Incidental Findings in Medical Imaging Ethical Considerations Best

Incidental Findings in Medical Imaging Ethical Considerations, Best Practices, and Lessons From Genomics Jake Bowling UNC School of Medicine Class of 2019 September 2018

Module Objectives This module will: 1. Define the term “incidental finding” and surrounding terminology. 2. Provide relevant clinical case examples. 3. Review the current radiology literature on incidental findings. 4. Define core principles in medical ethics and explore the ethical considerations of managing incidental findings. 5. Consider lessons from genomics, a field with many parallel considerations.

What is an “Incidental Finding”? • The term incidental finding (IF) refers to potentially significant medical information that is discovered unintentionally during an examination done for other purposes. • IF’s occur in a variety of settings, including routine medical care, clinical imaging, biomedical research, autopsy, and genetic testing. • Incidental imaging findings are commonly called “incidentalomas” because of their potential representation of cancerous masses or other dangerous lesions. • The American College of Radiology defines an incidental imaging finding as “an incidentally discovered mass or lesion, detected by CT, MRI, or other imaging modality performed for an unrelated reason. ”

A Growing Problem • Trends in utilization of diagnostic imaging vary by setting and modality. Rates have broadly declined since 2008 (Horný et al. , 2015), though some new evidence suggests an uptick since 2015 (Levin et al. , 2017). These trends are very difficult to study & interpret as many factors are influential. • However, it is clear that imaging resolution is rapidly increasing across modalities. With this “march of progress” comes an increasing ability to detect incidental findings (Lumbreras et al. , 2010). • In addition to this, our aging population contributes to higher rates of incidental findings over time (Pinato et al. , 2012).
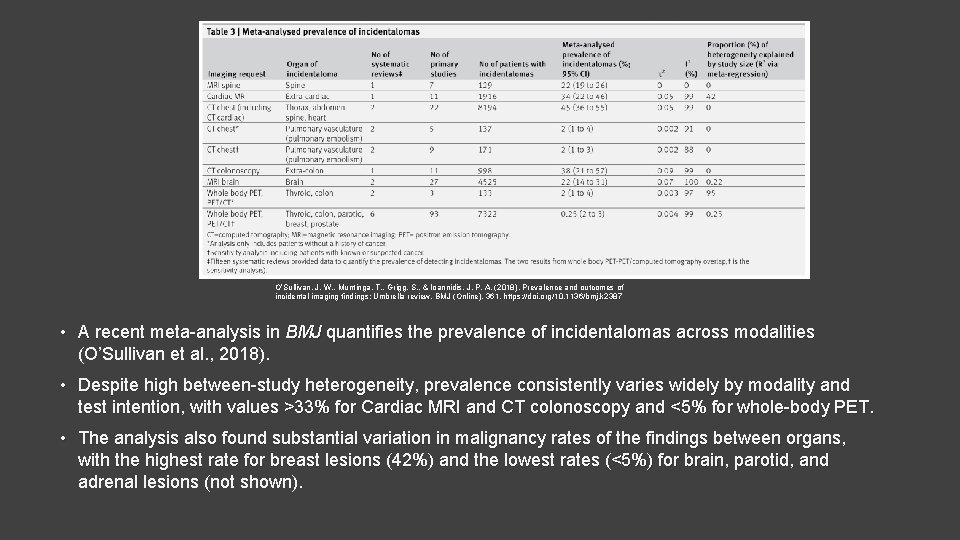
O’Sullivan, J. W. , Muntinga, T. , Grigg, S. , & Ioannidis, J. P. A. (2018). Prevalence and outcomes of incidental imaging findings: Umbrella review. BMJ (Online), 361. https: //doi. org/10. 1136/bmj. k 2387 • A recent meta-analysis in BMJ quantifies the prevalence of incidentalomas across modalities (O’Sullivan et al. , 2018). • Despite high between-study heterogeneity, prevalence consistently varies widely by modality and test intention, with values >33% for Cardiac MRI and CT colonoscopy and <5% for whole-body PET. • The analysis also found substantial variation in malignancy rates of the findings between organs, with the highest rate for breast lesions (42%) and the lowest rates (<5%) for brain, parotid, and adrenal lesions (not shown).

