Influence of the hydrocarbon rest upon reactivity Influence

Influence of the hydrocarbon rest upon reactivity
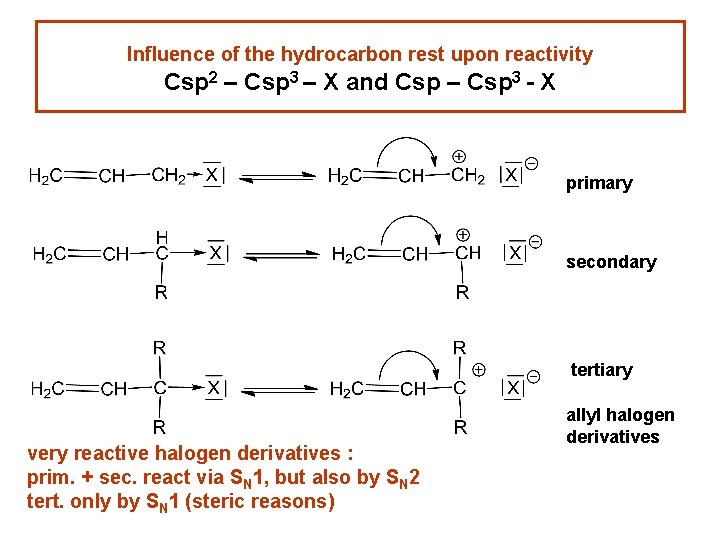
Influence of the hydrocarbon rest upon reactivity Csp 2 – Csp 3 – X and Csp – Csp 3 - X primary secondary tertiary very reactive halogen derivatives : prim. + sec. react via SN 1, but also by SN 2 tert. only by SN 1 (steric reasons) allyl halogen derivatives
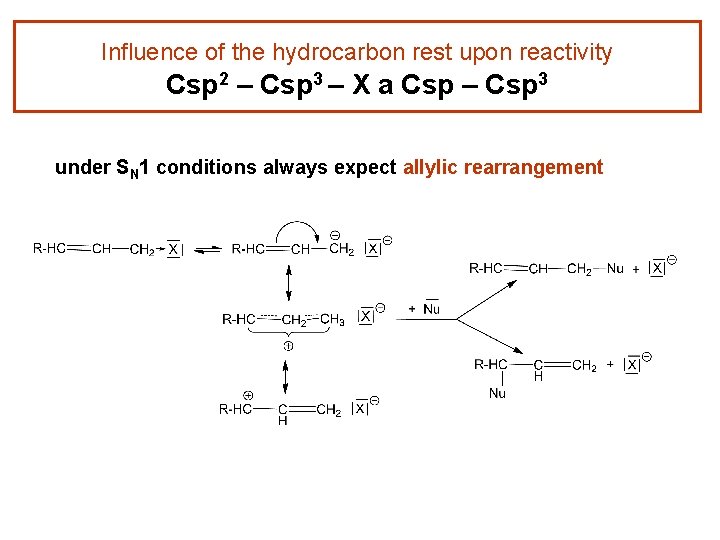
Influence of the hydrocarbon rest upon reactivity Csp 2 – Csp 3 – X a Csp – Csp 3 under SN 1 conditions always expect allylic rearrangement
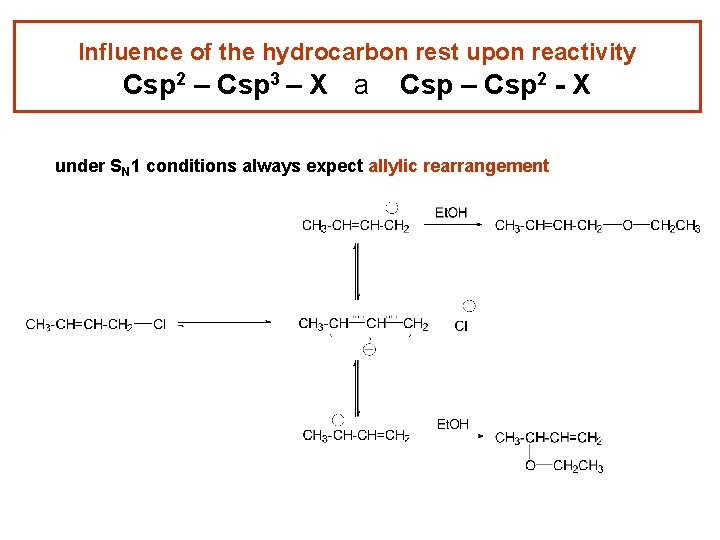
Influence of the hydrocarbon rest upon reactivity Csp 2 – Csp 3 – X a Csp – Csp 2 - X under SN 1 conditions always expect allylic rearrangement
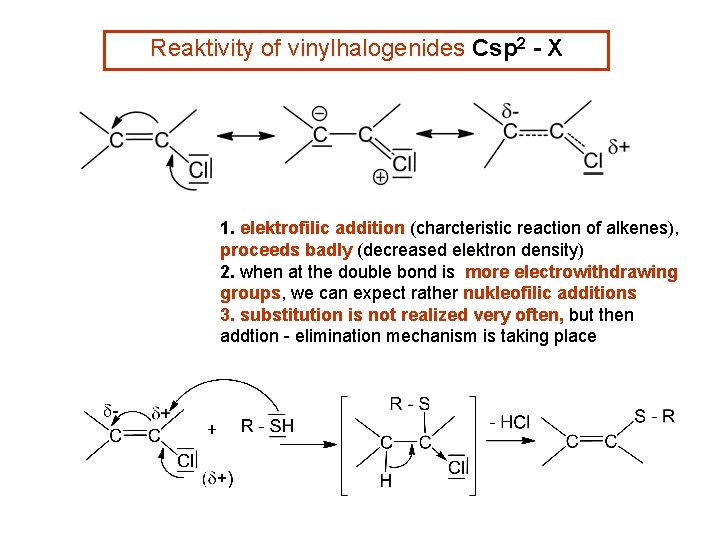
Reaktivity of vinylhalogenides Csp 2 - X 1. elektrofilic addition (charcteristic reaction of alkenes), proceeds badly (decreased elektron density) 2. when at the double bond is more electrowithdrawing groups, we can expect rather nukleofilic additions 3. substitution is not realized very often, but then addtion - elimination mechanism is taking place

Reaktivity of aromatic halogen derivatives 1. The characteristic reactions for aromatic systems are elektrofilic aromatic substitution, they are realized at halogen substituted aromatic system to ortho- a paraposition. Such an aromat has got lower electron density than unsubstituted benzene (it behaves as worse nucleophile)
