Individual antimicrobial processes Common sterilizers and disinfectants Sterilizers

Individual antimicrobial processes

Common sterilizers and disinfectants • Sterilizers – Physical • Dry heat (> 160 o. C) • Most heat (>121 o. C) • Ionizing radiation – Chemical • Gaseous chemicals • Disinfectants – Physical • Ultraviolet radiation • Hydrostatic pressure – Chemical • • Surface-active agents Phenolic compounds Iodine and iodine compounds Peoxygen compounds Chlorine species (free chlorine, chloramines) Chlorine dioxide Ozone

Sterilizers

Dry heat (I) • Mechanism: protein denaturation, enzyme inhibition, and RNA and DNA breakdown • Protein coagulation (complete denaturation) (e. g. egg albumin) – – – 50 % water: 56 o. C 25 % water: 74 -80 o. C 18 % water: 80 -90 o. C 6 % water: 145 o. C 0 % water: 160 -170 o. C • Time-temperature in sterilization with dry air (to inactivate bacterial spores) – – – 170 o. C for 60 min 160 o. C for 120 min 150 o. C for 150 min 140 o. C for 180 min 121 o. C for overnight

Dry heat (II) • Advantages – Deep penetration – Less corrosivity • Disadvantages – High temperature – Long sterilization period – Deterioration of materials • Used only for those materials that can not be sterilized by moist heat: petroleum, oil, powers, sharp instruments, and glassware

Moist heat (I) • Mechanism: protein denaturation, enzyme inhibition, RNA and DNA breakdown • Advantage – Low temperature and short sterilization period (121 o. C for 15 -30 min) • Disadvantage – Less penetration – Moisture damage
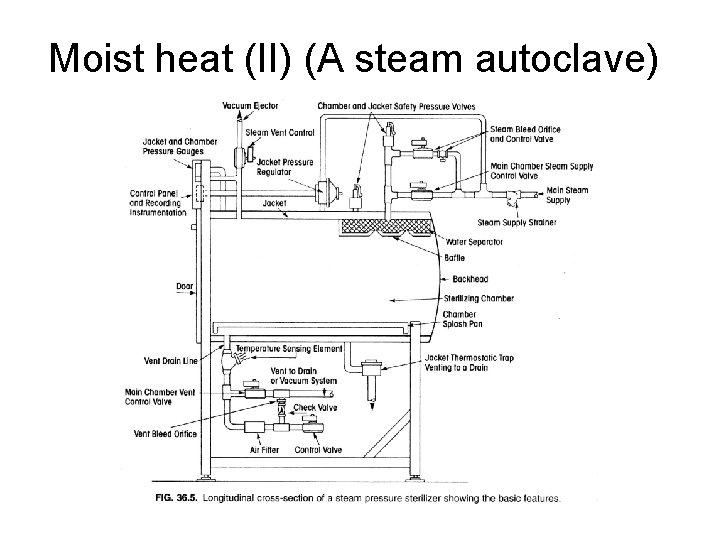
Moist heat (II) (A steam autoclave)