Isn’t More Information Always Good? • The upsides are obvious: increased detection and reporting of IF’s can and has saved lives by prompting early interventions. But that potential must be weighed against numerous potential costs. 1. Financial: Workup of incidentalomas uses increasing amounts of health care resources, and has the potential to affect patients’ insurance status. • For example, a May 2009 CMS noncoverage decision for CT colonography cited a high rate of incidental findings leading to expensive workup (Jensen et al. , 2009). 2. Physical: diagnostic workup and intervention is not without physical risk to patients. 3. Emotional: information asymmetry in healthcare means that even the most benign incidentalomas will cause patients significant anxiety (Kole, J. , & Fiester, A. , 2013). Uncertainty in management causes provider anxiety as well.

Key Ethical Principles • Beauchamp & Childress (2001) put forth an influential framework of four nonhierarchical core principles to be weighed against one another with attention paid to the scope of each: 1. Autonomy: the right of a patient to refuse or choose their treatment. 2. Beneficence: the value of doing good, or acting in the patient’s best interest. 3. Non-maleficence: ”Do no harm”. 4. Justice: Scarce health care resources should be distributed fairly (e. g. , treating similar cases similarly). • Additional values are commonly considered: • Respect for persons: concept of all persons having a right to autonomy (physicians, family members, etc. ) • Utility: the value of promoting more good than harm, on the balance. • Confidentiality of patient-provider relations. • Truthfulness: a component of informed consent, a value of not withholding information or promoting misinformation.

Case 1 • Joan, a healthy 30 year old female, presents to the E. D. mid-day after 6 hours of nausea, vomiting, bloating, and lower-quadrant abdominal pain. She at a new seafood restaurant last night. • Her provider orders a contrasted abdominal CT to rule out appendicitis. She agrees to the exam after receiving information about potential contrast reactions and about risks of radiation. • Her CT is negative for appendicitis, but does reveal this small lesion which is noted in the radiology report as a “small circumscribed lesion of unknown significance”. • Joan is discharged with Zofran and a copy of the imaging report and is told to follow up with her PCP on the finding. She tries to obtain more information about the finding, clearly fixated on the word “lesion”, but her providers have moved on to other patients. Image courtesy of A. Prof Frank Gaillard, <a href="https: //radiopaedia. org/">Radiopaedia. org</a>. From the case <a href="https: //radiopaedia. org/cases/2600">r. ID: 2600</a>
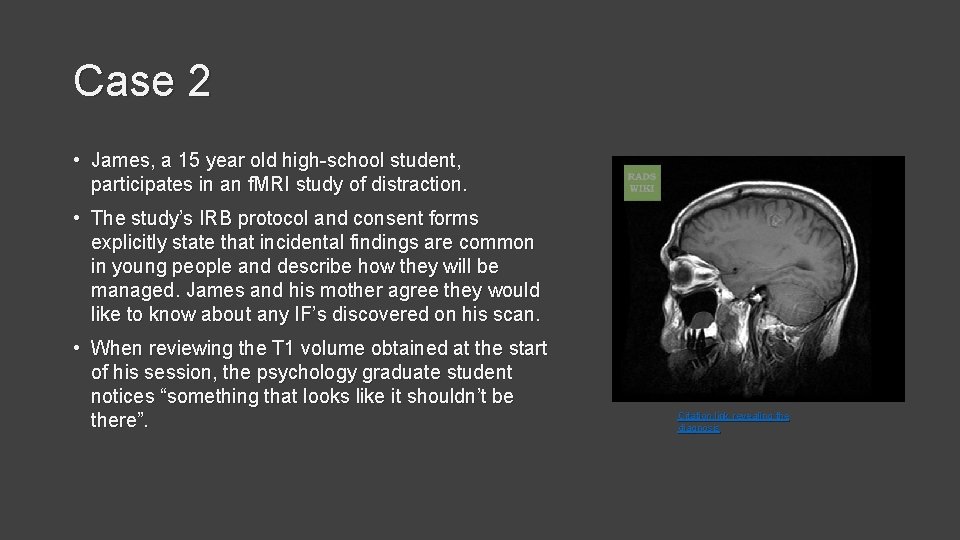
Case 2 • James, a 15 year old high-school student, participates in an f. MRI study of distraction. • The study’s IRB protocol and consent forms explicitly state that incidental findings are common in young people and describe how they will be managed. James and his mother agree they would like to know about any IF’s discovered on his scan. • When reviewing the T 1 volume obtained at the start of his session, the psychology graduate student notices “something that looks like it shouldn’t be there”. Citation link revealing the diagnosis

