Infrared spectroscopy of hydrogenbonded clusters of protonated histidine

Infrared spectroscopy of hydrogen-bonded clusters of protonated histidine Department of Chemistry, School of Science, Kitasato University, Japan Makoto Kondo, Yasutoshi Kasahara, Haruki Ishikawa
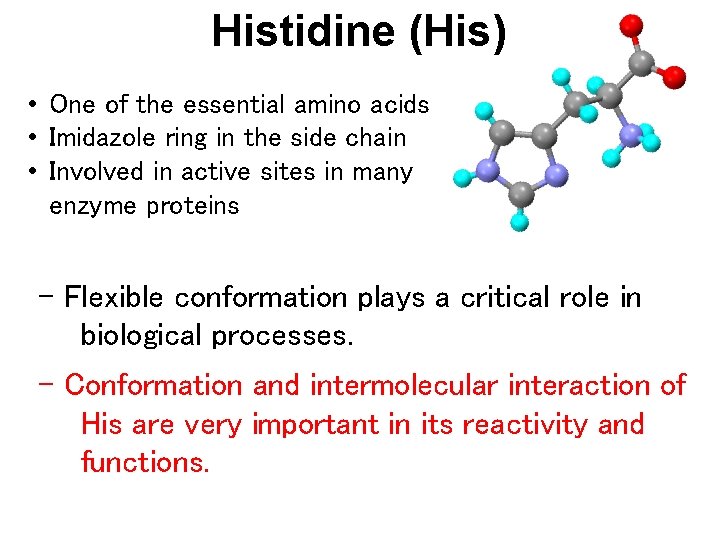
Histidine (His) • One of the essential amino acids • Imidazole ring in the side chain • Involved in active sites in many enzyme proteins - Flexible conformation plays a critical role in biological processes. - Conformation and intermolecular interaction of His are very important in its reactivity and functions.
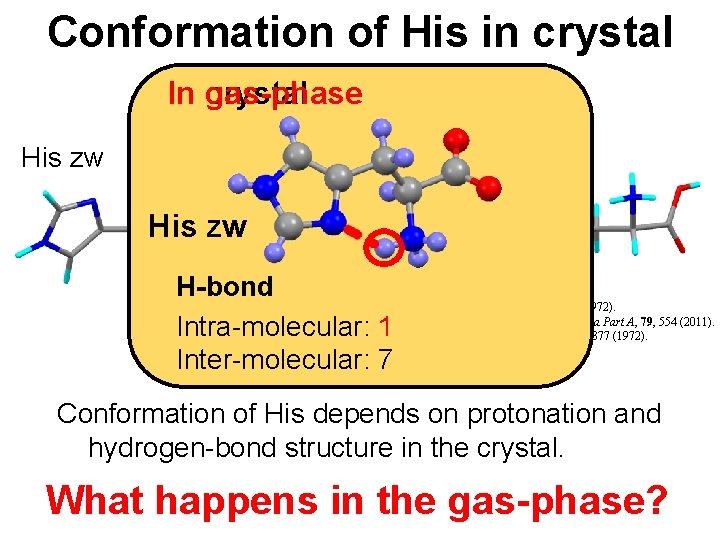
Conformation of His in crystal In gas-phase crystal His zw His HCl His 2 HCl His zw H-bond Intra-molecular: 1 Inter-molecular: 7 K. Oda, Acta. Cryst. , B 28, 639 (1972). A. B. Ahmed, Spectrochimica Acta Part A, 79, 554 (2011). J. J. Madden, Acta. Cryst. , B 28, 2377 (1972). Conformation of His depends on protonation and hydrogen-bond structure in the crystal. What happens in the gas-phase?
