Infrared Spectroscopy of Protonated BenzeneWater Complexes Biswajit Bandyopadhyay

Infrared Spectroscopy of Protonated Benzene-Water Complexes Biswajit Bandyopadhyay, Tim Cheng and Michael A. Duncan Department of Chemistry, University of Georgia, Athens, GA, 30602 www. arches. uga. edu/~maduncan/

Motivation • Proton transfer is ubiquitous in chemistry and biology. • Bridged proton systems are key intermediates in proton transfer reactions. • Protonated benzene-water complexes provide a simple system to study hydrophobic-hydrophilic interface. • Protonated benzene complex and protonated water clusters have been studied by our group previously.
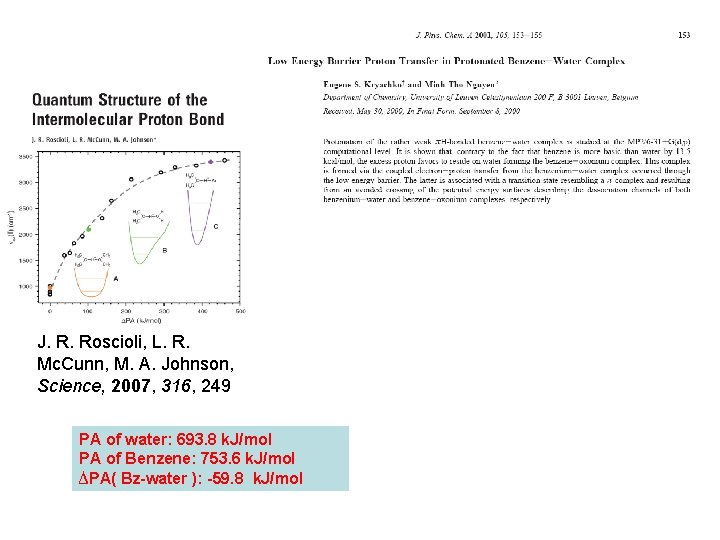
J. R. Roscioli, L. R. Mc. Cunn, M. A. Johnson, Science, 2007, 316, 249 PA of water: 693. 8 k. J/mol PA of Benzene: 753. 6 k. J/mol ∆PA( Bz-water ): -59. 8 k. J/mol
