Infrared Spectra and Calculated Binding Energies of butyrolactone

Infrared Spectra and Calculated Binding Energies of γbutyrolactone Dimers and Trimers 69 TH ANNUAL INTERNATIONAL SYMPOSIUM ON MOLECULAR SPECTROSCOPY ERIC WILLIS, CHRIS BAUMANN JUNE 19, 2014 RI 07
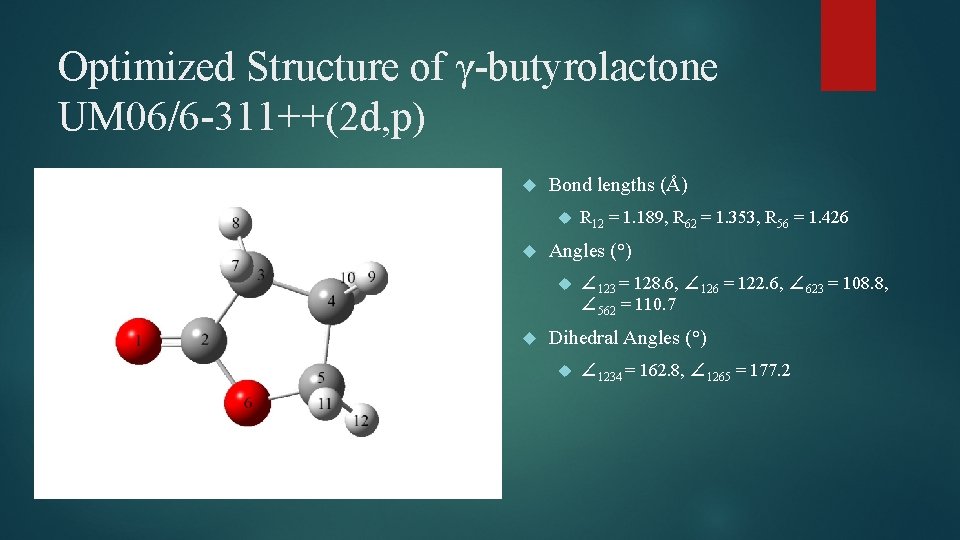
Optimized Structure of γ-butyrolactone UM 06/6 -311++(2 d, p) Bond lengths (Å) Angles (°) R 12 = 1. 189, R 62 = 1. 353, R 56 = 1. 426 ∠ 123 = 128. 6, ∠ 126 = 122. 6, ∠ 623 = 108. 8, ∠ 562 = 110. 7 Dihedral Angles (°) ∠ 1234 = 162. 8, ∠ 1265 = 177. 2
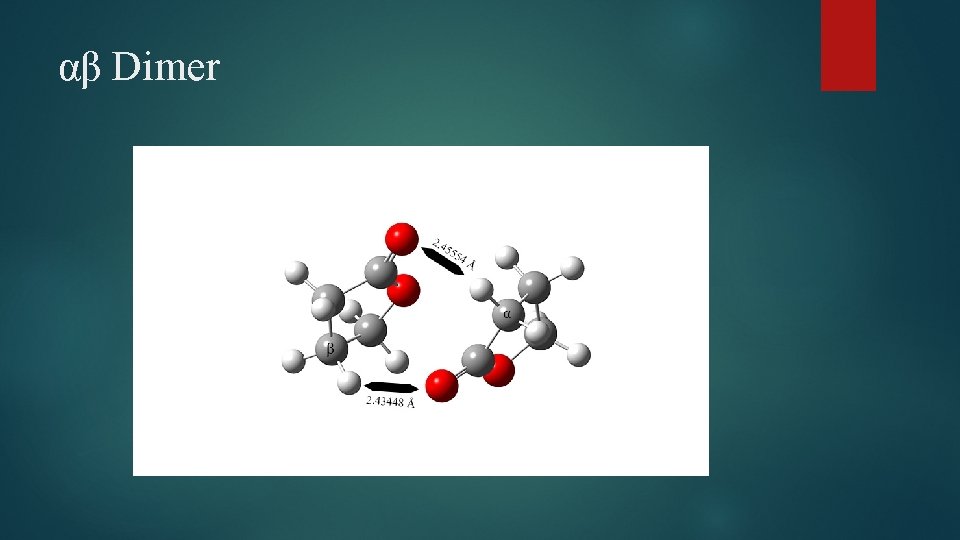
αβ Dimer
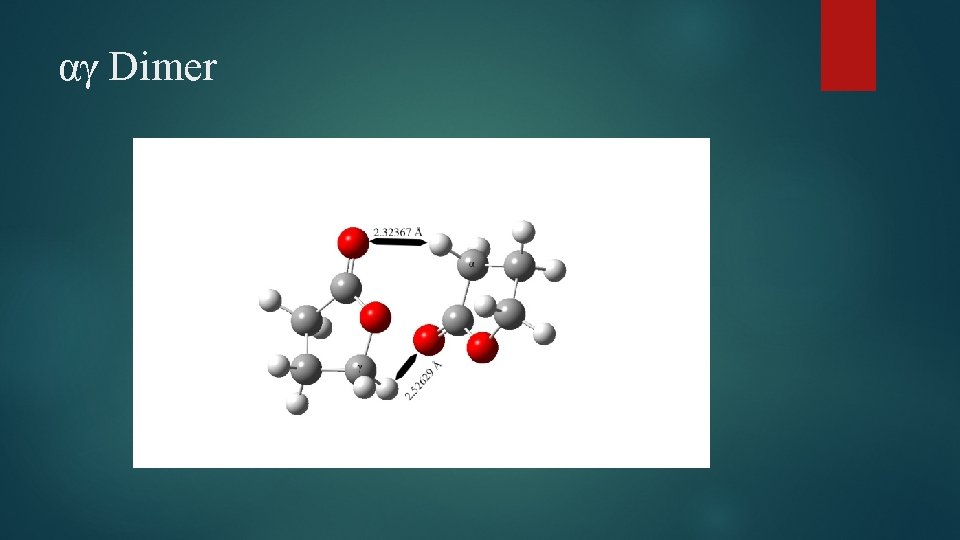
αγ Dimer
