Individualization of Advanced Colorectal Cancer Treatment HER2 Positive














- Slides: 43

Individualization of Advanced Colorectal Cancer Treatment: HER-2 Positive Patients Cagatay Arslan, M. D. Assoc. Prof. 1, 2 1 Bahcesehir University, Faculty of Medicine, Department of Internal Medicine, Istanbul 2 Izmir Medical Park Hospital, Department of Medical Oncology, Izmir; Turkey

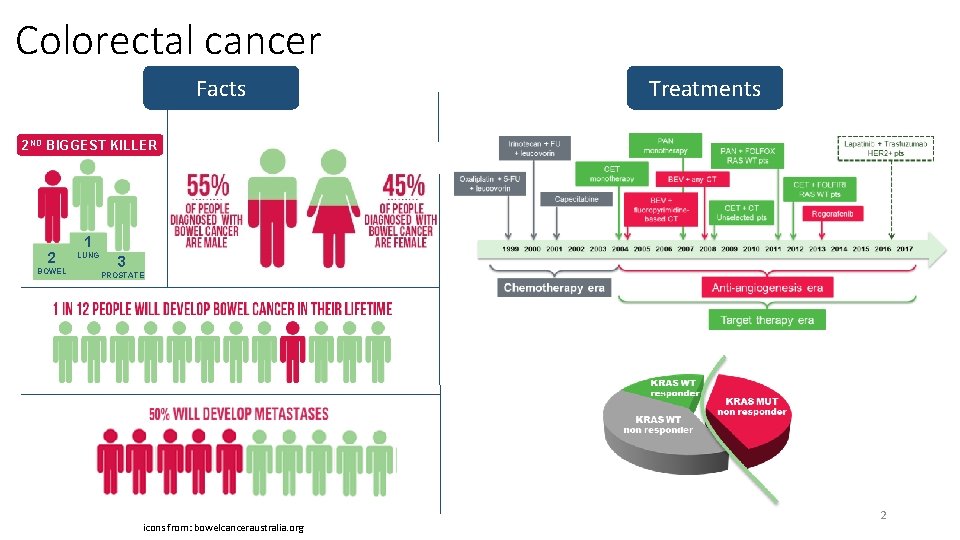
Colorectal cancer Facts Treatments 2 ND BIGGEST KILLER 2 BOWEL 1 LUNG 3 PROSTATE icons from: bowelcanceraustralia. org 2

Relevance of HER 2 in Metastatic Colorectal Cancer • HER 2 is an oncogenic driver, and a well-established therapeutic target in breast and gastric cancers • 2 -5% of metastatic colorectal cancer (CRC) tumors is driven by amplification or mutation of HER 2 • The role of HER 2 as a biomarker for prognosis in CRC remains uncertain • Independent studies documented substantial clinical benefit in patients treated with biomarker-driven HER 2 -targeted therapies Siena et al, Annals of Oncology 29: 1108– 1119, 2018

Relevance of HER 2 in Metastatic Colorectal Cancer • HER 2 is a negative predictor of response to EGFR-targeted treatments • HER 2 -positive CRC pts might have fewer treatment options and carry an inferior prognosis • HER 2 overexpression is usually caused by gene amplification • HER 2 is the only member of the EGFR family that does not bind ligands • It is activated via homo- or hetero-dimerization with other ligandbound receptors • The strongest mitogenic signals created by HER 2–HER 3 heterodimers Siena et al, Annals of Oncology 29: 1108– 1119, 2018

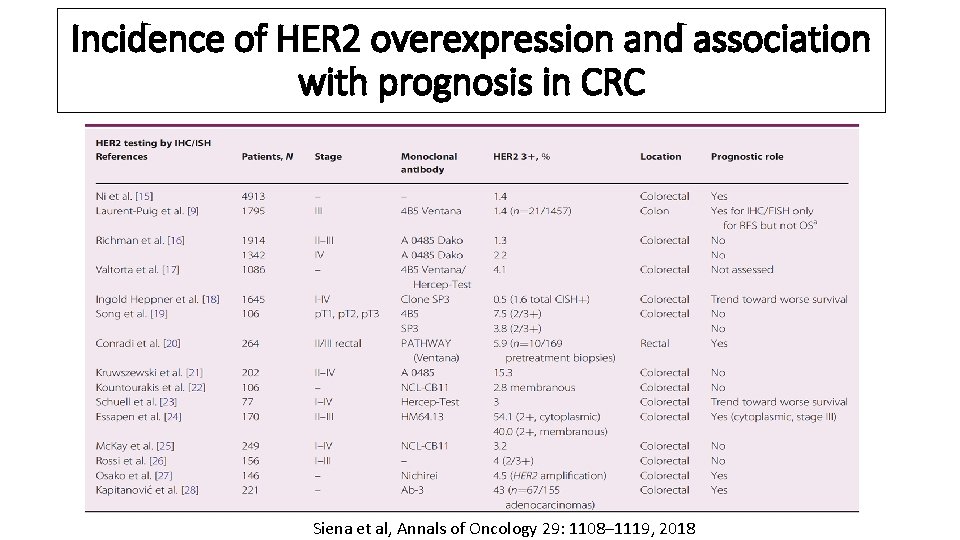
Incidence of HER 2 overexpression and association with prognosis in CRC Siena et al, Annals of Oncology 29: 1108– 1119, 2018

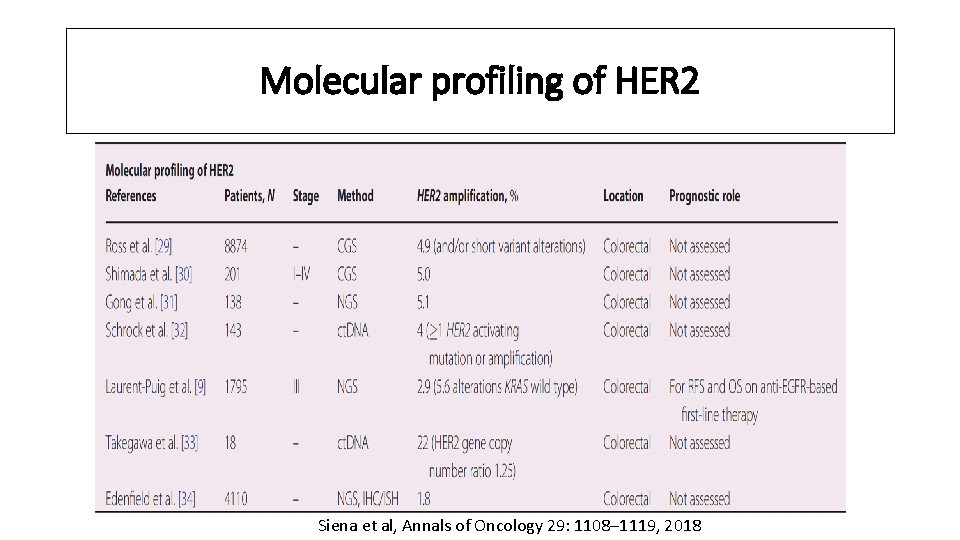
Molecular profiling of HER 2 Siena et al, Annals of Oncology 29: 1108– 1119, 2018

HER 2 by primary tumor location Siena et al, Annals of Oncology 29: 1108– 1119, 2018

Studies addressing the predictive role of HER 2 to EGFR-targeted therapies in m. CRC Siena et al, Annals of Oncology 29: 1108– 1119, 2018

Clinical studies exploiting HER 2 as a target for m. CRC Siena et al, Annals of Oncology 29: 1108– 1119, 2018

