INDUSTRIALLY IMPORTANT MATERIALS BS c Chemistry Semester 6

INDUSTRIALLY IMPORTANT MATERIALS BS. c Chemistry Semester 6 JOFRIN J

CARBIDES �Carbides are the binary compounds in which carbon has combined with elements of lower or equal electronegativity than carbon.

Types of carbides Carbides are mainly classified into three � 1. Ionic carbides � 2. Covalent carbides � 3. Interstitial or metallic carbides

IONIC CARBIDES �Ionic carbides are formed by the combination of carbon with alkali, alkaline earth metals, Sc, Ag, Thorium and aluminium. �These carbides are usually prepared by heating the metals or metal oxide with carbon or CO 2

IONIC CARBIDES Ionic carbides are further classified into three. a) Acetylides b) Methanides c) Allylides
![IONIC CARBIDES Acetylides �These carbides yield acetylene on hydrolysis. �They contain [C 2 ]2 IONIC CARBIDES Acetylides �These carbides yield acetylene on hydrolysis. �They contain [C 2 ]2](http://slidetodoc.com/presentation_image_h/185ac5a93a5c8fc04799452461d2223b/image-6.jpg)
IONIC CARBIDES Acetylides �These carbides yield acetylene on hydrolysis. �They contain [C 2 ]2 - group. �Example: Calcium carbide Ca. C 2+ 2 H 2 O------ Ca(OH)2+ C 2 H 2(acetylene) �Since these carbides yield acetylene on hydrolysis, they are called acetylides
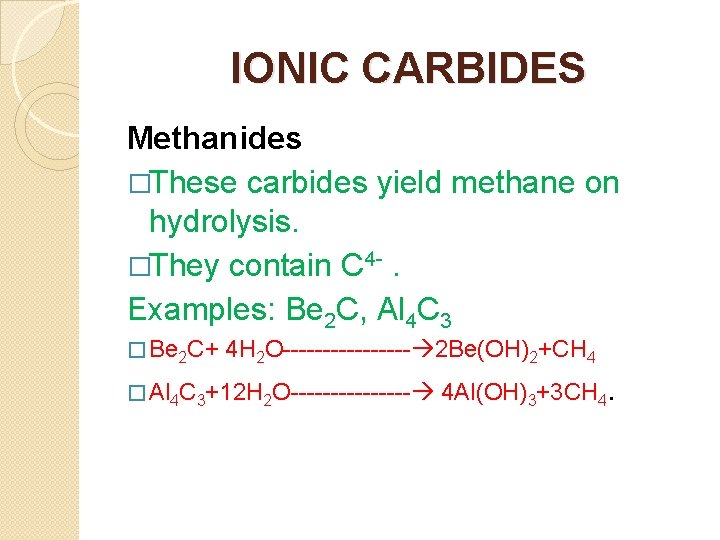
IONIC CARBIDES Methanides �These carbides yield methane on hydrolysis. �They contain C 4 -. Examples: Be 2 C, Al 4 C 3 � Be 2 C+ 4 H 2 O-------- 2 Be(OH)2+CH 4 � Al 4 C 3+12 H 2 O-------- 4 Al(OH)3+3 CH 4.
![IONIC CARBIDES �Allylides �These ionic carbides on hydrolysis yield allylene. �[C 3 ]4 - IONIC CARBIDES �Allylides �These ionic carbides on hydrolysis yield allylene. �[C 3 ]4 -](http://slidetodoc.com/presentation_image_h/185ac5a93a5c8fc04799452461d2223b/image-8.jpg)
IONIC CARBIDES �Allylides �These ionic carbides on hydrolysis yield allylene. �[C 3 ]4 - is the common group found in these type of carbides. �Example: Mg 2 C 3 � Mg 2 C 3 +4 H 2 O-------- 2 Mg(OH)2+ C 3 H 4.

COVALENT CARBIDES �Binary compounds of carbon with elements of similar electronegativity are called covalent carbides. Examples: Silicon carbide (Si. C) or Carborandum & Boron carbide (B 4 C 3).

INTERSTITIAL / METALLIC CARBIDES �In these carbides, small carbon atoms occupy interstitial positions in the crystal lattices of metals. So they are also called interstitial carbides Examples : Ti. C, Zr. C, VC, W 2 C These carbides are also extremely hard as diamond. It has been estimated that the radius of the metal atom should be greater than 1. 3 A 0 to form the interstitial carbides. So metals like Cr, Fe, Ni, Co which possess smaller atomic radii than 1. 3 A 0 do not form interstitial carbides.