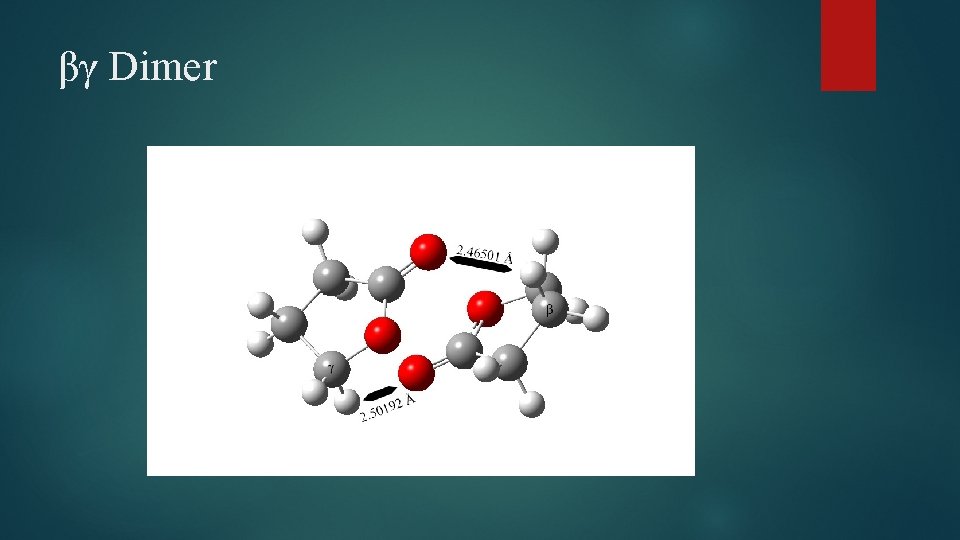
βγ Dimer
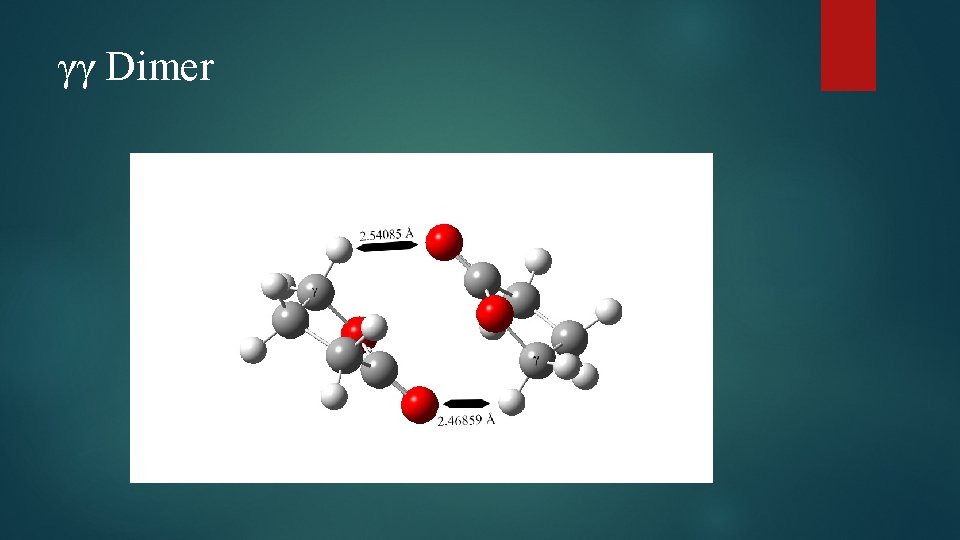
γγ Dimer

Dimer Structural Changes Elongation of C=O bond observed in all species Cγ – O bond elongated in all species Carbonyl C-O bond shortened in case of γγ and βγ dimers Several bond angle alterations observed in dimers Centered around carbonyl group and ring oxygen Dihedral angle changes indicating changes in ring puckering Increased puckering on one side, decreased on the other
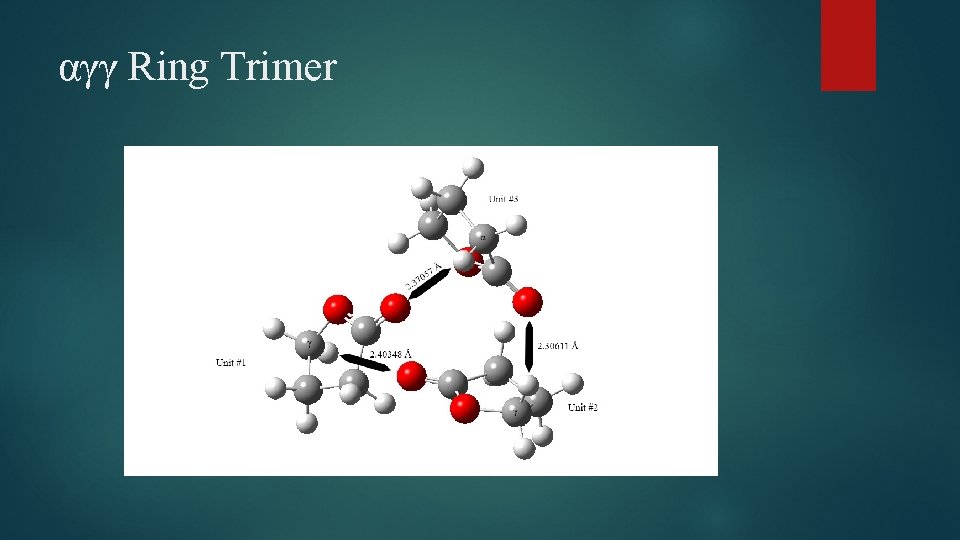
αγγ Ring Trimer
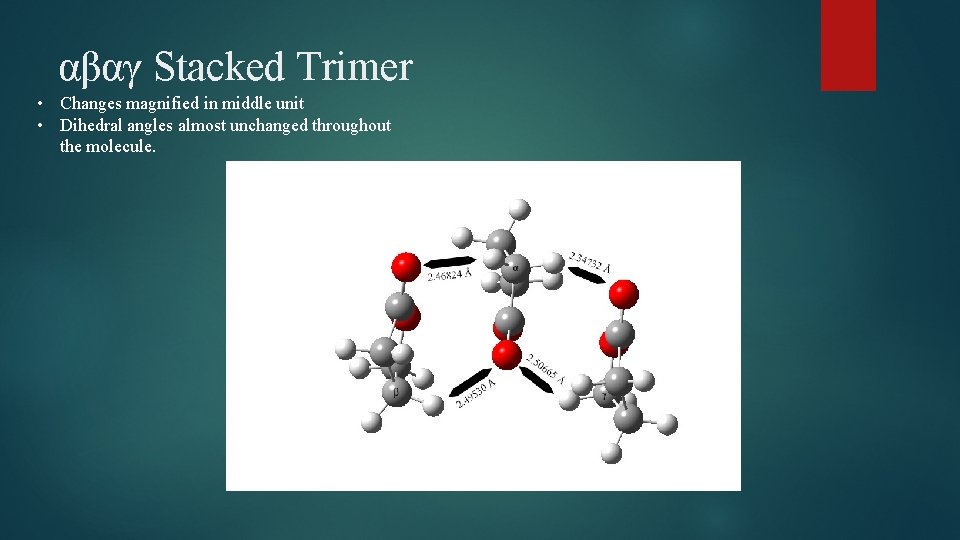
αβαγ Stacked Trimer • Changes magnified in middle unit • Dihedral angles almost unchanged throughout the molecule.
