Influenza Vaccination of Pregnant Women Bernard Gonik M

Influenza Vaccination of Pregnant Women Bernard Gonik, M. D. Professor and Fann Srere Chair of Perinatal Medicine Wayne State University School of Medicine Detroit, Michigan 38 th National Immunization Conference

Influenza Infection • 10% of U. S. adults annually • Similar range in pregnancy (2 -22%) Neuzil etal Infect Dis Clin N Am 2001; 15: 123 Griffiths etal J Epidemiol Comm Health 1980; 34: 124

Influenza Morbidity/Mortality in Pregnancy • Pandemics of 1918 and 1957 – 50% complicated by pneumonia – 50% mortality rate • Recent flu case reports and SARS data in pregnancy show higher risks • Mixed data for fetus/neonate with both short and long -term concerns Harris JAMA 1919; 72 -978 Freeman etal AJOG 1959; 78: 172 Gonik etal IDSOG (abstract) 2003

Perinatal Outcomes Associated with Respiratory Diseases in Pregnancy • • 8 flu seasons, 297 admissions (5. 1: 1000) Significant comorbidities (per 1000) • • Asthma (59. 7) ≥ age 35 (11. 2) 3 rd trimester (7. 3) No difference in perinatal outcomes Hartert etal AJOG 2003; 189: 1705
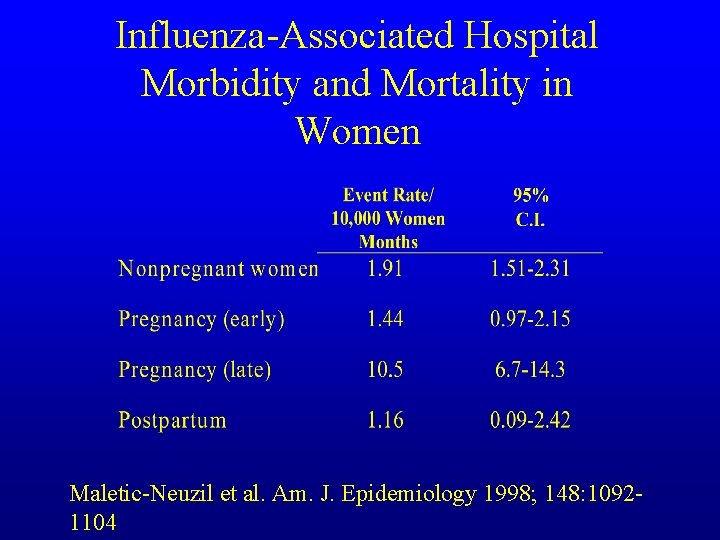
Influenza-Associated Hospital Morbidity and Mortality in Women Maletic-Neuzil et al. Am. J. Epidemiology 1998; 148: 10921104

Current CDC/ACIP Recommendations • All women who will be in the 2 nd and 3 rd trimester during the flu season, and those with underlying high risk conditions regardless of pregnancy stage, should be vaccinated (endorsed by ACOG Committee Opinion No. 282, January 2003) • Trivalent inactivated influenza vaccine should be used • Live-attenuated vaccine is contraindicated

Presumed Vaccine Safety • Collaborative Perinatal Project (1960’s) • 2291 doses • Swine flu vaccine studies (1970’s) • 290 doses • No reported fetal/neonatal adverse events
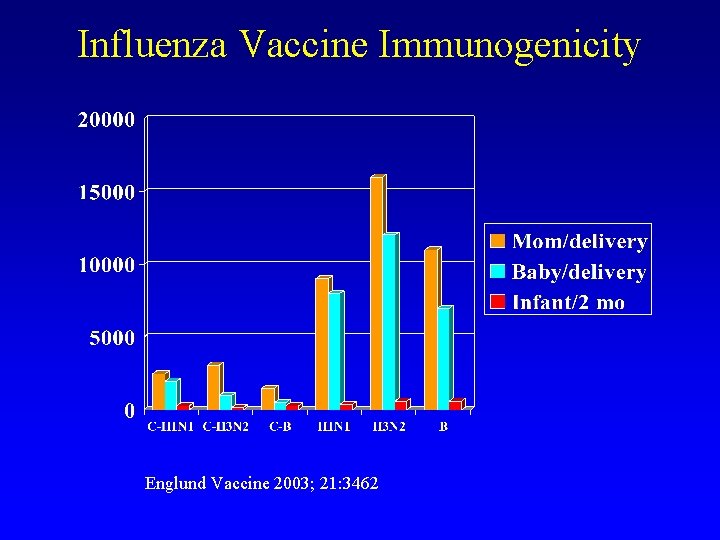
Influenza Vaccine Immunogenicity Englund Vaccine 2003; 21: 3462

