Influenza Sentinel Site Surveillance Training Addis Ababa Ethiopia

Influenza Sentinel Site Surveillance Training Addis Ababa, Ethiopia: February, 2010 Council of State and Territorial Epidemiologists United States Centers for Disease Control and Prevention

Part 1: Background and Objectives

Outline Background Purpose of the Training Guiding Principles Objectives of sentinel surveillance Topics covered in the guidance

Global Burden of Respiratory Infections Influenza: 3 – 5 million cases of severe illness 250, 000 – 500, 000 deaths Lower Respiratory Infections: Leading cause of death in low-income countries 3 rd leading cause worldwide

Need for Improved Surveillance Historically, influenza surveillance data collection: Virologic data for vaccine selection Limited epidemiologic data Lacked international standards Remaining gaps in understanding: Epidemiology, burden of disease Social factors, clinical risk factors Climatic factors

Global Surveillance for Influenza WHO Global Influenza Surveillance Network (GISN) International, laboratory-based (NICs) surveillance network Provides virus strain information to select seasonal vaccine • National Influenza Centres (NICs) Laboratories designated by national Ministries of Health and recognized by WHO to participate in GISN 131 NICs, in 102 countries (as of Feb. 4, 2010)

Pandemic H 1 N 1 (2009) Timely virologic and epidemiologic monitoring at national level: o Track progression of pandemic o Track impact of pandemic o Convey an accurate perspective on severity and risk to population o Prioritize country-specific risk groups for intervention resources
Relatedness of Seasonal and Influenza Pandemic Preparedness Seasonal Influenza Preparedness Pandemic Monitoring

Surveillance Needs During a Pandemic Is the situation changing? o Total amount of ILI, proportion of outpatient ILI due to influenza o Laboratory data to look at proportion of subtypes circulating in the community o Systematic sampling of viruses by age How severe is the disease and is it changing? o SARI hospitalizations o Deaths (and ICU admissions) from sentinel hospitals o Clinical picture of hospitalized cases Is the virus changing? o Drug resistance, drift and shift from laboratories o Comparison of viruses from less and more severe cases

Why is Monitoring the Severity/Virulence of a Pandemic Important? Inform how aggressively we think about interventions, for example: School closures Stopping public gatherings Vaccination priorities Antivirals priorities Mitigation, gain time to procure vaccine

Role of Sentinel Surveillance in Broader Monitoring Systems Reporting of qualitative indicators Aggregate reporting of laboratory-confirmed cases Aggregate reporting of syndromes ILI/SARI/Mortality Embedded sentinel sites to efficiently monitor o Virus characteristics o Severity and change in severity o Risk factors for severe illness o Hospital impact

Routine Value of Sentinel Surveillance Routinely produce useful epidemiologic and virologic data o Identify priority groups for intervention (severe outcomes) o Accurately characterize circulating viruses o Monitor seasonality to prepare for influenza season o Compare provinces, countries and regions o Estimate burden of disease Establish standards for surveillance reporting, collection and analysis Provide a platform for the study of influenza and other respiratory pathogens

Guiding Principles: Short and Long Term Value Integration into national systems Standard case definitions for comparisons (ILI, SARI) Efficiently monitor pandemic severity, high risk groups, and impact Ongoing surveillance of influenza and other pathogens High quality data in limited amounts
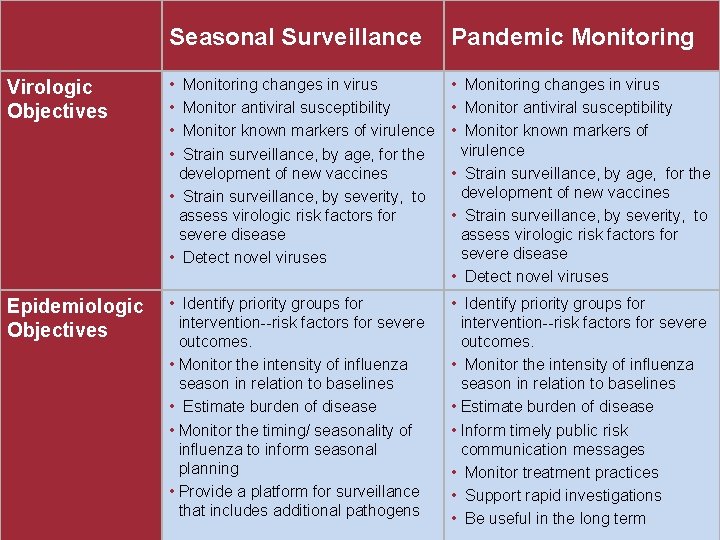
Seasonal Surveillance Pandemic Monitoring Virologic Objectives • Monitoring changes in virus • Monitor antiviral susceptibility • Monitor known markers of virulence • Strain surveillance, by age, for the development of new vaccines • Strain surveillance, by severity, to assess virologic risk factors for severe disease • Detect novel viruses • Monitoring changes in virus • Monitor antiviral susceptibility • Monitor known markers of virulence • Strain surveillance, by age, for the development of new vaccines • Strain surveillance, by severity, to assess virologic risk factors for severe disease • Detect novel viruses Epidemiologic Objectives • Identify priority groups for intervention--risk factors for severe outcomes. • Monitor the intensity of influenza season in relation to baselines • Estimate burden of disease • Monitor the timing/ seasonality of influenza to inform seasonal planning • Provide a platform for surveillance that includes additional pathogens • Identify priority groups for intervention--risk factors for severe outcomes. • Monitor the intensity of influenza season in relation to baselines • Estimate burden of disease • Inform timely public risk communication messages • Monitor treatment practices • Support rapid investigations • Be useful in the long term

Topics Covered in Training Toolkit to establish a few high quality sentinel IP/OP sites Criteria for selecting sentinel SARI and ILI sites Methods to avoid bias in the selection of cases for testing Epidemiologic data collection forms Procedures for laboratory specimens Report templates for weekly and annual data summaries Techniques for routine monitoring of the surveillance system Pandemic support functions

Part 2: Case Definitions
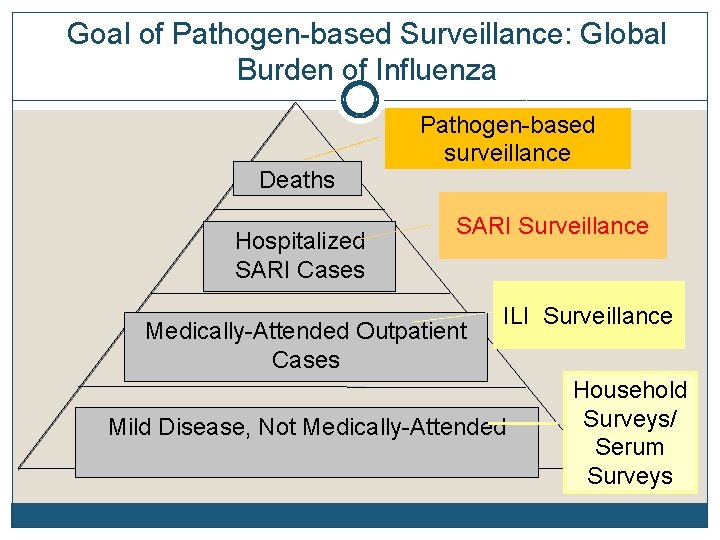
Goal of Pathogen-based Surveillance: Global Burden of Influenza Deaths Hospitalized SARI Cases Pathogen-based surveillance SARI Surveillance Medically-Attended Outpatient Cases ILI Surveillance Mild Disease, Not Medically-Attended Household Surveys/ Serum Surveys