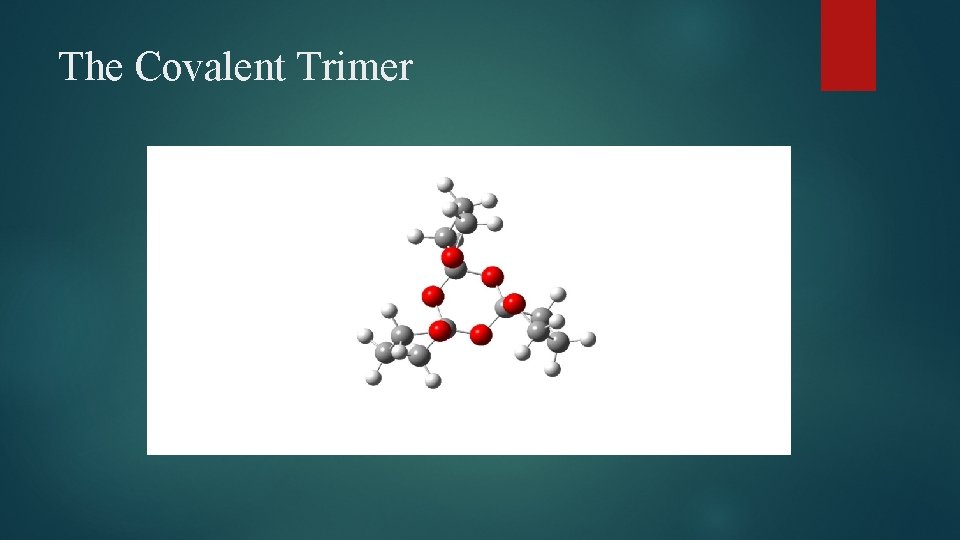
The Covalent Trimer
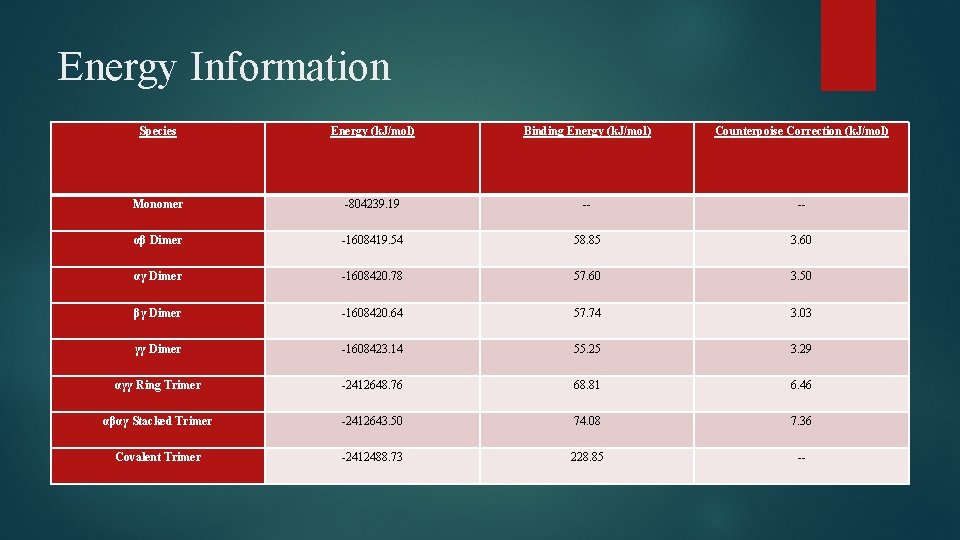
Energy Information Species Energy (k. J/mol) Binding Energy (k. J/mol) Counterpoise Correction (k. J/mol) Monomer -804239. 19 -- -- αβ Dimer -1608419. 54 58. 85 3. 60 αγ Dimer -1608420. 78 57. 60 3. 50 βγ Dimer -1608420. 64 57. 74 3. 03 γγ Dimer -1608423. 14 55. 25 3. 29 αγγ Ring Trimer -2412648. 76 68. 81 6. 46 αβαγ Stacked Trimer -2412643. 50 74. 08 7. 36 Covalent Trimer -2412488. 73 228. 85 --

Matrix Isolation Process GBL deposited on nitrogen at 20 K Cooled down to 12 K Photolysis Annealing
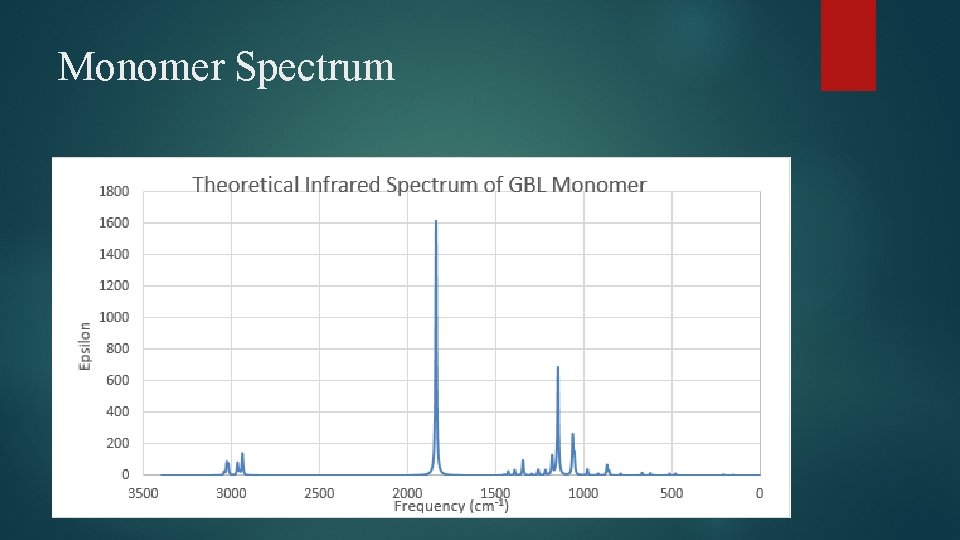
Monomer Spectrum
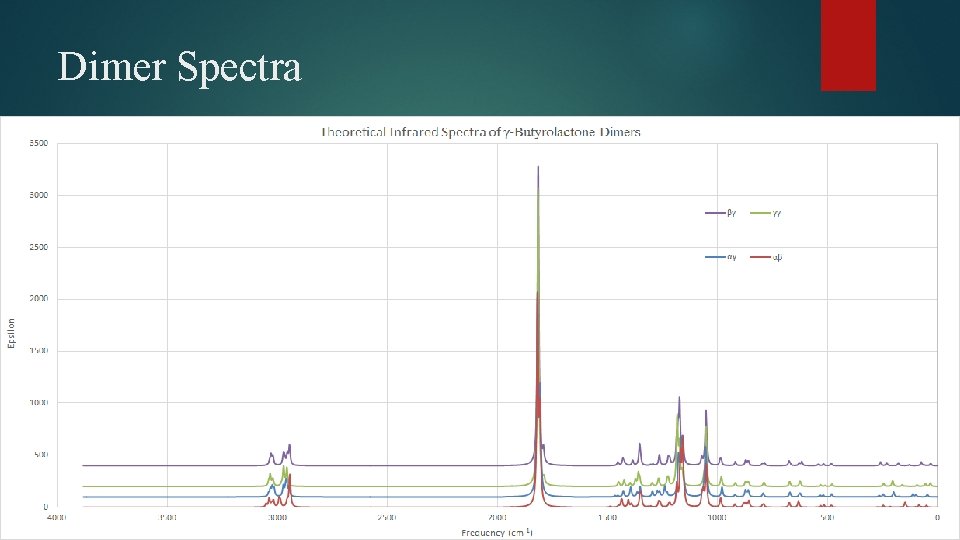
Dimer Spectra
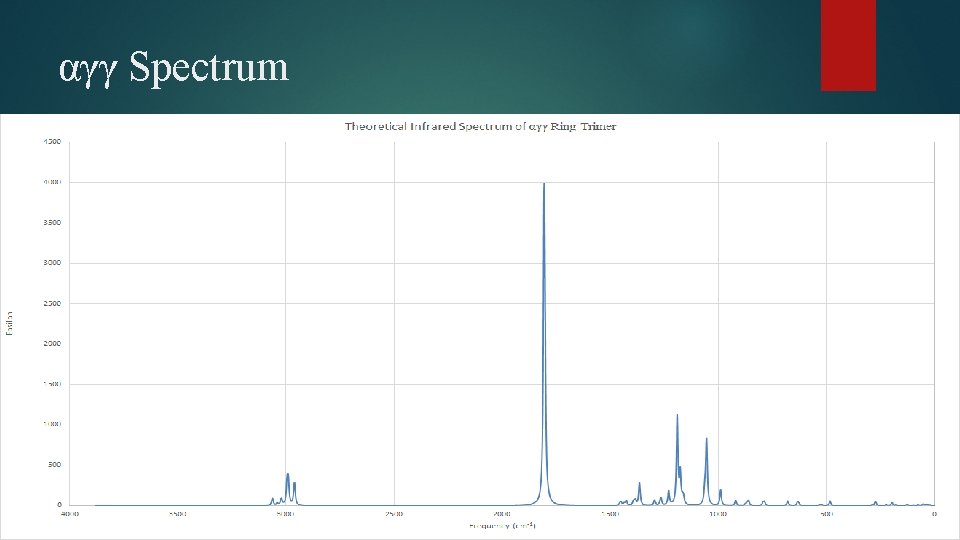
αγγ Spectrum
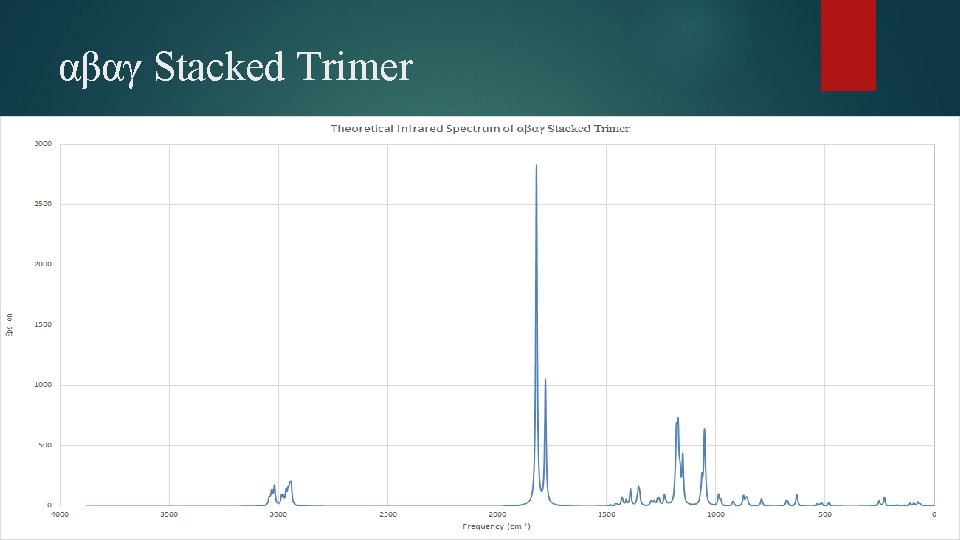
αβαγ Stacked Trimer
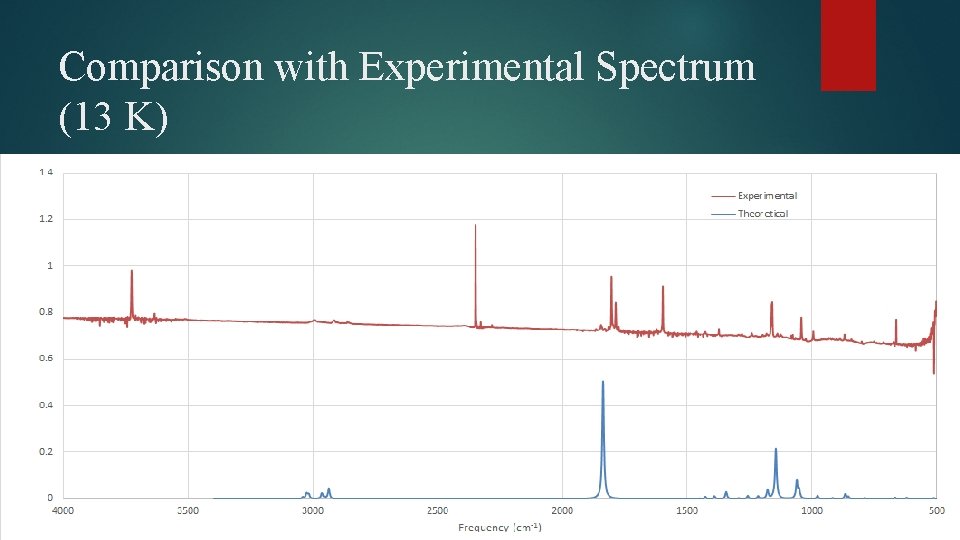
Comparison with Experimental Spectrum (13 K)
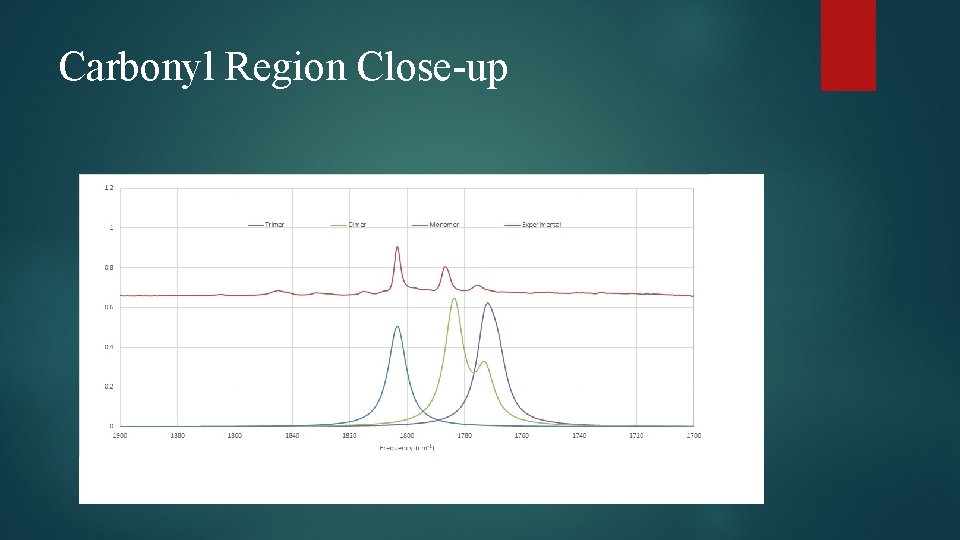
Carbonyl Region Close-up
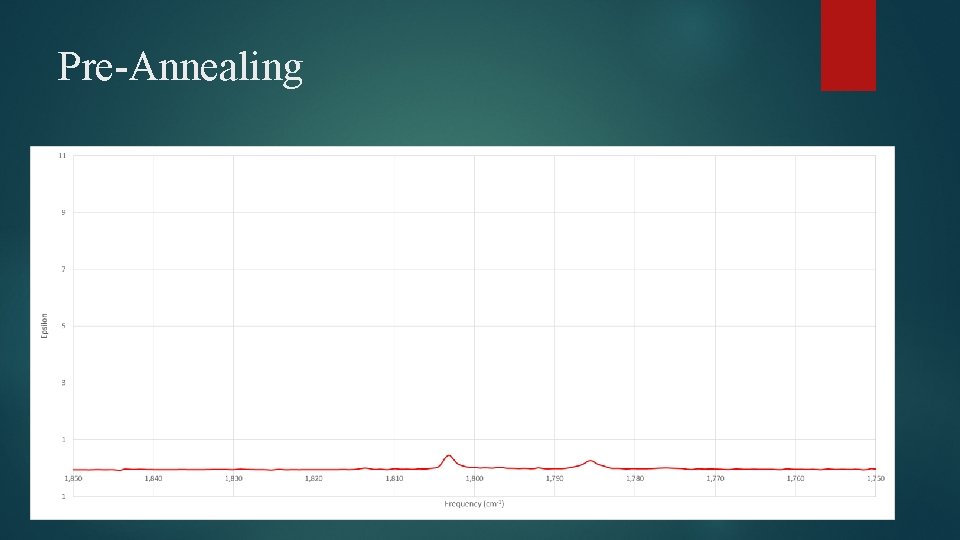
