InfraRed IR Spectroscopy or Vibrational spectroscopy Applied Chemistry
Infra-Red (IR) Spectroscopy or Vibrational spectroscopy Applied Chemistry Course: CHY 101
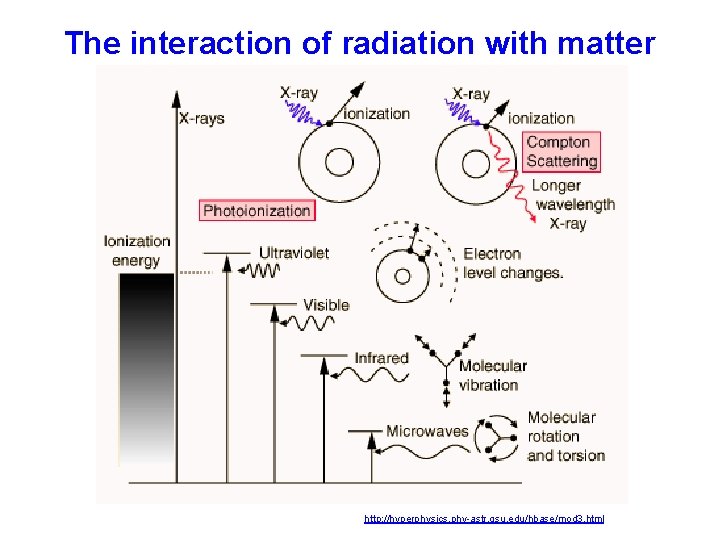
The interaction of radiation with matter http: //hyperphysics. phy-astr. gsu. edu/hbase/mod 3. html
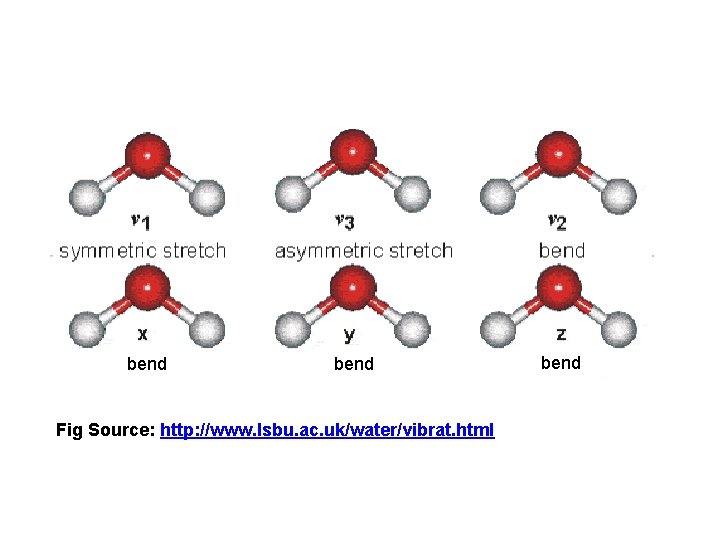
bend Fig Source: http: //www. lsbu. ac. uk/water/vibrat. html bend

Stretching and Bending Types of Molecular Vibrations: Vibrations fall into the basic categories of stretching and bending. q Stretching vibration involves a continuous change in the inter-atomic distance along the axis of the bond between two atoms. q Bending vibrations are characterized by a change in the angle between two bonds and are of four types: scissoring, rocking, wagging, and twisting.
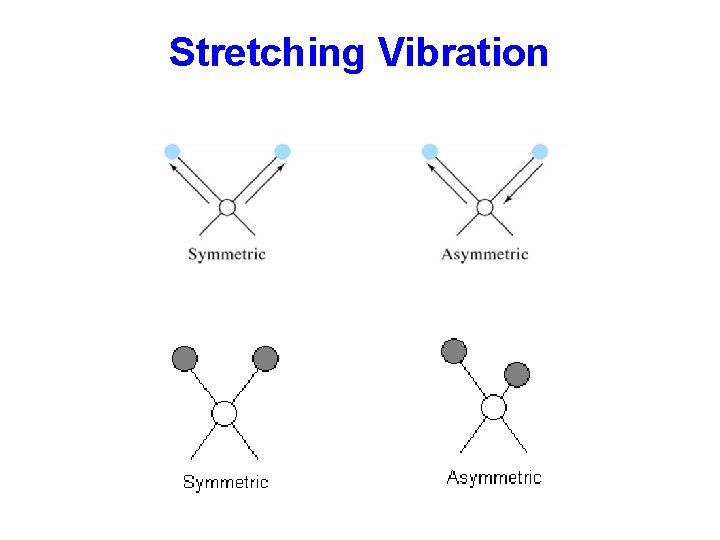
Stretching Vibration
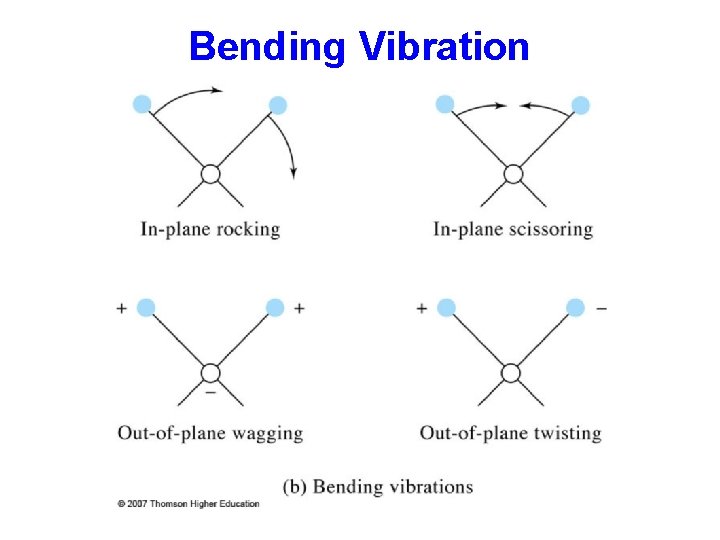
Bending Vibration
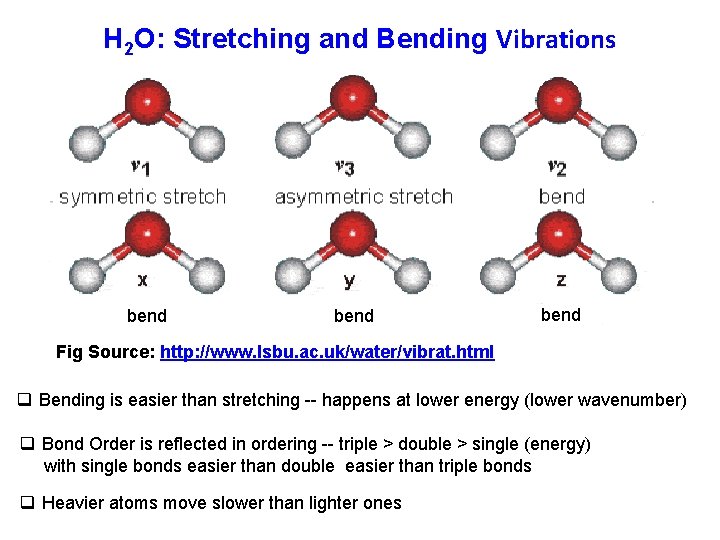
H 2 O: Stretching and Bending Vibrations bend Fig Source: http: //www. lsbu. ac. uk/water/vibrat. html q Bending is easier than stretching -- happens at lower energy (lower wavenumber) q