Ionizing radiation (I) • Electromagnetic radiations: γ radiation, xray, and electrons • Particle radiations: α radiation, β radiation, meson, positron, neutrino • Mechanism: single or double-strand breakage in DNA
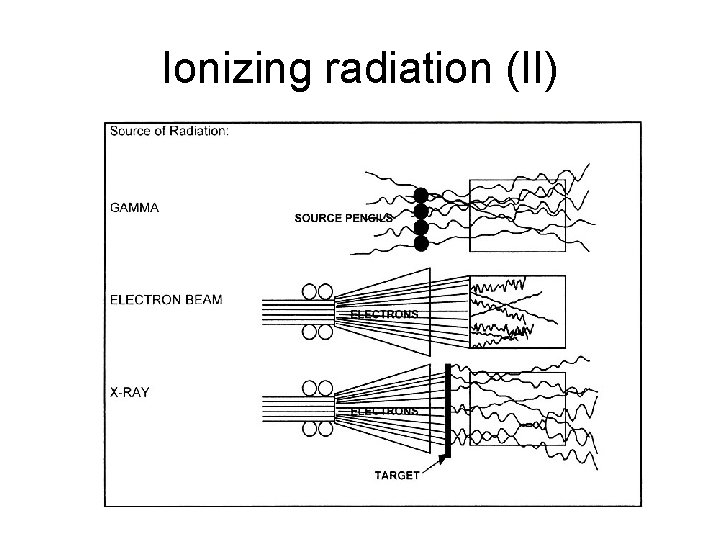
Ionizing radiation (II)
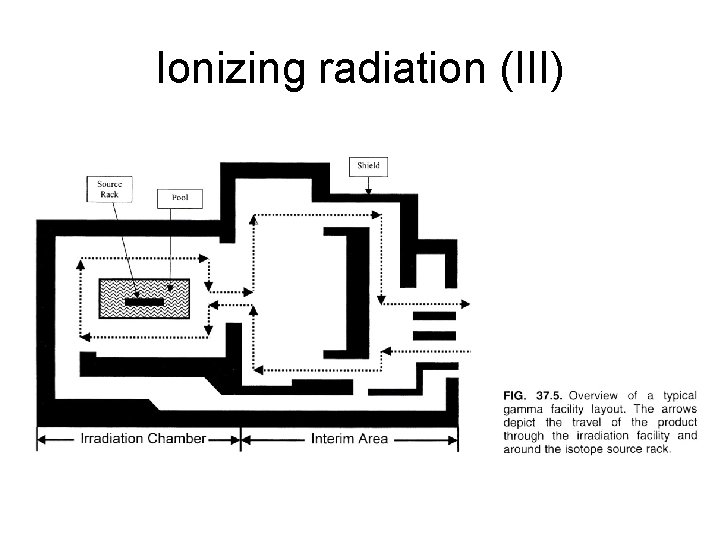
Ionizing radiation (III)
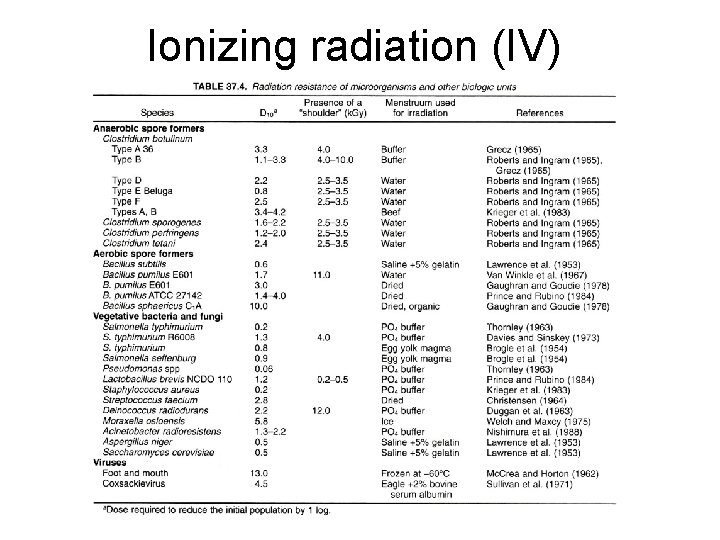
Ionizing radiation (IV)

Ionizing radiation (V)
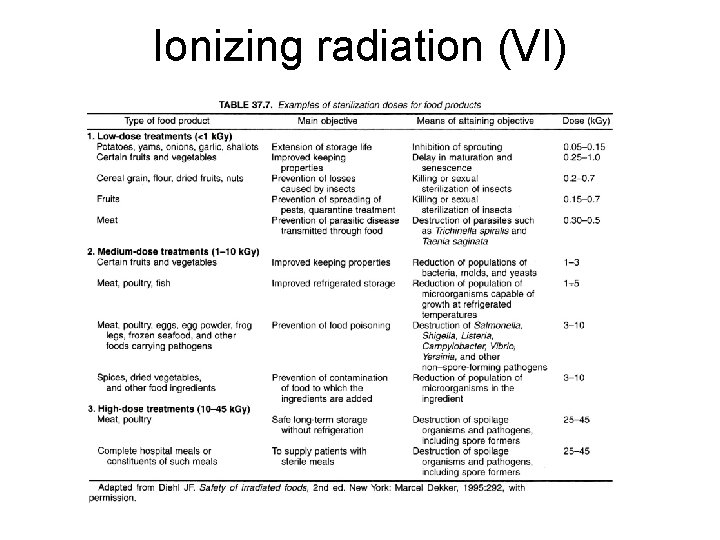
Ionizing radiation (VI)

