Infrared Spectroscopy of Metal Ions and Clusters Inorganic





- Slides: 26

Infrared Spectroscopy of Metal Ions and Clusters: Inorganic Chemistry in the Gas Phase Michael A. Duncan Department of Chemistry, University of Georgia, Athens, GA 30602 maduncan@uga. edu http: //www. arches. uga. edu/~maduncan Department of Energy Air Force Office of Scientific Research Allen Ricks, Tim Cheng, Biswajit Bandyopadyay, Zach Reed

Infrared spectroscopy of ions and clusters: Metal complexes, protonated species and carbocations M+(H 2 O)n, M 2+(H 2 O)n M+(CO)n, , M+(CO 2)n M+(acetone)n M+(benzene)n H+(H 2 O)n H+(CO 2)n H+(H 2)n H+(N 2)n H+(C 2 H 2)n H+(benzene)n C 2 H 3 + , C 3 H 5 + , C 4 H 9 + , … IR spectroscopy probes fundamental bonding interactions and structures. Ligand vibrational shifts Structures, isomers Coordination spheres Metal spin states Solvation Intracluster reactions Proton transfer intermediates Astrophysical ions

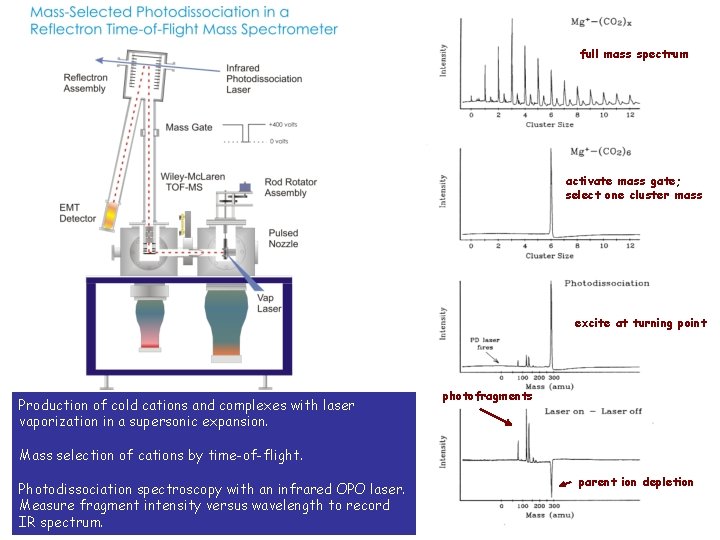
full mass spectrum activate mass gate; select one cluster mass excite at turning point Production of cold cations and complexes with laser vaporization in a supersonic expansion. photofragments Mass selection of cations by time-of-flight. Photodissociation spectroscopy with an infrared OPO laser. Measure fragment intensity versus wavelength to record IR spectrum. parent ion depletion

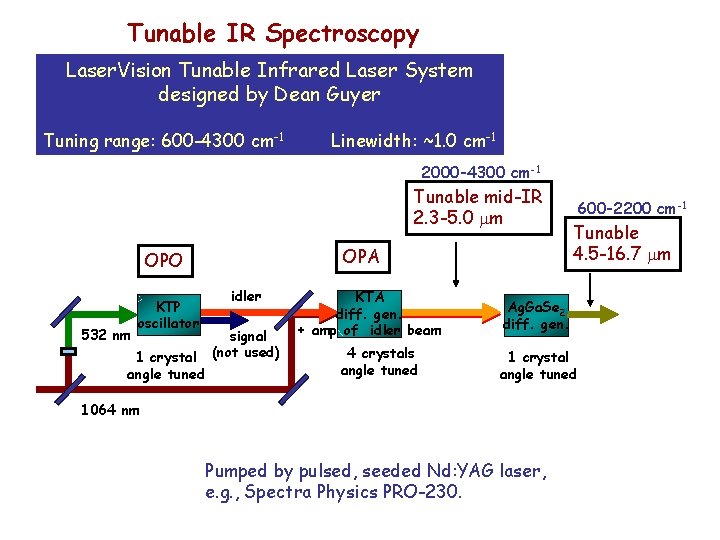
Tunable IR Spectroscopy Laser. Vision Tunable Infrared Laser System designed by Dean Guyer Tuning range: 600 -4300 cm-1 Linewidth: ~1. 0 cm-1 2000 -4300 cm-1 Tunable mid-IR 2. 3 -5. 0 m OPA OPO 532 nm KTP oscillator 1 crystal angle tuned idler signal (not used) KTA diff. gen. + amp of idler beam 4 crystals angle tuned 600 -2200 cm-1 Tunable 4. 5 -16. 7 m Ag. Ga. Se 2 diff. gen. 1 crystal angle tuned 1064 nm Pumped by pulsed, seeded Nd: YAG laser, e. g. , Spectra Physics PRO-230.