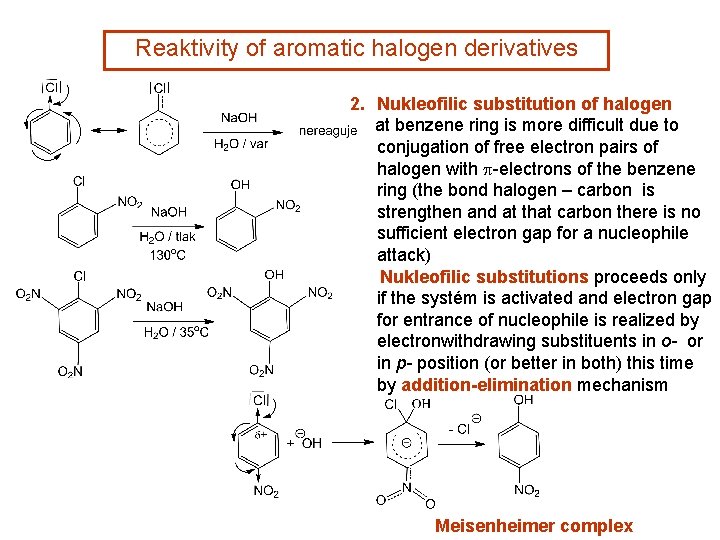
Reaktivity of aromatic halogen derivatives 2. Nukleofilic substitution of halogen at benzene ring is more difficult due to conjugation of free electron pairs of halogen with p-electrons of the benzene ring (the bond halogen – carbon is strengthen and at that carbon there is no sufficient electron gap for a nucleophile attack) Nukleofilic substitutions proceeds only if the systém is activated and electron gap for entrance of nucleophile is realized by electronwithdrawing substituents in o- or in p- position (or better in both) this time by addition-elimination mechanism Meisenheimer complex
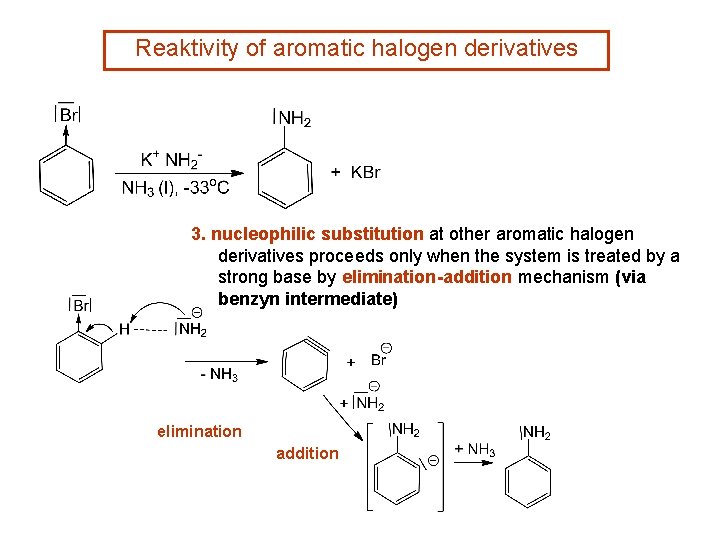
Reaktivity of aromatic halogen derivatives 3. nucleophilic substitution at other aromatic halogen derivatives proceeds only when the system is treated by a strong base by elimination-addition mechanism (via benzyn intermediate) elimination addition
![Reaktivity of aromatic halogen derivatives Důkaz benzynu [4+2] cykloadice dokázala strukturu Reaktivity of aromatic halogen derivatives Důkaz benzynu [4+2] cykloadice dokázala strukturu](http://slidetodoc.com/presentation_image/5447fe5f479066b48dc62799bf9517b7/image-9.jpg)
Reaktivity of aromatic halogen derivatives Důkaz benzynu [4+2] cykloadice dokázala strukturu
- Slides: 9