Pre-Annealing
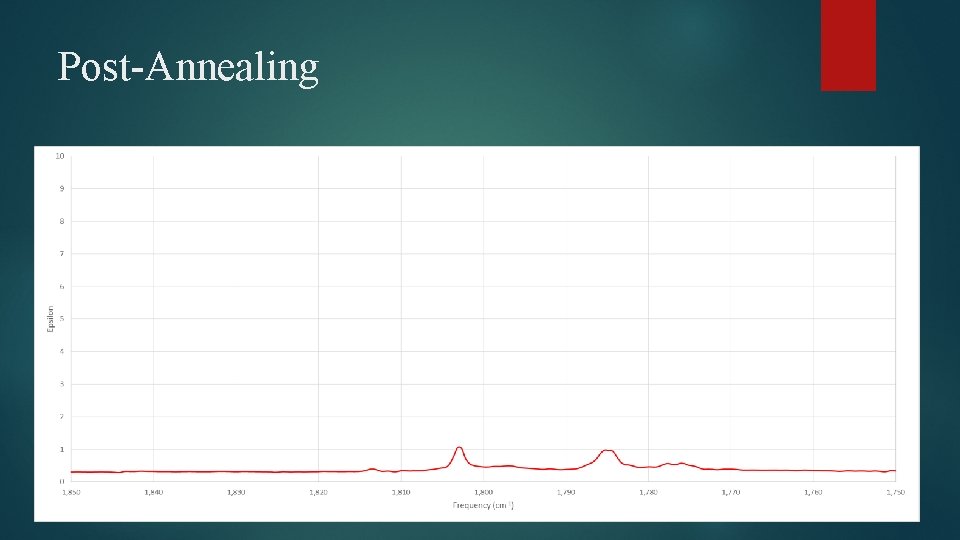
Post-Annealing

Results of Experimental Spectral Analysis Dimer and trimer peaks can be differentiated, however individual dimer and trimer structures can’t be resolved Relative amount of dimer and trimer increases with annealing Photolysis affects ratio as well
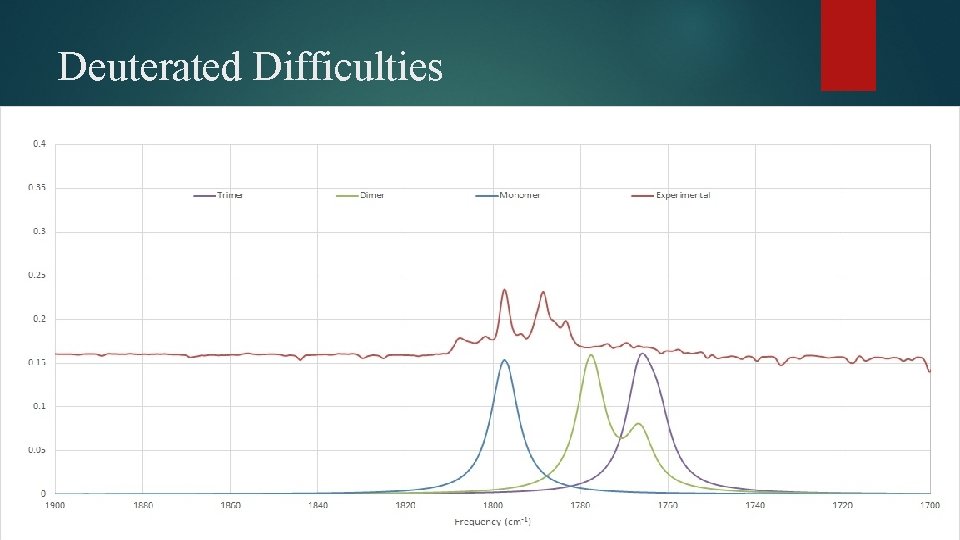
Deuterated Difficulties
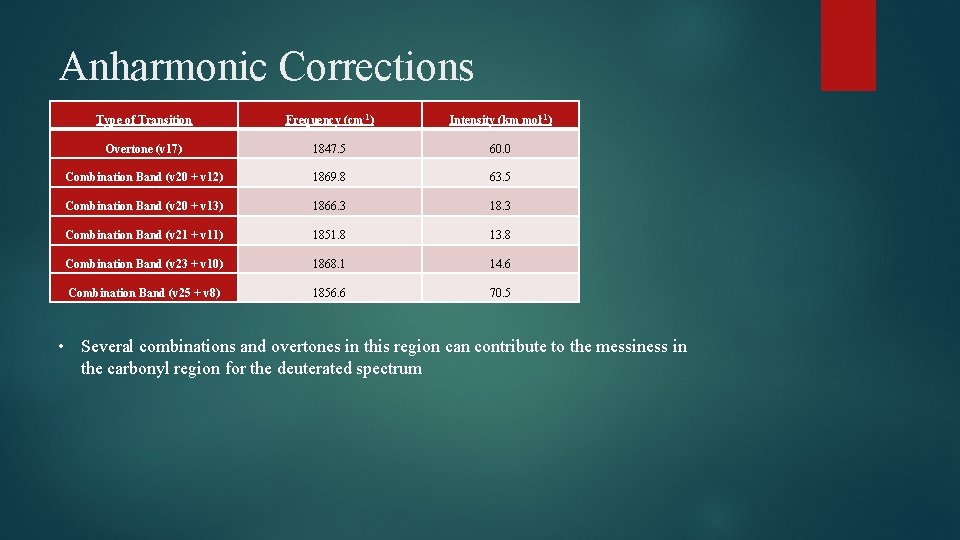
Anharmonic Corrections Type of Transition Frequency (cm-1) Intensity (km mol-1) Overtone (v 17) 1847. 5 60. 0 Combination Band (v 20 + v 12) 1869. 8 63. 5 Combination Band (v 20 + v 13) 1866. 3 18. 3 Combination Band (v 21 + v 11) 1851. 8 13. 8 Combination Band (v 23 + v 10) 1868. 1 14. 6 Combination Band (v 25 + v 8) 1856. 6 70. 5 • Several combinations and overtones in this region can contribute to the messiness in the carbonyl region for the deuterated spectrum
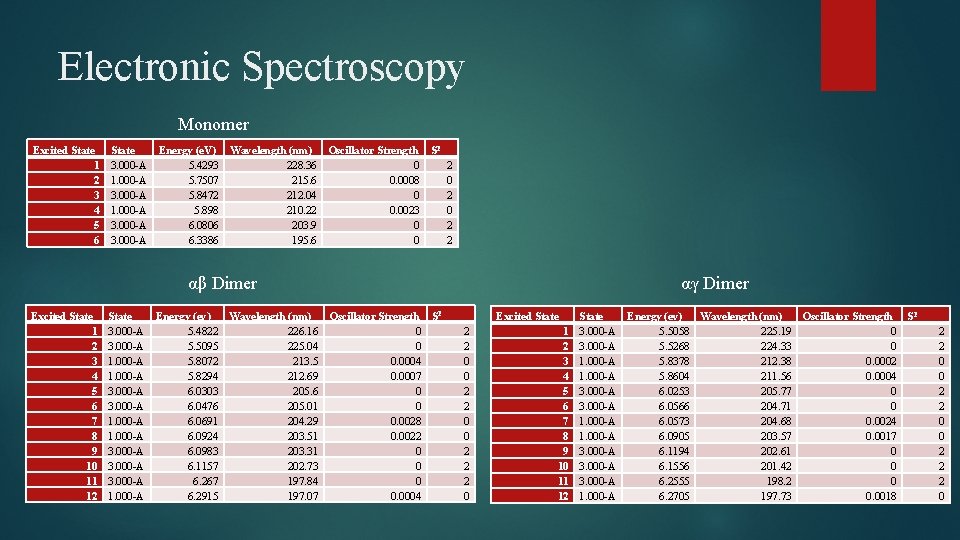