SARI Surveillance Should be priority/minimum basic influenza surveillance for countries with limited resources Recommend adding this measure in countries with existing outpatient surveillance
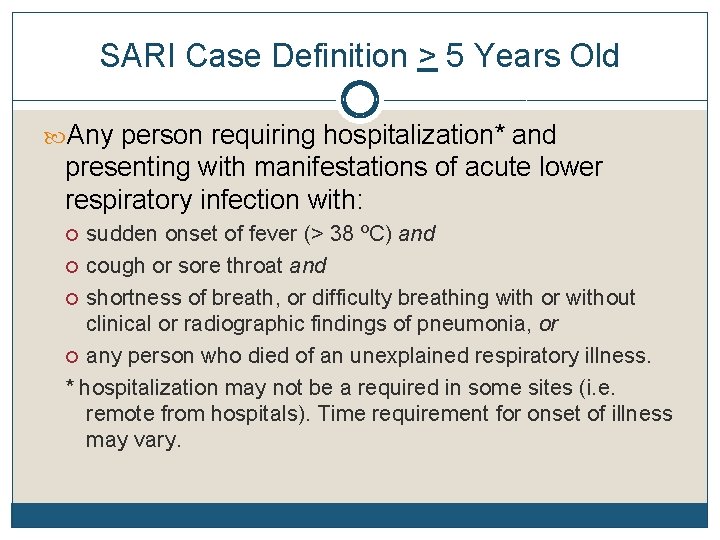
SARI Case Definition > 5 Years Old Any person requiring hospitalization* and presenting with manifestations of acute lower respiratory infection with: sudden onset of fever (> 38 ºC) and cough or sore throat and shortness of breath, or difficulty breathing with or without clinical or radiographic findings of pneumonia, or any person who died of an unexplained respiratory illness. * hospitalization may not be a required in some sites (i. e. remote from hospitals). Time requirement for onset of illness may vary.

SARI Case Definition < 5 Years Old IMCI case definition for pneumonia: Any child aged 2 months to 5 years with cough or difficulty breathing and: Breathing faster than 40 breaths / minute (ages 1 – 5 years) Breathing faster than 50 breaths / minute (ages 2 – 12 months) *Note that infants less than 2 months of age with fast breathing of 60 breaths or more per minute should be referred for serious bacterial infection.

SARI Case Definition < 5 Years Old IMCI case definition for severe pneumonia: Any child aged 2 months to 5 years with cough or difficulty breathing and any of the following: Unable to drink or breastfeed, or Vomits everything, or Convulsions, or Lethargic or unconscious, or Chest indrawing or stridor in a calm child

ILI Case Definition A person with: o sudden onset of fever >38°C, and o cough or sore throat in the absence of other diagnosis

Strengths Weaknesses ILI SARI • Less resource intensive as a case definition than SARI. • Captures hospitalized component of influenza. • Will capture cases with exacerbations of chronic conditions, not just those with pneumonia. • Fever requirement may • Will overlook those that do overlook persons without not present with fever, such as elderly and • Will overlook those that do immune-compromised. not seek care at a hospital. • Not ideal if being used to • Fever requirement makes it monitor viral respiratory more specific, less sensitive pathogens other than influenza.

Considerations When Using the SARI Case Definition Fever requirement: may exclude elderly and immunocompromised Option: do not include fever, or require a measured fever May detect non-influenza viral pathogens Will increase resource demands Should record presence or absence of measured fever on the swab form for comparison to other countries Ensure consistent application of case definitions across all sites

Experiences with SARI Surveillance Early data from Member States in 5 WHO regions suggests routine surveillance for SARI will…. …achieve virologic objectives of seasonal influenza surveillance (pct. positive similar to ILI during influenza season) …provide epidemiologic and virologic data on severe influenza infections …provide a basis for monitoring severe respiratory disease during a pandemic …serve as a platform for assessing burden of multiple viral respiratory pathogens

Prioritizing the Focus of the Surveillance System Simple Model Budget: Low, no existing surveillance Surveillance for: SARI as a minimum standard Epidemiologic data collection (i. e. , denominator data = total number of SARI or ILI seen) Virologic testing Small number of well-run sentinel sites preferred to large number of poorly-run sites.

Prioritizing the Focus of the Surveillance System Intermediate Model Budget: Medium Surveillance for: SARI and outpatient ILI Virologic testing Epidemiologic data collection (i. e. denominator data)

Prioritizing the Focus of the Surveillance System Advanced Model Budget: High Surveillance for: SARI and ILI surveillance Virologic testing Epidemiologic data collection (i. e. denominator data) Multiple pathogens, possibly reduce fever requirement in inpatient setting as well

Part 3: Mechanics of Sentinel Surveillance

Outline Sentinel site selection Case sampling strategies Epidemiologic data collection and forms

Selection of Sentinel Sites

What is sentinel surveillance? One or more designated health care facilities that routinely collect epidemiologic information and laboratory specimens from patients presenting an illness consistent with a specified case definition The system provides an efficient way to obtain high-quality data on relatively common conditions from a manageable number of locations

Why Sentinel Surveillance? For monitoring/ identification of: Identify target groups: For antiviral use and timing That might have greatest impact on transmission Strains to include in vaccine Appropriate management practices Hospital staffing and procurement needs

Key Attributes of Sentinel Sites Efficient data collection High quality data Limited number of well-chosen sites “A few high quality data are better than a lot of bad data”