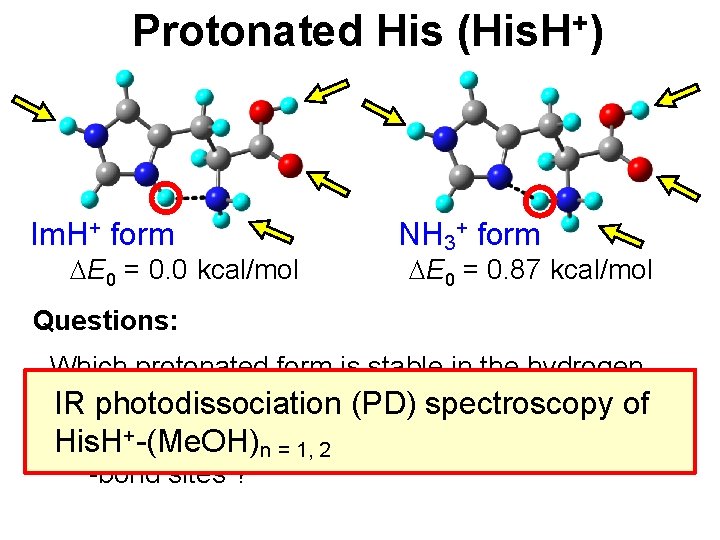
Protonated His (His. H+) Im. H+ form DE 0 = 0. 0 kcal/mol NH 3+ form DE 0 = 0. 87 kcal/mol Questions: - Which protonated form is stable in the hydrogencluster ? IR bonded photodissociation (PD) spectroscopy of +-(Me. OH) - Is there any relation the protonation and H His. H n = 1, between 2 -bond sites ?
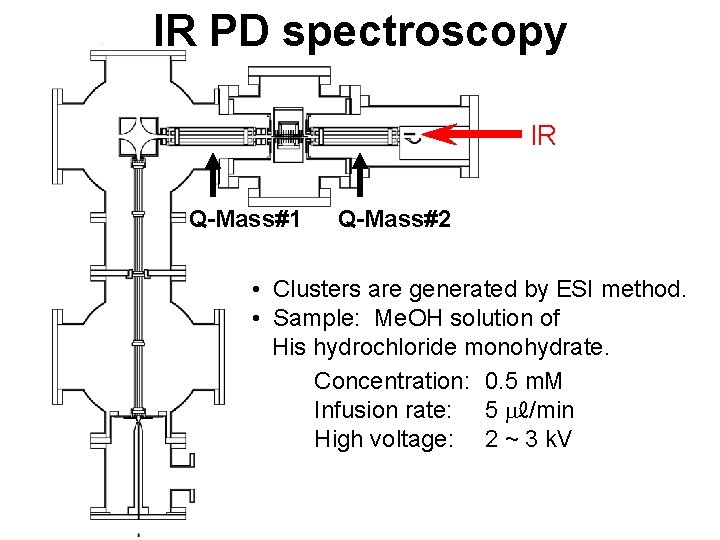
IR PD spectroscopy IR Q-Mass#1 Q-Mass#2 • Clusters are generated by ESI method. • Sample: Me. OH solution of His hydrochloride monohydrate. Concentration: 0. 5 m. M Infusion rate: 5 mℓ/min High voltage: 2 ~ 3 k. V