Gaseous chemical sterilization (I) • Alkylating agents (Alkylation of DNA) – – Ethylene oxide Propylene oxide Formaldehyde Beta-propiolactone • Oxidizing agents (Oxidation of proteins and nucleic acids) – – Hydrogen peroxide Peracetic acid Chlorine dioxide Ozone

Gaseous chemical sterilization (II)
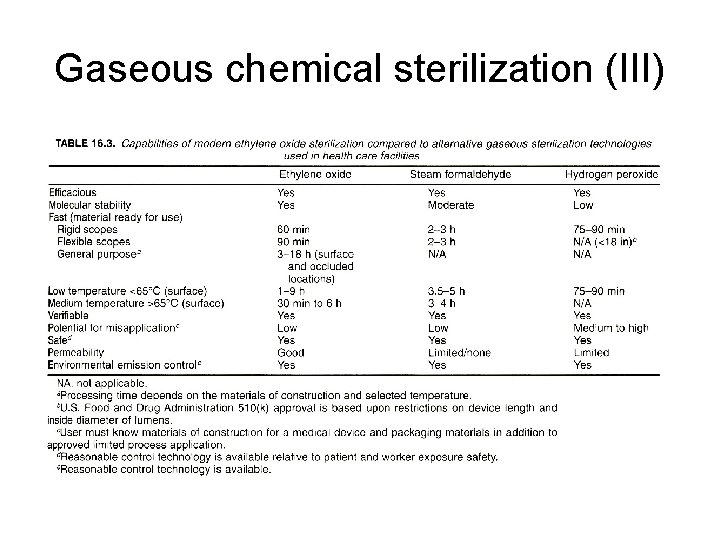
Gaseous chemical sterilization (III)

(Chemical) disinfectants

Surface-active agents • Amphiphilic compounds • Anionic, cationic, nonionic, and amphoterics • Cationic surfactants: Quaternary ammonium compounds – Basic structure: • One nitrogen atom • Four carbon atoms covalently linked to the nitrogen atom • An anion eletrostatically linked to the nitrogen atom – Mechanism: Protein denaturation, enzyme inhibition, and disruption of cytoplasmic membrane
Quaternary ammonium compounds (I)
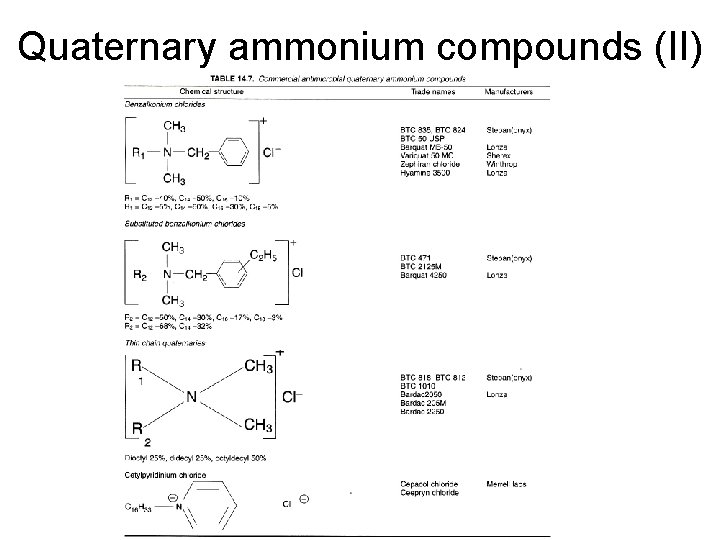
Quaternary ammonium compounds (II)

Quaternary ammonium compounds (III)

Quaternary ammonium compounds (III) • Advantages – – Low toxicity Low corrosivity Stable at high temperature and wide p. H range Relatively tolerable with organic load • Disadvantage – Not effective against viruses, protozoa, and spores – Less effective at low temperature – Inhibited by most anionics and hard water salts

Quaternary ammonium compounds (IV) (applications) • General surface disinfectant • Industrial application (hot water in large commercial laundry) • Swimming pool water? ? ? Drinking water (emergency situation)? ? ?