Practical Considerations • As the cases demonstrate, the optimal and ethical handling of individual IF’s depends on many factors specific to the situation. 1. Patient preferences 2. Pretest probability of the patient for potential abnormal findings 3. Specific test performed/modality used 4. The finding itself: appearance, organ, distribution 5. Test setting: clinical (inpatient versus outpatient), research 6. Patient age (minor versus adult) 7. Contractual & legal obligations: IRB protocol, consent form, local statute 8. Presence/absence of published consensus guidelines or “standard of care”

Efforts Within Radiology • The largest effort within clinical radiology toward consensus in management of IF’s is the ACR’s Incidental Findings Committee (IFC). • Starting within the field of Body Imaging in 2006, the IFC was formed to develop guidelines for the management of IF’s analogous to existing guidelines for pulmonary nodules and other findings (Berland et al. , 2010). • The committee currently cites the following five goals of its efforts (IFC main page): 1. Reducing risks to patients from additional unnecessary examinations, including the risks of radiation and risks associated with interventional procedures; 2. Limiting the costs of managing incidental findings to patients and the health care system; 3. Achieving greater consistency in recognizing, reporting, and managing incidental findings, as a component of formal quality improvement efforts; 4. Providing guidance to radiologists who are concerned about the risk for litigation for missing incidental findings that later prove to be clinically important; and 5. Helping focus research efforts to lead to an evidence based approach to incidental findings.

IFC White Papers • The IFC has published 15 white papers on various significant IF’s to date. The current list of papers is available here: https: //publish. smartsheet. com/42 d 18 e 874 a 164318 a 0 f 702481 f 2 fbb 70 • Topics include: 1. Abdominal/Pelvic CT/MRI: adrenal, liver, pancreas, biliary, splenic, nodal, adnexal, vascular findings 2. Head & Neck CT/MRI/PET: thyroid nodules, pituitary findings 3. Thoracic CT: mediastinal and cardiovascular findings • The IFC maintains a proposal form where anyone can propose a new IF of-interest for the committee to study https: //app. smartsheet. com/b/form/2 b 87 c 01 e 7 f 2 a 4 b 13 ac 911 f 43 c 5 efca 93

The Case for a Revised Consent Process • Kole & Fiester (2013) argue that a “more robust consent process is needed to enable patients to anticipate incidentalomas and to correctly perceive the associated risks”. • Informed consent has a narrow legal meaning, but more commonly invokes an ethical duty to ensure a patient’s substantial understanding of an intervention including short- and long-term consequences, alternatives, a risk -benefit analysis, and provider recommendations. • While practices vary among institutions, it is not standard practice to discuss the potential for IF’s with patients before common imaging tests. • A 2006 survey of 113 radiology chairpersons asked what risks their department explains to a patient before a CT (Lee et al. , 2006). • 66% of institutions had consent guidelines in place. 84% of respondents reported regular consenting about contrast reactions, 15% for radiation risk, and 10% for risks to pregnant women and nephrotoxicity. 0% of respondents reported consenting for incidental findings.

The Case for a Revised Consent Process • Kole & Fiester’s arguments for a revised consent process include: • Consenting for IF’s is routine for non-diagnostic imaging of research subjects. This is supported by international multidisciplinary guidelines, such as those from the Council for International Organizations of Medical Sciences in 2002. United States law dictates that for research, “informed consent requires a description of all reasonably foreseeable risks and benefits” (Wolf et al. , 2008). • Evidence from research imaging suggests a large majority of persons still favor imaging and receiving ”any and all” information obtained after being consented about IF’s (Kirschen et al. , 2006). • Many types of IF’s have a high enough prevalence and large enough foreseeable consequences that information about them must be disclosed under the “reasonable person” consent standard (Beauchamp & Childress, 2001). • Incorporating potential IF’s into standard consent forms judiciously (i. e. , only for tests with highly prevalent rates of findings, such as cardiac MRI) would be minimally disruptive to institutional proceedings.