IR Photodissociation Spectroscopy and Rare Gas Tagging The density of mass-selected ions is too low for absorption spectroscopy. Therefore, we use photodissociation, with sensitive TOF mass spectrometer detection. However, the bonds in our ions are often too strong (D 0>20 kcal/mol) to be broken with infrared light (e. g. , C-H stretch of 3100 cm -1 = 9 kcal/mol). Our laser power is too low for multiphoton absorption. In larger complexes, external ligands (not in contact with ion) can be eliminated. For others, we attach a weakly bound (1 -2 kcal) “tag” atom to enhance fragmentation. The tag elimination (only when light is absorbed) provides indirect evidence of absorption. Use computations on ion with & without tag to reveal the degree of perturbation. Ar + Mass selected ion + h (tunable IR) + Fragment ion after argon elimination detected vs IR frequency

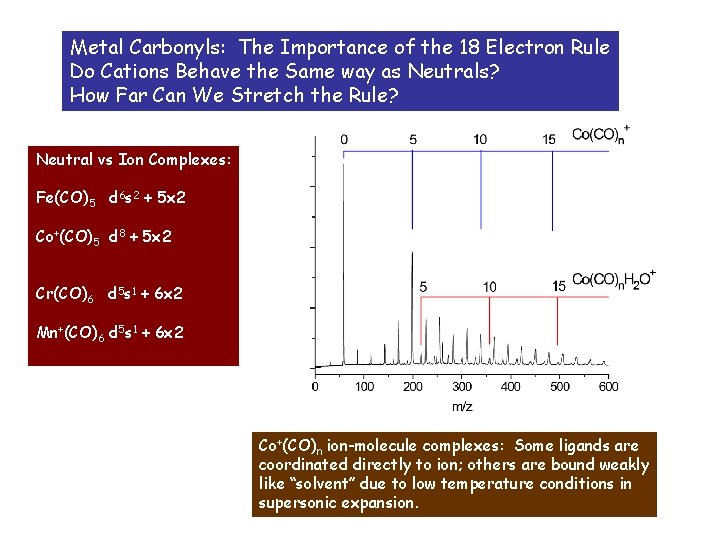
Metal Carbonyls: The Importance of the 18 Electron Rule Do Cations Behave the Same way as Neutrals? How Far Can We Stretch the Rule? Neutral vs Ion Complexes: Fe(CO)5 d 6 s 2 + 5 x 2 Co+(CO)5 d 8 + 5 x 2 Cr(CO)6 d 5 s 1 + 6 x 2 Mn+(CO)6 d 5 s 1 + 6 x 2 Co+(CO)n ion-molecule complexes: Some ligands are coordinated directly to ion; others are bound weakly like “solvent” due to low temperature conditions in supersonic expansion.

