Infrared Spectroscopy of Protonated BenzeneWater Nanoclusters Hydronium Zundel

Infrared Spectroscopy of Protonated Benzene-Water Nanoclusters: Hydronium, Zundel and Eigen at a Hydrophobic Interface Tim C. Cheng Biswajit Bandyopadhyay Michael A. Duncan Department of Chemistry, University of Georgia, Athens, GA 30602 maduncan@uga. edu http: //www. arches. uga. edu/~maduncan National Science Foundation

Are clusters relevant for interfaces?
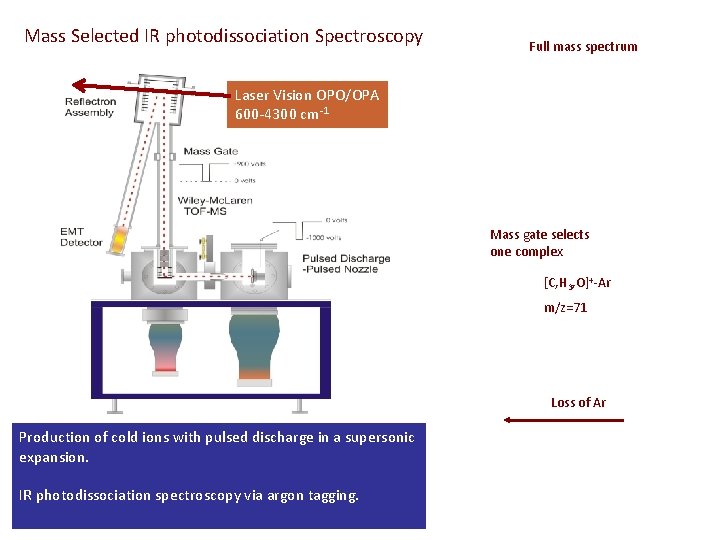
Mass Selected IR photodissociation Spectroscopy Full mass spectrum Laser Vision OPO/OPA 600 -4300 cm-1 5 μs Mass gate selects one complex [C, H 3, O]+-Ar m/z=71 Loss of Ar Production of cold ions with pulsed discharge in a supersonic expansion. IR photodissociation spectroscopy via argon tagging.