Influenza Vaccine Usage • Pregnancy • Reproductive-age women • ≥ age 65 Englund Vaccine 2003; 21: 3460 <10% 16% 66%
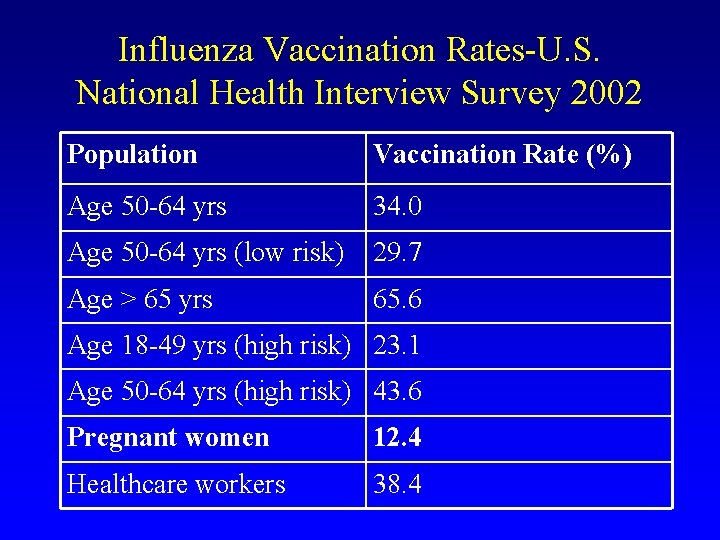
Influenza Vaccination Rates-U. S. National Health Interview Survey 2002 Population Vaccination Rate (%) Age 50 -64 yrs 34. 0 Age 50 -64 yrs (low risk) 29. 7 Age > 65 yrs 65. 6 Age 18 -49 yrs (high risk) 23. 1 Age 50 -64 yrs (high risk) 43. 6 Pregnant women 12. 4 Healthcare workers 38. 4
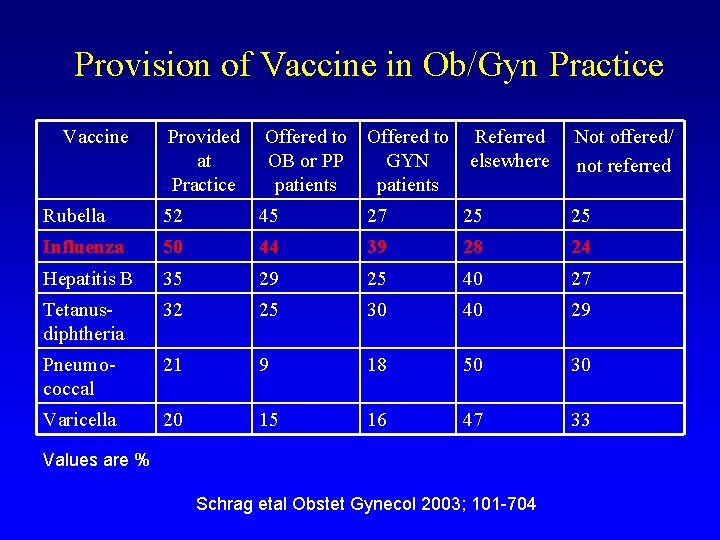
Provision of Vaccine in Ob/Gyn Practice Vaccine Provided at Practice Offered to OB or PP GYN patients Referred elsewhere Rubella 52 45 27 25 25 Influenza 50 44 39 28 24 Hepatitis B 35 29 25 40 27 Tetanusdiphtheria 32 25 30 40 29 Pneumococcal 21 9 18 50 30 Varicella 20 15 16 47 33 Values are % Schrag etal Obstet Gynecol 2003; 101 -704 Not offered/ not referred

Reasons OB/GYN Practices Not Offering Flu Vaccine (56%) • • Reimbursement 52% No pt literature 46% Liability 45% Ambiguous guidelines 30% • Patient refusal* 26% • • Vaccine efficacy 25% Lack of time 20% Vaccine safety 17% Patient’s don’t need 14% *Yeager study – 71% accepted Schrag etal Obset Gynecol 2003; 101: 704 Yeager etal Am J Perinatal 1999; 16: 283

Influenza Vaccination in Pregnancy Survey • 242 postpartum women; 113 OB physicians • Estimated vaccine rate 8% • Physicians more likely to give vaccine – Received vaccine – Know CDC guidelines – Have vaccine in office • Patients more likely to receive vaccine – Previous history of flu/vaccine – Know additional risk – Discussed with OB physician • Physician/Patient discussion (74% vs. 22%) Silverman etal J Repro Med 2001; 46: 989

Ob/Gyn VPD Knowledge, Attitude and Practice Patterns • • • 840 Michigan ACOG Fellows (RR 43%) Acknowledged VPD responsibility 85% Screen for VPD 10 -19% Gave vaccines 6 -25% VPD knowledge 7 -57% (better with females, < years in practice, knowledge and PCP designation) Gonik etal Obstet Gynecol 2000; 98: 81