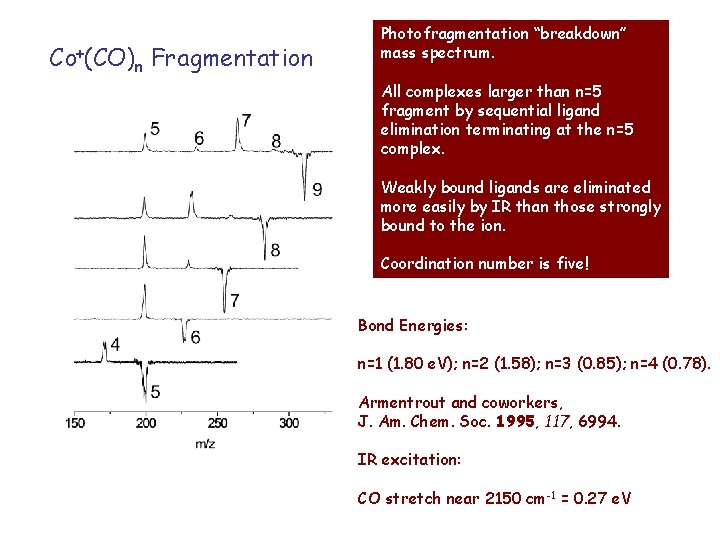
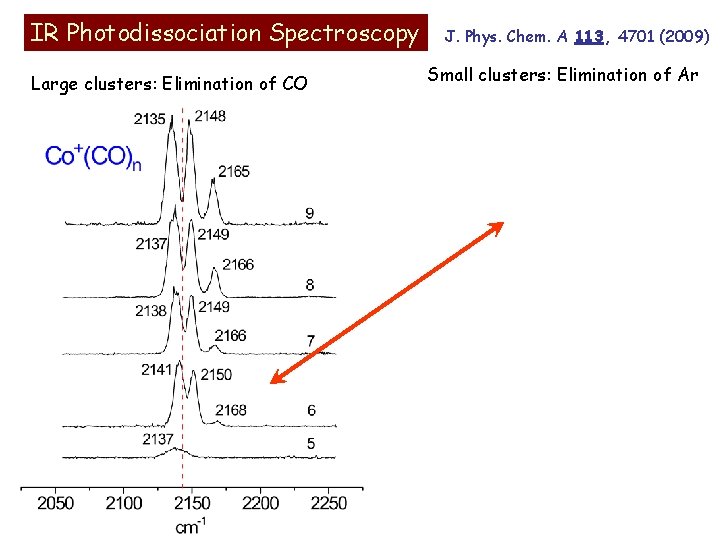
Co+(CO)n Fragmentation Photofragmentation “breakdown” mass spectrum. All complexes larger than n=5 fragment by sequential ligand elimination terminating at the n=5 complex. Weakly bound ligands are eliminated more easily by IR than those strongly bound to the ion. Coordination number is five! Bond Energies: n=1 (1. 80 e. V); n=2 (1. 58); n=3 (0. 85); n=4 (0. 78). Armentrout and coworkers, J. Am. Chem. Soc. 1995, 117, 6994. IR excitation: CO stretch near 2150 cm-1 = 0. 27 e. V

IR Photodissociation Spectroscopy Large clusters: Elimination of CO J. Phys. Chem. A 113, 4701 (2009) Small clusters: Elimination of Ar

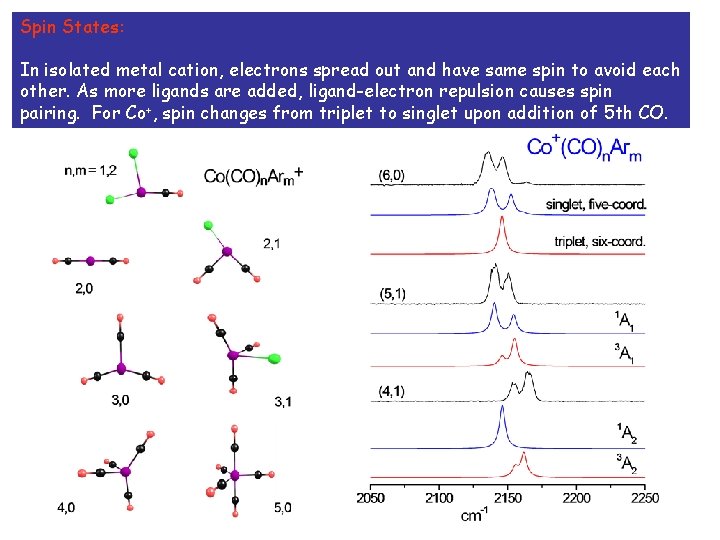
Spin States: In isolated metal cation, electrons spread out and have same spin to avoid each other. As more ligands are added, ligand-electron repulsion causes spin pairing. For Co+, spin changes from triplet to singlet upon addition of 5 th CO.