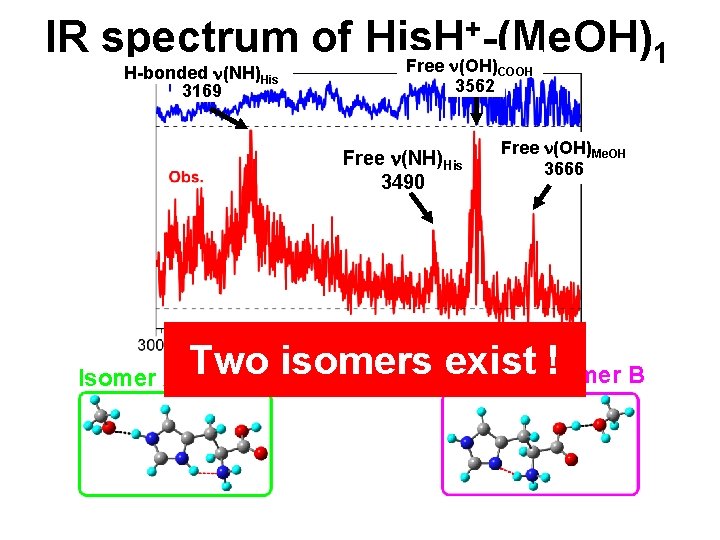
+-(Me. OH) IR spectrum of His. H 1 Free n(OH) H-bonded n(NH)His 3169 3562 Free n(NH)His 3490 Isomer A COOH Free n(OH)Me. OH 3666 Two isomers exist Isomer ! B
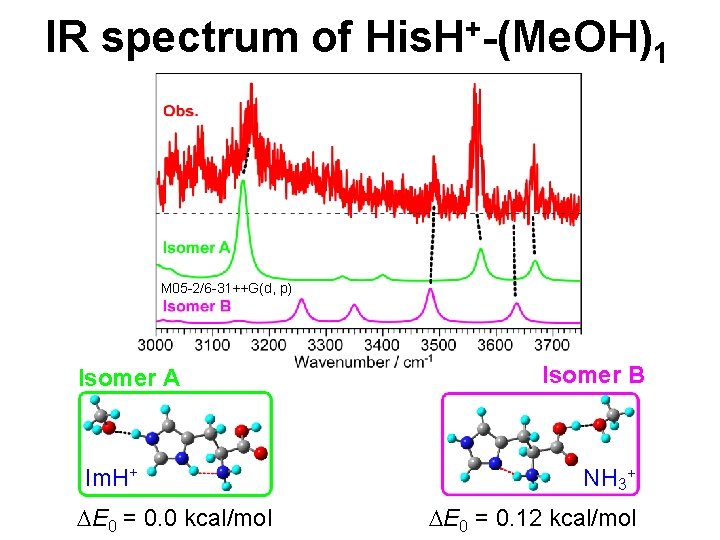
IR spectrum of His. H+-(Me. OH)1 M 05 -2/6 -31++G(d, p) Isomer A Im. H+ DE 0 = 0. 0 kcal/mol Isomer B NH 3+ DE 0 = 0. 12 kcal/mol
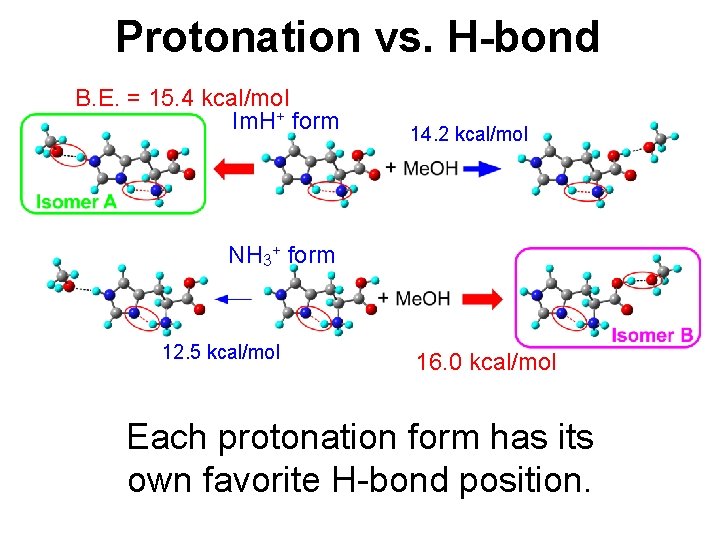
Protonation vs. H-bond B. E. = 15. 4 kcal/mol Im. H+ form 14. 2 kcal/mol NH 3+ form 12. 5 kcal/mol 16. 0 kcal/mol Each protonation form has its own favorite H-bond position.