The Efficiency of Sentinel Sites Systematic testing of inpatient and outpatient (SARI and ILI) cases Representation of viruses by age and severity Efficient collection of demographic and epidemiologic data that is linked to virologic data Can yield measures of disease burden Valuable seasonally as well as in a pandemic Can support efforts to monitor surge on hospitals
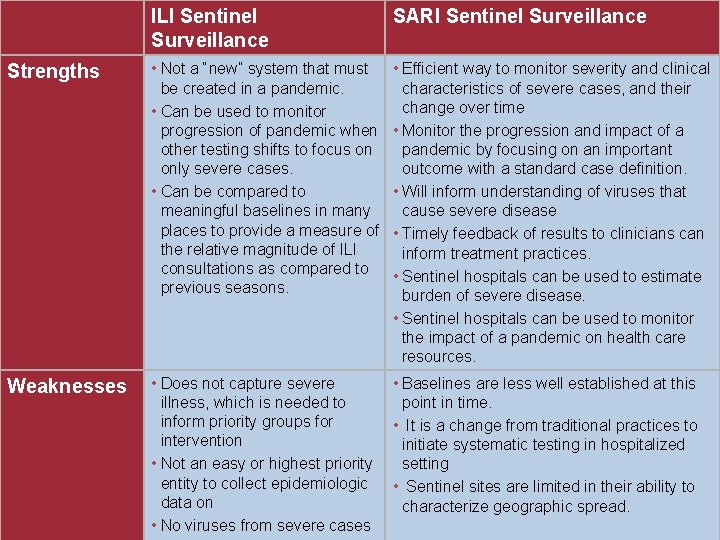
ILI Sentinel Surveillance SARI Sentinel Surveillance Strengths • Not a “new” system that must be created in a pandemic. • Can be used to monitor progression of pandemic when other testing shifts to focus on only severe cases. • Can be compared to meaningful baselines in many places to provide a measure of the relative magnitude of ILI consultations as compared to previous seasons. • Efficient way to monitor severity and clinical characteristics of severe cases, and their change over time • Monitor the progression and impact of a pandemic by focusing on an important outcome with a standard case definition. • Will inform understanding of viruses that cause severe disease • Timely feedback of results to clinicians can inform treatment practices. • Sentinel hospitals can be used to estimate burden of severe disease. • Sentinel hospitals can be used to monitor the impact of a pandemic on health care resources. Weaknesses • Does not capture severe • Baselines are less well established at this illness, which is needed to point in time. inform priority groups for • It is a change from traditional practices to intervention initiate systematic testing in hospitalized • Not an easy or highest priority setting entity to collect epidemiologic • Sentinel sites are limited in their ability to data on characterize geographic spread. • No viruses from severe cases

Ideal Characteristics of Sentinel Sites Feasibility People that are motivated to run the system Political willingness for a site to participate Sufficient staffing and laboratory capacity to maintain sampling and testing during healthcare surge Efficient data management and transmission capability

Ideal Characteristics of Sentinel Sites Patient representativeness All ages Wide range of medical conditions For SARI surveillance, general or community hospitals are preferable to specialty care hospitals For ILI surveillance, general outpatient clinics are often appropriate

Ideal Characteristics of Sentinel Sites Quantifiable population denominator o Facilitates estimates of burden of disease o Requires understanding of population served by the sentinel site Large referral hospitals may underestimate community incidence and require extra staff Difficult to test a large proportion of all SARI cases identified If calculating rates, may be difficult to assess the total population served by the surveillance site

Placement of Sentinel Sites Population representativeness Ethnicity Socioeconomics Climatic representativeness Climate affects virus activity (transmission and viability in environment)
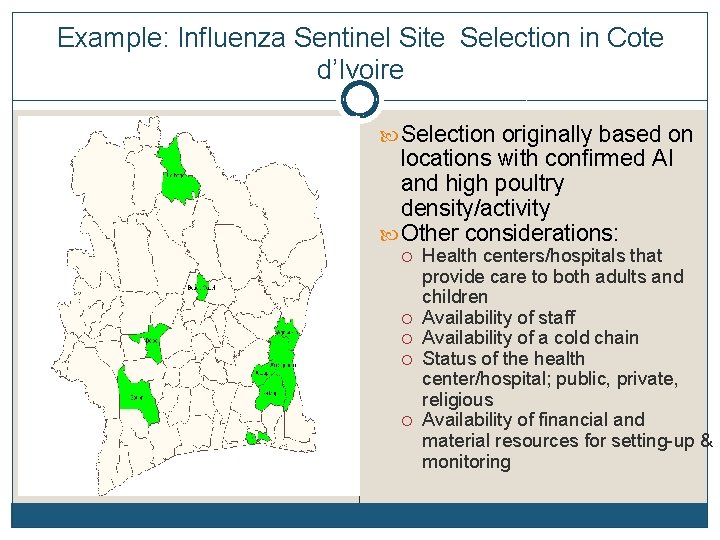
Example: Influenza Sentinel Site Selection in Cote d’Ivoire Selection originally based on locations with confirmed AI and high poultry density/activity Other considerations: Health centers/hospitals that provide care to both adults and children Availability of staff Availability of a cold chain Status of the health center/hospital; public, private, religious Availability of financial and material resources for setting-up & monitoring
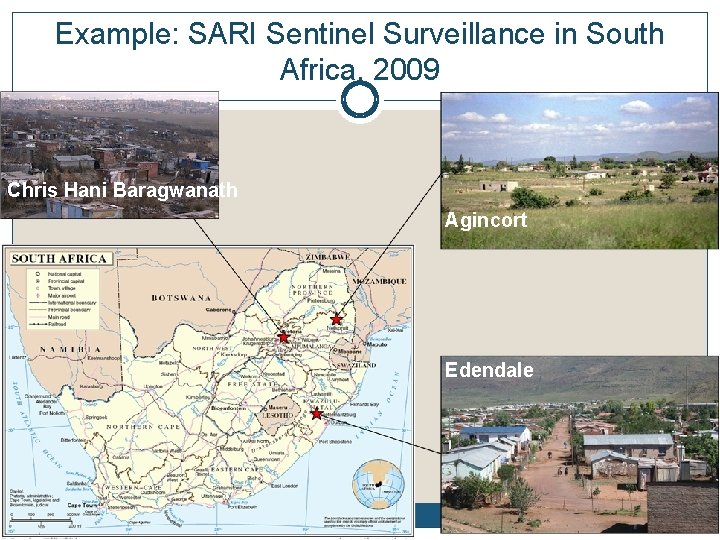
Example: SARI Sentinel Surveillance in South Example: SARI sentinel sites in South Africa, 2009 Chris Hani Baragwanath Agincort Edendale
Example: Sentinel Site Selection in Ukraine

Integration into National Clinical Reporting Systems Ideally, sentinel sites could be integrated: Adopt standard case definitions Establish smaller number of sentinel sites within the broader universal reporting system Assure high-quality data at these sites through more intensive training and oversight Systematic laboratory testing only at sentinels Broader clinical reporting provides valuable indicator of geographic spread within a country
Integration into National Clinical Reporting Systems ILI/SARI Syndrome Reporting ILI/SARI Case-based epi data collection, laboratory testing ILI/SARI Syndrome Reporting ILI/SARI Case-based epi data collection, laboratory testing

Selection of SARI Cases It is preferable to collect data and specimens from all or most SARI cases from a few facilities rather than a small sample of SARI cases from multiple facilities Logistically feasible Less bias If not possible, an unbiased sampling protocol should be established