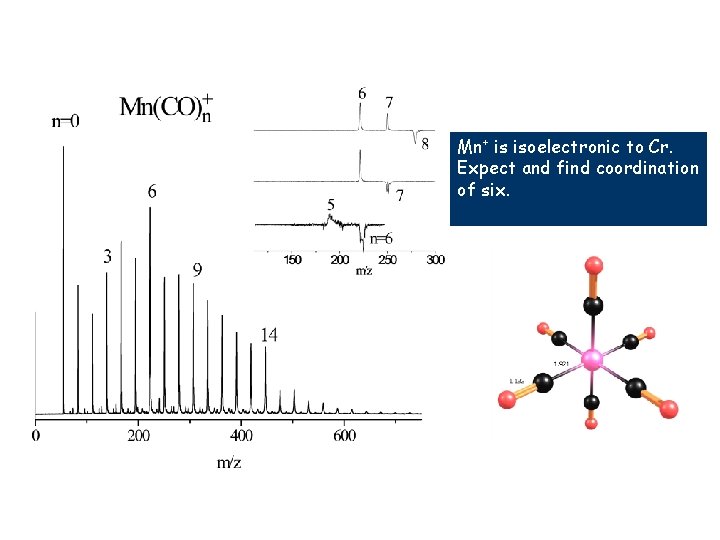
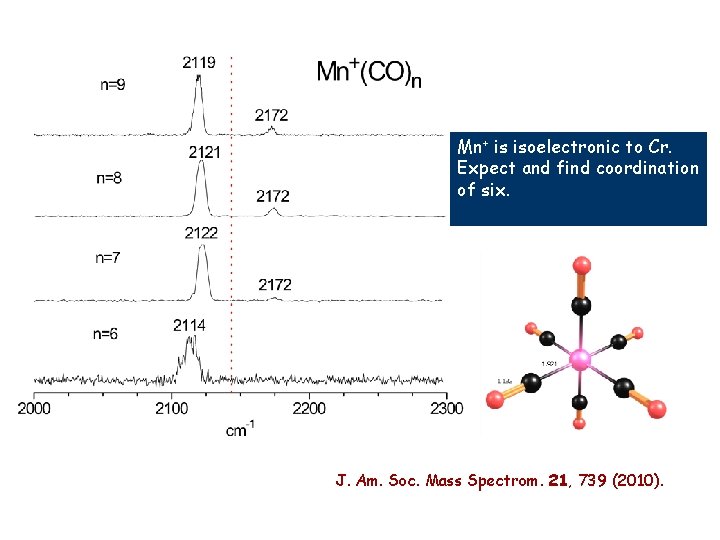
Mn+ is isoelectronic to Cr. Expect and find coordination of six.

Mn+ is isoelectronic to Cr. Expect and find coordination of six. J. Am. Soc. Mass Spectrom. 21, 739 (2010).

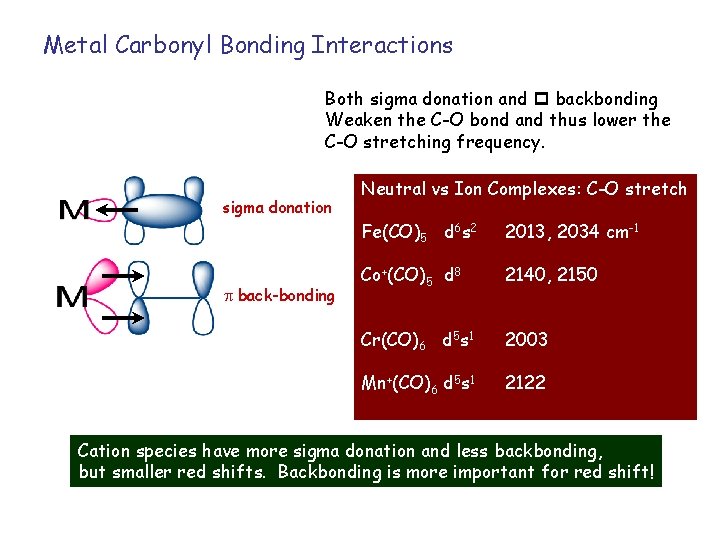
Metal Carbonyl Bonding Interactions Both sigma donation and p backbonding Weaken the C-O bond and thus lower the C-O stretching frequency. sigma donation back-bonding Neutral vs Ion Complexes: C-O stretch Fe(CO)5 d 6 s 2 2013, 2034 cm-1 Co+(CO)5 d 8 2140, 2150 Cr(CO)6 d 5 s 1 2003 Mn+(CO)6 d 5 s 1 2122 Cation species have more sigma donation and less backbonding, but smaller red shifts. Backbonding is more important for red shift!

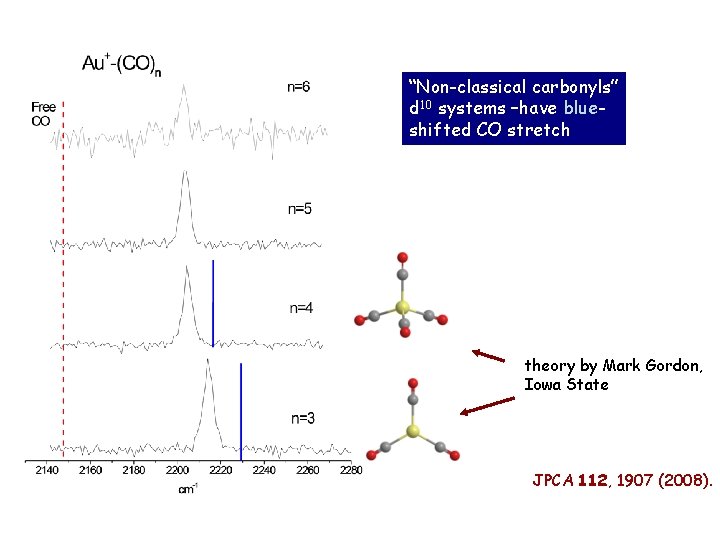
“Non-classical carbonyls” d 10 systems –have blueshifted CO stretch theory by Mark Gordon, Iowa State JPCA 112, 1907 (2008).

