Infrared photodissociation spectroscopy of protonated methanol cluster ions




- Slides: 14

Infrared photodissociation spectroscopy of protonated methanol cluster ions Kensuke Tono and Koichi Tsukiyama Department of Chemistry & IR FEL Research Center Tokyo University of Science

Infrared photodissociation spectroscopy of H+(CH 3 OH)n (n = 5 ~ 8) Kensuke Tono and Koichi Tsukiyama Department of Chemistry & IR FEL Research Center Tokyo University of Science

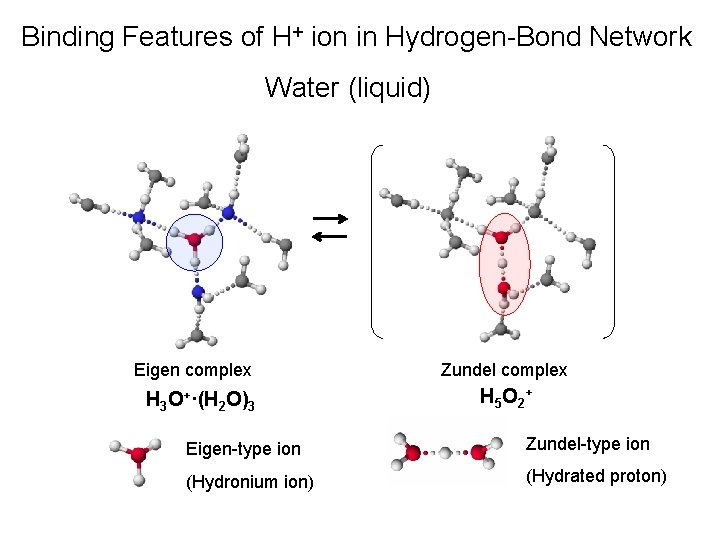
Binding Features of H+ ion in Hydrogen-Bond Network Water (liquid) Eigen complex H 3 O+·(H 2 O)3 Zundel complex H 5 O 2 + Eigen-type ion Zundel-type ion (Hydronium ion) (Hydrated proton)

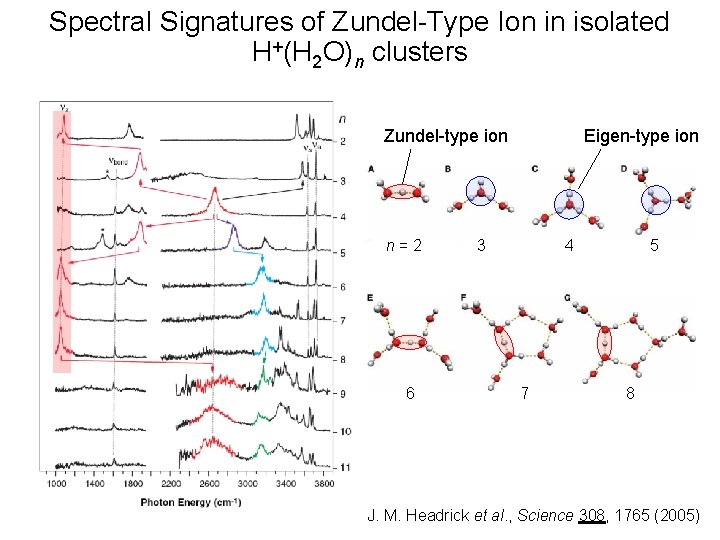
Spectral Signatures of Zundel-Type Ion in isolated H+(H 2 O)n clusters Eigen-type ion Zundel-type ion n=2 6 3 4 7 5 8 J. M. Headrick et al. , Science 308, 1765 (2005)