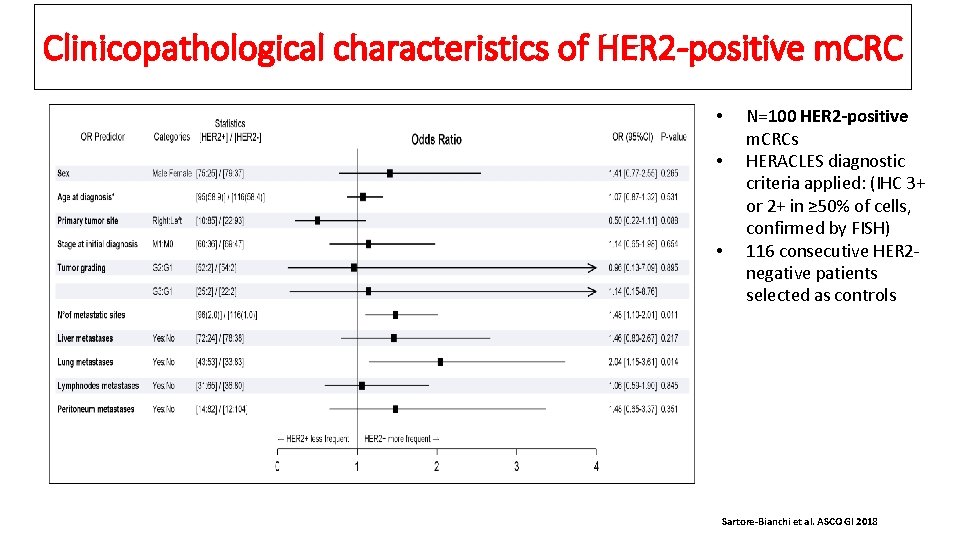
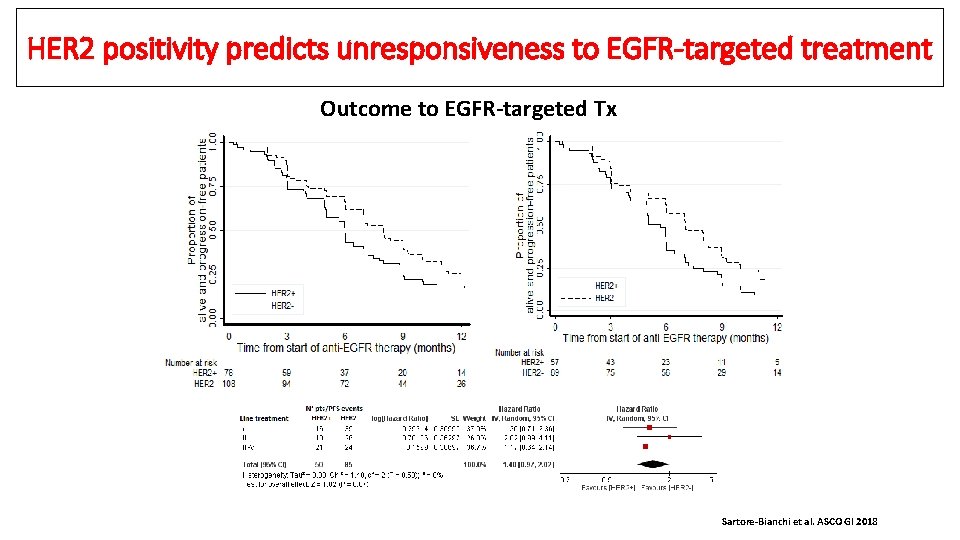
Clinicopathological characteristics of HER 2 -positive m. CRC • • • N=100 HER 2 -positive m. CRCs HERACLES diagnostic criteria applied: (IHC 3+ or 2+ in ≥ 50% of cells, confirmed by FISH) 116 consecutive HER 2 negative patients selected as controls Sartore-Bianchi et al. ASCO GI 2018

HER 2 positivity predicts unresponsiveness to EGFR-targeted treatment Outcome to EGFR-targeted Tx Sartore-Bianchi et al. ASCO GI 2018


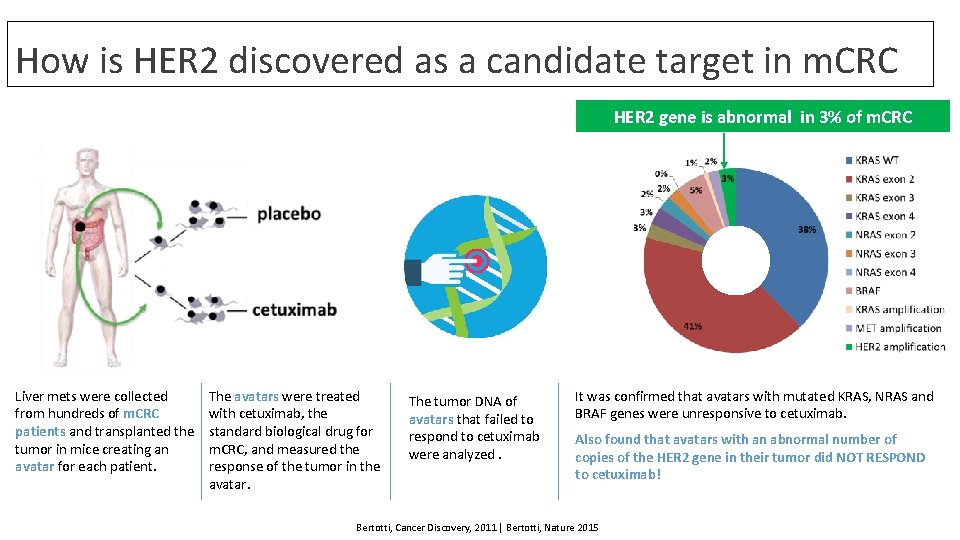
How is HER 2 discovered as a candidate target in m. CRC HER 2 gene is abnormal in 3% of m. CRC Liver mets were collected from hundreds of m. CRC patients and transplanted the tumor in mice creating an avatar for each patient. The avatars were treated with cetuximab, the standard biological drug for m. CRC, and measured the response of the tumor in the avatar. The tumor DNA of avatars that failed to respond to cetuximab were analyzed. It was confirmed that avatars with mutated KRAS, NRAS and BRAF genes were unresponsive to cetuximab. Also found that avatars with an abnormal number of copies of the HER 2 gene in their tumor did NOT RESPOND to cetuximab! Bertotti, Cancer Discovery, 2011 | Bertotti, Nature 2015

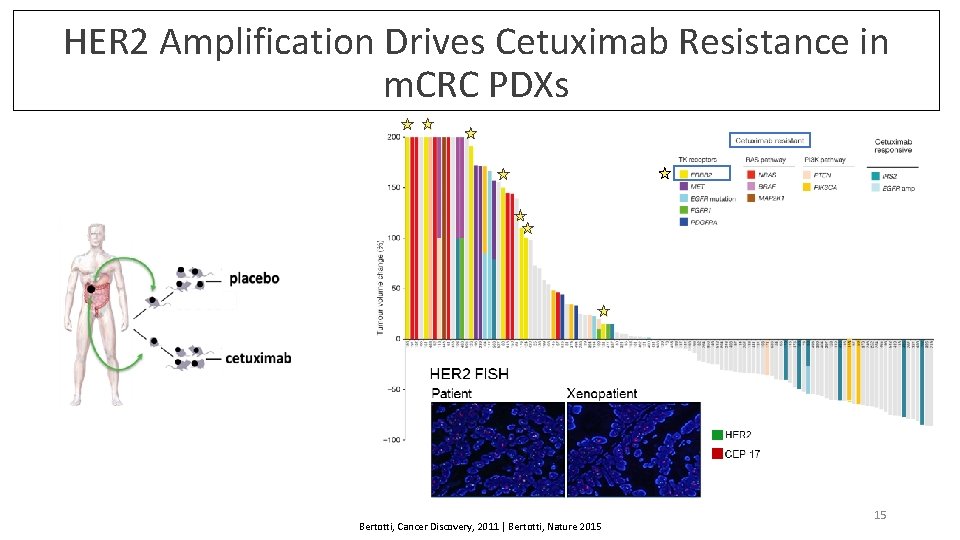
HER 2 Amplification Drives Cetuximab Resistance in m. CRC PDXs Bertotti, Cancer Discovery, 2011 | Bertotti, Nature 2015 15