Educational Interventions • Grand Rounds-styled lectures (532 providers/16 programs) • Office-based in-services (15 sessions) • Joint venture with CDC, MDCH, M-ACOG, MSU extension program, WSU • Resource materials/contacts

Gonik et al. AJOG 2001; 185: S 162
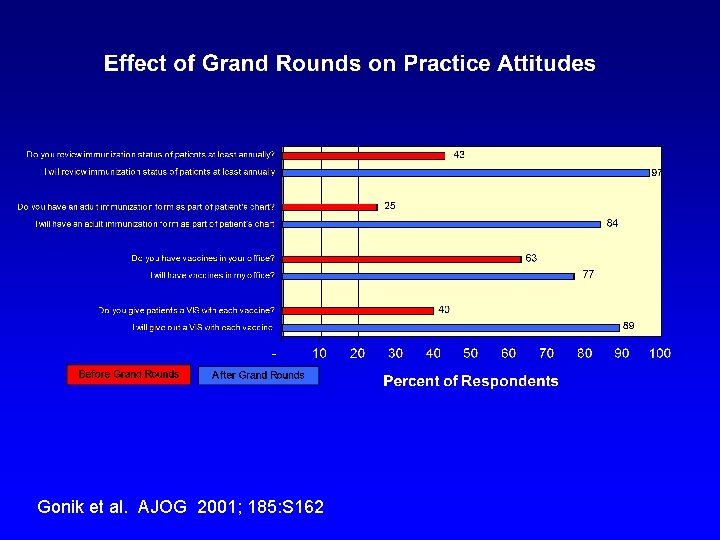
Gonik et al. AJOG 2001; 185: S 162
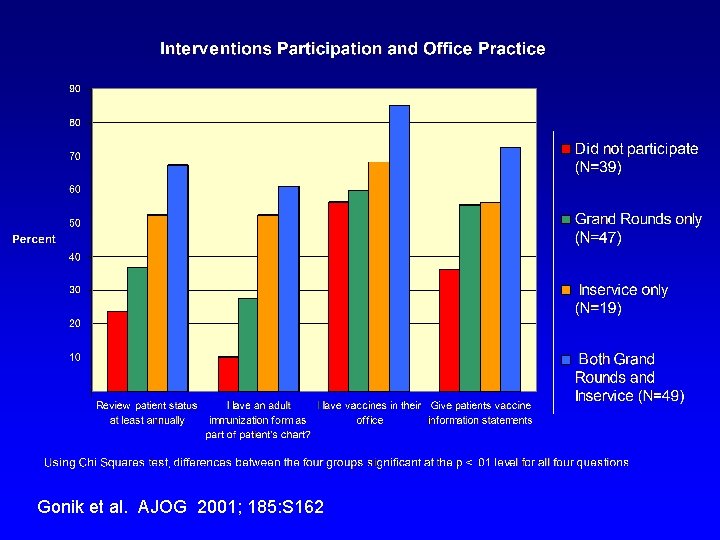
Gonik et al. AJOG 2001; 185: S 162
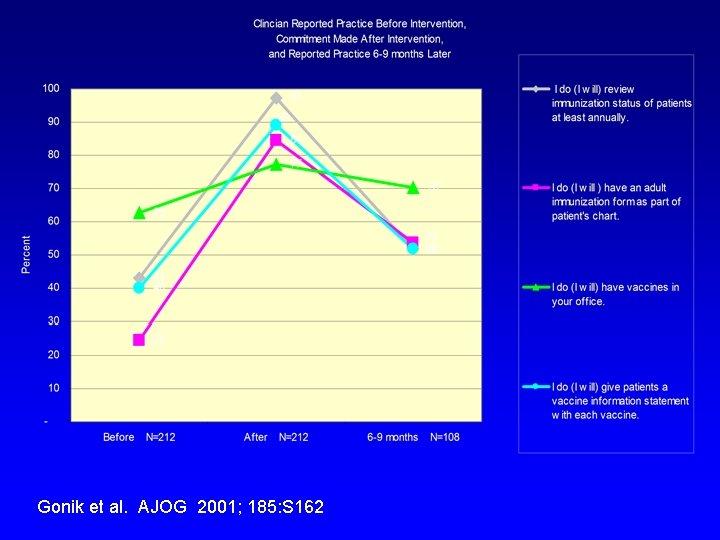
Gonik et al. AJOG 2001; 185: S 162

Take Home Messages • Educate HCP/Patient • • Vaccine self-benefits CDC recommendations Pregnancy-specific risks Passive antibody transfer to neonate • Encourage office vaccine program • Billing, coding • Storage, documentation • VIS
- Slides: 20