Phenol compounds (I) • Structure • Mechanism – Bacteria: denaturation of proteins, inhibition of enzymes, damages on plasma membrane – Viruses and fungi: Unknown
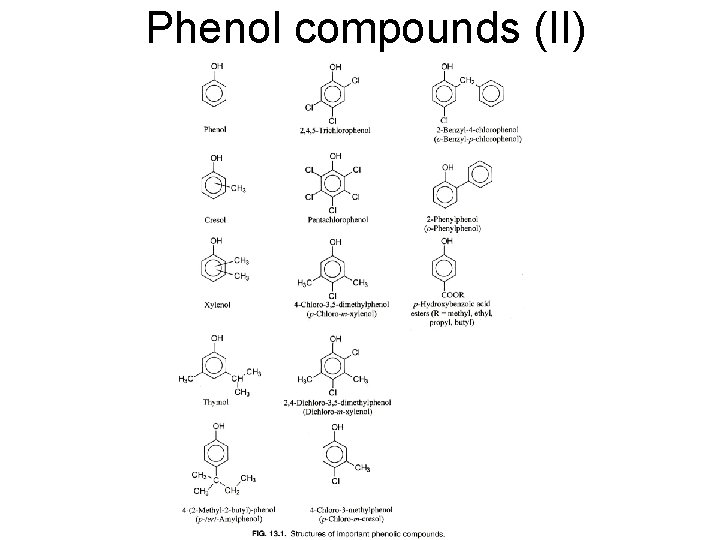
Phenol compounds (II)
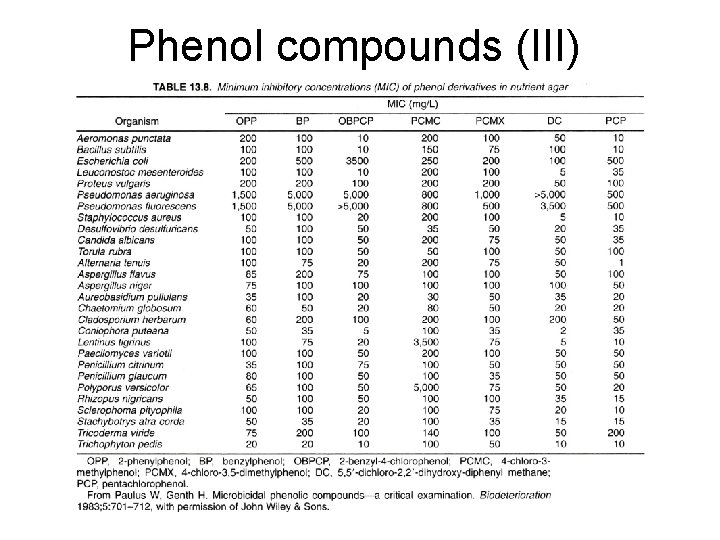
Phenol compounds (III)

Phenol compounds (IV) • Advantages – Effective against viruses, bacteria, and fungi – Stable in concentrate – Tolerable for organic load and hard water • Disadvantages – – Not effective against spores High toxicity Not effective at low temperature Incompatible with nonionic and cation surfactants

Phenol compounds (IV) • General surface disinfectant • Gemicidal soaps and lotions, antiseptics, preservatives in cosmetics, and mouthwash preparation (Listerine)

Iodine and iodine compounds (I) • Chemistry • Antimicrobial agents: I 2 (free iodine), HOI (hypoiodous acid), H 2 OI+ (Iodine cation) • Mechanism: protien denaturation, damages in plasma membrane, and nucleic acid breakdown
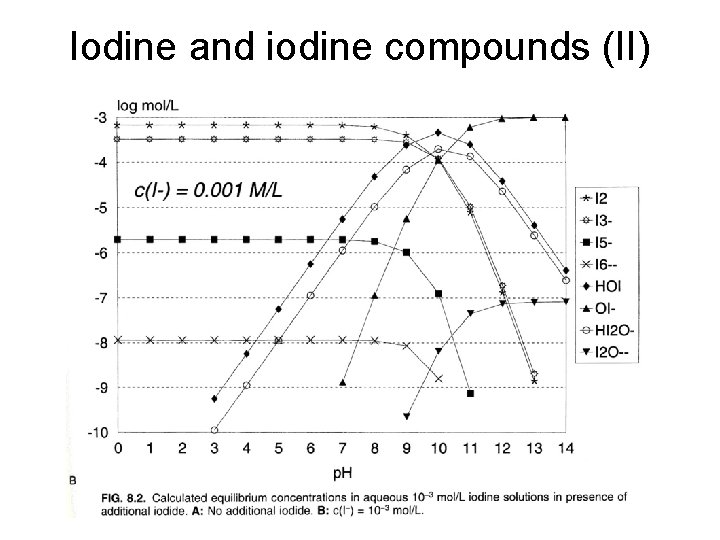
Iodine and iodine compounds (II)