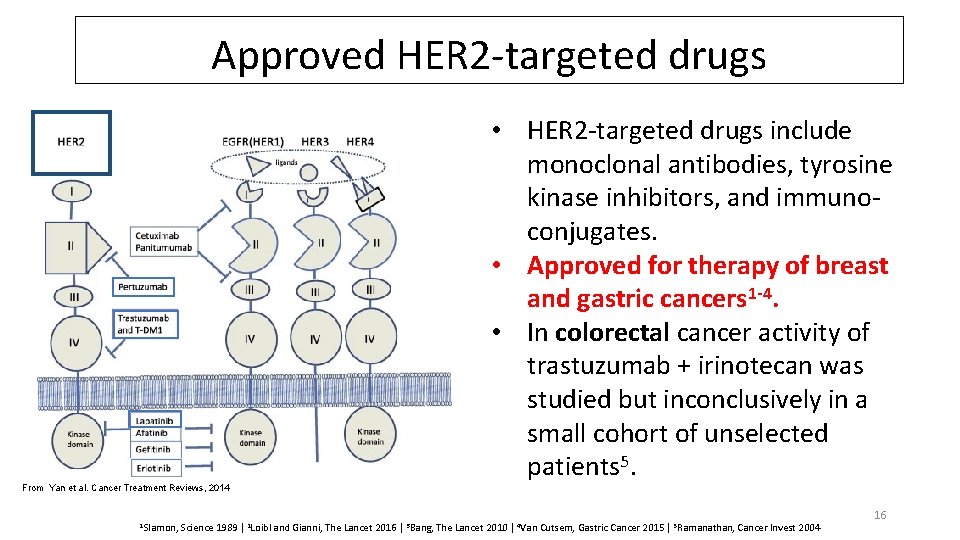
Approved HER 2 -targeted drugs From Yan et al. Cancer Treatment Reviews, 2014 1 Slamon, • HER 2 -targeted drugs include monoclonal antibodies, tyrosine kinase inhibitors, and immunoconjugates. • Approved for therapy of breast and gastric cancers 1 -4. • In colorectal cancer activity of trastuzumab + irinotecan was studied but inconclusively in a small cohort of unselected patients 5. Science 1989 | 2 Loibl and Gianni, The Lancet 2016 | 3 Bang, The Lancet 2010 | 4 Van Cutsem, Gastric Cancer 2015 | 5 Ramanathan, Cancer Invest 2004 16

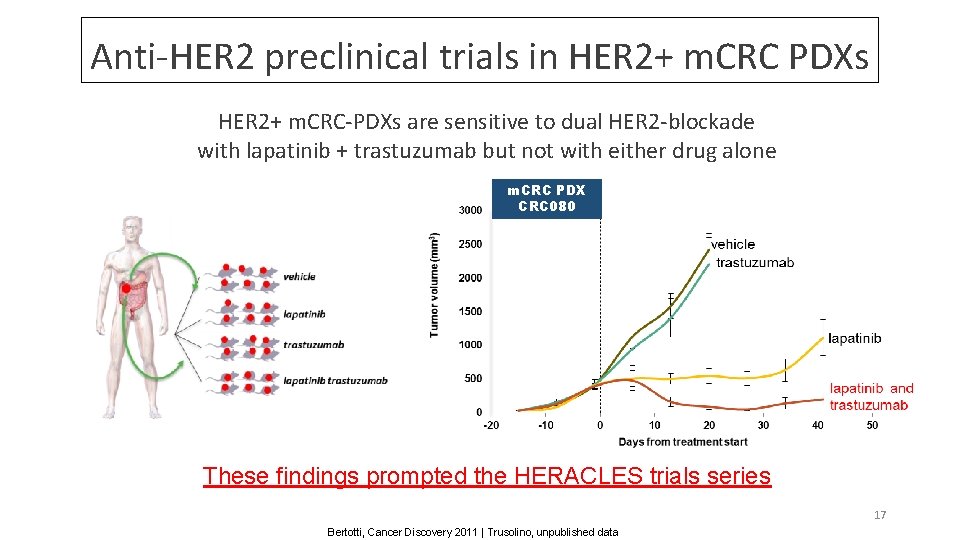
Anti-HER 2 preclinical trials in HER 2+ m. CRC PDXs HER 2+ m. CRC-PDXs are sensitive to dual HER 2 -blockade with lapatinib + trastuzumab but not with either drug alone m. CRC PDX CRC 080 These findings prompted the HERACLES trials series 17 Bertotti, Cancer Discovery 2011 | Trusolino, unpublished data

HERACLES* Project Seven studies in HER 2+ m. CRC patients Two DIAGNOSTICS studies Three THERAPEUTICS phase II trials HERACLES DGX developed lab criteria for screening HER 2+ m. CRC patients 1 HERACLES NOMO is developing a nomogram for clinical pre-screening of HER 2+ m. CRC patients ° HERACLES A proved the activity of trastuzumab + lapatinib in any line chemorefractory, anti EGFR resistant, m. CRC pts 2 HERACLES B* is testing trastuzumab-emtansine (T-DM 1) + pertuzumab in chemorefractory m. CRC pts, 2 nd/3 rd line° HERACLES RESCUE* is testing T-DM 1 monotherapy in m. CRC pts progressing on HERACLES A therapy° °Ongoing studies Two TRANSLATIONAL studies H-MUTATION uncovered HER 2 mutations as drivers of primary resistance to anti EGFR therapy and potential targets 3 H-RESISTANCE is an NGS study of primary and acquired resistance to HER 2 -inhibitors° °Ongoing studies 18 *HER 2 Amplification for Colo-Recta. L Cancer Enhanced Stratification || 1 Valtorta Modern Pathology 2015 | 2 Sartore-Bianchi et al. Lancet Oncology 2016 | 3 Kavuri et al. Cancer Discovery 2015

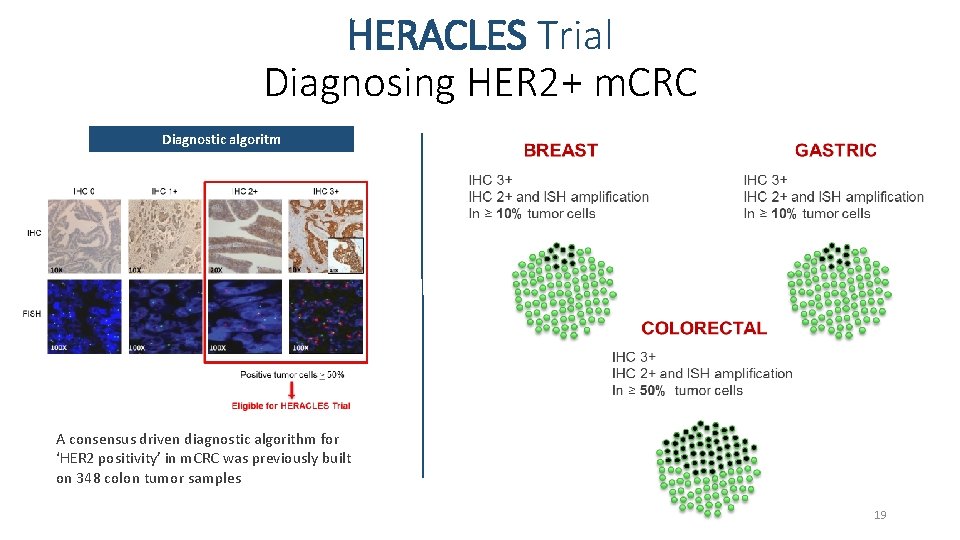
HERACLES Trial Diagnosing HER 2+ m. CRC Diagnostic algoritm A consensus driven diagnostic algorithm for ‘HER 2 positivity’ in m. CRC was previously built on 348 colon tumor samples 19

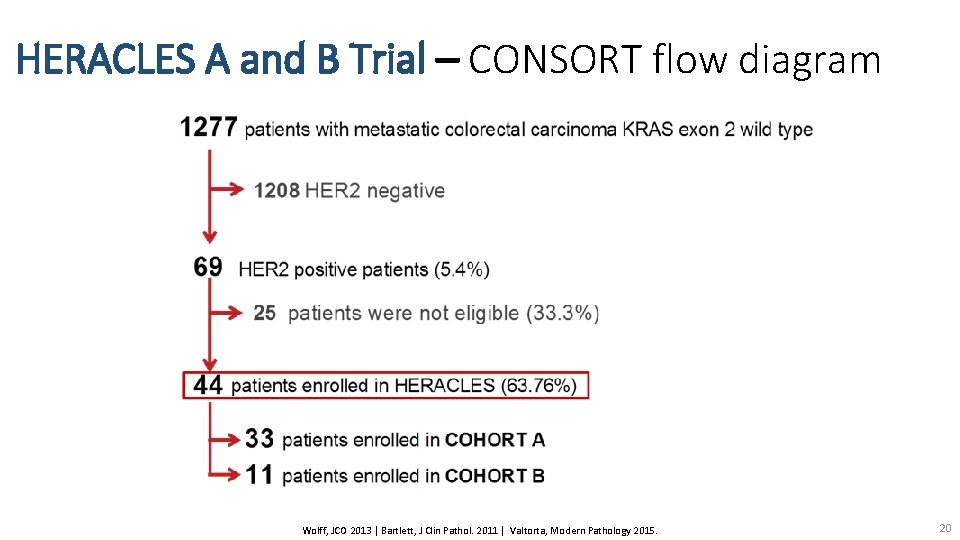
HERACLES A and B Trial – CONSORT flow diagram Wolff, JCO 2013 | Bartlett, J Clin Pathol. 2011 | Valtorta, Modern Pathology 2015. 20