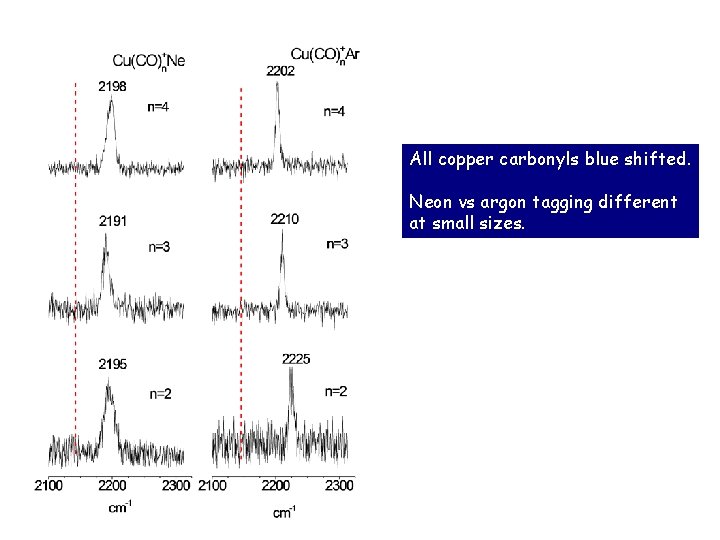
All copper carbonyls blue shifted. Neon vs argon tagging different at small sizes.

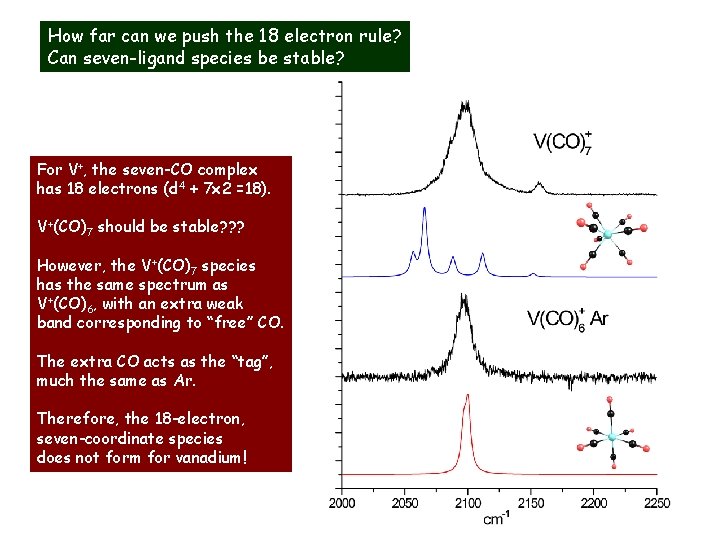
How far can we push the 18 electron rule? Can seven-ligand species be stable? For V+, the seven-CO complex has 18 electrons (d 4 + 7 x 2 =18). V+(CO)7 should be stable? ? ? However, the V+(CO)7 species has the same spectrum as V+(CO)6, with an extra weak band corresponding to “free” CO. The extra CO acts as the “tag”, much the same as Ar. Therefore, the 18 -electron, seven-coordinate species does not form for vanadium!

