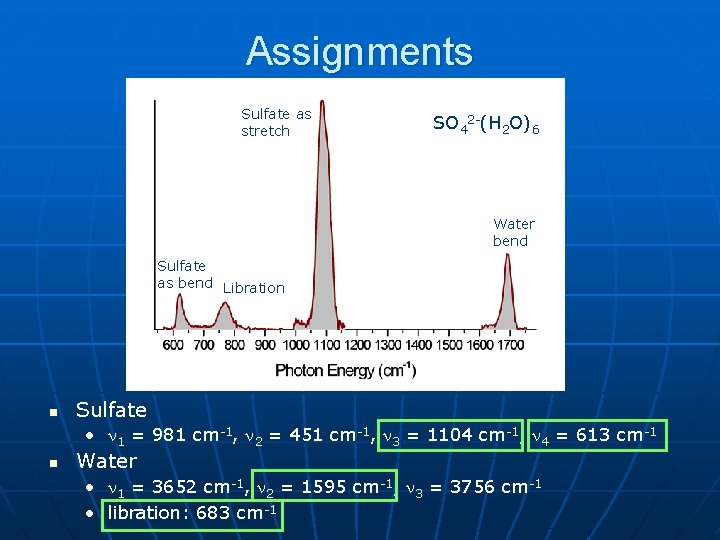
Assignments Sulfate as stretch SO 42 -(H 2 O)6 Water bend Sulfate as bend Libration n Sulfate • 1 = 981 cm-1, 2 = 451 cm-1, 3 = 1104 cm-1, 4 = 613 cm-1 n Water • 1 = 3652 cm-1, 2 = 1595 cm-1, 3 = 3756 cm-1 • libration: 683 cm-1
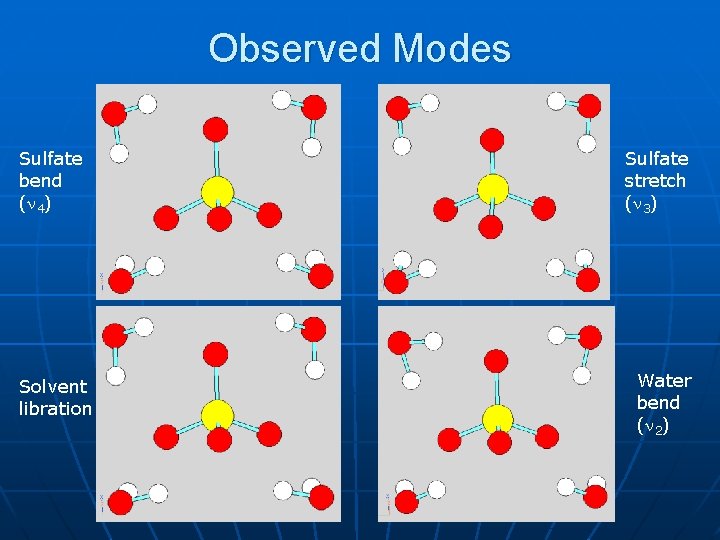
Observed Modes Sulfate bend ( 4) Solvent libration Sulfate stretch ( 3) Water bend ( 2)
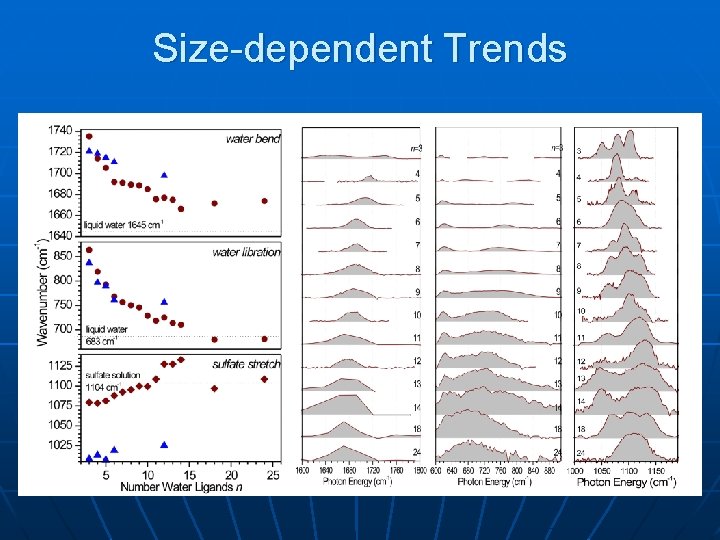
Size-dependent Trends
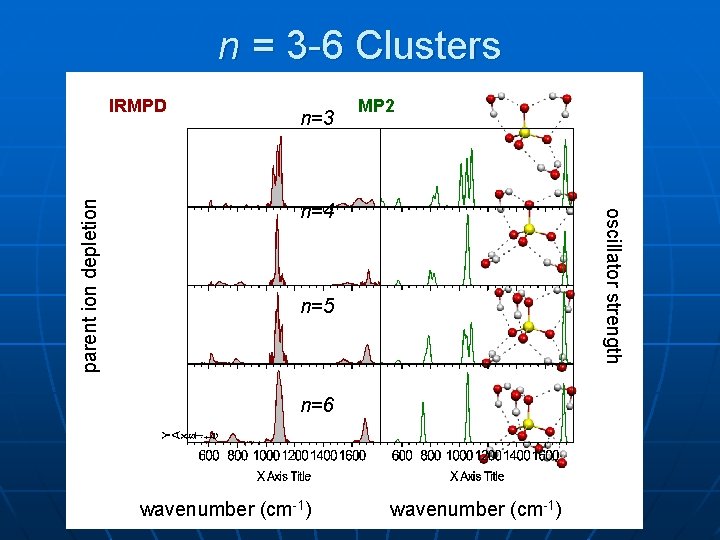
n = 3 -6 Clusters n=3 MP 2 n=4 oscillator strength parent ion depletion IRMPD n=5 n=6 wavenumber (cm-1)
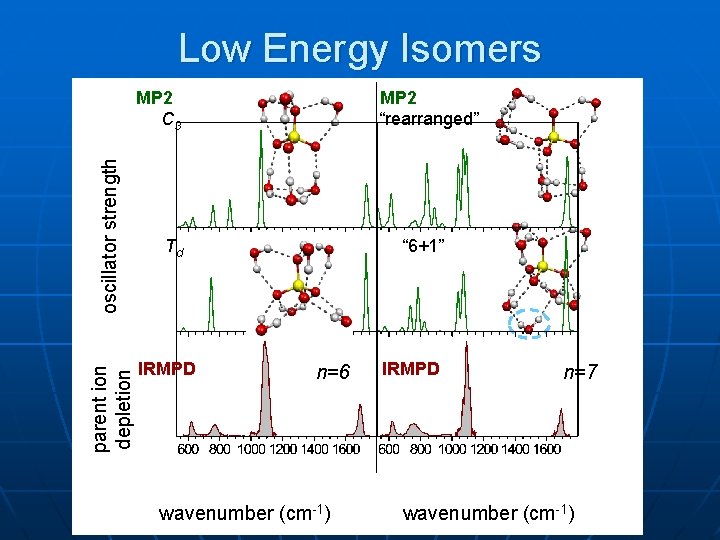
