Indicators Indicators w Indicators are a substance that

Indicators

Indicators w Indicators are a substance that change color in the presence of (whatever they check for) w They do this because of Le Châtelier’s principle. All you need an equilibrium reaction with different colored products and reactants. w The pen used to check for counterfeit money is a starch indicator

How an acid base indicator works w A generic indicator will follow this reaction, HId is the reactant indicator, and ID- is its product w H[ID] + H 2 O H 3 O+ + [ID]- w The color differences are important, HId is one color and Id- is a different color! w in an acidic solution (high H 3 O+) you see reactant w H[ID] + H 2 O H 3 O+ + [ID]- w in a basic solution (low H 3 O+) you see product + w H[ID] + H O + [ID] 3 2

Acid Base indicators w Acid base indicators change color at certain p. H levels w They don’t have to change at 7 (most don’t) w Universal indicator solution (phenolphthalein, bromthymol blue and methyl red dissolved in ethanol and water) changes color at each integral p. H value
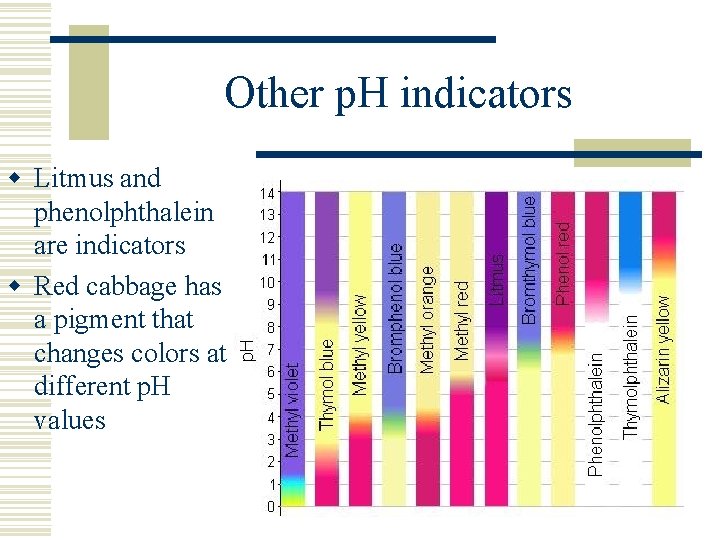
Other p. H indicators w Litmus and phenolphthalein are indicators w Red cabbage has a pigment that changes colors at different p. H values

Buffers w Buffers are solutions that don’t change in p. H when acids or bases are added. w They use weak acids/bases and Le Châtelier’s principle. w WA = weak acid w HWA + H 2 O H 3 O+ + WA-

What it does w p. H = mol H 3 O+/L H 2 O w adding H 3 O+ should increase [H 3 O+] w However, this forces the equation to the left, decreasing H 3 O+ and increasing H 2 O w so the [H 3 O+] remains constant
![w Removing H 3 O+ (adding a base) should decrease [H 3 O+] w w Removing H 3 O+ (adding a base) should decrease [H 3 O+] w](http://slidetodoc.com/presentation_image_h/330da8abf4cfba666d0ce64e918285ee/image-8.jpg)
w Removing H 3 O+ (adding a base) should decrease [H 3 O+] w However, this forces the equation to the right, increasing H 3 O+ and decreasing H 2 O w So again, there is no change to [H 3 O+] w There is a breaking point where the p. H will change.

What does this have to do with my life? w Your blood is a buffered solution w The p. H must remain between 7. 35 -7. 45 w Outside of that range can kill you w below this range is called acidosis w above is called alkalosis

Buffered Products w medications (Bufferin) w Shampoos, body soaps w All are buffered to be near your body p. H so they won’t cause a major disruption.
- Slides: 10