Selection of ILI Cases Large number of cases at outpatient sentinel sites is likely Total number of ILI cases seen is very important to collect Collect specimens and case-based epidemiologic data from only a sample of ILI cases Select cases for laboratory testing in as unbiased a manner as logistically possible

Sampling Methods for Respiratory Specimen and Casebased Epidemiologic Data Collection Select all cases for testing Select every xth case for testing Select all cases on certain days of the week for testing Select the first x cases on a certain day of the week

Selecting All Cases For Testing Minimizes bias Requires the most resources May not be feasible in many settings More feasible for sentinel surveillance focused on hospitalized cases (SARI)

Selecting Every Xth Case For Testing Less likely to be biased than methods that only select cases on the same day or at the same time Fewer resources than sampling all cases Frequently used for hospital-based (SARI) surveillance

Selecting All Cases On Certain Days Of The Week For Testing Does introduces bias o Patient volume and mix is not the same every day of the week o Day(s) of the week should be alternated Reduces logistical challenges of transport to laboratory

Selecting the First X Cases On a Certain Day of the Week Can introduce bias by day-of-week or time-of-day Commonly used for ILI surveillance where General Practitioners (GP’s) require convenience sampling models Consider local health seeking behaviors Not desirable for burden estimation of for SARI surveillance
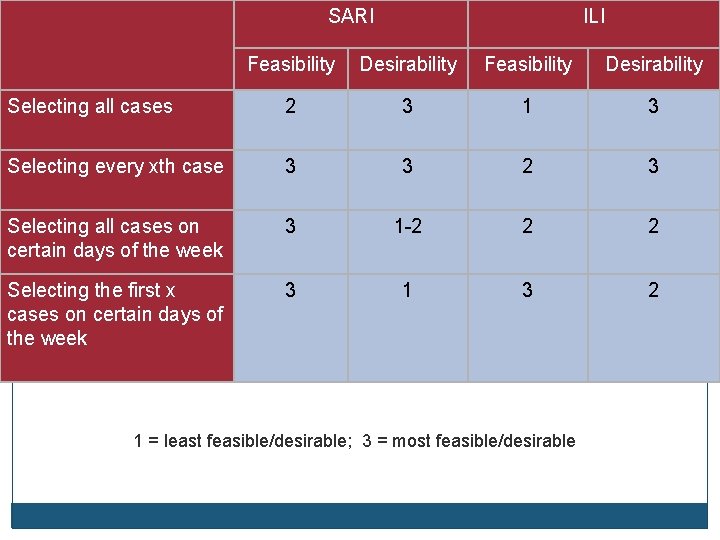
SARI ILI Feasibility Desirability Selecting all cases 2 3 1 3 Selecting every xth case 3 3 2 3 Selecting all cases on certain days of the week 3 1 -2 2 2 Selecting the first x cases on certain days of the week 3 1 3 2 1 = least feasible/desirable; 3 = most feasible/desirable

Epidemiologic Data Collection

Data Collection Tools Forms filled out by designated sentinel site staff At a minimum, collect data 4 weeks before and after typical peak months Assign each patient a unique identification number to link epidemiologic and laboratory data Recorded on data collection form, swab forms, and laboratory specimen

Minimal Data Elements Unique identification number Patient demographics Minimal clinical information including relevant dates of onset and clinical symptoms Pre-existing medical conditions (for SARI only) Vaccine and antiviral use

Case-Based Data Collection Forms: SARI Swab Form For all SARI cases tested for influenza o Should be completed as soon as possible after identification Send one copy to laboratory with the specimen, one to the national surveillance centre, keep original at the sentinel site


Case-Based Data Collection Forms: Outpatient Swab Form For all ILI cases tested for influenza Complete as soon as possible after selection for laboratory testing Send one copy to laboratory with the specimen, one to the national surveillance centre, keep original at the sentinel site


SARI Weekly Aggregate Data Form ID number of sentinel site, and focal point New SARI cases during week New inpatients during week Number of SARI cases selected for influenza testing Number of SARI deaths during the week Ages: 0 -4, 5 -14, 15 -29, 30 -64, 65+, Total
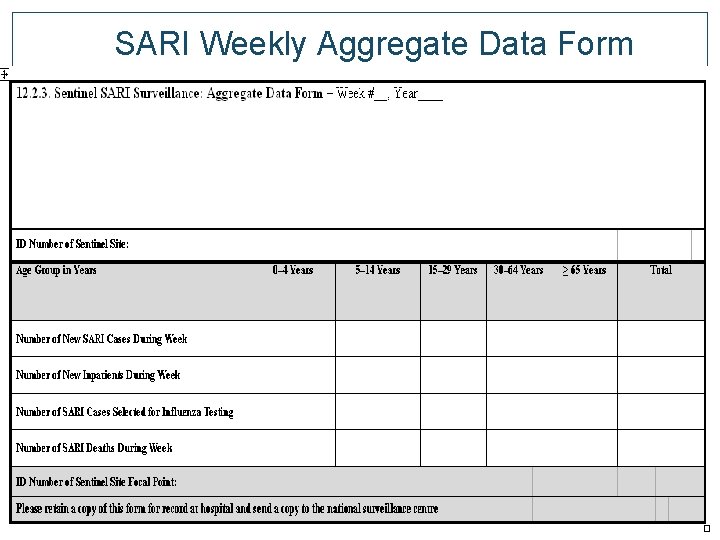
SARI Weekly Aggregate Data Form

ILI Weekly Aggregate Data Form ID number of sentinel site, and focal point Day(s) of week surveillance was undertaken New ILI cases during week New outpatients during week Number of ILI cases selected for influenza testing Ages: 0 -4, 5 -14, 15 -29, 30 -64, 65+, Total

Unique Identification Numbers Important for linkage between laboratory and epidemiologic information Numbers should be standardized throughout the country Assigned when SARI or Outpatient Swab forms are filled out Same number used on any forms or specimens for each patient

Identification Number Example 001 / 09 / 1 / 0001 Sentinel Site Year SARI or ILI Example: 1 = SARI 2 = ILI Case Number