HERACLES Trial ‘Proof of Concept’ phase II trial 2 -sequential cohort design HERACLES A HERACLES B Final analysis presented Preliminary data presented Pertuzumab iv 840 mg load and then 420 mg q 21 Trastuzumab Emtasine (T-DM 1) iv 3. 6 mg/kg q 21 Trastuzumab iv 4 mg/kg load and then 2 mg/kg q 7 Lapatinib po 1000 mg qd endpoints and statistics § Primary: Response § Secondary: Time to Progression and Safety § Translational: Liquid biopsy and deep sequencing of tumor biopsy at progression to study heterogeneity and resistance 1 Fleming § Sample size: 6 responses out of 27 patients needed to declare the study positive § Sensitivity analysis: for HER 2 gene copy number cut-off § Last patient in: Heracles B March, 2017 § Data lock: 28 th February, 2017 A’Hern, Statistics in Medicine 2001 21

HERACLES A treatment and assessments Therapy with: - Trastuzumab iv 4 mg/kg load and then 2 mg/kg qw - Lapatinib po 1000 mg qd PD Tumor assessments CE-CT scan baseline, q 8 weeks, until progression Translational assessments HER 2 ct. DNA (plasma) HER 2 ECD (serum) baseline, q 2 weeks, and at progression baseline, q 8 weeks, and at progression NGS Custom Panel (plasma, tumor tissue) baseline and at progression re-biopsy if possible

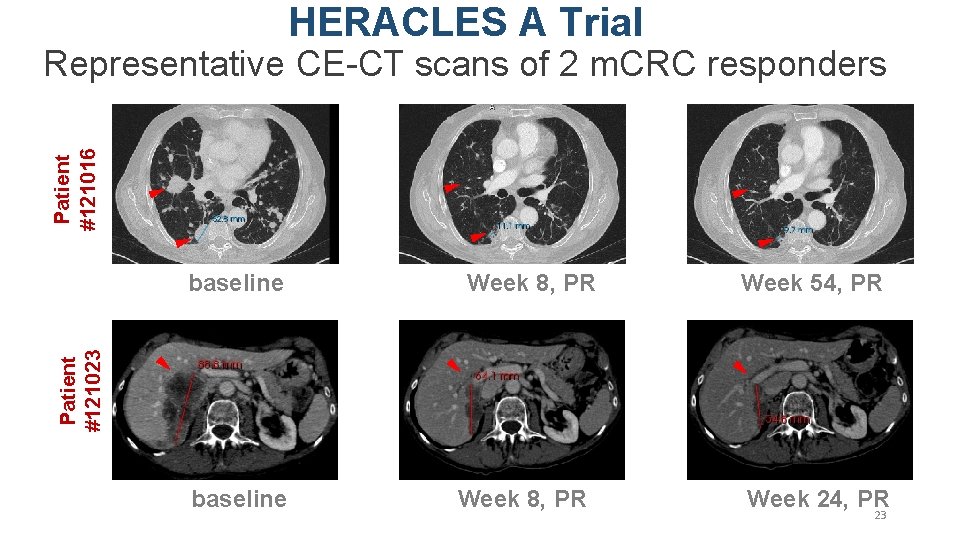
HERACLES A Trial Patient #121016 Representative CE-CT scans of 2 m. CRC responders Week 8, PR Week 54, PR Patient #121023 baseline Week 8, PR Week 24, PR 23

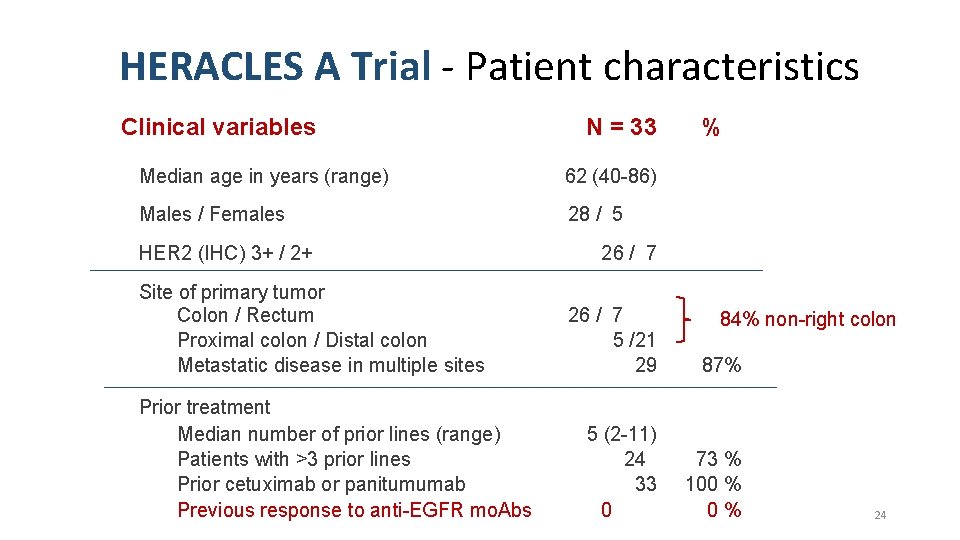
HERACLES A Trial - Patient characteristics Clinical variables N = 33 Median age in years (range) 62 (40 -86) Males / Females 28 / 5 HER 2 (IHC) 3+ / 2+ Site of primary tumor Colon / Rectum Proximal colon / Distal colon Metastatic disease in multiple sites Prior treatment Median number of prior lines (range) Patients with >3 prior lines Prior cetuximab or panitumumab Previous response to anti-EGFR mo. Abs % 26 / 7 5 /21 29 87% 5 (2 -11) 24 33 0 73 % 100 % 0% 84% non-right colon 24

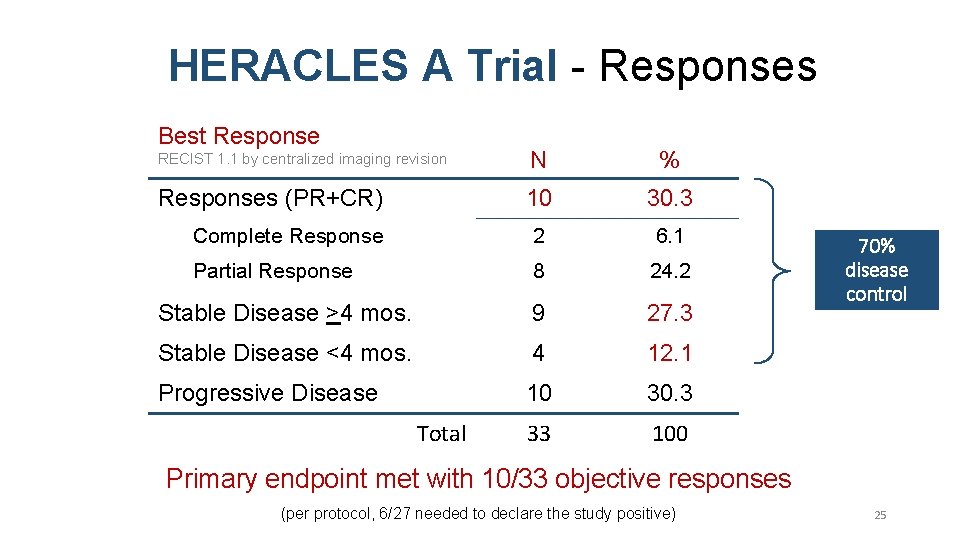
HERACLES A Trial - Responses Best Response N % 10 30. 3 Complete Response 2 6. 1 Partial Response 8 24. 2 Stable Disease >4 mos. 9 27. 3 Stable Disease <4 mos. 4 12. 1 Progressive Disease 10 30. 3 33 100 RECIST 1. 1 by centralized imaging revision Responses (PR+CR) Total 70% disease control Primary endpoint met with 10/33 objective responses (per protocol, 6/27 needed to declare the study positive) 25