Low Energy Isomers MP 2 “rearranged” parent ion depletion oscillator strength MP 2 C 3 Td IRMPD “ 6+1” n=6 wavenumber (cm-1) IRMPD n=7 wavenumber (cm-1)
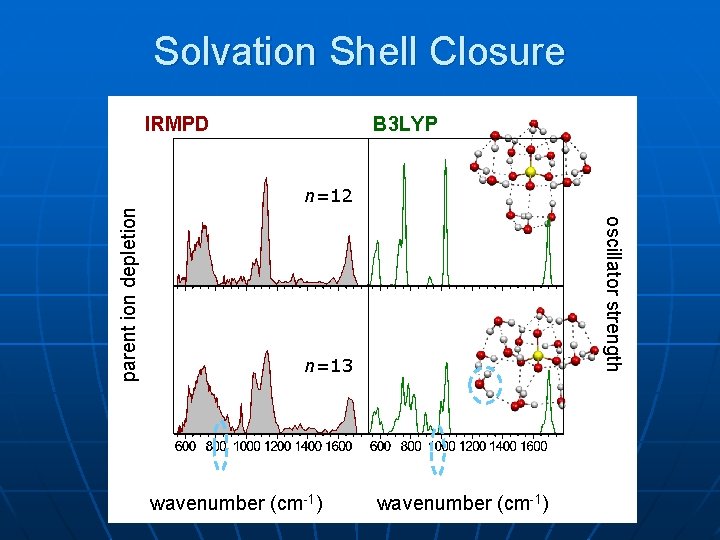
Solvation Shell Closure B 3 LYP n=12 oscillator strength parent ion depletion IRMPD n=13 wavenumber (cm-1)
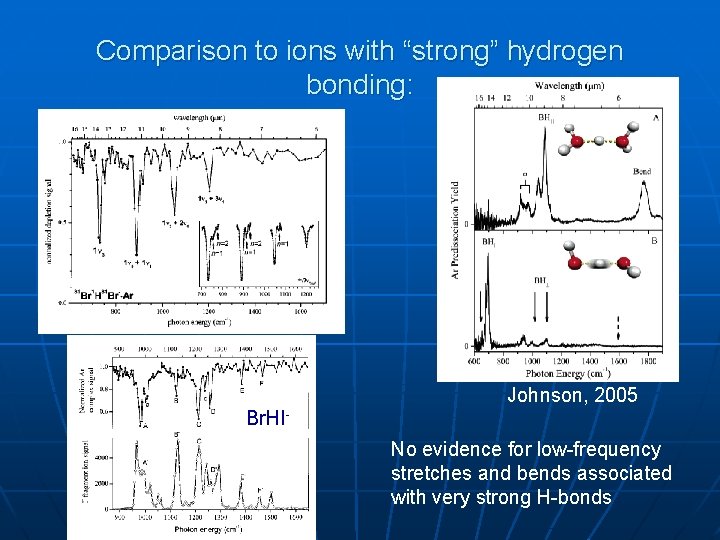
Comparison to ions with “strong” hydrogen bonding: Johnson, 2005 Br. HINo evidence for low-frequency stretches and bends associated with very strong H-bonds

Hydrated sulfate summary: n n n First IR spectra of multply-charged anion in gas phase Triply-degenerate 3 mode of sulfate and librational modes of