Bond Order is reflected in ordering -- triple > double > single (energy) with single bonds easier than double easier than triple bonds q Heavier atoms move slower than lighter ones


Selection Rules Selection rule: q A molecule will absorb infrared radiation if the change in vibrational states is associated with a change in the dipole moment ( ) of the molecule. q Vibrations which do not change the dipole moment are Infrared Inactive (homonuclear diatomics). Homonuclear diatomic molecules (O 2, N 2, H 2, Cl 2) – IR Inactive Heteronuclear diatomic molecules (HF, HCl) – IR Active Dipole moment is greater when electronegativity difference between the atoms in a bond is greater. Some electronegativity values are: H 2. 2; C 2. 55; N 3. 04; O 3. 44; F 3. 98; P 2. 19; S 2. 58; Cl 3. 16
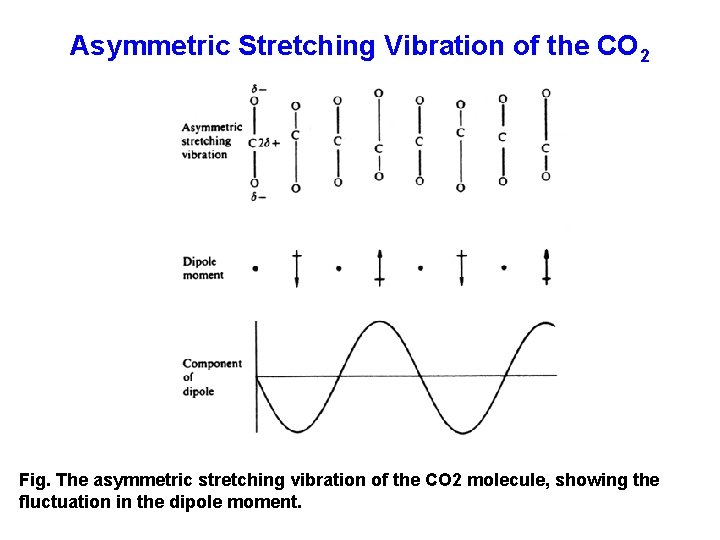
Asymmetric Stretching Vibration of the CO 2 Fig. The asymmetric stretching vibration of the CO 2 molecule, showing the fluctuation in the dipole moment.
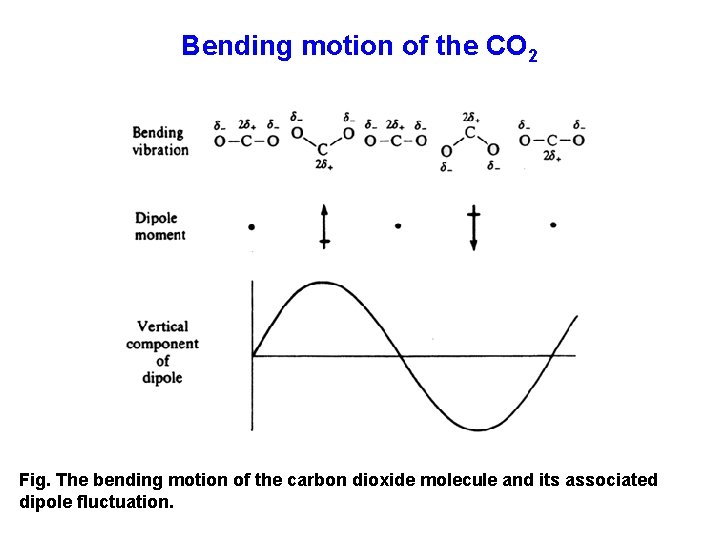
Bending motion of the CO 2 Fig. The bending motion of the carbon dioxide molecule and its associated dipole fluctuation.
- Slides: 11