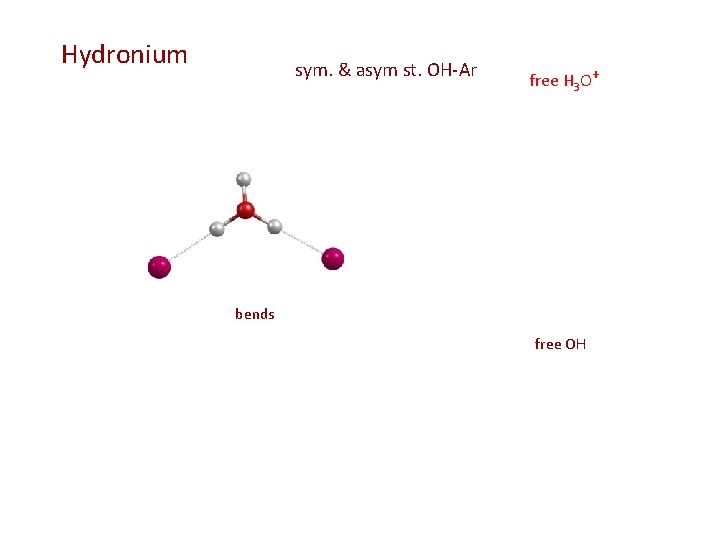
Hydronium sym. & asym st. OH-Ar free H 3 O+ bends free OH
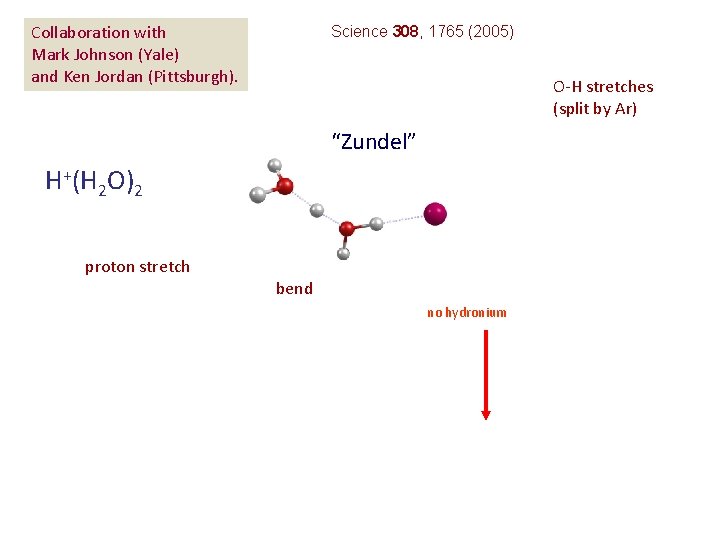
Collaboration with Mark Johnson (Yale) and Ken Jordan (Pittsburgh). Science 308, 1765 (2005) O-H stretches (split by Ar) “Zundel” H+(H 2 O)2 proton stretch bend no hydronium
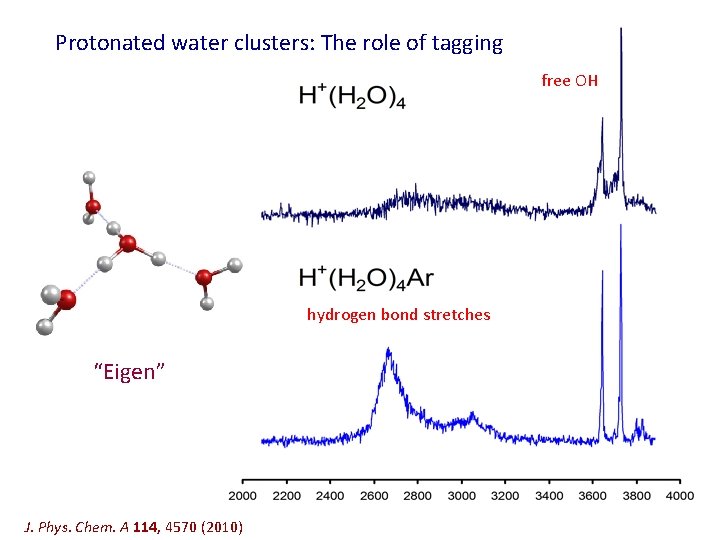
Protonated water clusters: The role of tagging free OH hydrogen bond stretches “Eigen” J. Phys. Chem. A 114, 4570 (2010)
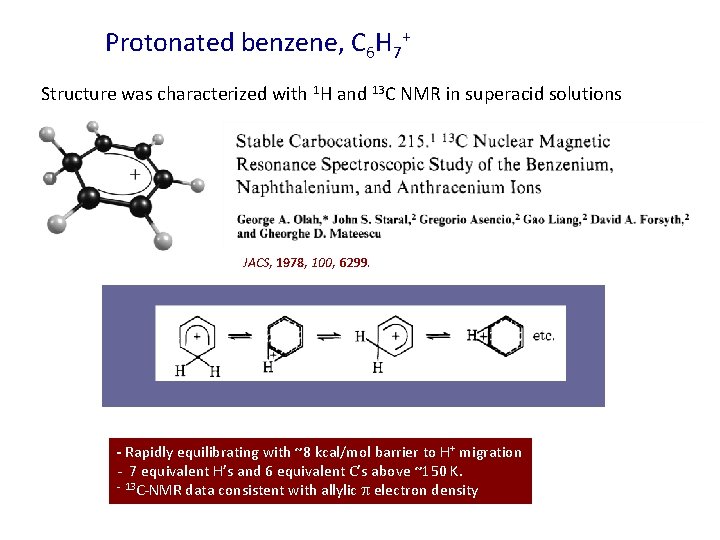
Protonated benzene, C 6 H 7+ Structure was characterized with 1 H and 13 C NMR in superacid solutions JACS, 1978, 100, 6299. - Rapidly equilibrating with ~8 kcal/mol barrier