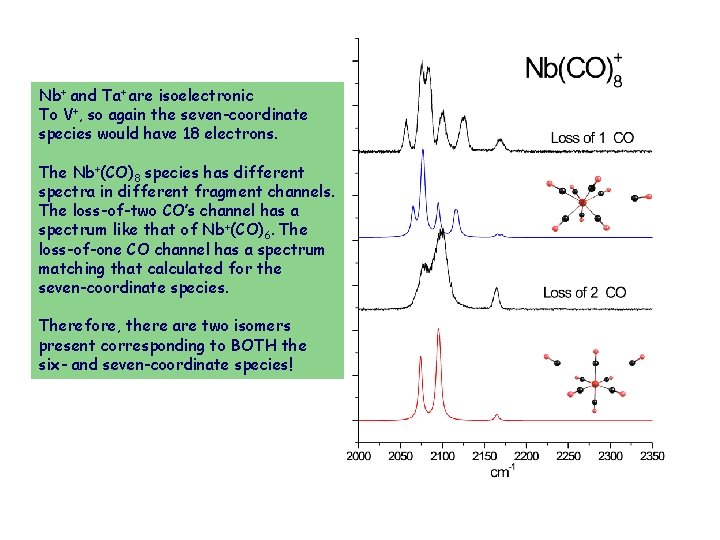
Nb+ and Ta+ are isoelectronic To V+, so again the seven-coordinate species would have 18 electrons. The Nb+(CO)8 species has different spectra in different fragment channels. The loss-of-two CO’s channel has a spectrum like that of Nb+(CO)6. The loss-of-one CO channel has a spectrum matching that calculated for the seven-coordinate species. Therefore, there are two isomers present corresponding to BOTH the six- and seven-coordinate species!

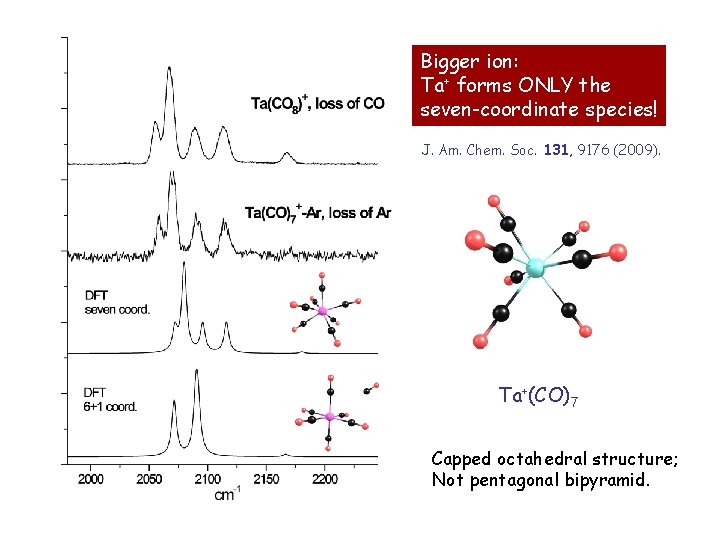
Bigger ion: Ta+ forms ONLY the seven-coordinate species! J. Am. Chem. Soc. 131, 9176 (2009). Ta+(CO)7 Capped octahedral structure; Not pentagonal bipyramid.

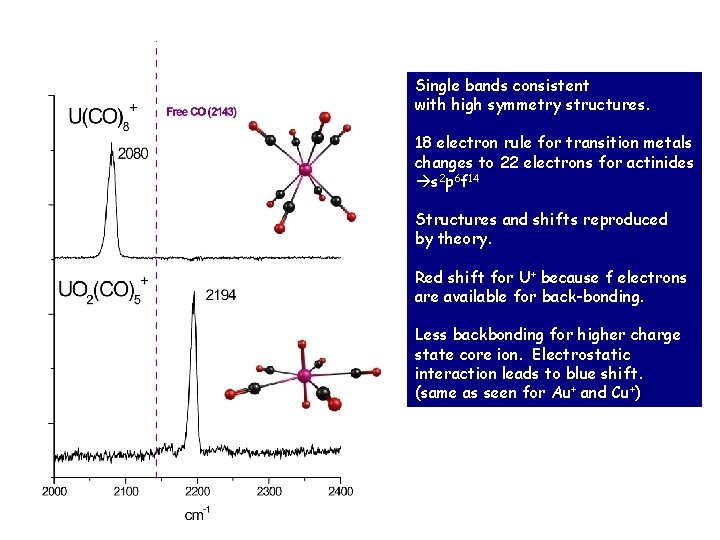
Single bands consistent with high symmetry structures. 18 electron rule for transition metals changes to 22 electrons for actinides s 2 p 6 f 14 Structures and shifts reproduced by theory. Red shift for U+ because f electrons are available for back-bonding. Less backbonding for higher charge state core ion. Electrostatic interaction leads to blue shift. (same as seen for Au+ and Cu+)