Direct Reporting of Findings • Radiologists’ favorability of directly disclosing imaging findings to patients has followed a sliding scale based on severity of findings; most are in favor for normal results, while support falls for severe findings (Levitsky et al. , 1993). • Amber & Fiester (2013) argue that when a direct provider-patient relationship has been entered, radiologists have ethical (and likely legal) obligations to disclose all severities of findings. • They propose a paradigm for disclosure whereby the obligation for disclosure follows a sliding scale based on the diagnostic certainty of the finding rather than the perceived severity. • Evidence on different types of IF’s thus guides the obligation for disclosure. Findings which are exceedingly benign and do not require follow up are a prime opportunity for disclosure. IF’s with less data on implications are better left to referring clinicians, who have access to more clinical data to correlate with the finding.

Research: A Special Case • IF’s are also commonly discovered during research studies that use medical imaging modalities. • However, research imaging protocols typically differ from clinical protocols, leading to incomplete characterization of IF’s. • Further, research data sets are commonly analyzed by people not trained in radiologic diagnosis, and are re-analyzed by secondary researchers years later, complicating the management of IF’s in research. • A tangential literature exists on the management of IF’s in human subjects research. • It focuses largely on MRI Brain, which is commonly used for nonclinical purposes because of its extra-clinical applications and relative safety for participants. • Prevalence rates of IF’s in research unsurprisingly are comparable to those for tests done in clinical settings. MRI Brain studies report a prevalence of 13 -84%, and CT colonography studies 15 -89% (Wolf et al. , 2008).

Research: A Special Case • A researcher’s obligations under current standards differ from those of a radiologist working in a clinical setting. • First, several international laws govern a researcher’s obligations to promote the health, well-being, and rights of participants, to obtain voluntary and thorough informed consent, and to comply with Institutional Review Board and Compliance Assurance policies. • In the U. S. , these include the Nuremberg Code (1947), the Declaration of Helsinki (1964), and the Federal Policy for the Protection of Human Subjects (1979). • Also, there is near-unanimous agreement that protocols for managing IF’s should be addressed in the research design, subject to IRB approval, and outlined in the consent process (King 2018). • Several groups, including a Working Group on Incidental Findings sponsored by the NIH, have produced guidelines for researchers for the management of IF’s (Illes et al. , 2008, Wolf 2008). A large burden falls on the IRB and the consent process.

Research: Unanswered Questions • Despite legal and consensus agreement that IF’s should be thoroughly addressed in human subjects research, many implementation details remain unanswered. • One relevant one is: should research teams be required to include a trained radiologist? If not, when should they decide it is appropriate to seek consultation for an IF? • f. MRI, for example, is often being used by a Ph. D. or graduate student in psychology or a related field without training in radiologic diagnosis. • On this question, researchers’ opinions range widely, from mandatory radiologic review of all scans (Phillips et al. , 2015) to only seeking radiologic consultation for “obviously life-threatening” findings (Royal & Peterson, 2008). • Practices will evolve as more data emerges on specific rates of different IF’s with specific imaging protocols.

Pediatrics: A Special Case • IF’s in children represent a special case because of the dual rights of children and their parents/guardians. • In addition, young children experience different pathologies than adults, and thus the IF risk-benefit calculation changes. • Though the ACR’s IF Committee has not commented to-date, some reports have attempted to quantify pediatric IF’s. • In the Netherlands Generation-R study, 3, 966 children from a general pediatric population (mean age 10. 1, 8. 6 -11. 9) underwent 3 T MRI which was read by a Neuroradiologist (Vernooij et al. , 2017). • An IF was present in 25. 6% of children, though it required follow-up in only 0. 43% of children. • Common findings included pineal gland arachnoid cysts, and developmental venous abnormalities.

Pediatrics: A Special Case • In clinical and research settings, pediatric IF’s should be addressed in the consent process on a situational basis just as with adults. • In research, federal regulations broadly use a “minimal risk” standard for acceptable studies, and potential for IF’s should be considered as contributing to such (Wolf et al. , 2008). • Ideally, the adolescent and their caretakers will agree on a desire to receive information about IF’s. However, Wolf and co-authors (2008) consider the situation where wishes conflict. • When an IF is “grave or life-threatening”, a child’s asserted right not-to-know should be overridden. • Less ominous IF’s will require “case-by-case evaluation” and potential consultation with a hospital ethics committee. • IF’s suggesting sensitive conditions, such as substance abuse, pregnancy, or physical abuse, should be given special consideration in minors.