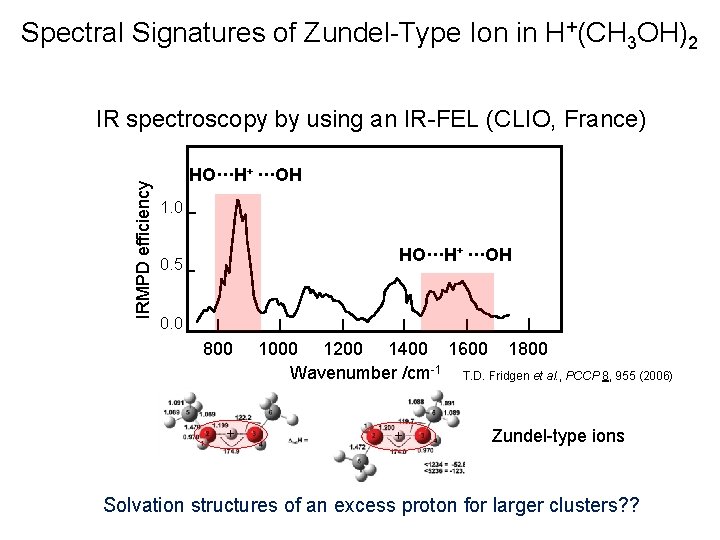
Spectral Signatures of Zundel-Type Ion in H+(CH 3 OH)2 IRMPD efficiency IR spectroscopy by using an IR-FEL (CLIO, France) HO···H+ ···OH 1. 0 HO···H+ ···OH 0. 5 0. 0 800 + 1000 1200 1400 1600 1800 Wavenumber /cm-1 T. D. Fridgen et al. , PCCP 8, 955 (2006) + Zundel-type ions Solvation structures of an excess proton for larger clusters? ?

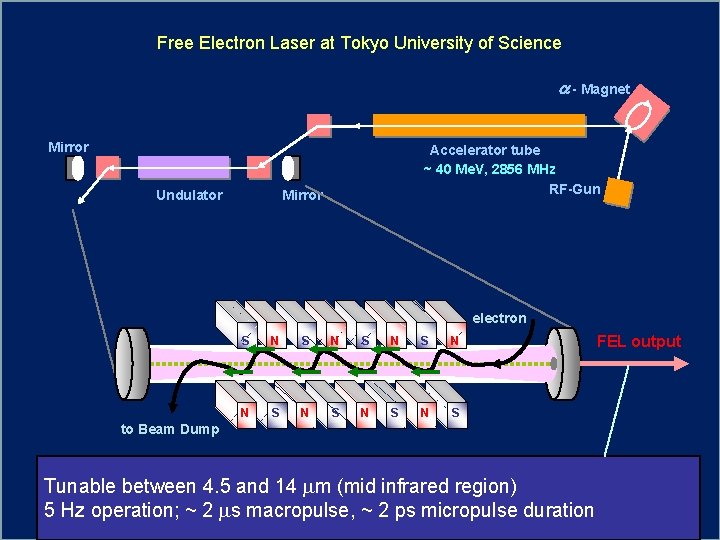
Free Electron Laser at Tokyo University of Science a - Magnet Mirror Undulator Accelerator tube ~ 40 Me. V, 2856 MHz RF-Gun Mirror electron S N S N S to Beam Dump Tunable between 4. 5 and 14 mm (mid infrared region) 5 Hz operation; ~ 2 ms macropulse, ~ 2 ps micropulse duration FEL output

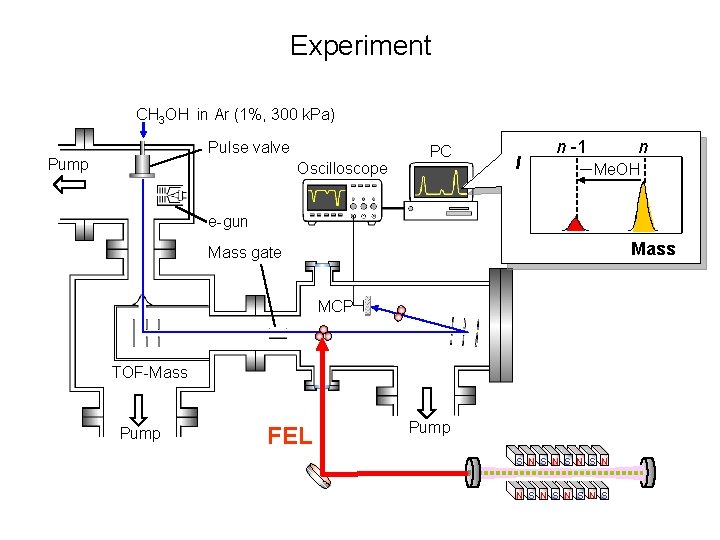
Experiment CH 3 OH in Ar (1%, 300 k. Pa) Pulse valve Pump Oscilloscope PC I n -1 n -Me. OH e-gun Mass gate MCP TOF-Mass Pump FEL Pump S N S N S