Part 4: Roles and Responsibilities Within the Sentinel Surveillance System
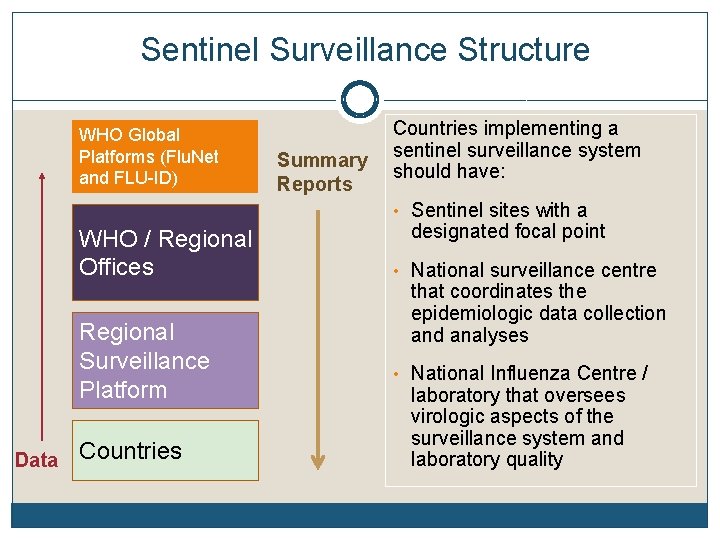
Sentinel Surveillance Structure WHO Global Platforms (Flu. Net and FLU-ID) Summary Reports Countries implementing a sentinel surveillance system should have: • Sentinel sites with a WHO / Regional Offices Regional Surveillance Platform Data Countries designated focal point • National surveillance centre that coordinates the epidemiologic data collection and analyses • National Influenza Centre / laboratory that oversees virologic aspects of the surveillance system and laboratory quality

Sentinel Site Focal Point Ensures that staff at the site carry out the following: Manage and transmit Adhere to case definitions epidemiologic data Adhere to unbiased Track daily number of systematic sampling SARI and ILI cases, and strategies number selected for Collect respiratory laboratory testing specimens Assure timeliness of data reporting and specimen Package, store, and collection and transport specimens Report SARI laboratory Assign unique ID number to test results to the clinician specimens and forms All data forms are filled out completely and accurately

National Surveillance Centre Organizational entity assigned to coordinate influenza surveillance in a country Should have a national surveillance focal point who is responsible for implementation and coordination of the national influenza sentinel site surveillance system May be located in different agencies or institutes in different countries

National Surveillance Centre Focal Point Responsibilities: Assist MOH with selection of sentinel sites Assist with decisions about ILI and SARI sampling and epidemiologic data collection Assure that sites have data collection forms and reporting mechanisms Assure that the data is analyzed correctly Disseminate the weekly and annual flu report Report data into the regional and global platforms Routinely monitor the sentinel surveillance system

National Influenza Centre (NIC) Recognized by WHO Participates in regional influenza network and GISN An up-to-date list of NICS can be found at: http: //www. who. int/csr/disease/influenza/centres/en/in dex. html

NIC Routine Responsibilities Provide technical support and guidance to sentinel sites Receive, register, and store specimens from SARI and ILI cases Perform analyses on seasonal viruses Perform subtyping of non-seasonal viruses, minimally Influenza A(H 5 N 1) Pandemic influenza A(H 1 N 1) 2009 Archive and store original specimens at least one year Share samples with WHO CCs Conduct antiviral susceptibility testing (where possible) Maintain linkage between site and laboratory ID numbers

NIC Responsibilities: Data Analysis and Reporting Communicate results of confirmatory tests Consolidate and analyze national laboratory data Report weekly national surveillance data into regional and global surveillance platforms Develop national diagnostic standards and assays, provide training, and organize quality assurance programs (where possible)
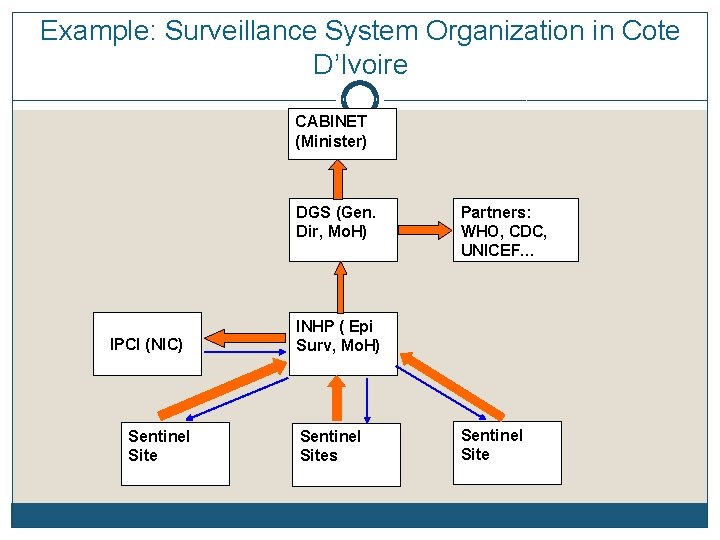
Example: Surveillance System Organization in Cote D’Ivoire CABINET (Minister) DGS (Gen. Dir, Mo. H) IPCI (NIC) Sentinel Site Partners: WHO, CDC, UNICEF… INHP ( Epi Surv, Mo. H) Sentinel Sites Sentinel Site
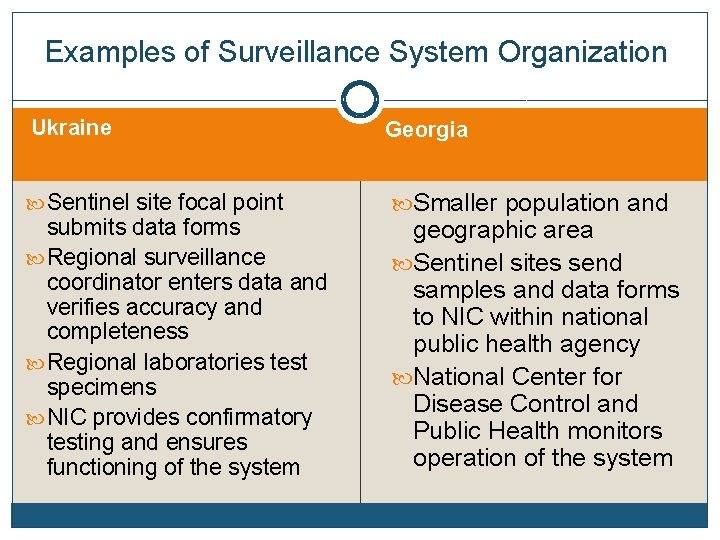
Examples of Surveillance System Organization Ukraine Georgia Sentinel site focal point Smaller population and submits data forms Regional surveillance coordinator enters data and verifies accuracy and completeness Regional laboratories test specimens NIC provides confirmatory testing and ensures functioning of the system geographic area Sentinel sites send samples and data forms to NIC within national public health agency National Center for Disease Control and Public Health monitors operation of the system

Notification of Novel Influenza Viruses through the IHR Mechanism Immediate notification to WHO through the IHR mechanism: Human infection with influenza A other than seasonal subtypes Any combination of H 2, or H 4 -H 16 with any of the N 1 -N 9 subtypes Includes avian influenza subtypes A(H 5 N 1), A(H 9 N 2), and A(H 7 N 7) Human infections of Influenza A viruses that grow poorly in cell culture or react poorly with standard WHO reagents in HAI assays May have same subtype as seasonal viruses, but be of animal origin (such as pandemic (H 1 N 1) 2009)

