INDUSTRIAL MICROBIOLOGY for diploma 4 STERILIZATION Prof Dr

INDUSTRIAL MICROBIOLOGY (for diploma) (4) STERILIZATION Prof. Dr. Waiel Farghaly Prof. of Microbiology/ Dept. Botany, Fac. Science

STERILIZATION OF THE FERMENTOR n Steam is the most practical method for fermentor sterilization. n In order to avoid microbial growth within the fermentor when not in use, crevices and rough edges are avoided in the construction of fermentors (pockets of media for growth of undesirable microorganisms). n Saturated steam should be used and should remain in contact with all parts of the fermentor for at least half an hour. n Pipes should be steam- sealed using saturated steam. The various probes (for dissolved oxygen, CO 2, p. H, foam, etc. ), should also be sterilized.

Sterilization of heat labile medium n Tyndalization (the temperature of the medium is raised to boiling on three consecutive days). n The resulting vegetative cells (from spores that survived the first two days) will be killed with the third day’s boiling. n Chemical sterilization and filtration of the medium may also be used.

Prevention of Phage Contamination n Phages are present in the air, water, soil, etc. The first rule in avoiding phage contamination is routine general cleanliness and asepsis. Pipes, fermentors, utensils, and media, should all be well sterilized. n Air filters should be replaced or sterilized regularly. n Aerosol sterilization of the factory with chlorine compounds, and other disinfectants, as well as UV irradiation of fermentation halls should be done routinely.

TREATMENT OF WASTES IN INDUSTRY n Wastes, unwanted materials, result inevitably from industrial activities in the same way as they also do in domestic ones. n Waste carried in water, whether from industry or from domestic activity is known as sewage. n Sewage from some chemical industries especially those dealing with manmade chemicals is not only less degradable but sometimes toxic to microorganisms and man.

METHODS FOR DETERMINING TREATMENT EFFICIENCY n Wastewaters are sampled analyzed particularly at the point of the discharge of the treated wastewater into rivers, streams and other natural bodies of water. n large numbers of aerobic microorganisms will develop to break down the organic matter. They will use up the available oxygen causing the death of fish and other aquatic life. n Anaerobic bacteria will develop following the exhaustion of oxygen and its activities result in foul odours.

Some of the methods for analyzing the organic matter are : 1 - Dissolved Oxygen (DO) 2 - The Biological or Biochemical Oxygen Demand (BOD) Tests 3 - Chemical Oxygen Demand (COD) 4 - Total Organic Carbon (TOC) 5 - Total Suspended Solids (TSS) n
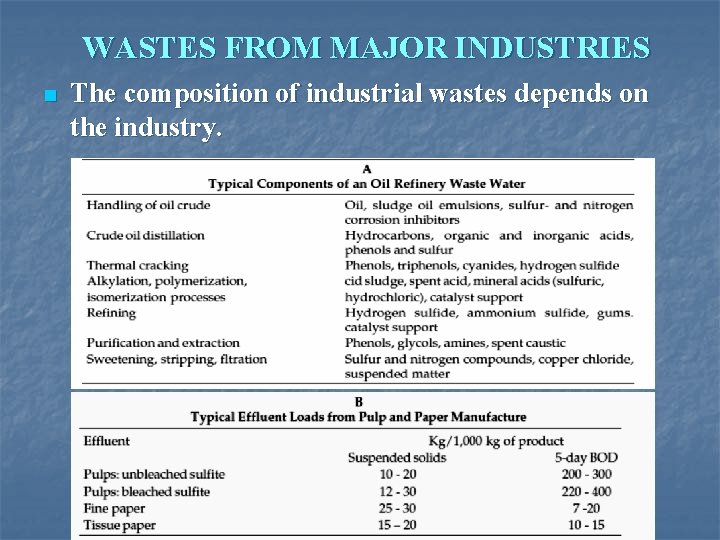
WASTES FROM MAJOR INDUSTRIES n The composition of industrial wastes depends on the industry.

SYSTEMS FOR THE TREATMENT OF WASTES The basic microbiological involvement in waste treatment (in aqueous environments) is as follows: (i) The degradable organic compounds in the wastewater (carbohydrates, proteins, fats, etc. ) are broken down mainly by aerobic bacteria and, to some extent, fungi. The result is an effluent with drastically reduced organic matter content. (ii) The materials difficult to digest form a sludge which must be removed from time to time and which is also treated separately. n n Therefore, there are two headings: aerobic breakdown of raw wastewater and anaerobic breakdown of sludge.

EXAMPLE: PHARMACEUTICAL INDUSTRY The wastes from a pharmaceutical industry represent a broad range of materials including easily degradable organic materials, and sometimes, some inorganic and even toxic compounds. n The used methods of disposal will depend on: (a) the cost of the disposal method (b) the location of the industry (c) the nature of the industry and its waste materials (d) the governmental regulations in the locality. n

n. Nature of Wastes: High BOD wastes may include: 1 - Easily degradable materials such as emulsion syrup, malt and tablet preparations with considerable amounts of carbohydrates. 2 - Acids including acetic, formic and sulfanilic, as well as HCl and H 2 SO 4 (should be neutralized before being allowed into the treatment system). 3 - Dissolved salts and many drugs with some being toxic or inhibitory to bacteria. n. Pre-treatment: 1 - Before treatment, acid (or alkali) is neutralized. 2 - Dissolved salts are removed usually by precipitation as calcium salts by lime addition (also neutralizes acidity). 3 - Chloride and sulfate may be removed by ion exchange or rendered non toxic by dilution with water. 4 - Volatile compounds are stripped by pre-aeration.

Treatment: n Before a routine is used within a treatment method, laboratory experiments are carried out to determine how much of the wastes may be efficiently handled within a given period. n It may often be necessary to segregate the wastes, treating the more easily biodegradable organic forms separately from those wastes rich in inorganic materials. This is because the latter may require ‘seeding’ or the development of specific microorganisms able to grow in and degrade them. n Seeding is achieved by shaking a sample of the waste with a soil sample long enough for a special flora to develop.
- Slides: 12