IR photodissociation action spectra of H+(CH 3 OH)n

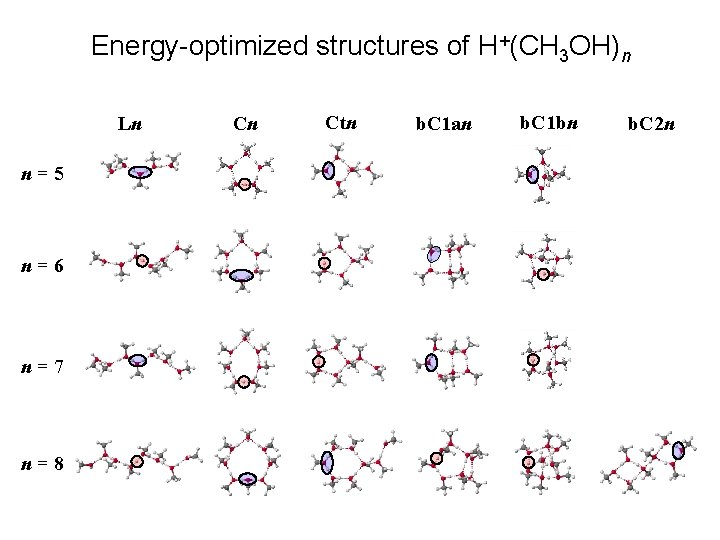
Energy-optimized structures of H+(CH 3 OH)n Ln n=5 n=6 n=7 n=8 Cn Ctn b. C 1 an b. C 1 bn b. C 2 n

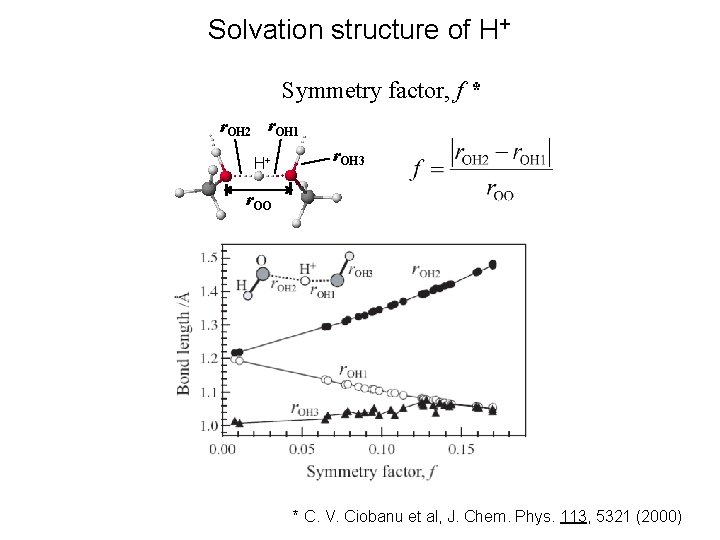
Solvation structure of H+ Symmetry factor, f * r. OH 2 r. OH 1 H+ r. OH 3 r. OO * C. V. Ciobanu et al, J. Chem. Phys. 113, 5321 (2000)

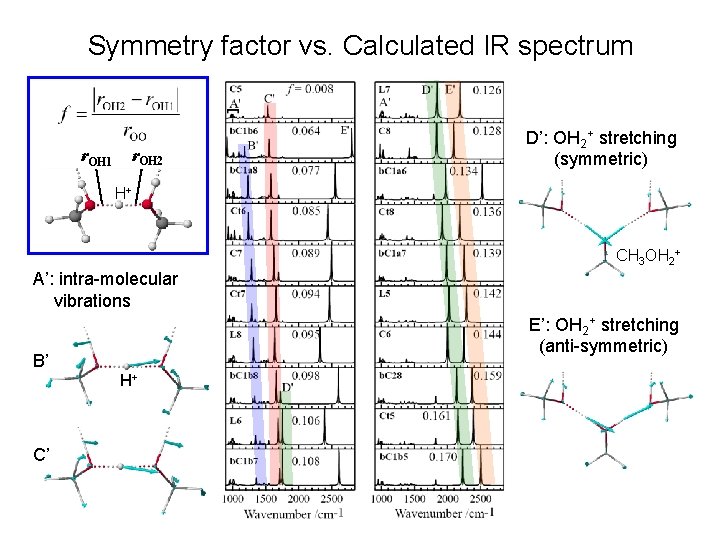
Symmetry factor vs. Calculated IR spectrum r. OH 1 r. OH 2 D’: OH 2+ stretching (symmetric) H+ CH 3 OH 2+ A’: intra-molecular vibrations E’: OH 2+ stretching (anti-symmetric) B’ H+ C’