Participating in WHO External Quality Assessment Project for the Detection of Influenza A Viruses by PCR To improve global laboratory capacity for influenza diagnosis To monitor quality and standards of performance of NICs around the world Requirements: Laboratories test a panel of simulated H 1, H 3, H 5 specimens twice per year Each laboratory receives performance report

Part 5: Data Reports and Analysis

Outline Measures of Burden Weekly Reports Annual Reports Reporting Outside the Country/Publications

Measures of Burden Incidence rate o o Number of influenza cases per 100, 000 population per time unit Allows for estimate of age-specific burden of disease International standard for reporting Incidence of influenza consultations also valuable to monitor seasonal intensity over time Proportion of SARI and ILI at sentinel sites that is caused by laboratory-confirmed influenza o Minimal standard

Example: The Netherlands General practitioners Collect specimens and data from a random sample of persons meeting ILI case definition Each maintains list of 2, 500 patients, which can be used for denominator data Incidence rate Calculated per 100, 000 per week

Minimal Analyses and Reports Generate weekly, at least 4 weeks prior and following typical peak months Disseminate to all relevant governmental partners, sentinel sites, and the public Virologic surveillance conducted outside flu season may be reported less frequently

Weekly SARI Data During the previous week: Number of new cases at each sentinel site Number of total new hospital admissions at each sentinel site Number of SARI cases selected for influenza testing % testing positive By influenza type and subtype Number of inpatient deaths due to SARI Number of sentinel SARI sites reporting Results for all sites stratified by the age groups: 0 -4, 5 -14, 15 -29, 30 -64, 65+
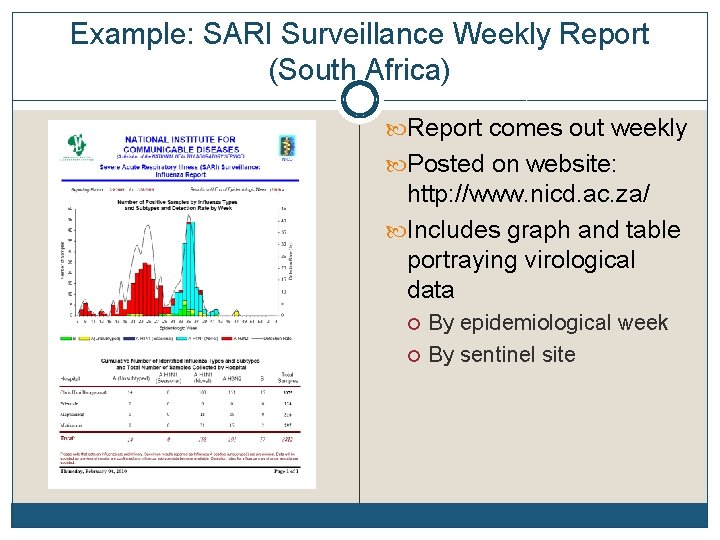
Example: SARI Surveillance Weekly Report (South Africa) Report comes out weekly Posted on website: http: //www. nicd. ac. za/ Includes graph and table portraying virological data By epidemiological week By sentinel site

Weekly Outpatient Data During the previous week: Number of new ILI cases reported Number of total outpatients seen at ILI sentinel site Number of ILI cases selected for influenza testing Percent of tested ILI positive for influenza By influenza type and subtype

Additional Data to Report If available, report: Weekly consultation rates for ILI, or SARI Antiviral resistance testing results http: //www. cdc. gov/flu/weekly/ • Data on geographical spread (map) • Level of detail will depend on number and location of sentinel sites

Annual Analyses and Reports

Annual Analyses SARI, ILI, and confirmed influenza cases by age and month Underlying medical conditions of SARI patients Number and percent of positive influenza cases who received vaccination in the current season. Number and percent of positive influenza cases exposed to antivirals 14 days before onset of symptoms Incidence rates
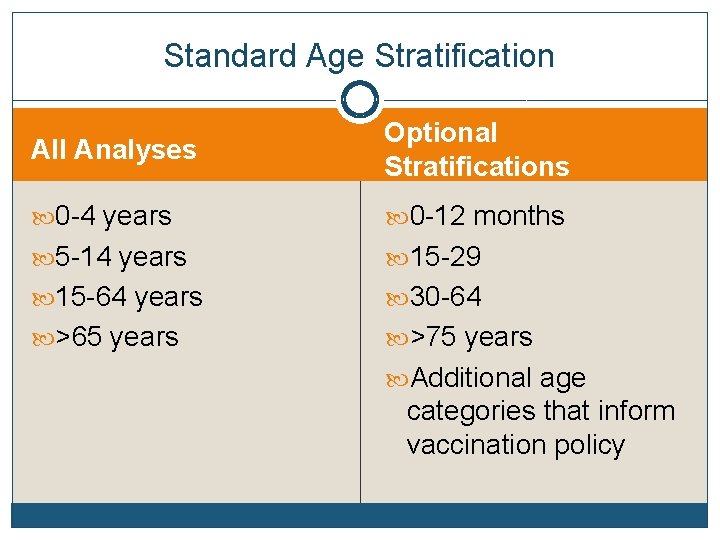
Standard Age Stratification All Analyses Optional Stratifications 0 -4 years 0 -12 months 5 -14 years 15 -29 15 -64 years 30 -64 >65 years >75 years Additional age categories that inform vaccination policy

Assessment of Seasonality Establish the baseline rate of influenza in a region/ country Inform the timing of vaccination and treatment

Example: Using Data in the UK Yearly guidance issued to doctors about when antiviral agents should be prescribed Recommendations are based off of surveillance data and are triggered when: o GP consultation rates rise above 30 consultations per 100, 000 population o Community-based influenza surveillance is above baseline

Reporting and Publication

Regional and Global Reporting for Seasonal Influenza Flu Season (4 weeks before and after typical peak influenza months): epidemiologic and virologic data should be reported regionally Non-flu season: only virologic data Can be used for a regularly published bulletin

Example: Euro. Flu Weekly Electronic Bulletin Detailed epidemiologic and virologic data for each country and Europe as a whole. Presented with: Maps Tables Graphs Pie charts National level reporting mechanism is important

Example: Euro. Flu as a Basis for Synthesizing Regional Information

Reports for Posting/Publication Public posting of routine analysis and data should be national surveillance system goal. Peer-reviewed publication is also important. May include: Routine analyses Special studies Informs international influenza control efforts Creates advocates for influenza interventions