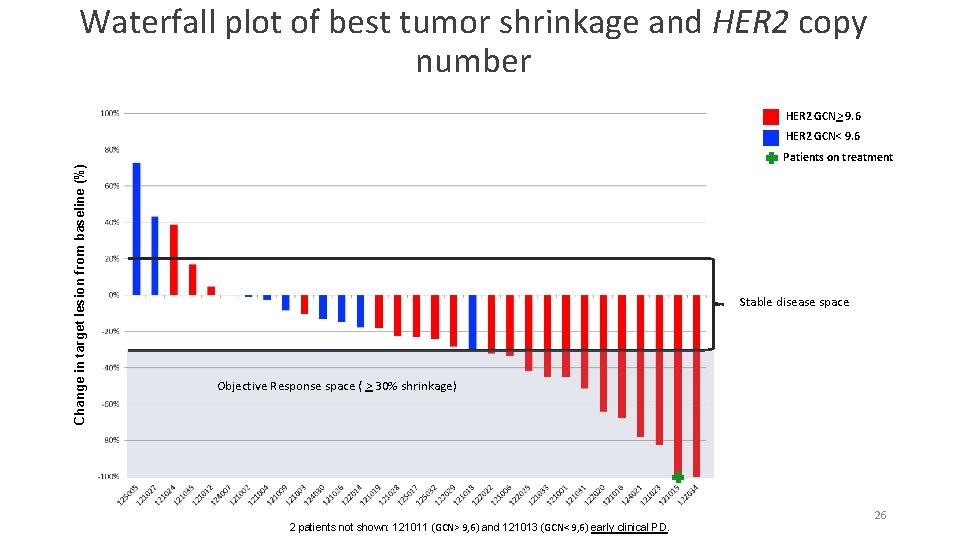
Waterfall plot of best tumor shrinkage and HER 2 copy number HER 2 GCN> 9. 6 Change in target lesion from baseline (%) HER 2 GCN< 9. 6 Patients on treatment Stable disease space Objective Response space ( > 30% shrinkage) 2 patients not shown: 121011 (GCN> 9, 6) and 121013 (GCN< 9, 6) early clinical PD. 26

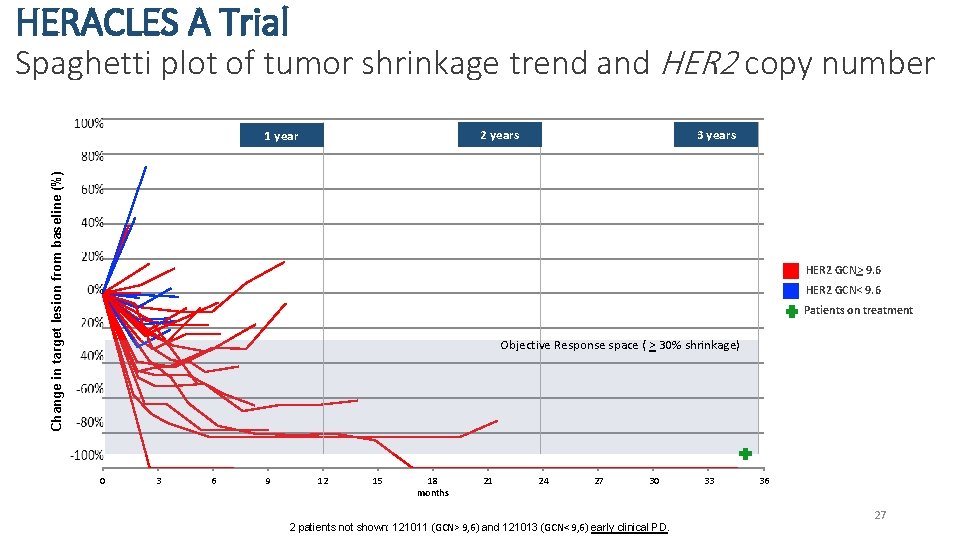
HERACLES A Trial Spaghetti plot of tumor shrinkage trend and HER 2 copy number 3 years 2 years Change in target lesion from baseline (%) 1 year HER 2 GCN> 9. 6 HER 2 GCN< 9. 6 Patients on treatment Objective Response space ( > 30% shrinkage) 0 3 6 9 12 15 18 months 21 24 27 30 2 patients not shown: 121011 (GCN> 9, 6) and 121013 (GCN< 9, 6) early clinical PD. 33 36 27

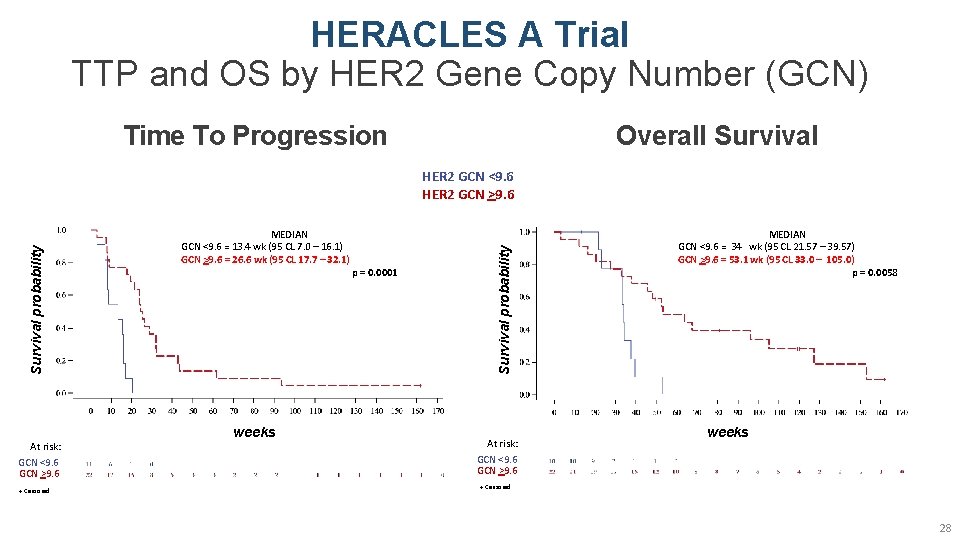
HERACLES A Trial TTP and OS by HER 2 Gene Copy Number (GCN) Time To Progression Overall Survival At risk: GCN <9. 6 GCN >9. 6 + Censored MEDIAN GCN <9. 6 = 13. 4 wk (95 CL 7. 0 – 16. 1) GCN >9. 6 = 26. 6 wk (95 CL 17. 7 – 32. 1) weeks p = 0. 0001 Survival probability HER 2 GCN <9. 6 HER 2 GCN >9. 6 At risk: GCN <9. 6 GCN >9. 6 MEDIAN GCN <9. 6 = 34 wk (95 CL 21. 57 – 39. 57) GCN >9. 6 = 53. 1 wk (95 CL 33. 0 – 105. 0) p = 0. 0058 weeks + Censored 28