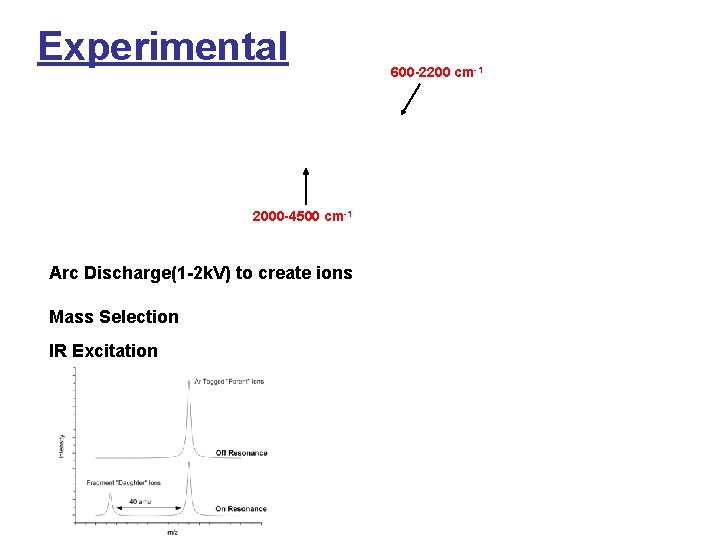
Experimental 2000 -4500 cm-1 Arc Discharge(1 -2 k. V) to create ions Mass Selection IR Excitation 600 -2200 cm-1
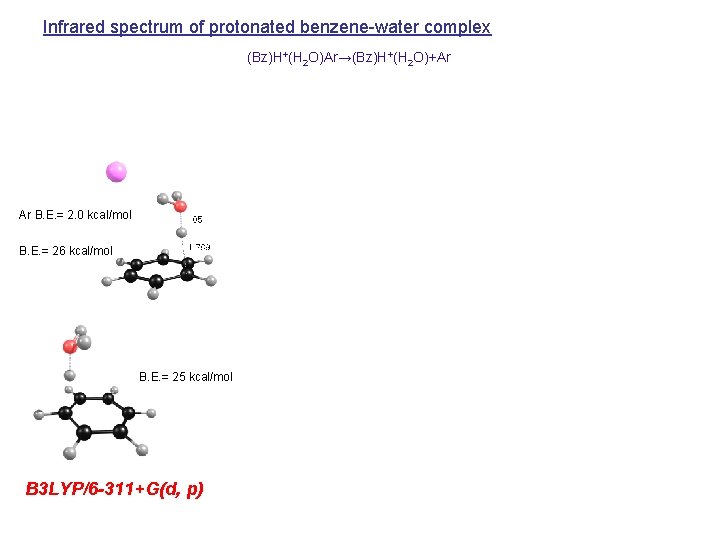
Infrared spectrum of protonated benzene-water complex (Bz)H+(H 2 O)Ar→(Bz)H+(H 2 O)+Ar Ar B. E. = 2. 0 kcal/mol B. E. = 26 kcal/mol B. E. = 25 kcal/mol B 3 LYP/6 -311+G(d, p)
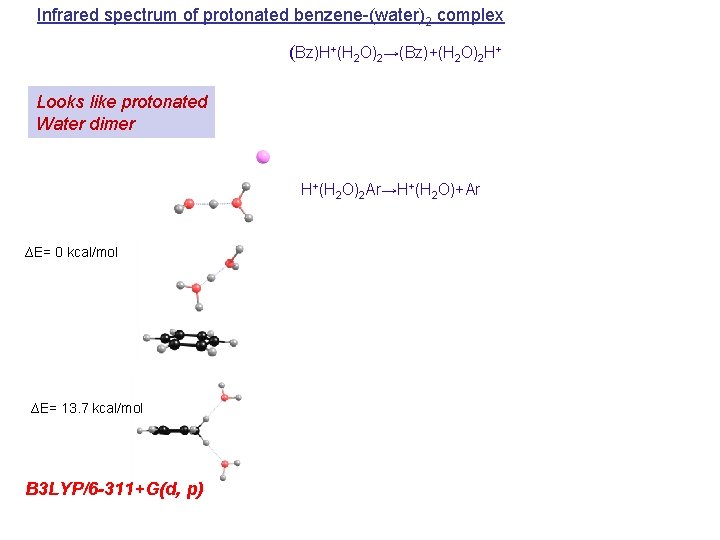
Infrared spectrum of protonated benzene-(water)2 complex (Bz)H+(H 2 O)2→(Bz)+(H 2 O)2 H+ Looks like protonated Water dimer H+(H 2 O)2 Ar→H+(H 2 O)+Ar ∆E= 0 kcal/mol ∆E= 13. 7 kcal/mol B 3 LYP/6 -311+G(d, p)
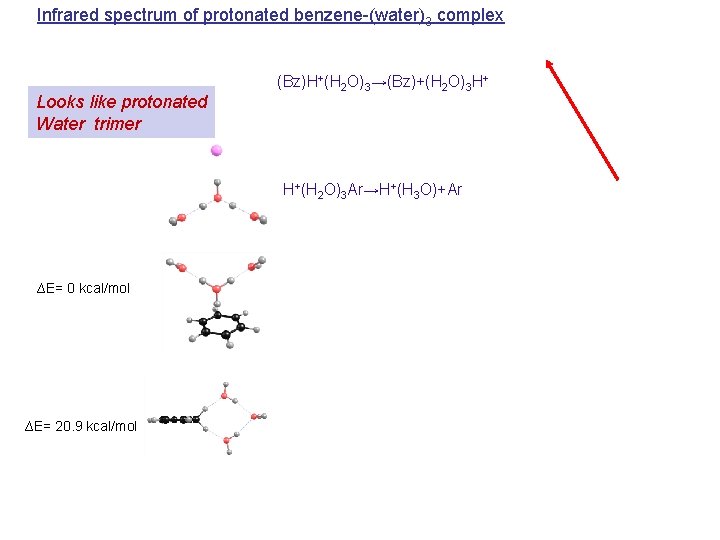
Infrared spectrum of protonated benzene-(water)3 complex Looks like protonated Water trimer (Bz)H+(H 2 O)3→(Bz)+(H 2 O)3 H+ H+(H 2 O)3 Ar→H+(H 3 O)+Ar ∆E= 0 kcal/mol ∆E= 20. 9 kcal/mol