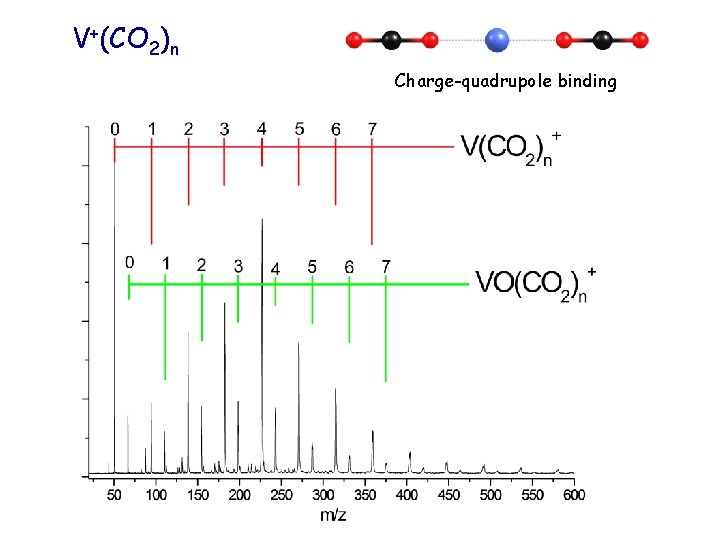
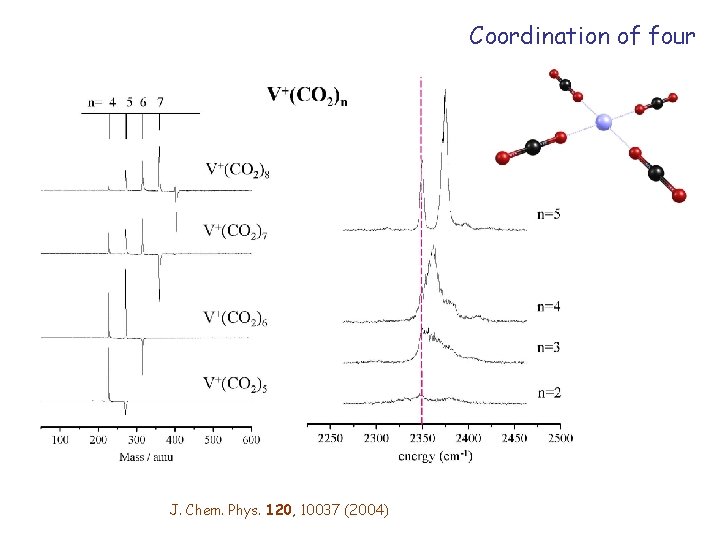
V+(CO 2)n Charge-quadrupole binding

Coordination of four J. Chem. Phys. 120, 10037 (2004)

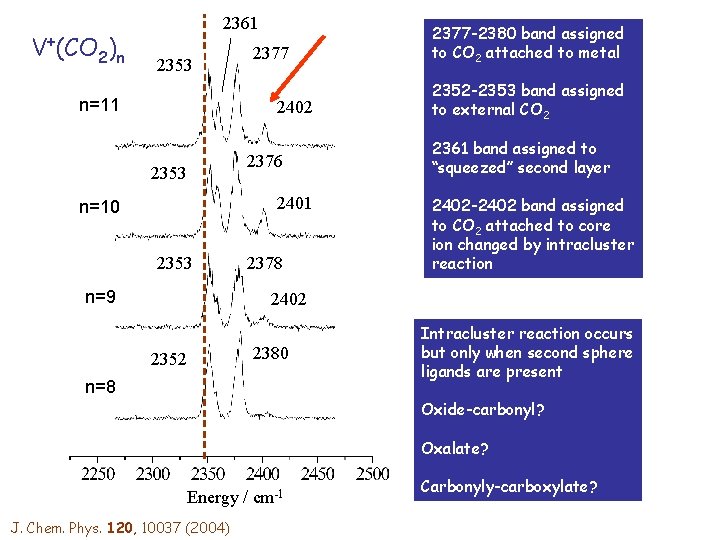
V+(CO 2)n 2361 2353 n=11 2377 2402 2353 2376 2401 n=10 2353 n=9 2378 2377 -2380 band assigned to CO 2 attached to metal 2352 -2353 band assigned to external CO 2 2361 band assigned to “squeezed” second layer 2402 -2402 band assigned to CO 2 attached to core ion changed by intracluster reaction 2402 2352 2380 n=8 Intracluster reaction occurs but only when second sphere ligands are present Oxide-carbonyl? Oxalate? Energy / cm-1 J. Chem. Phys. 120, 10037 (2004) Carbonyly-carboxylate?