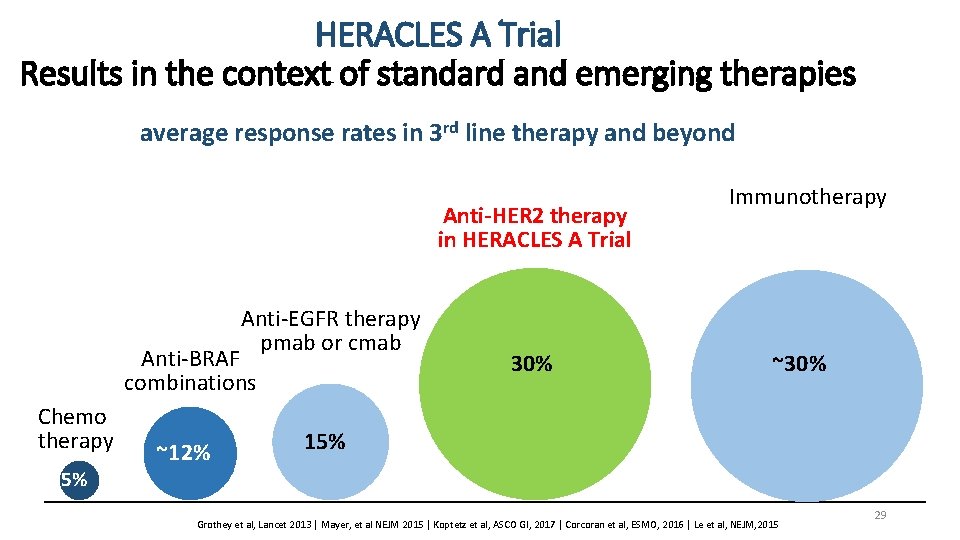
HERACLES A Trial Results in the context of standard and emerging therapies average response rates in 3 rd line therapy and beyond Anti-HER 2 therapy in HERACLES A Trial Anti-EGFR therapy pmab or cmab Anti-BRAF combinations Chemo therapy 5% ~12% 30% Immunotherapy ~30% 15% Grothey et al, Lancet 2013 | Mayer, et al NEJM 2015 | Koptetz et al, ASCO GI, 2017 | Corcoran et al, ESMO, 2016 | Le et al, NEJM, 2015 29

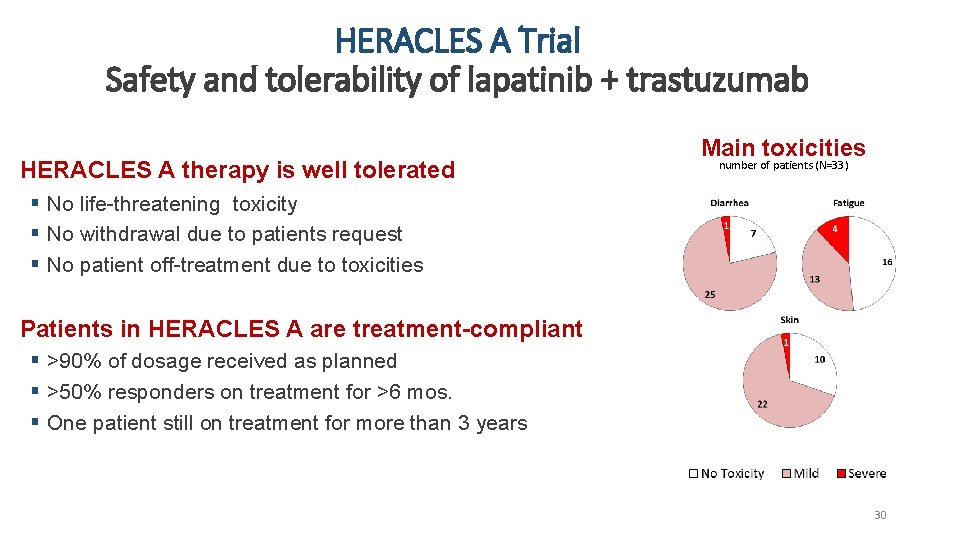
HERACLES A Trial Safety and tolerability of lapatinib + trastuzumab HERACLES A therapy is well tolerated Main toxicities number of patients (N=33) § No life-threatening toxicity § No withdrawal due to patients request § No patient off-treatment due to toxicities Patients in HERACLES A are treatment-compliant § >90% of dosage received as planned § >50% responders on treatment for >6 mos. § One patient still on treatment for more than 3 years 30

HERACLES A Trial - Conclusions ü Anti HER 2 treatment is effective in HER 2+ m. CRC: 30% ORR, 70% Clinical benefit ü Activity well compare to immumotherapy in MSI-H m. CRC patients ü Estimated number of HER 2+ m. CRCs is comparable to other malignancies with actionable molecular targets (i. e. mut+ NSCLC, CML, GIST) and to MSI-H patients in CRC (3%). ü HER 2+ m. CRC are primarily resistant to cetuximab or panitumumab, supporting the use of HERACLES therapy in anti-EGFR-naive patients. HERACLES A, a precision medicine trial, has documented that dual HER 2 -targeted therapy is a new valuable treatment option for HER 2+ m. CRC. Results on the first 27 patients have been published in: Sartore-Bianchi, Lancet Oncology 2016

HERACLES A treatment and assessments Therapy with: - Trastuzumab iv 4 mg/kg load and then 2 mg/kg/qw - Lapatinib po 1000 mg/qd PD Tumor assessments CE-CT scan baseline, q 8 weeks, until progression Translational assessments HER 2 ct. DNA (plasma) HER 2 ECD (serum) baseline, q 2 weeks, and at progression baseline, q 8 weeks, and at progression NGS Custom Panel (plasma, tumor tissue) baseline and at progression re-biopsy if possible

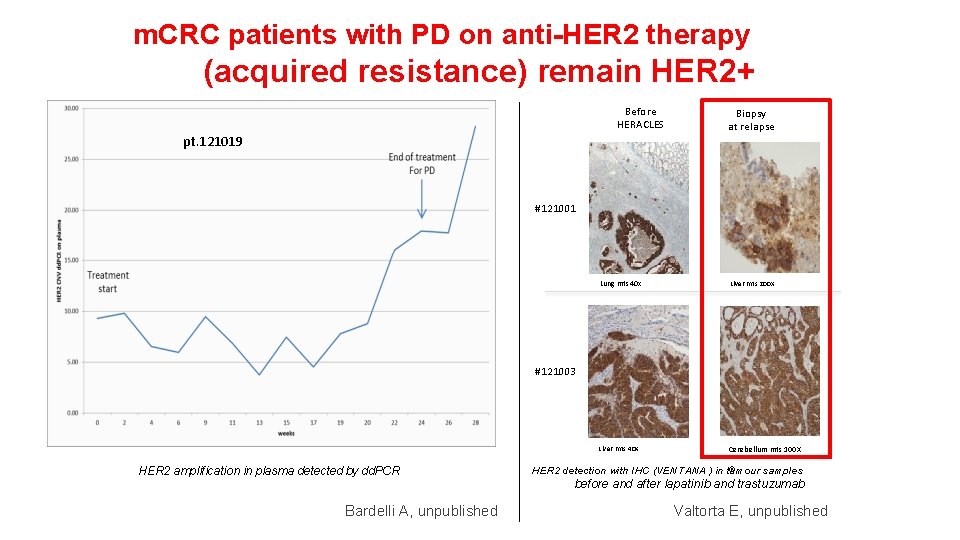
m. CRC patients with PD on anti-HER 2 therapy (acquired resistance) remain HER 2+ Before HERACLES pt. 121019 Biopsy at relapse #121001 Lung mts 40 x Liver mts 200 X Liver mts 40 X Cerebellum mts 100 X #121003 HER 2 amplification in plasma detected by dd. PCR Bardelli A, unpublished ® HER 2 detection with IHC (VENTANA ) in tumour samples before and after lapatinib and trastuzumab Valtorta E, unpublished

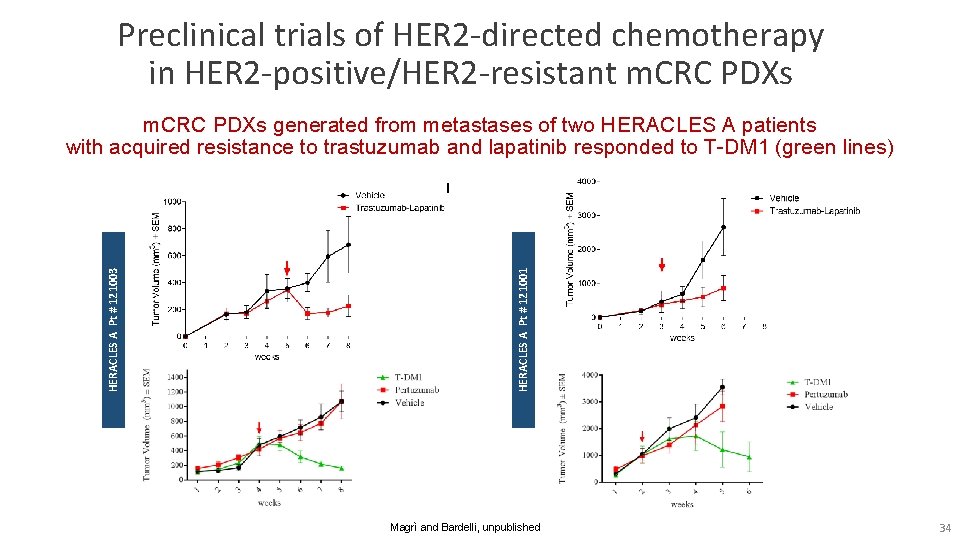
Preclinical trials of HER 2 -directed chemotherapy in HER 2 -positive/HER 2 -resistant m. CRC PDXs generated from metastases of two HERACLES A patients with acquired resistance to trastuzumab and lapatinib responded to T-DM 1 (green lines) DM 240847 HERACLES A Pt # 121001 HERACLES A Pt # 121003 RR 251239 Magrì and Bardelli, unpublished 34