Iodine and iodine compounds (III) • Advantage – Effective against viruses, bacteria, fungi, protozoa and bacterial/fungal spores – Low toxicity – Very stable in concentrate • Disadvantages – – Chemical hazard (staining) Less effective at low temperature Relatively corrosive at high temperature Expensive
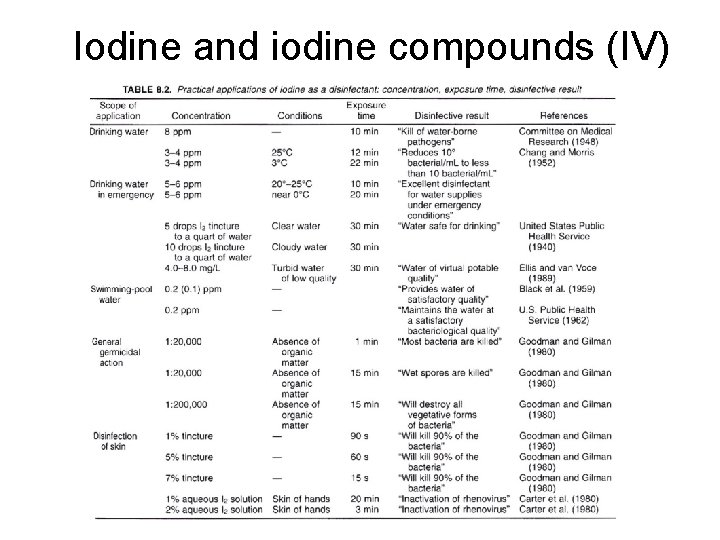
Iodine and iodine compounds (IV)

