INDUSTRIAL MICROBIOLOGY for diploma 8 YEAST Prof Dr

INDUSTRIAL MICROBIOLOGY (for diploma) (8) YEAST Prof. Dr. Waiel Farghaly Prof. of Microbiology/ Dept. Botany, Fac. Science

n Eukaryotic unicellular microfungi that are widely distributed in the natural environment. n Around 800 yeast species are known, but this represents only a fraction of yeast biodiversity on Earth. n The most widely exploited and studied yeast species is Saccharomyces cerevisiae, commonly referred to as “baker’s yeast”. n They are used in the production of foods, beverages, and pharmaceuticals

Natural habitats of yeast communities n Yeasts can be isolated from terrestrial, aquatic, and aerial samples. Yeast communities are also found in association with plants, animals, and insects. n Preferred yeast habitats are plant tissues, but a few species are found in commensal or parasitic relationships with animals. n Some yeasts, most notably Candida albicans, are opportunistic human pathogens. n Several species of yeast may be isolated from specialized or extreme environments (i. e. high sugar or salt concentrations, low temperature, intestinal tracts of animals, etc).
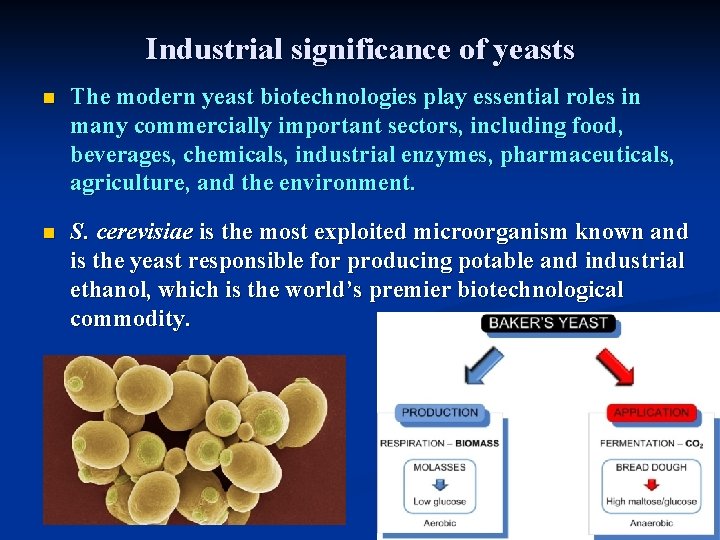
Industrial significance of yeasts n The modern yeast biotechnologies play essential roles in many commercially important sectors, including food, beverages, chemicals, industrial enzymes, pharmaceuticals, agriculture, and the environment. n S. cerevisiae is the most exploited microorganism known and is the yeast responsible for producing potable and industrial ethanol, which is the world’s premier biotechnological commodity.

INDUSTRIAL ETHANOL PRODUCTION n The process starts by milling a feedstock, such as sugar cane, sweetcorn, or cheap cereal grains, and then adding dilute sulfuric acid, or fungal alpha amylase enzymes, to breakdown the starches into complex sugars. A gluco amylase is then added to break the complex sugars down into simple sugars. After this, yeasts are added to convert the simple sugars to ethanol, which is then distilled off to obtain ethanol up to 96% in concentration. C 6 H 12 O 6 2 CH 3 CH 2 OH + 2 CO 2 Glucose Ethanol + Carbon dioxide
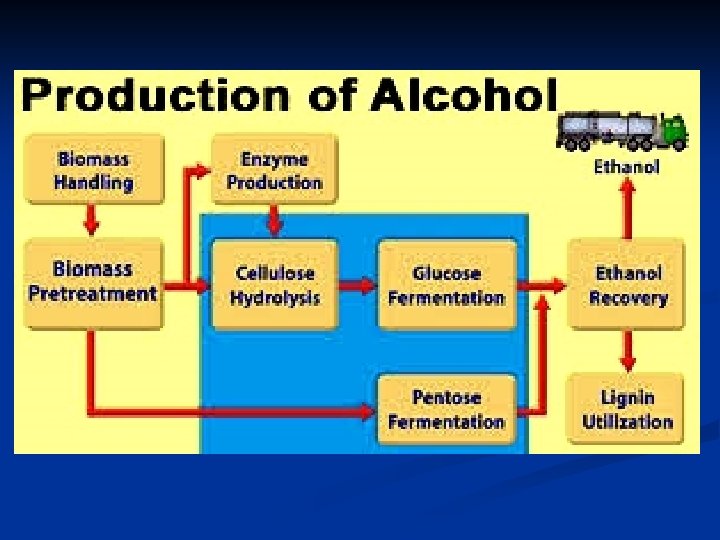