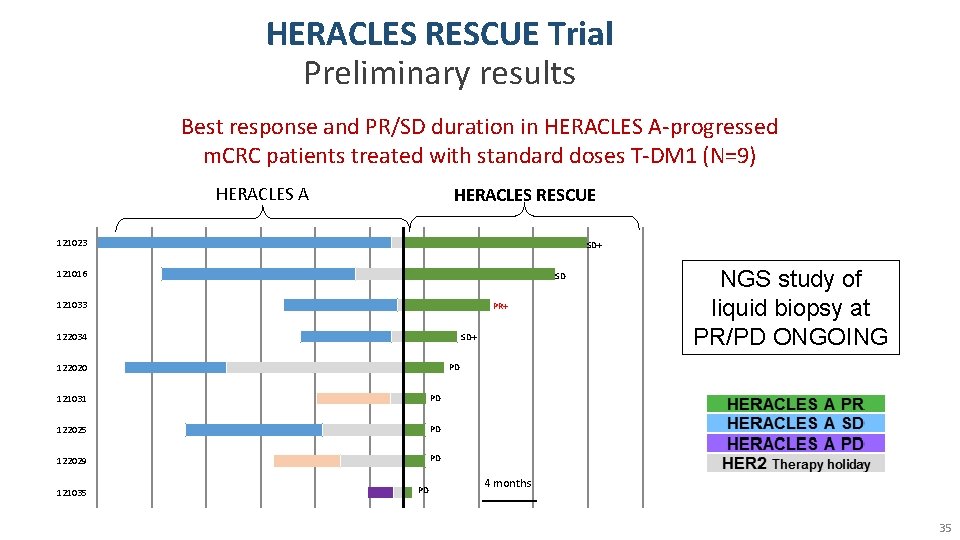
HERACLES RESCUE Trial Preliminary results Best response and PR/SD duration in HERACLES A-progressed m. CRC patients treated with standard doses T-DM 1 (N=9) HERACLES A HERACLES RESCUE 121023 SD+ 121016 SD 121033 PR+ 122034 SD+ 122020 PD 121031 PD 122025 PD 122029 PD 121035 NGS study of liquid biopsy at PR/PD ONGOING PD 4 months 35

HERACLES-B: Pertuzumab + TDM-1 HER-2 +, RAS wt m. CRC pts progressed after fluoropyrimidine, oxaliplatin and irinotecan Phase II Recruiting centers Primary endpoint: ORR (Recist 1. 1) Target accrual: 27 pts • Niguarda – Milano Pertuzumab + TDM-1 • Fond. Piemonte Oncol. – Candiolo • Policl. S. Orsola Malpighi – Bologna • SUN – Napoli • IOV – Padova • Campus Biomedico – Roma PD • Molinette - Torino Siena S. et al, https: //clinicaltrials. gov

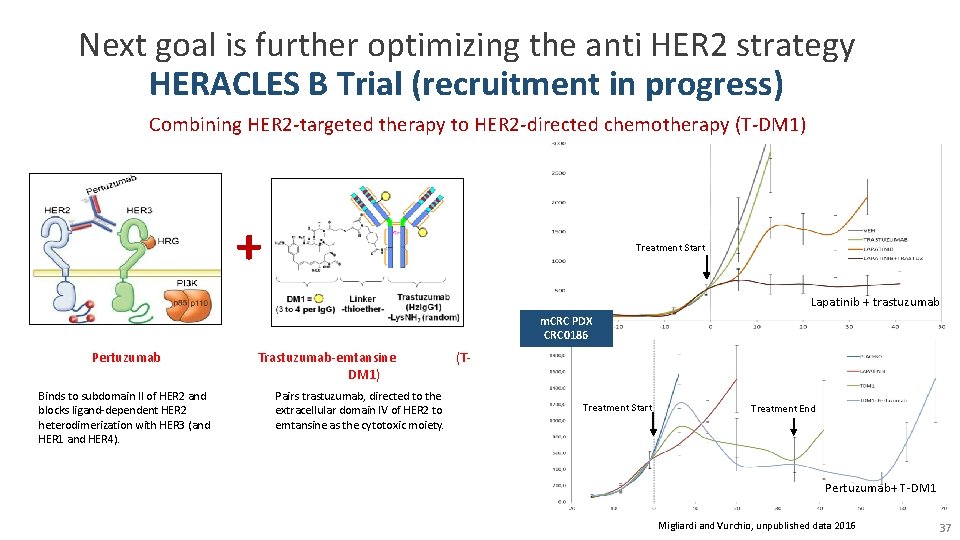
Next goal is further optimizing the anti HER 2 strategy HERACLES B Trial (recruitment in progress) Combining HER 2 -targeted therapy to HER 2 -directed chemotherapy (T-DM 1) + Treatment Start Lapatinib + trastuzumab m. CRC PDX CRC 0186 Pertuzumab Binds to subdomain II of HER 2 and blocks ligand-dependent HER 2 heterodimerization with HER 3 (and HER 1 and HER 4). Trastuzumab-emtansine DM 1) Pairs trastuzumab, directed to the extracellular domain IV of HER 2 to emtansine as the cytotoxic moiety. (T- Treatment Start Treatment End Pertuzumab+ T-DM 1 Migliardi and Vurchio, unpublished data 2016 37

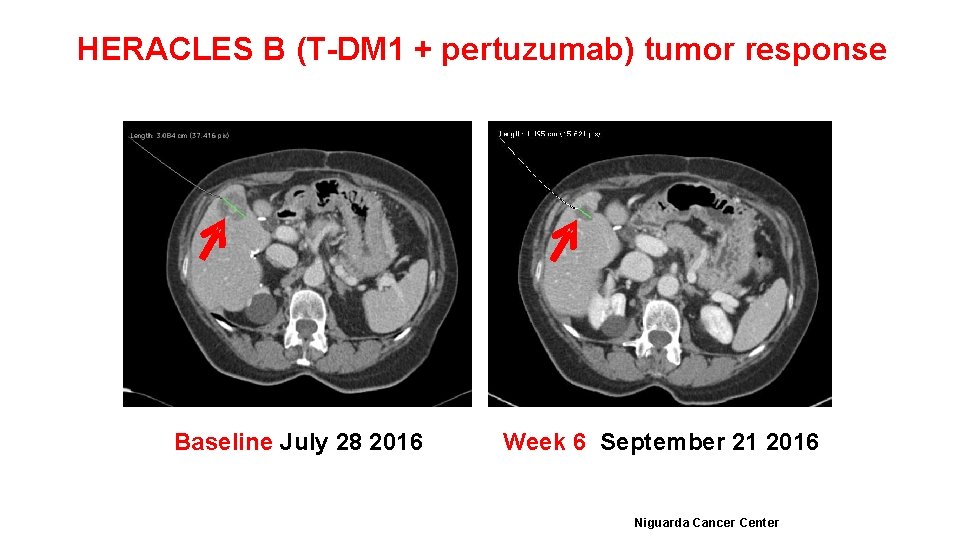
HERACLES B (T-DM 1 + pertuzumab) tumor response Baseline July 28 2016 Week 6 September 21 2016 Niguarda Cancer Center