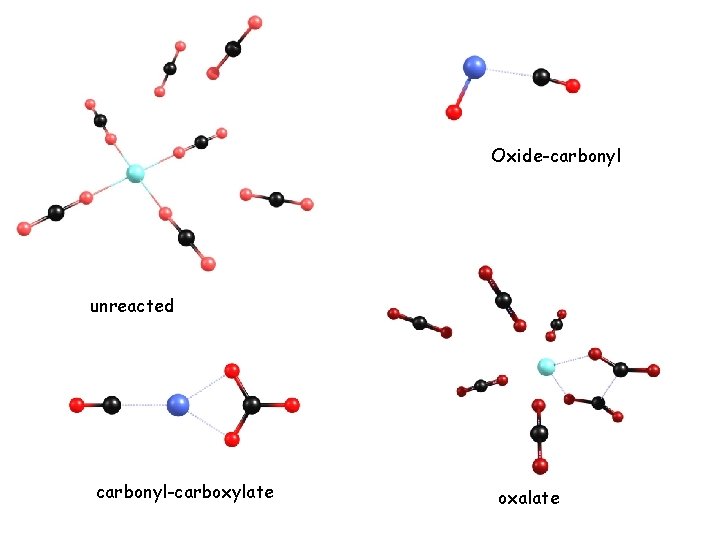
Oxide-carbonyl unreacted carbonyl-carboxylate oxalate

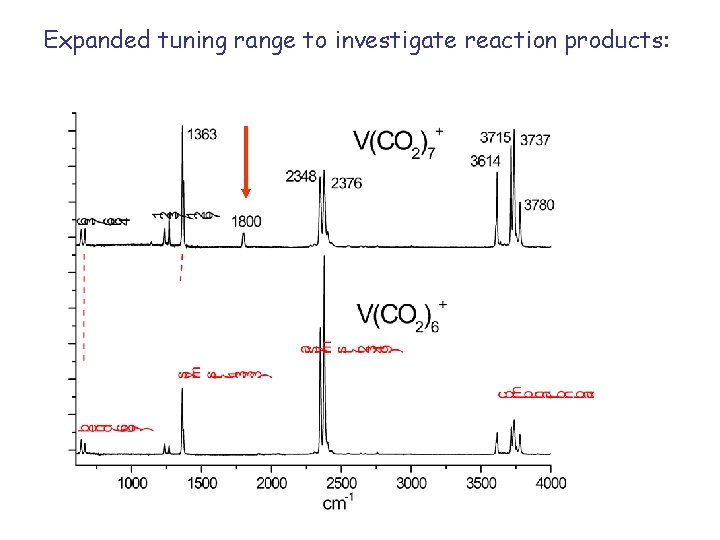
Expanded tuning range to investigate reaction products:

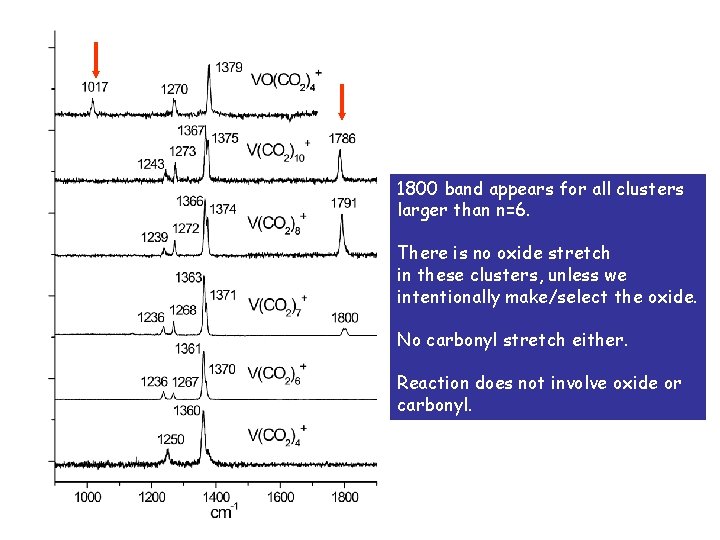
1800 band appears for all clusters larger than n=6. There is no oxide stretch in these clusters, unless we intentionally make/select the oxide. No carbonyl stretch either. Reaction does not involve oxide or carbonyl.

Oxalate moiety has two vibrational bands in about the right places to explain the reaction. Why does it only happen when second-sphere ligands are present? Associated with spin change quintet triplet?

Conclusions Coordination numbers, spin states and ligand shifts for metal carbonyls. Intracluster reactions.