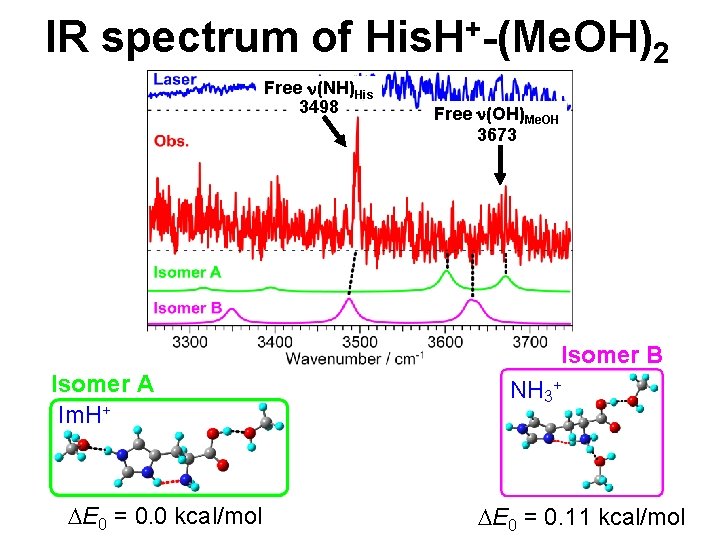
IR spectrum of His. H+-(Me. OH)2 Free n(NH)His 3498 Free n(OH)Me. OH 3673 Isomer B Isomer A Im. H+ DE 0 = 0. 0 kcal/mol NH 3+ DE 0 = 0. 11 kcal/mol

Summary • The structures of His. H+-(Me. OH)n (n = 1, 2) were determined by IR PD spectroscopy and the DFT calculation. Q. Which protonated form is stable in the hydrogen-bonded cluster ? A. Im. H+ form is the most stable for n = 1 and 2. But, the energy difference is reduced by the solvation. Q. Is there any relation between the protonation and H-bond sites ? A. Yes. Each protonation form has its own favorite H-bond position.

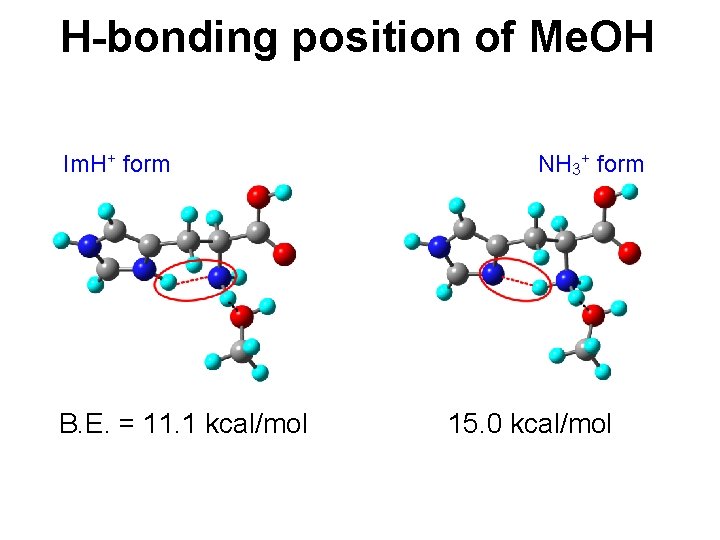
H-bonding position of Me. OH Im. H+ form B. E. = 11. 1 kcal/mol NH 3+ form 15. 0 kcal/mol
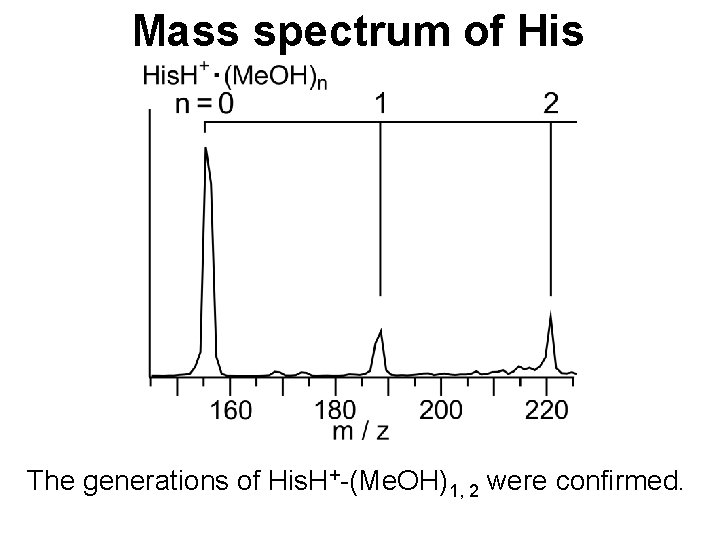
Mass spectrum of His The generations of His. H+-(Me. OH)1, 2 were confirmed.