Genomics • Genomics is the interdisciplinary science focused on the structure, function, mapping, and editing of genomes, an organism’s complete set of DNA. • High-throughput sequencing (HTS) methods, such as Sanger Sequencing, have become broadly available in the last two decades, allowing rapid sequencing of entire genomes and exomes. • The large amount of unfiltered data generated by genome sequencing comes with increased detection of IF’s, more commonly called “unsolicited” or “secondary” findings in the genomics literature. • For example, a sequencing done for personalized pancreatic cancer treatment may reveal a Lynch Syndrome mutation. • In genomics, variants discovered that are poorly understood are termed “Variants of Uncertain Clinical Significance (VUS). ” • Depending on the thresholds and modalities used, prevalence rates for IF’s can be quite similar in the two fields.

Genomics & Imaging: A Shared Ethic • Wouters et al. (2017) argue for a need for a combined ethical debate between the two fields, to preserve the public trust and ensure people perceive healthcare policies as fair across domains. • Management of IF’s are debated in both literatures in an isolated way, though each uses the other in analogies when justifying policies.

Genomics & Imaging: A Shared Ethic • Wouters et al. (2017) identify six ethical issues that pertain to both medical imaging and genetics. 1. Terminology • Varying terms in the literatures, such as “incidental” findings, “secondary” findings, “unsolicited” findings, and “variants of uncertain significance” convey subtly different meanings to patients. 2. Benefits and Risks • The same risk-benefit calculation applies to both fields. Revealing actionable or life-saving information versus causing undue anxiety, financial hardship, or physical harm through unnecessary treatment. • In genomics in particular, findings are also relevant to genetic relatives.

Genomics & Imaging: A Shared Ethic 3. Autonomy and Consent • The consent process is traditionally more explicit and thorough with regard to IF’s in genomics. • Ethicists have argued for a tiered consent process in which findings are divided into categories (Dondorp & De Wert, 2013) and decisions are staggered over time (Newson 2017), to minimize the effects of information overload and ensure people make decisions in line with their true values. • Consideration of the autonomy of genetic relatives is more critical in genomics. 4. Return of Adult-Onset Findings in Children • Genetic variants are innate, though they often do not cause disease until later in life. This applies to some, but not all, radiologic findings. • The ACGM has recommended an intentional search for and disclosure of a list of actionable results for both adult- and child-onset conditions, with an opt-out option. This represents an all-or-nothing approach to disclosure (Green et al. , 2013). • Applying the influential ethical argument for a child’s “right to an open future” (Feinberg, 1980) would suggest that in certain situations, adult-onset findings should be withheld for a time from the child (though delayed disclosure does raise technical difficulties related to the storage of the information).

Genomics & Imaging: A Shared Ethic 5. Uncertainty • In both fields, the clinical significance of a finding is commonly unclear. • A distinction can be made between informational and inherent uncertainty (Babrow et al. , 1998). • Informational uncertainty comes from a gap in data that will foreseeably be addressed with further research, such as the prevalence of a certain finding on abdominal CT. • Inherent uncertainty can be reduced with more precise testing methods, but cannot be eliminated. • Both types of uncertainty should be addressed in the consent process. 6. Filters • Imaging IF’s are often readily visible to radiologists (e. g. , a large mass), but genetic variants are usually discovered by computers. • Arguments can be made for a duty to search for certain IF’s, but once one has been found moral obligations to act change. • This seemingly inherent distinction between imaging and genetics may become blurred as artificial intelligence systems are implemented in clinical radiology, which could be programmed to search for or ignore various IF’s.

Case 1 Revisited • Joan’s abdominal CT done to rule out appendicitis revealed an adrenal mass. • In 2017, the ACR’s Incidental Findings Committee released a paper on management of adrenal masses incidentally detected on CT or MRI (Mayo-Smith et al. , 2017). • The paper includes a management algorithm (right), in which features including size, features such as presence/absence of calcification, patient cancer history, and progression from prior imaging are considered toward actions of various further workup versus no follow-up needed. Image courtesy of A. Prof Frank Gaillard, <a href="https: //radiopaedia. org/">Radiopaedia. org</a>. From the case <a href="https: //radiopaedia. org/cases/2600">r. ID: 2600</a>