Part 6: System Monitoring

Outline Suggested Quality Indicators Timeliness Completeness Validity

Timeliness Indicators Expected dates of data reporting from sentinel site to next administrative level compared to actual dates Some examples are time elapsed from: Specimen collection to arrival at the laboratory Laboratory receipt of specimens to laboratory processing and testing Laboratory confirmation results to inclusion of results in Weekly Influenza Reports Laboratory confirmation results for individual SARI cases to notification of the sentinel site focal point Laboratory results for individual SARI cases by the sentinel site focal point to notification of the patient’s doctor

Quantification of Timeliness Indicators Percent of time site achieves targets for specific time intervals e. g. 90% of specimens received at the laboratory within 48 hours of specimen collection Percent of samples tested within a target time frame e. g. 90% of tested specimens have results within 1 week Percent of sites achieving time targets or time lag averages e. g. 90% of sentinel sites package and transport 90% of specimens within 48 hours of collection Percent of time that doctors receive laboratory results for SARI cases within 48 hours of confirmatory test completion

Completeness Percentage of swab forms received from each site with complete data Percentage of total expected swab forms that are received Percentage of total expected cases that have specimens submitted to the laboratory

Validity: Data Audits Ensures Validity by determining the following: o Case definitions are understood and adhered to o Cases are counted appropriately (no duplicates) o Reported cases meet the case definition o Uniform sampling procedures are being followed o Laboratory equipment and staff are sufficient o Data values are being recorded correctly (can be compared to gold-standard of chart-review) o Sentinel sites and doctors are receiving timely feedback and SARI test results

Validity: Compare to Expected Results Number of cases reported each month at site Number of specimens submitted each month at each site Percent of specimens positive for influenza each month at each site Number and percent of SARI and ILI cases tested Does something look out of the ordinary?

Comprehensive System Evaluations Conduct in addition to routine monitoring When to conduct At start-up At regular intervals If aberrations in expected results are observed

Part 7: Additional Uses of the System and Surveillance Data

Outline Estimating disease burden Establishing baselines and thresholds Pandemic detection, planning, and monitoring

Overview Make decisions about health resource allocation Identification of risk groups for targeted intervention Monitoring of the impact of intervention programs, including vaccination

Estimating Disease Burden

Estimates of Disease Burden Disease burden = amount and impact of influenza in the population represented by a sentinel site(s) Basic measures o o Incidence rates of hospitalized SARI caused by laboratoryconfirmed influenza Estimates of excess mortality Direct costs of medical care and treatment Indirect costs due to lost time from employment or costs incurred by family members of a sick person
Importance of Disease Burden Assist policy makers Decisions about allocation of resources by putting influenza in perspective relative to other diseases Judge cost-benefit ratio of interventions Stimulate or target future surveillance activities

Estimating Catchment Area Sources of population denominator data: Patient lists from sentinel physicians Well circumscribed municipalities of medium size where sentinel sites capture a large portion of admissions/outpatients Health Utilization Survey can be used if: Well-defined community Site does not receive a large proportion of cases from distant areas See guidance document for more details

Establishing Baselines and Thresholds

Baseline Incidence Baseline = level of influenza activity typically seen outside the seasonal epidemic Rise above baseline indicates start of seasonal epidemic Serves as an objective threshold for public health action Occasionally influenza activity does not exceed the baseline threshold

Pandemic Detection, Planning, and Monitoring

Sentinel Surveillance Supports Public Health Response Establishes infrastructure for: Prioritizing investigation of severe respiratory illness cases Creating clinical specimen collection and transport network Building expertise in confirmatory testing for human and novel influenza viruses Strengthening relationships between laboratory, epidemiologic, and clinical personnel

Sentinel Systems and Early Warning Sentinel systems are generally not adequate for detection of unusual events or outbreaks early in their course

The Role of the Sentinel System in Monitoring a Pandemic Monitoring the course of a pandemic will use existing routine influenza surveillance data describing: Location of the virus Trend in cases Severity of pandemic Changes in virulence

Part 8: Laboratory Specimen Processing

Outline Specimen collection Specimen storage and packaging Specimen testing Shipment of specimens

Specimen Collection

Collecting Respiratory Specimens For direct detection of antigens or nucleic acids, and for virus isolation (culture) o o o Take no more than 7 days after onset of symptoms Ideally take within 3 days Preferably before antiviral therapy Record time from illness onset to specimen collection Give patient verbal explanation of reason for specimen collection and how it will be collected

Specimen Tube Labeling Patient unique identifier Specimen date Type of specimen in the tube Hazard label

Upper Respiratory Specimens Best for virus isolation and PCR Nasopharyngeal (NP) swab Nasopharyngeal aspirates or washes Nasal wash Alternatively, collect both Nasal swab Throat swab Combine in single vial of VTM

Materials Required Personal protective equipment Swabs Tongue depressors Plastic vials, such as cryovials, containing 2 -3 ml of virus transport medium (VTM) stored at 4°C (supplied by the NIC) Serum collection: collection tubes, alcohol, gauze, non-heparin treated needles Alcohol and/or bleach Packaging materials for transport in country

Specimen Storage and Packaging

Specimen Storage Place in VTM immediately Store at 4°C, up to 24 hours For immunoflourescence: Refrigerate and process within 1 -2 hours For virus isolation: Refrigerate and inoculate into cell cultures as soon as possible If not processed within 48 -72 hours, freeze at or below -70°C

Specimen Storage Specimens may be divided into aliquots Freeze-thaw cycles should be minimized Do not store in a household freezer (-20°C) with a “defrost” cycle Keeping sample on ice up to a week is better than allowing freeze-thaw cycles Blood may be stored at room temperature overnight or incubated at 56°C for 30 minutes Remove serum and store at 4°C up to a week or put into long term storage at -20°C

Preparation of VTM Add 10 g veal infusion broth and 2 g bovine albumin fraction V to 400 ml sterile distilled water Add 0. 8 ml gentamicin sulfate solution (50 mg/ml) and 3. 2 ml amphotericin B (250 μg/ml) Sterilize by filtration Store unopened for up to one year

Packaging 3 layers of packaging Compliance with P 650 requirements for infectious substances in UN 3373 category B 1 st layer Watertight specimen tube (no more than 500 m. L) Absorbent material 2 nd layer Watertight container (ziploc or hard plastic) 3 rd layer Rigid outer packaging

Packaging May need to include ice packs or dry ice Example: P 650 packaging system in combination with Safetybag

Specimen Testing

Specimen Processing BSL-2 laboratory Class II microbiological safety cabinet for procedures that may give rise to infectious aerosols Seasonal influenza: disposable gloves and gown Suspect avian influenza / severe respiratory pathogens: BSL-2 laboratory with BSL-3 practices, or BSL-3 laboratory
Non-molecular Influenza Detection and Subtyping Viral isolation Eggs Cell culture Followed by HAI assay using WHO reagent kit Immunofluorescence assay 1918 influenza virions. CDC/ Dr. Terrence Tumpey Initial screen to select specimens for viral isolation Low sensitivity Requires living cells ELISA Low sensitivity