Electronic Spectroscopy Monomer Excited State 1 2 3 4 5 6 State 3. 000 -A 1. 000 -A 3. 000 -A Energy (e. V) Wavelength (nm) Oscillator Strength S 2 5. 4293 228. 36 0 2 5. 7507 215. 6 0. 0008 0 5. 8472 212. 04 0 2 5. 898 210. 22 0. 0023 0 6. 0806 203. 9 0 2 6. 3386 195. 6 0 2 αβ Dimer Excited State 1 2 3 4 5 6 7 8 9 10 11 12 State 3. 000 -A 1. 000 -A 3. 000 -A 1. 000 -A Energy (ev) Wavelength (nm) Oscillator Strength S 2 5. 4822 226. 16 0 5. 5095 225. 04 0 5. 8072 213. 5 0. 0004 5. 8294 212. 69 0. 0007 6. 0303 205. 6 0 6. 0476 205. 01 0 6. 0691 204. 29 0. 0028 6. 0924 203. 51 0. 0022 6. 0983 203. 31 0 6. 1157 202. 73 0 6. 267 197. 84 0 6. 2915 197. 07 0. 0004 αγ Dimer Excited State 2 2 0 0 2 2 2 0 1 2 3 4 5 6 7 8 9 10 11 12 State 3. 000 -A 1. 000 -A 3. 000 -A 1. 000 -A Energy (ev) Wavelength (nm) Oscillator Strength S 2 5. 5058 225. 19 0 5. 5268 224. 33 0 5. 8378 212. 38 0. 0002 5. 8604 211. 56 0. 0004 6. 0253 205. 77 0 6. 0566 204. 71 0 6. 0573 204. 68 0. 0024 6. 0905 203. 57 0. 0017 6. 1194 202. 61 0 6. 1556 201. 42 0 6. 2555 198. 2 0 6. 2705 197. 73 0. 0018 2 2 0 0 2 2 2 0