to H+ migration - 7 equivalent H’s and 6 equivalent C’s above ~150 K. - 13 C-NMR data consistent with allylic electron density
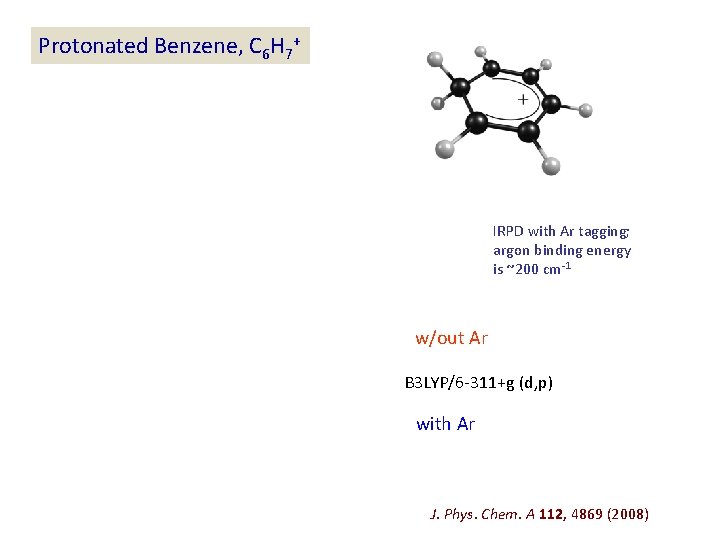
Protonated Benzene, C 6 H 7+ IRPD with Ar tagging; argon binding energy is ~200 cm-1 w/out Ar B 3 LYP/6 -311+g (d, p) with Ar J. Phys. Chem. A 112, 4869 (2008)
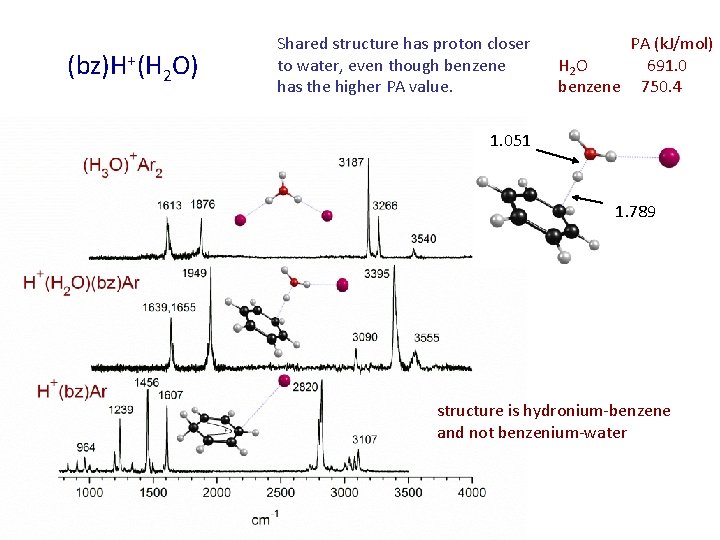
(bz)H+(H 2 O) Shared structure has proton closer to water, even though benzene has the higher PA value. PA (k. J/mol) H 2 O 691. 0 benzene 750. 4 1. 051 1. 789 structure is hydronium-benzene and not benzenium-water
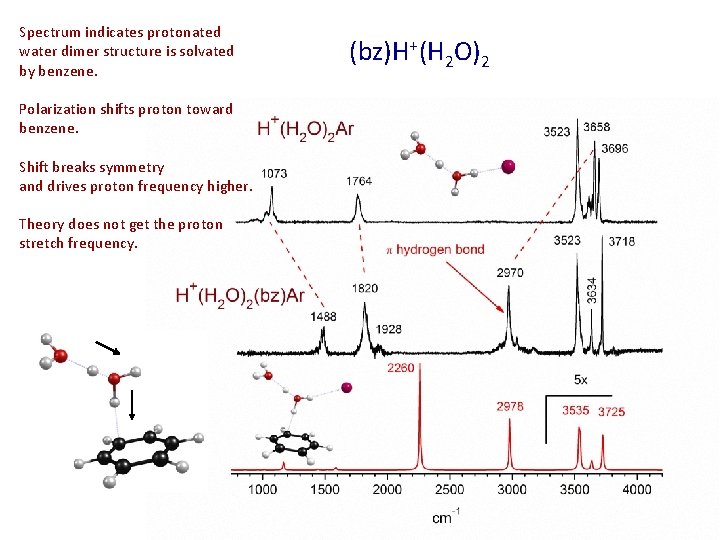