Case 1 Revisited: Areas for Improvement • Abdominal CT carries a high risk of incidental findings in general, and 3 -7% of adults are estimated to have incidental adrenal masses, most of which are benign. • The possibility of incidental findings should be addressed in Joan’s consent process. Ideally this would be written into hospital protocol. • Because of a relatively low level of informational uncertainty in this case (a management algorithm exists), disclosure of the IF is indicated. The radiology report should cite the IFC’s paper and include the recommended follow-up action, if any. Image courtesy of A. Prof Frank Gaillard, <a href="https: //radiopaedia. org/">Radiopaedia. org</a>. From the case <a href="https: //radiopaedia. org/cases/2600">r. ID: 2600</a>

Case 2 Revisited • James’ research scan revealed an asymptomatic cavernous venous malformation. • His study’s research team did not include a radiologist, but, as outlined in the study’s IRB, his images were sent to a predetermined Neuroradiology division for further analysis. • In accordance with their wishes, James and his mother received a copy of the radiology report, and eventually contacted neurosurgery, who recommended conservative management. https: //www. wikidoc. org/index. php/File: Cavernousmalformation-05. jpg

Takeaways • Rates vary widely by modality and population, but incidental findings are common and will be encountered frequently. • A robust consent process including the possibility of IF’s and standardized reporting procedures on their management when available are beneficial to patients. • Obligations differ in clinical and research contexts, and in the case of minors. • Literature on IF’s is emerging in radiology, in bioethics, and in genomics and should be consulted to guide best practices.

Suggested Readings • ACR Incidental Findings Committee Papers: • https: //publish. smartsheet. com/42 d 18 e 874 a 164318 a 0 f 702481 f 2 fbb 70 • Wolf, et al. “Managing Incidental Findings in Human Subjects Research- Analysis and Recommendations” • https: //www. ncbi. nlm. nih. gov/pubmed/18547191 • Brown, Stephen, “Professional Norms Regarding How Radiologists Handle Incidental Findings” • https: //www. ncbi. nlm. nih. gov/pubmed/23545084 • O’Sullivan, et al. “Prevalence and outcomes of incidental imaging findings: umbrella review” • https: //www. bmj. com/content/361/bmj. k 2387 • Kalia, et al. “Recommendations for reporting of secondary findings in clinical exome and genome sequencing, 2016 update (ACMG SF v 2. 0): a policy statement of the American College of Medical Genetics and Genomics” • https: //www. nature. com/articles/gim 201619

References • Amber, I. , & Fiester, A. (2013, March). Communicating findings: A justification and framework for direct radiologic disclosure to patients. American Journal of Roentgenology. https: //doi. org/10. 2214/AJR. 12. 9468 Roentgenology. https: //doi. org/10. 2214/AJR. 12. 9468 • Babrow, A. S. , Kasch, C. R. , & Ford, L. A. (1998). The Many Meanings of Uncertainty in Illness: Toward a Systematic Accounting. Health Communication, 10(1), 1– 23. https: //doi. org/10. 1207/s 15327027 hc 1001_1 Communication, 10 • Beauchamp T. L. , & Childress, J. (2001). Principles of Medical Ethics. Oxford University Press, 454. Retrieved from http: //books. google. co. uk/books? id=_14 H 7 MOw 1 o 4 C&lpg=PR 9&ots=1 u. Zk 3 Kzp. Tx&dq=Beauchamp and Childress; Principles Biomedical Ethics, OUP, 5 th edition 2001&lr&pg=PP 1#v=onepage&q&f=false • Berland, L. L. , Silverman, S. G. , Gore, R. M. , Mayo-Smith, W. W. , Megibow, A. J. , Yee, J. , … Taylor, A. J. (2010). Managing incidental findings on abdominal CT: White paper of the ACR incidental findings committee. Journal of the American College of Radiology. Elsevier. https: //doi. org/10. 1016/j. jacr. 2010. 06. 013 • Council for International Organizations of Medical Sciences & World Health Organization. International ethical guidelines for biomedical research involving human subjects. , Bulletin of medical ethics § (2002). Retrieved from http: //www. ncbi. nlm. nih. gov/pubmed/14983848 • Dondorp, W. J. , & De Wert, G. M. (2013). Policy: The ‘thousand-dollar genome’: An ethical exploration. European 21, S 6– Dondorp, W. J. , & De Wert, G. M. (2013). Policy: The ‘thousand-dollar genome’: An ethical exploration. European Journal of Human Genetics, 21 S 26. https: //doi. org/10. 1038/ejhg. 2013. 73 • Feinberg, J. , The Child's Right to an Open Future. In W. Aiken and H. La. Follette (eds. ). Whose Child? Totowa, NJ: Rowman & Littlefield (1980): 124 -53. See also Joel Feinberg. The Moral Limits of the Criminal Law: Harm to Self. New York; Oxford: Oxford University Press (1986): 325 -326. • Green, R. C. , Berg, J. S. , Grody, W. W. , Kalia, S. S. , Korf, B. R. , Martin, C. L. , … Biesecker, L. G. (2013). ACMG recommendations for reporting of incidental findings in clinical exome and genome sequencing. Genetics 15(7), 565– 574. https: //doi. org/10. 1038/gim. 2013. 73 incidental findings in clinical exome and genome sequencing. Genetics in Medicine, 15 • Horný, M. , Burgess, J. F. , & Cohen, A. B. (2015). Advanced Imaging Utilization Trends in Privately Insured Patients From 2007 to 2013. Journal of the American College of Radiology, 12(12), 1380– 1387. e 4. https: //doi. org/10. 1016/j. jacr. 2015. 09. 013 Radiology, 12