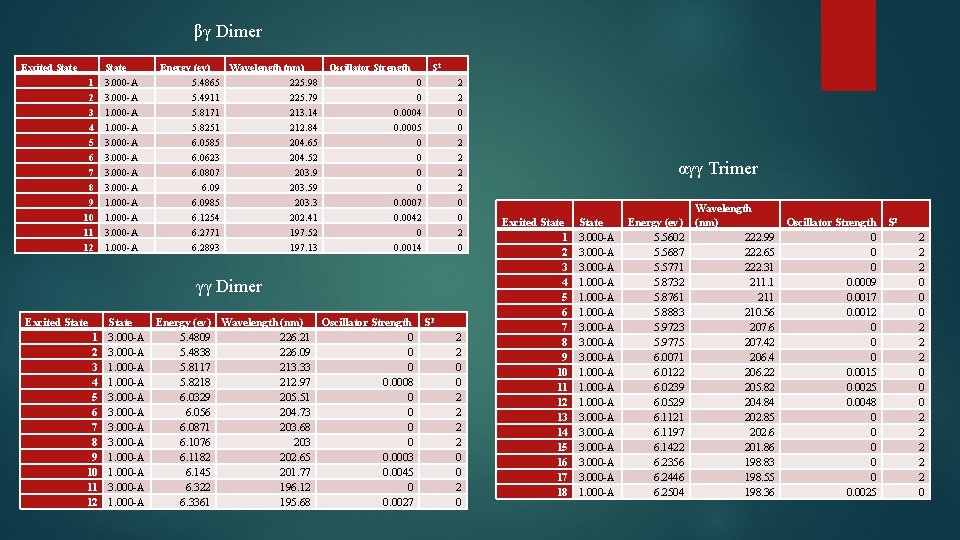
βγ Dimer Excited State 1 2 3 4 State 3. 000 -A 1. 000 -A Energy (ev) 5. 4865 5. 4911 5. 8171 5. 8251 Wavelength (nm) 225. 98 225. 79 213. 14 212. 84 5 6 7 8 9 10 11 12 3. 000 -A 1. 000 -A 6. 0585 6. 0623 6. 0807 6. 0985 6. 1254 6. 2771 6. 2893 204. 65 204. 52 203. 9 203. 59 203. 3 202. 41 197. 52 197. 13 Oscillator Strength S 2 0 0 0. 0004 0. 0005 2 2 0 0 0 0. 0007 0. 0042 0 0. 0014 2 2 0 0 2 0 γγ Dimer Excited State 1 2 3 4 5 6 7 8 9 10 11 12 State 3. 000 -A 1. 000 -A 3. 000 -A 1. 000 -A Energy (ev) Wavelength (nm) Oscillator Strength S 2 5. 4809 226. 21 0 5. 4838 226. 09 0 5. 8117 213. 33 0 5. 8218 212. 97 0. 0008 6. 0329 205. 51 0 6. 056 204. 73 0 6. 0871 203. 68 0 6. 1076 203 0 6. 1182 202. 65 0. 0003 6. 145 201. 77 0. 0045 6. 322 196. 12 0 6. 3361 195. 68 0. 0027 2 2 0 0 2 0 αγγ Trimer Excited State 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 State 3. 000 -A 1. 000 -A 3. 000 -A 1. 000 -A Energy (ev) 5. 5602 5. 5687 5. 5771 5. 8732 5. 8761 5. 8883 5. 