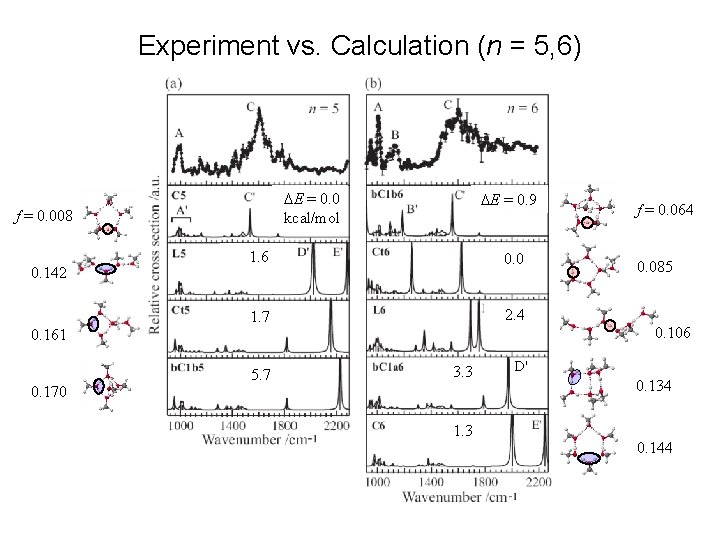
Experiment vs. Calculation (n = 5, 6) DE = 0. 0 kcal/mol f = 0. 008 0. 142 DE = 0. 9 1. 6 0. 0 1. 7 2. 4 0. 170 0. 085 0. 106 0. 161 5. 7 f = 0. 064 3. 3 1. 3 D' 0. 134 0. 144

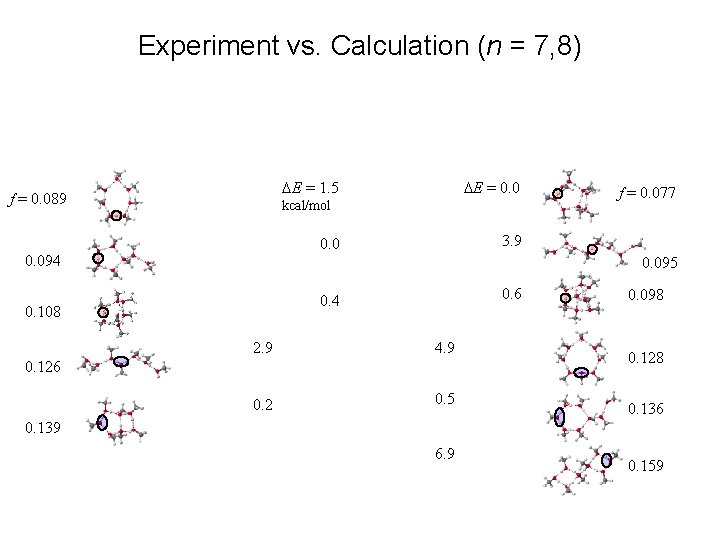
Experiment vs. Calculation (n = 7, 8) DE = 1. 5 f = 0. 089 DE = 0. 0 kcal/mol 3. 9 0. 094 f = 0. 077 0. 095 0. 6 0. 4 0. 108 2. 9 4. 9 0. 2 0. 5 0. 126 0. 098 0. 128 0. 136 0. 139 6. 9 0. 159

Conclusions l. Photodissociation spectra of H+(CH 3 OH)n (n = 5 -8) were measured in the mid-IR range of 900 -2300 cm-1. l. Spectral signature of the Zundel-type structure emerges between 1200 and 1600 cm-1. l. Zundel-based isomers are dominantly produced. l. Eigen-based isomers coexist with the Zundel-based ones in the large size clusters (n ≥ 6).