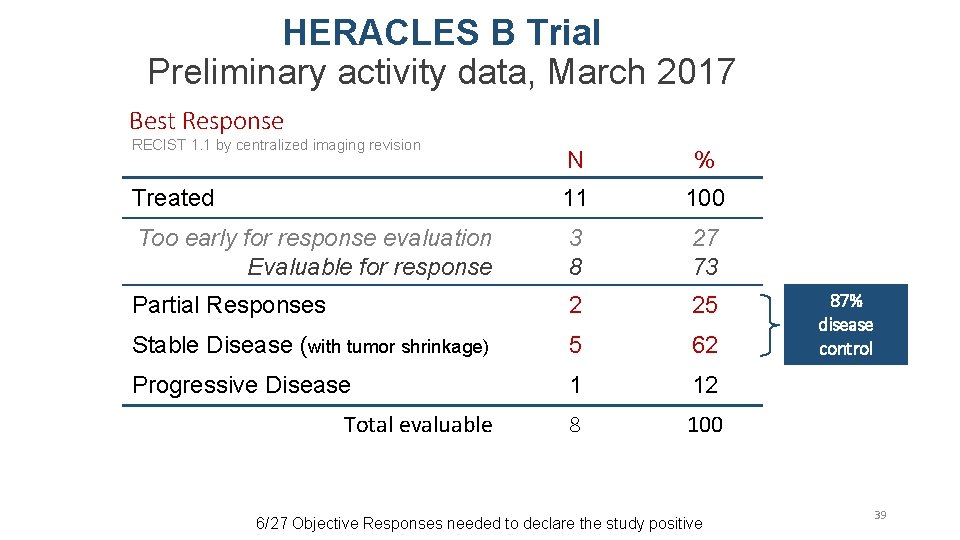
HERACLES B Trial Preliminary activity data, March 2017 Best Response RECIST 1. 1 by centralized imaging revision N % Treated 11 100 Too early for response evaluation Evaluable for response 3 8 27 73 Partial Responses 2 25 Stable Disease (with tumor shrinkage) 5 62 Progressive Disease 1 12 8 100 Total evaluable 6/27 Objective Responses needed to declare the study positive 87% disease control 39

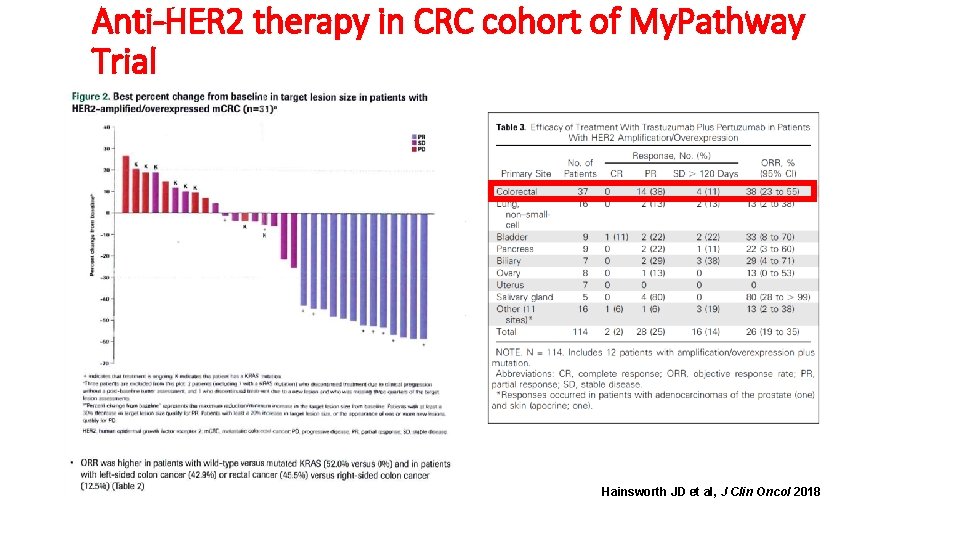
Anti-HER 2 therapy in CRC cohort of My. Pathway Trial Hainsworth JD et al, J Clin Oncol 2018

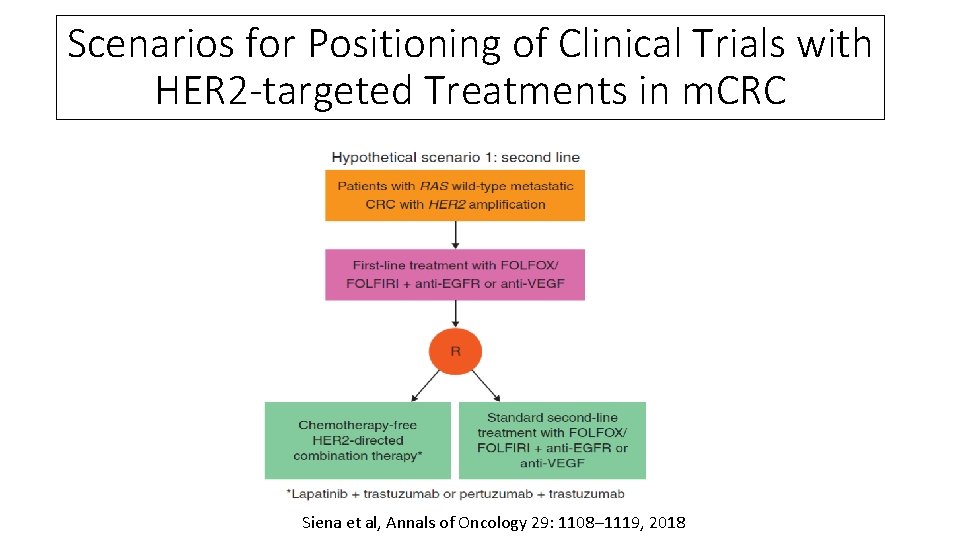
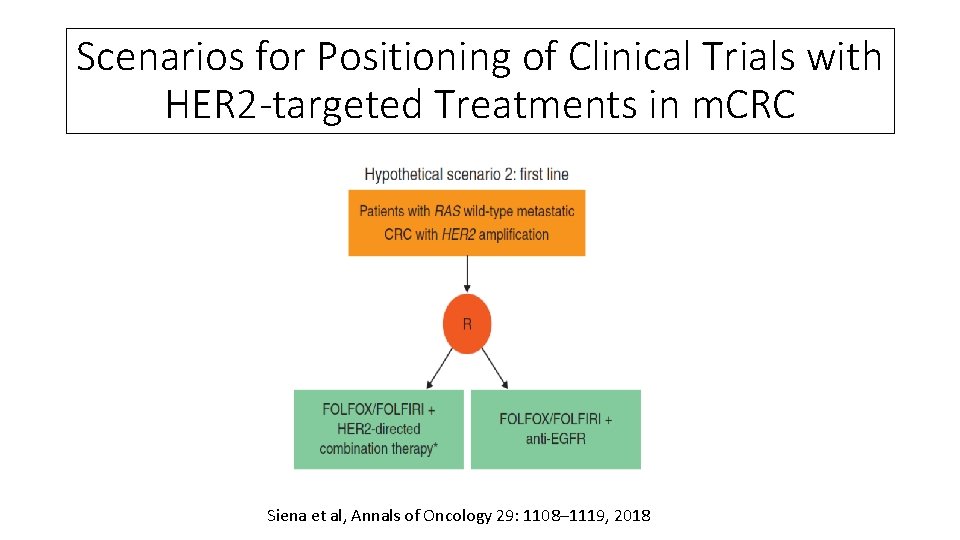
Scenarios for Positioning of Clinical Trials with HER 2 -targeted Treatments in m. CRC Siena et al, Annals of Oncology 29: 1108– 1119, 2018

Scenarios for Positioning of Clinical Trials with HER 2 -targeted Treatments in m. CRC Siena et al, Annals of Oncology 29: 1108– 1119, 2018

Conclusions • HER-2 is a negative predictor for anti. EGFR m. Ab efficacy • Trastuzumab + Lapatinib is a new alternative for HER-2 positive pretreated m. CRC pts (RAS wild type ) • ORR 32%, Control of Disease 76%, TTP 5. 5 months • Number of HER-2 positive m. CRC pts is comparable to GIST or CML • TDM-1 and TDM-1 plus pertuzumab results are awaited • Primary resistance of HER-2 positive m. CRC pts to anti-EGFR m. Abs opens the door to 1 st and 2 nd line treatment with anti. HER-2 tx’s

Special thanks to; Prof. Salvatore Siena for sharing their most recent data and his slides Thank You!