9723 5. 9775 6. 0071 6. 0122 6. 0239 6. 0529 6. 1121 6. 1197 6. 1422 6. 2356 6. 2446 6. 2504 Wavelength (nm) Oscillator Strength S 2 222. 99 0 222. 65 0 222. 31 0 211. 1 0. 0009 211 0. 0017 210. 56 0. 0012 207. 6 0 207. 42 0 206. 4 0 206. 22 0. 0015 205. 82 0. 0025 204. 84 0. 0048 202. 85 0 202. 6 0 201. 86 0 198. 83 0 198. 55 0 198. 36 0. 0025 2 2 2 0 0 0 2 2 2 0
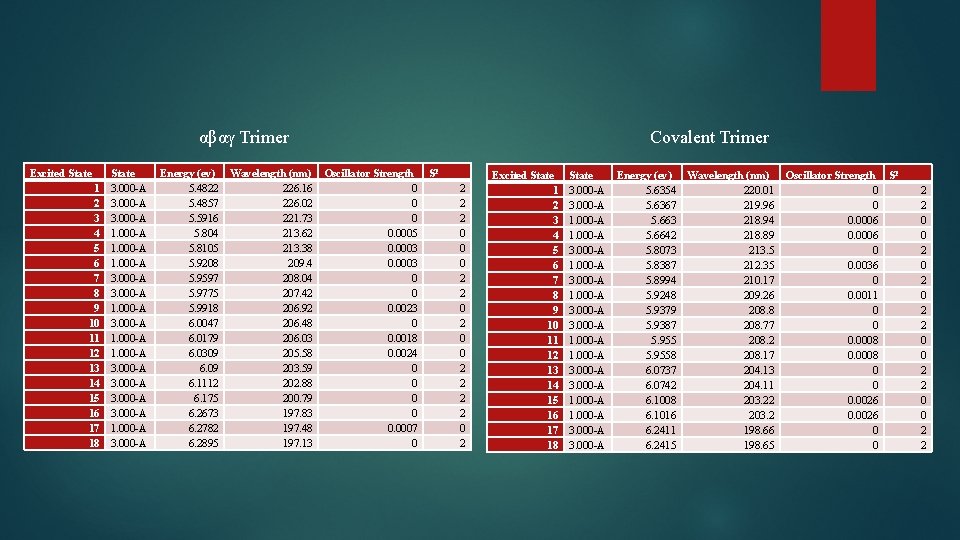
αβαγ Trimer Excited State 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 State 3. 000 -A 1. 000 -A 3. 000 -A Energy (ev) Wavelength (nm) Oscillator Strength S 2 5. 4822 226. 16 0 5. 4857 226. 02 0 5. 5916 221. 73 0 5. 804 213. 62 0. 0005 5. 8105 213. 38 0. 0003 5. 9208 209. 4 0. 0003 5. 9597 208. 04 0 5. 9775 207. 42 0 5. 9918 206. 92 0. 0023 6. 0047 206. 48 0 6. 0179 206. 03 0. 0018 6. 0309 205. 58 0. 0024 6. 09 203. 59 0 6. 1112 202. 88 0 6. 175 200. 79 0 6. 2673 197. 83 0 6. 2782 197. 48 0. 0007 6. 2895 197. 13 0 Covalent Trimer 2 2 2 0 0 0 2 2 0 2 Excited State 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 State 3. 000 -A 1. 000 -A 3. 000 -A Energy (ev) Wavelength (nm) Oscillator Strength S 2 5. 6354 220. 01 0 5. 6367 219. 96 0 5. 663 218. 94 0. 0006 5. 6642 218. 89 0. 0006 5. 8073 213. 5 0 5. 8387 212. 35 0. 0036 5. 8994 210. 17 0 5. 9248 209. 26 0. 0011 5. 9379 208. 8 0 5. 9387 208. 77 0 5. 955 208. 2 0. 0008 5. 9558 208. 17 0. 0008 6. 0737 204. 13 0 6. 0742 204. 11 0 6. 1008 203. 22 0. 0026 6. 1016 203. 2 0. 0026 6. 2411 198. 66 0 6. 2415 198. 65 0 2 2 0 0 2 2

Summary/Conclusions Structural changes observed in monomer units when dimer and trimer species are formed Dimers and trimers can be detected experimentally using vibrational spectroscopy However, individual dimer and trimer species cannot be resolved Complicated nature of carbonyl region may be due to combination bands and overtones

What’s Next? GBL + H 2 O complexes Selective photodissociation of trimers and dimers as a possibility More accurate calculations Creating more trimer structures and higher multimers
- Slides: 28