References • Illes, J. , Kirschen, M. P. , Edwards, E. , Bandettini, P. , Cho, M. K. , Ford, P. J. , … Seto, B. (2008). Practical approaches to incidental findings in brain imaging research. Neurology. Lippincott Williams and Wilkins. https: //doi. org/10. 1212/01. wnl. 0000280469. 17461. 94 imaging research. Neurology. Lippincott Williams and Wilkins. • Jensen, T. S. , Salive, M. E. , Larson, W. , Jones, S. , & Chin, J. (2009). Decision Memo for Screening Computed Tomography Colonography (CTC) for Colorectal Cancer (CAG-00396 N). https: //www. cms. gov/medicare-coverage-database/details/nca-decisionmemo. aspx? NCAId=220&ver=14&Nca. Name=Screening+Computed+Tomography+Colonography+(CTC)+for+Colorectal+Cancer&bc=BEAAAAAAEA AA&&fromdb=true • King, M. (2018). A Social Constructivism Decision-Making Approach to Managing Incidental Findings in Neuroimaging Research. Journal of Ethics and Behavior, 28(5): 393– 410. doi: 10. 1080/10508422. 2017. 1306445. • Kirschen, M. P. , Jaworska, A. , & Illes, J. (2006). Subjects’ expectations in neuroimaging research. Journal 23(2), 205 Kirschen, M. P. , Jaworska, A. , & Illes, J. (2006). Subjects’ expectations in neuroimaging research. Journal of Magnetic Resonance Imaging, 23 – 209. https: //doi. org/10. 1002/jmri. 20499 • Kole, J. , & Fiester, A. (2013). Incidental findings and the need for a revised informed consent process. American 201(5), Kole, J. , & Fiester, A. (2013). Incidental findings and the need for a revised informed consent process. American Journal of Roentgenology, 201 1064– 1068. https: //doi. org/10. 2214/AJR. 13. 11138 • Lee, C. I. , Flaster, H. V. , Haims, A. H. , Monico, E. P. , & Forman, H. P. (2006). Diagnostic CT scans: Institutional informed consent guidelines and practices at academic medical centers. American 187(2), 282– 287. https: //doi. org/10. 2214/AJR. 05. 0813 practices at academic medical centers. American Journal of Roentgenology, 187 • Levin, D. C. , Parker, L. , & Rao, V. M. (2017). Recent Trends in Imaging Use in Hospital Settings: Implications for Future Planning. Journal of the American College of Radiology, 14(3), 331– 336. https: //doi. org/10. 1016/j. jacr. 2016. 08. 025 Radiology, 14 • Levitsky, D. B. , Frank, M. S. , Richardson, M. L. , & Shneidman, R. J. (1993). How should radiologists reply when patients ask about their diagnoses? A survey of radiologists’ and clinicians’ preferences. American 161(2), 433– 436. https: //doi. org/10. 2214/ajr. 161. 2. 8333389 survey of radiologists’ and clinicians’ preferences. American Journal of Roentgenology, 161 • Lumbreras, B. , Donat, L. , & Hernández-Aguado, I. (2010). Incidental findings in imaging diagnostic tests: a systematic review. The British Journal of Radiology, 83(988), 276– 289. https: //doi. org/10. 1259/bjr/98067945 Radiology, 83