Peroxygen compounds (I) • Hydrogen peroxide, peracetic acids, perfomic acid, and perpropionic acids • Mechanism: hydroxyl radicals: proteins, lipids, and DNA • Very effective on most microbes including spores
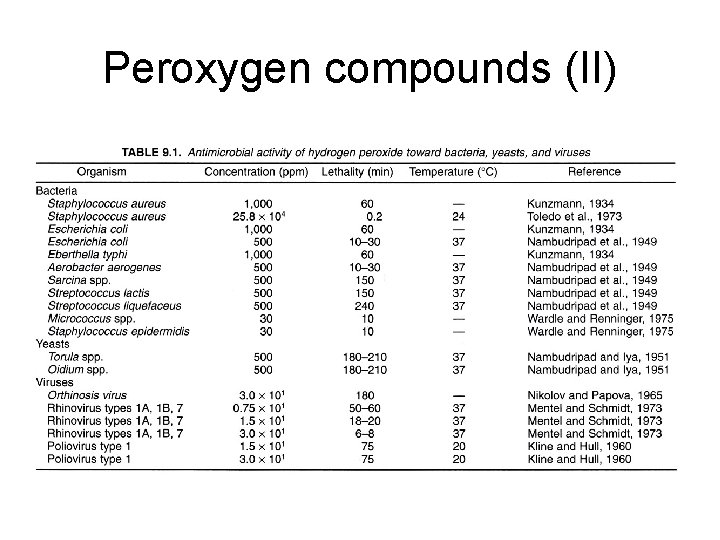
Peroxygen compounds (II)
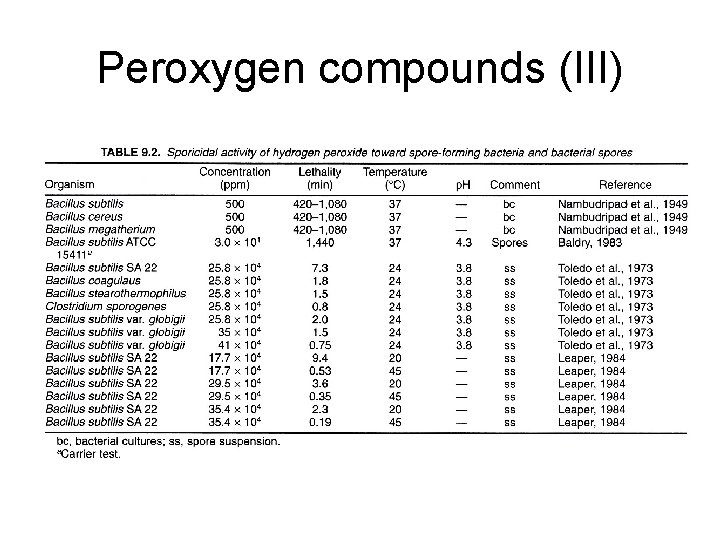
Peroxygen compounds (III)
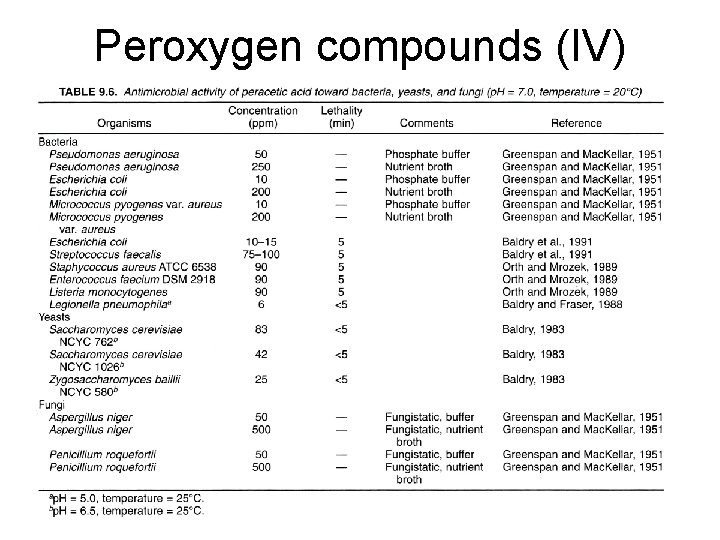
Peroxygen compounds (IV)
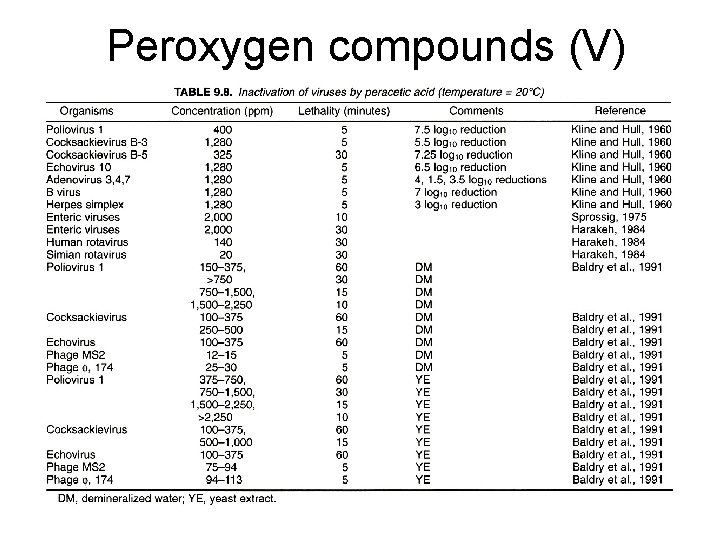
Peroxygen compounds (V)

Peroxygen compounds (VI) • Advantages – – – Strong, fast-acting Effective against most microbes including spores No toxicity No environmental concern Effective over wide p. H (up to 7. 5) and temperature ranges (40 o. F – 150 o. F) – Stable in concentrate – Tolerable for organic load • Disadvantages – Limited stability at use solution – Corrosive on soft surfaces (brass, copper, and mild and galvanized steel)

Peroxygen compounds (VII) • Excellent surface disinfectants • Industrial water systems (Legionella control) • Wastewater disinfection • Antiseptics • Cold sterilization of phamatheuticals (emulsions, hydrogels, ointments, and powders)

To be continued
- Slides: 40