water are very sensitive to solvation structure 1 st six water molecules bridged between two O atoms on sulfate • Subsequent waters: water-water H-bonds n Direct spectroscopic evidence for 2 nd solvent shell starting at n=13

Many Thanks! Mark Abel Paul Crider Etienne Garand Scott Goncher Graham Griffin Aster Kammrath Beth Kautzman Jeong Hyun Kim David Moore Brad Parsons Thomas Pfeifer Sean Sheehan Niels Sveum Dave Szpunar Jan Verlet Chia Wang Terry Yen Jia Zhou Knut Asmis (FHI/Berlin)
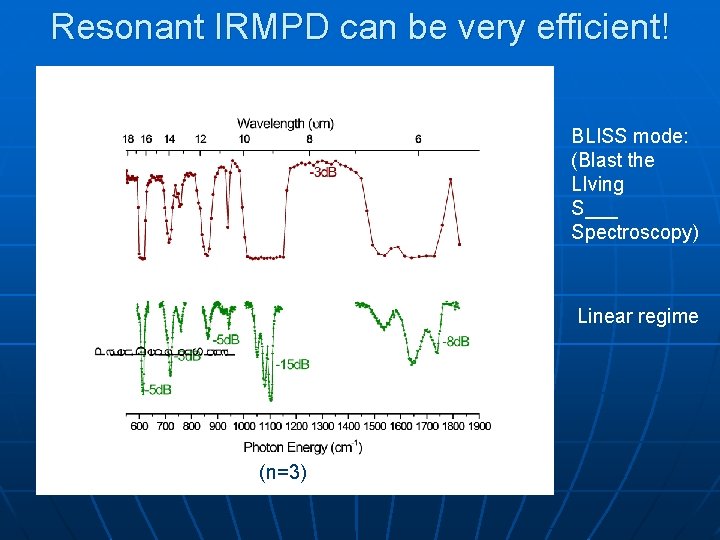
Resonant IRMPD can be very efficient! BLISS mode: (Blast the LIving S___ Spectroscopy) Linear regime (n=3)
- Slides: 22