Increasing Access to Next Generation Injectables Working Meeting

Increasing Access to Next Generation Injectables Working Meeting Dakar, Senegal Purpose and Process of WHO guideline development James Kiarie Coordinator Department of Reproductive Health & Research, WHO

WHO Guidelines, Derivative Documents & tools Family Planning Medical Eligibility Criteria 5 th edition 2015 Selected Practice Recommendations 3 rd edition in 2016 The Medical Eligibility Criteria (MEC) Wheel (new) Guide to family planning for community health care providers and their clients (to be updated) Ensuring Human Rights with Contraceptive Service Delivery (An Implementation Guide) Decision-Making Tool 2 (to be updated) Global Handbook To be updated in 2017

The Medical Eligibility Criteria for Contraceptive Use q q 3 Filename WHO’s Evidence-based guidance to develop and implement family planning guidelines for national programmes. Provides thorough information and guidance on the safety of various contraceptive methods for use in the context of specific health conditions and characteristics.

WHO MEC guidance on use of hormonal contraception for women at high risk of HIV q 2014 guidance: no restriction on any HC method, with a clarification for progestin-only injectables: – “Women at high risk of HIV infection should be informed that progestogen-only injectables may or may not increase their risk of HIV acquisition. Women and couples at high risk of HIV acquisition considering progestogen-only injectables should also be informed about and have access to HIV preventive measures, including male and female condoms. ” q 4 2017 guidance: expected soon, stay tuned!

MEC REVIEW CIRE Continuous Identification and Review of Evidence GSG Guideline Steering Group GDG Guideline Development Group WHO Approves and Issues Guideline 5 Review and synthesis of published literature may recommend systematic review or refer to GSG Assessment of the evidence and decision No review, immediate review or at next review of MEC Review evidence and recommendations based on GRADE (Grades of Recommendation Assessment, Development and Evaluation) Approval by the Guideline Steering Committee (GRC) and the ADGO

Quality of the Evidence The extent to which one can be confident that an estimate of effect or association is correct. High, further research is very unlikely to change our confidence in the estimate of effect • Moderate, further research is likely to have an important impact on our confidence in the estimate and may change the estimate • Low, further research is very likely to have an important impact on our confidence in the estimate and is likely to change the estimate • Very low, any estimate of effect is very uncertain • 6

MEC Recommendations 1. 2. 3. quality of available evidence desirable and undesirable effects values and preferences q q 7 Category 1: no restriction on use Category 2: advantages generally outweigh theoretical or proven risks Category 3: theoretical or proven risks usually outweigh advantages Category 4: unacceptable health risk

WHAT NEXT q q q 8 Communication of WHO statement on HC and HIV Introduction of guidance into National guidelines and practice The ECHO trial Filename

Communication of WHO statement on HC and HIV Publication in early March on WHO website: http: //www. who. int/reproductivehealth/publications/gen eral/rhr_09_02/en/ q – Full statement 10 -15 pages – Web story – FAQ – Press release Pre release webinars with stakeholders, Ministries of Health and UN organizations that will be expected to respond to the issue q Key objective is to get out the an accurate message q 9 Filename

Introduction of guidance into National guidelines and practice q q q 10 Will be supported by WHO AFRO and WHO HQ Planning meetings of WHO Country offices and ministries of health from April 2017 Key objective is for program managers, providers and clients to receive correct evidence based message Filename

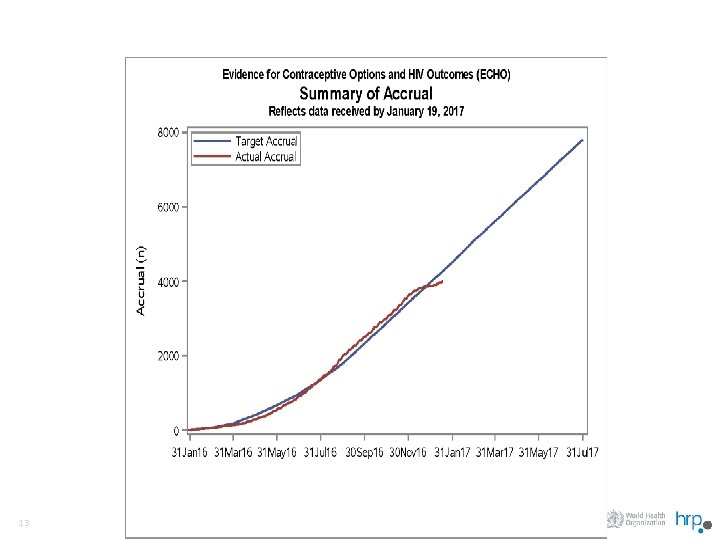
Update on the Evidence for Contraceptive Options and HIV Outcomes (ECHO) Trial A Multi-Center, Open-Label, Randomised Clinical Trial Comparing HIV Incidence and Contraceptive Benefits in 7800 Women using Depot Medroxyprogesterone Acetate (DMPA), Levonorgestrel (LNG) Implant, and Copper Intrauterine Devices (IUD) q A randomized trial, if done well, wil: – Provide clear guidance for policymakers and programs; – Help to formulate clear counselling messages for clinicians; and – Permitting women to make fully informed choices. 11

ECHO Team Currently funded by the Bill & Melinda Gates Foundation, the Swedish International Development Cooperation Agency, the Medical Research Council of South Africa, and the United Nations Population Fund, Contraceptive supplies supported by the Government of South Africa and the United 12 States Agency for International Development.
13

Thank you For more information, Follow us on Twitter @HRPresearch @jkiarie 0733 Website who. int/reproductivehealth 14
- Slides: 14