infrared Infrared radiation extends from the nominal red









- Slides: 10

infrared Infrared radiation extends from the nominal red edge of the visible spectrum at 700 nanometers (nm) to 1 mm. Name Gamma ray X-ray Ultraviolet Visible Infrared Microwave Radio Wavelength less than 0. 01 nm – 10 nm – 380 nm– 700 nm – 1 mm – 1 meter 1 mm – 100, 000 km

ISO 20473 scheme Designation Abbreviation Wavelength Near-Infrared NIR 0. 78– 3 µm Mid-Infrared MIR 3– 50 µm Far-Infrared FIR 50– 1000 µm

Natural infrared • At zenith, sunlight provides an irradiance of just over 1 kilowatt per square meter at sea level. Of this energy, 527 watts is infrared radiation, 445 watts is visible light, and 32 watts is ultraviolet radiation.

Infrared spectroscopy • Infrared spectroscopy is a simple and reliable technique widely used in both organic and inorganic chemistry, in research and industry.

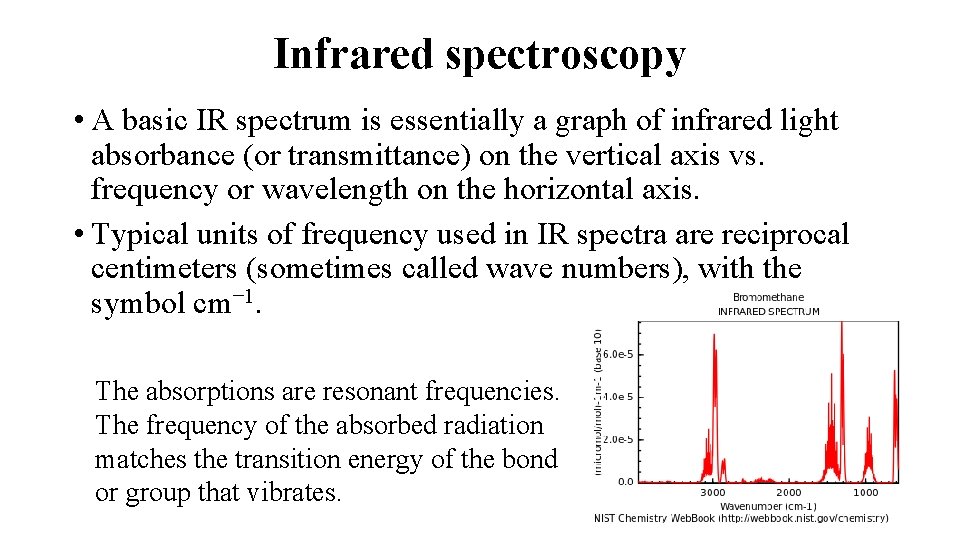
Infrared spectroscopy • A basic IR spectrum is essentially a graph of infrared light absorbance (or transmittance) on the vertical axis vs. frequency or wavelength on the horizontal axis. • Typical units of frequency used in IR spectra are reciprocal centimeters (sometimes called wave numbers), with the symbol cm− 1. The absorptions are resonant frequencies. The frequency of the absorbed radiation matches the transition energy of the bond or group that vibrates.

Number of vibrational modes • A molecule can vibrate in many ways, and each way is called a vibrational mode. For molecules with N number of atoms in them, linear molecules have 3 N – 5 degrees of vibrational modes, whereas nonlinear molecules have 3 N – 6 degrees of vibrational modes (also called vibrational degrees of freedom). As an example H 2 O, a non-linear molecule, will have 3 × 3 – 6 = 3 degrees of vibrational freedom, or modes.

• The atoms in a CH 2 X 2 group, commonly found in organic compounds and where X can represent any other atom, can vibrate in nine different ways. Six of these involve only the CH 2 portion: symmetric and antisymmetric stretching, scissoring, rocking, wagging and twisting, as shown below. (Note, that because CH 2 is attached to X 2 it has 6 modes, unlike H 2 O, which only has 3 modes. The rocking, wagging, and twisting modes do not exist for H 2 O, since they are rigid body translations and no relative displacements exist. )

Practical IR spectroscopy • The infrared spectrum of a sample is recorded by passing a beam of infrared light through the sample. When the frequency of the IR is the same as the vibrational frequency of a bond, absorption occurs. • Analysis of the position, shape and intensity of peaks in this spectrum reveals details about the molecular structure of the sample. • To take the infrared spectrum of a sample, it is necessary to measure both the sample and a "reference" (or "control").

Absorption bands • IR spectroscopy is often used to identify structures because functional groups give rise to characteristic bands both in terms of intensity and position (frequency).

傅立葉轉換紅外線儀(FTIR) • A common laboratory instrument that uses this technique is a Fourier transform infrared (FTIR) spectrometer. • A data-processing technique called Fourier transform turns the raw data into the desired result (the sample's spectrum): Light output as a function of infrared wavelength (or equivalently, wavenumber).