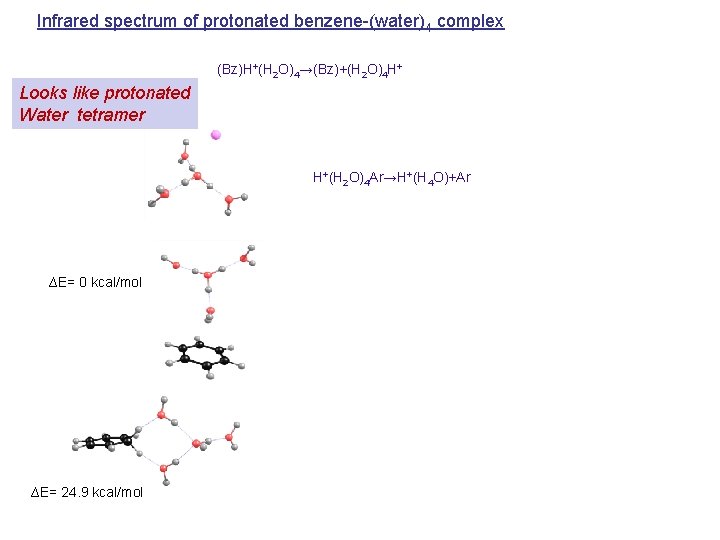
Infrared spectrum of protonated benzene-(water)4 complex (Bz)H+(H 2 O)4→(Bz)+(H 2 O)4 H+ Looks like protonated Water tetramer H+(H 2 O)4 Ar→H+(H 4 O)+Ar ∆E= 0 kcal/mol ∆E= 24. 9 kcal/mol
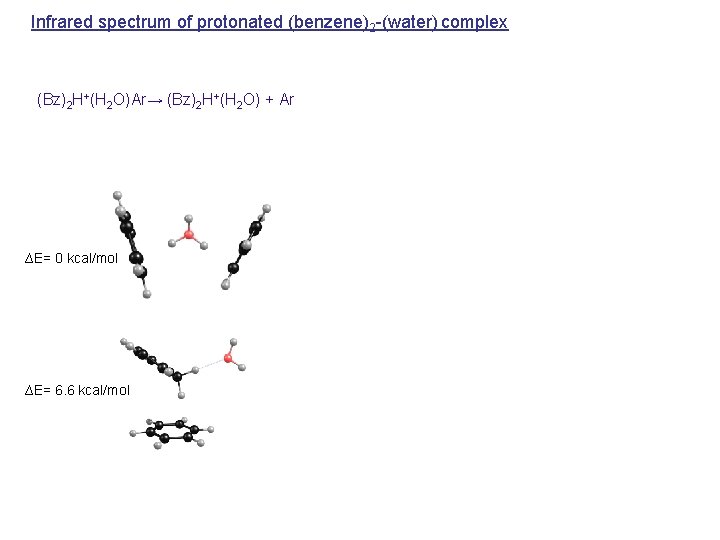
Infrared spectrum of protonated (benzene)2 -(water) complex (Bz)2 H+(H 2 O)Ar→ (Bz)2 H+(H 2 O) + Ar ∆E= 0 kcal/mol ∆E= 6. 6 kcal/mol
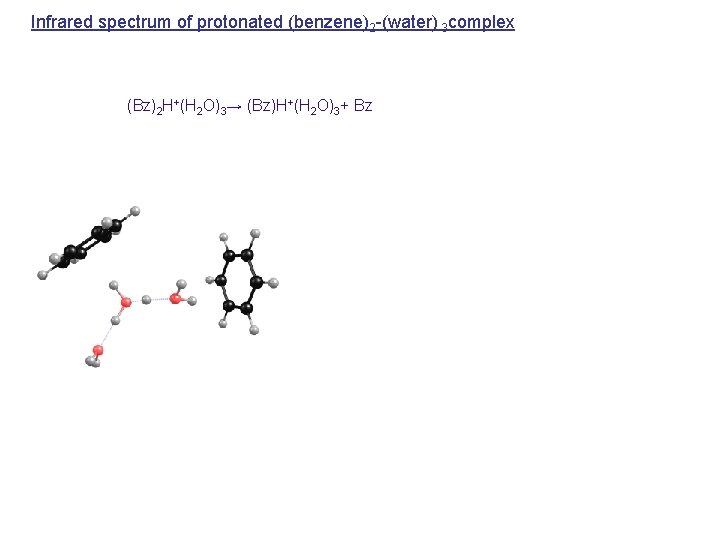
Infrared spectrum of protonated (benzene)2 -(water) 3 complex (Bz)2 H+(H 2 O)3→ (Bz)H+(H 2 O)3+ Bz

Conclusions • Various isomers are possible but the location of the proton is determined by the solvation energy • Structures consist primarily of protonated water clusters interacting with neutral benzene. Acknowledgement • • Michael A. Duncan Gary E. Douberly Allen M. Ricks NSF for funding
- Slides: 11