References • Mayo-Smith, W. W. , Song, J. H. , Boland, G. L. , Francis, I. R. , Israel, G. M. , Mazzaglia, P. J. , … Pandharipande, P. V. (2017). Management of Incidental Adrenal Masses: A White Paper of the ACR Incidental Findings Committee. Journal 14(8), 1038– 1044. Masses: A White Paper of the ACR Incidental Findings Committee. Journal of the American College of Radiology, 14 https: //doi. org/10. 1016/j. jacr. 2017. 05. 001 • Newson, A. J. (2017). Whole genome sequencing in children: Ethics, choice and deliberation. Journal 43(8), 540– 542. Newson, A. J. (2017). Whole genome sequencing in children: Ethics, choice and deliberation. Journal of Medical Ethics, 43 https: //doi. org/10. 1136/medethics-2016 -103943 • O’Sullivan, J. W. , Muntinga, T. , Grigg, S. , & Ioannidis, J. P. A. (2018). Prevalence and outcomes of incidental imaging findings: Umbrella review. BMJ 361. . O’Sullivan, J. W. , Muntinga, T. , Grigg, S. , & Ioannidis, J. P. A. (2018). Prevalence and outcomes of incidental imaging findings: Umbrella review. BMJ (Online), 361 https: //doi. org/10. 1136/bmj. k 2387 • Phillips, J. P. , Cole, C. , Gluck, J. P. , Shoemaker, J. M. , Petree, L. E. , Helitzer, D. L. , … Holdsworth, M. T. (2015). Stakeholder opinions and ethical perspectives support complete disclosure of incidental findings in mri research. Ethics 25(4), 332– 350. https: //doi. org/10. 1080/10508422. 2014. 938338 support complete disclosure of incidental findings in mri research. Ethics and Behavior, 25 • Pinato, D. J. , Stavraka, C. , Tanner, M. , Esson, A. , Jacobson, E. W. , Wilkins, M. R. , & Libri, V. (2012). Clinical, Ethical and Financial Implications of Incidental Imaging Findings: Experience from a Phase I Trial in Healthy Elderly Volunteers. PLo. S 7(11). https: //doi. org/10. 1371/journal. pone. 0049814 Imaging Findings: Experience from a Phase I Trial in Healthy Elderly Volunteers. PLo. S ONE, 7 (11). https: //doi. org/10. 1371/journal. pone. 0049814 • Royal, J. M. , & Peterson, B. S. (2008). The risks and benefits of searching for incidental findings in MRI research scans. In Journal of Law, Medicine and Ethics(Vol. 36, pp. 305– 314). https: //doi. org/10. 1111/j. 1748 -720 X. 2008. 00274. x • Vernooij, M. W. , Ikram, M. A. , Tanghe, H. L. , Vincent, A. J. P. E. , Hofman, A. , Krestin, G. P. , … van der Lugt, A. (2007). Incidental findings on brain MRI in the general population. The 357(18), 1821– 1828. https: //doi. org/10. 1056/NEJMoa 070972 general population. The New England Journal of Medicine, 357 • Wolf, S. M. , Lawrenz, F. P. , Nelson, C. a, Kahn, J. P. , Cho, M. K. , Clayton, E. W. , … Wilfond, B. S. (2008). Managing incidental findings in human subjects research: analysis and recommendations. The 36(2), 219– 48, 211. research: analysis and recommendations. The Journal of Law, Medicine & Ethics A Journal of the American Society of Law, Medicine & Ethics, 36 https: //doi. org/10. 1111/j. 1748 -720 X. 2008. 00266. x • Wouters, R. H. P. , Cornelis, C. , Newson, A. J. , Bunnik, E. M. , & Bredenoord, A. L. (2017). Scanning the body, sequencing the genome: Dealing with unsolicited findings. Bioethics , 31 31(9), 648– 656. https: //doi. org/10. 1111/bioe. 12375 findings. Bioethics, • Uncited images all licensed online as “Free to Share and Use”.
- Slides: 33