Applications of carbides �Since they are very hard they are used as abrasive, drilling tools, cutting tools. �As reducing agents in metallurgy �Aluminium carbide is used for producing methane while calcium carbide is used to produce acetylene. �Carbides are used as furnace lining �Boron carbide is used for cutting diamond, making lamp filaments, drilling holes in rocks

NITRIDES �Nitrides are binary compounds of nitrogen with elements which are less electronegative than nitrogen. �Depending on the bond between the metal and nitrogen nitrides have been classified as �(a) Ionic nitrides �(b) covalent nitrides �(c) Interstitial nitides

IONIC NITRIDES �These nitrides are formed by the elements like Mg, Ca, Ba, Sr, Zn, Cd, Lithium etc. �They contain the common group N 3�Eg: Ca 3 N 2, Li 3 N � 6 Li+ N 2 ----------- 2 Li 3 N � 3 Mg+N 2 ----------- Mg 3 N 2 �These nitrides on hydrolysis yield NH 3. �Eg: Li 3 N+3 H 2 O-------- 3 Li. OH+NH 3

COVALENT NITRIDES �These nitrides are formed sharing electrons between nitrogen and elements like Boron, Carbon, Aluminium, Silicon , Fe etc. �Eg: cyanogen (CN)2, disulphur dinitrogen (S 2 N 2), (S 4 N 4).

NITRIDES �INTERSTITIAL NITRIDES �Transition metals of group 3, 4, 5 absorb nitrogen on heating into their lattice to form interstitial nitrides. �Eg: Sc. N, Ti. N, VN

BORIDES �These are binary compounds of boron with transition metals. There a large group of borides with different stoichiometries. �(i) Boron rich borides (MB to MB 66) �(ii) Metal rich borides. These borides range from M 2 B to M 10 B 11

BORIDES �Applications of Borides (March 2014) � 1. Boron coated reaction vessels are used in making crucibles. � 2. Borides are used as high temperature semi-conductors � 3. Zr. B 2 and Cr. B 2 find application as turbine blades and rocket nozzles. � 4. Lanthanide hexaborides are good thermionic emitters.

GRAPHITE Graphite is an allotropic form of carbon. It has a two dimensional structure. In Graphite each carbon is sp 2 hybridized. Each carbon atom uses three hybrid orbitals to form three covalent bonds with three neighbouring carbon atoms. Clearly the structure of graphite is a two dimensional sheet like; consisting of hexagonal rings of carbon atoms fused together. Distance between carbon atoms is 142 pm and the distance between sheets are 340 pm. The large distance between the layers points to the formation of intercalation or lamellar compounds.

. . . . �The fourth valence electron in the unhybrid orbital of each carbon atom remains free. �These electrons can move from one carbon to another carbon under an applied field. �These free electrons make the graphite a good conductor of electricity

Properties of graphite. . . . �It is slowly oxidised to carbon dioxide by chromic acid �It is oxidised by Conc. Nitric acid to give graphitic acid C 11 H 4 O 5 �When graphite is treated with potassium permanganate and conc. Sulphuric acid, graphite oxide is formed. �It reacts with Fluorine at 5000 C to give carbon mono fluoride.

Intercalation compounds or lamellar compounds of graphite with alkali metals �Graphite reacts with potassium metal at 3000 C to give intercalation compound having the formula C 8 K � 8 C+K---------- C 8 K �In the same method we can prepare C 8 Rb, C 8 Cs. �The compound C 8 M (M=K, Rb, Cs ) is formed through four stages. �First C 48 M is formed. This then converted into C 36 M , then to C 24 M and finally to C 8 M.

Intercalation compounds of graphite with metal halides �These lamellar compounds are prepared by reacting metal halides in presence of the free halogens with graphite. �The intercalated halides are lewis acids such as Fe. Cl 3, Be. Cl 2, Ni. Cl 2, Al. Cl 3, Sb. Cl 5 � Graphite + x. Fe. Cl 3 ------- Graphite(Fe. Cl 3)x. �The product formed is a charge transfer complex. �The d-orbitals of Fe 3+ ion receive charge from the graphite π system.

CARBON MONOFLUORIDE �Fluorine reacts with graphite at 5000 C to give carbon monofluoride. (C 2 F and C 4 F are also formed) � C+F 2 ------- CFx (x= 0. 68 to 0. 99). �The colour of CFx depends on the fluorine content. �The colour deepens with decrease in fluorine content. When x=0. 99 the colour is white. �But it may turn to black when x=0. 7
- Slides: 23