n The potential quantity of ethanol that could be produced from cellulose is over an order of magnitude larger than that producible from corn. In contrast to the corn-to-ethanol conversion, the cellulose-to ethanol route involves little or no contribution to the greenhouse effect. In brief, there are three stages for the production of ethanol from biomass: 1 - The polymeric substrates are broken down to monosaccharides through physical, chemical, or enzymatic techniques, as appropriate. 2 - Microbial (generally yeast) fermentation converts the sugars to alcohol. 3 - Alcohol is recovered by distillation (as a constant-boiling mixture of 95. 6% ethanol and 4. 4% water, by volume). n
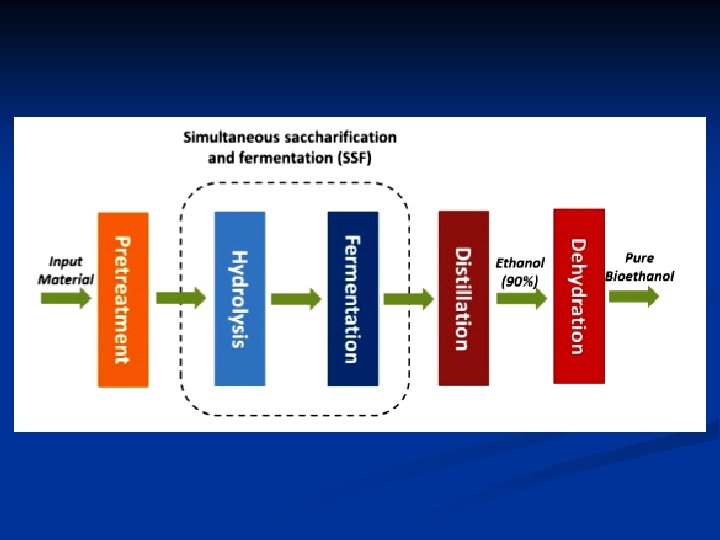

Ethanol Tolerance n Because the separation of ethanol from water (during stage III, alcohol recovery) accounts for much of the energy used in the production process, the higher the concentration of ethanol, the lower the distillation cost per liter of product. n Ethanol is toxic to yeast cells at concentrations between 8% and 18% by weight, depending on the strain and the metabolic state of the culture. n Yeast fermentation is totally inhibited by ethanol concentrations of about 11% by volume.

Flocculence and cell recycling n Cells can be collected at the end of one batch fermentation to be used as the inoculum for the next (cell recycling). n By using large amounts of yeast cells recovered in this manner, cell concentrations in the fermenter can be raised from a few grams dry weight/ L to tens of grams per liter. n Because of this increase in the cell concentration, cell recycle may increase the amount of alcohol produced per unit volume even when inhibition by ethanol decreases the specific productivity of individual cells. n Collecting cells by centrifugation or filtration costs more than any savings that result from such recycling. It is important then to find inexpensive ways of separation.

Stillage n Stillage is the residue from the first distillation of fermented substrate (corn mash, sugarcane juice, etc. ). With sugarcane, about 12 L of stillage are produced for each liter of ethanol. n Depending on what is done with it, stillage is either a serious water- polluting waste or a source of valuable byproducts. Major components in the stillage from fermented sugarcane juice
- Slides: 11