Influenza Detection and Subtyping by RT-PCR Higher sensitivity Increased biosafety Uses low amounts of virus Viral isolation can be performed on known subtypes Cost effective Rapid Can provide results in 24 hours

RT-PCR is Recommended for Confirmatory Testing and to Select Specimens for Virus Isolation Real-time RT-PCR preferred Assays can be developed locally or obtained commercially Primers and probes must be validated at least once a year Can be performed in BSL-2 laboratory o Reduces chances that isolation of a novel virus (a BSL-3 practice) will be attempted in BSL-2 conditions
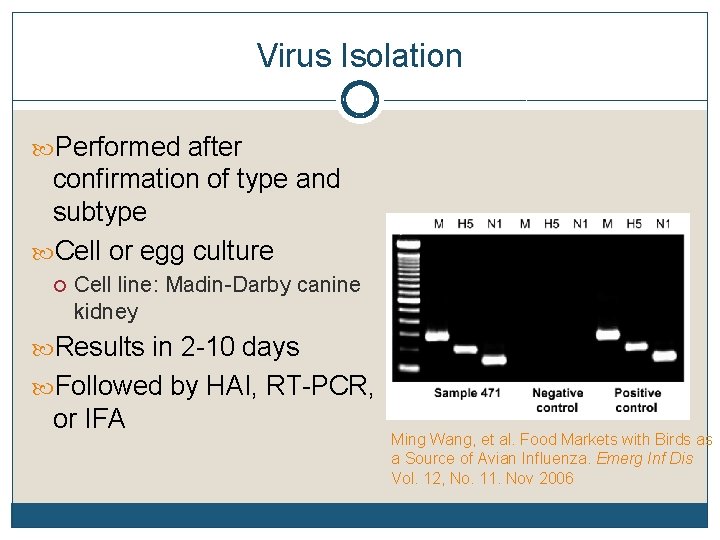
Virus Isolation Performed after confirmation of type and subtype Cell or egg culture Cell line: Madin-Darby canine kidney Results in 2 -10 days Followed by HAI, RT-PCR, or IFA Ming Wang, et al. Food Markets with Birds as a Source of Avian Influenza. Emerg Inf Dis Vol. 12, No. 11. Nov 2006

Principles for Viral Isolation BSL-2 laboratories test for: Seasonal influenza A(H 1 N 1) Seasonal influenza A(H 3 N 2) Influenza B Other respiratory viruses (RSV, adenovirus) BSL-3 laboratories test for: Influenza A, subtype not identified Influenza A, subtype identified as not being Human seasonal A(H 1 N 1) or Human seasonal A(H 3 N 2)
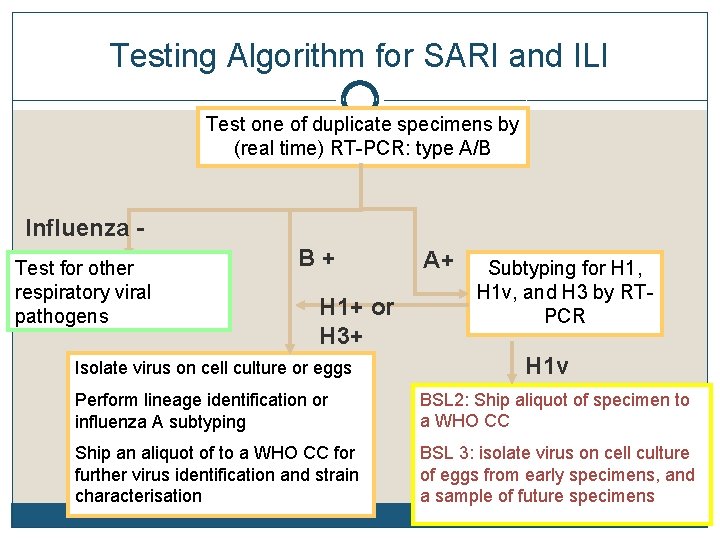
Testing Algorithm for SARI and ILI Test one of duplicate specimens by (real time) RT-PCR: type A/B Influenza Test for other respiratory viral pathogens B+ H 1+ or H 3+ Isolate virus on cell culture or eggs A+ Subtyping for H 1, H 1 v, and H 3 by RTPCR H 1 v Perform lineage identification or influenza A subtyping BSL 2: Ship aliquot of specimen to a WHO CC Ship an aliquot of to a WHO CC for further virus identification and strain characterisation BSL 3: isolate virus on cell culture of eggs from early specimens, and a sample of future specimens

Seasonal Influenza Serology Testing Use reagents from WHO CDC test kit Conduct HAI assay on paired sera (if available) Positive: 4 -fold rise in antibody titer Collect convalescent serum 14 days – 4 weeks after symptom onset Other considerations Important for retrospective analysis of outbreaks

Avian influenza A(H 5 N 1) Serology Testing Virus neutralization assay BSL-3 laboratory with BSL-3 standards Positive: 4 -fold or greater rise in neutralization antibody titer Paired serum samples Other considerations Single serum samples can be useful for detecting antibodies Used for: Confirmation when respiratory specimen testing and virus isolation are inconclusive Retrospective analysis of outbreaks

WHO kits and protocols for real time RT-PCR detection of pandemic (H 1 N 1) 2009 In response to the pandemic, WHO CC at CDC has developed and distributed kits for real-time RTPCR detection of pandemic (H 1 N 1) 2009.

Shipment of Specimens

Shipment of Specimens to WHO Collaborating Center Logistical support: WHO Global Shipment Project Covers 2 -3 shipments of seasonal viruses per season Covers novel viruses as necessary Uses World Courier Shipment initiated by completing booking form Requires export permit (standing permit recommended)

Shipment Regulations International regulations for air shipment Technical Instructions for the Safe Transport of Dangerous Goods by Air Dangerous Goods Regulations (DGR) may add further restrictions Shipping personnel IATA-certified training available to NIC staff Certificate valid for 12 months Encouraged, but not required for personnel shipping seasonal influenza pathogens

Shipping Specimens on Ice For fresh isolates Use ice packs Use insulated vials Delivery within 48 hours or refrigerate at 4°C For frozen isolates Ship on dry ice Include swab form and itemized list of contents between secondary and outer packaging

Specimens for WHO CC or WHO H 5 Reference Laboratories Forward to WHO CC for virus strain characterization and vaccine strain selection: Representative seasonal viruses from SARI and ILI cases A(H 3 N 2) A(H 1 N 1) Influenza B At beginning, peak and near end of season Dispatch immediately to WHO CC and/or WHO H 5 Reference Laboratory: The first specimens found to be positive for H 1 N 1 v Low-reacting viruses Specimens with influenza A viruses for which subtype was not identified Specimens with avian influenza or other novel viruses Include swab form and list of contents
- Slides: 146