Spectrum indicates protonated water dimer structure is solvated by benzene. Polarization shifts proton toward benzene. Shift breaks symmetry and drives proton frequency higher. Theory does not get the proton stretch frequency. (bz)H+(H 2 O)2
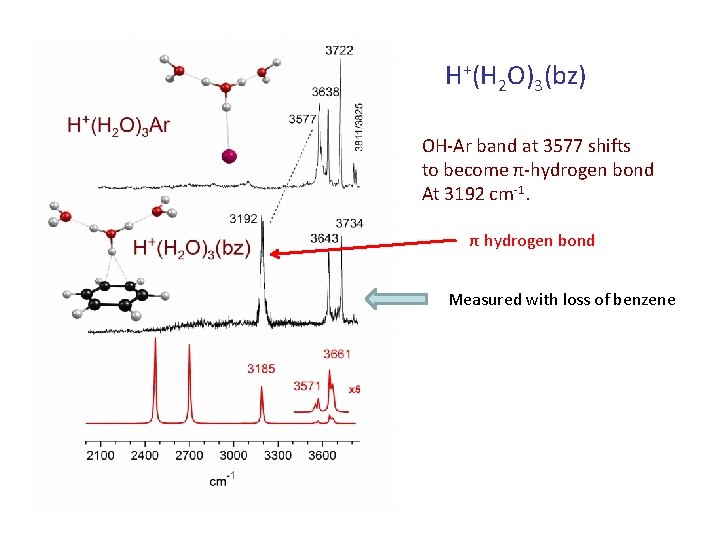
H+(H 2 O)3(bz) OH-Ar band at 3577 shifts to become π-hydrogen bond At 3192 cm-1. π hydrogen bond Measured with loss of benzene
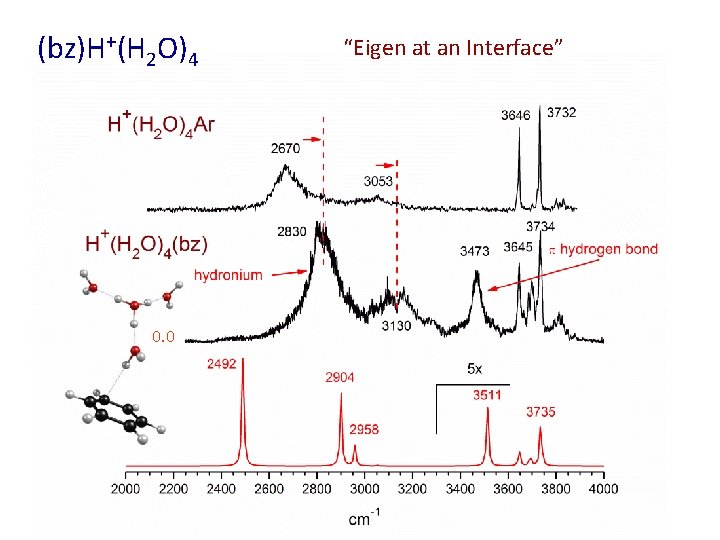
(bz)H+(H 2 O)4 0. 0 “Eigen at an Interface”
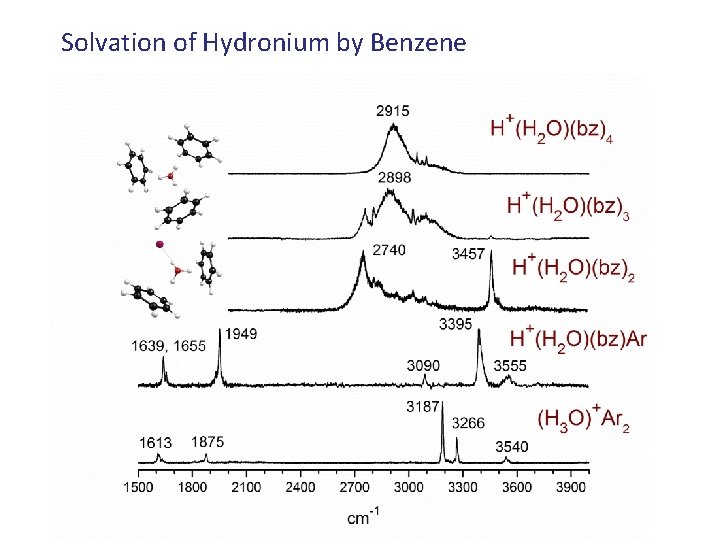
Solvation of Hydronium by Benzene
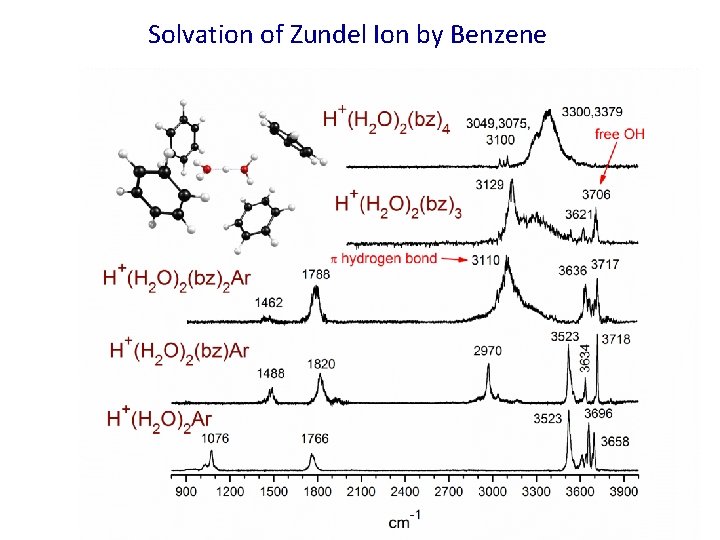
Solvation of Zundel Ion by Benzene

Conclusions Protonated water-benzene mixtures have proton on water Contact point between water and benzene is π-hydrogen bond Binding to benzene shifts frequencies of water clusters both at and