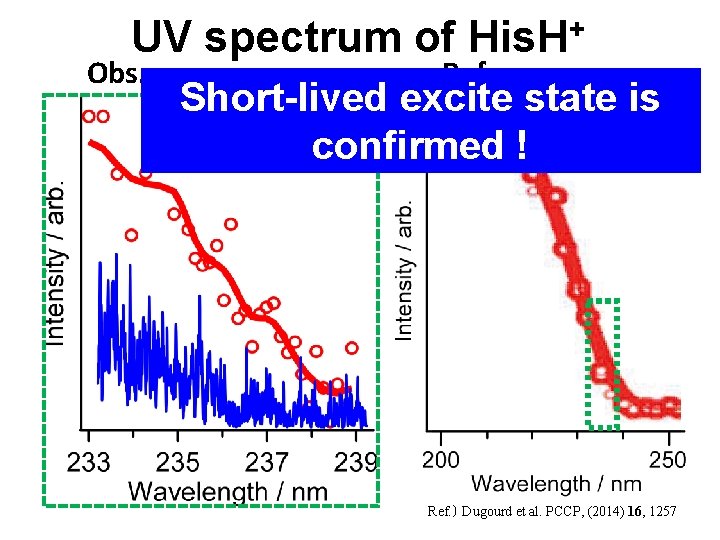
UV spectrum of His. H+ Obs. Ref. Short-lived excite state is confirmed ! Ref. ) Dugourd et al. PCCP, (2014) 16, 1257
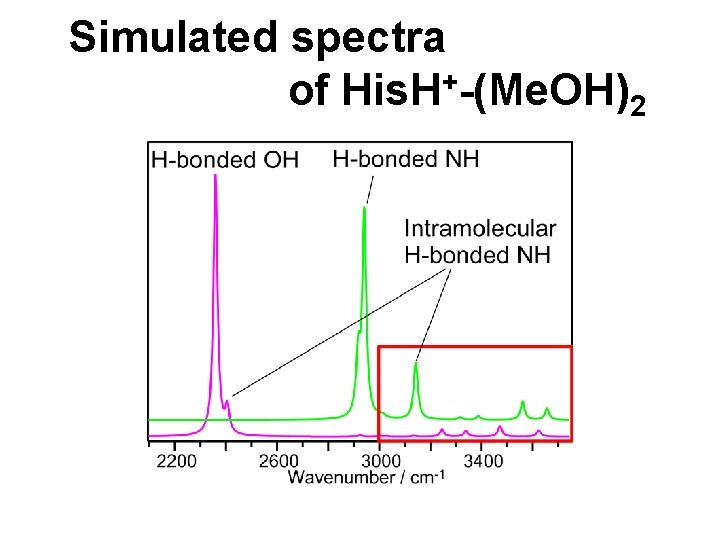
Simulated spectra + of His. H -(Me. OH)2
- Slides: 15