remote from contact point Clusters with more benzene distribute it around water ions rather than forming segregated domains
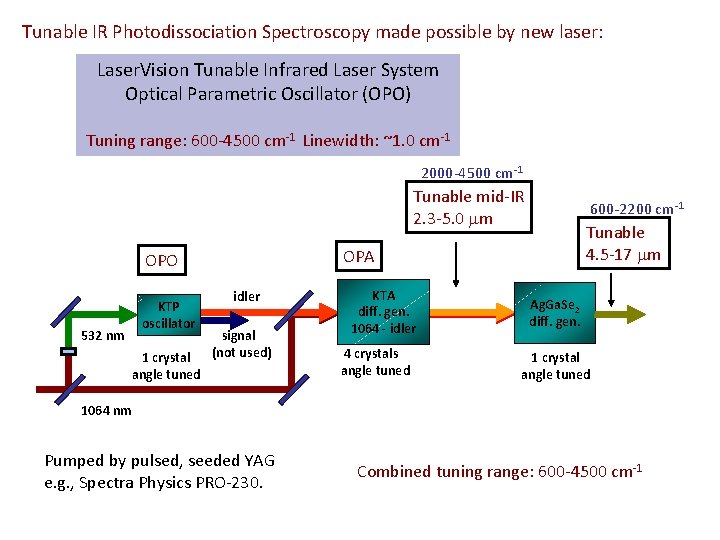
Tunable IR Photodissociation Spectroscopy made possible by new laser: Laser. Vision Tunable Infrared Laser System Optical Parametric Oscillator (OPO) Tuning range: 600 -4500 cm-1 Linewidth: ~1. 0 cm-1 2000 -4500 cm-1 Tunable mid-IR 2. 3 -5. 0 m 532 nm 1 crystal angle tuned Tunable 4. 5 -17 m OPA OPO KTP oscillator 600 -2200 cm-1 idler signal (not used) KTA diff. gen. 1064 - idler 4 crystals angle tuned Ag. Ga. Se 2 diff. gen. 1 crystal angle tuned 1064 nm Pumped by pulsed, seeded YAG e. g. , Spectra Physics PRO-230. Combined tuning range: 600 -4500 cm-1
- Slides: 16