INBORN ERRORS OF METABOLISM INHERITED METABOLIC DISORDERS DR

INBORN ERRORS OF METABOLISM (INHERITED METABOLIC DISORDERS) DR M. C UGONABO

DEFINITION • Rare group of genetic disorders 1 st reported by Sir. Archibald garrod in 1908. They’re characterized by specific blocks along biochemical pathways accumulation of the substrates, reduction or total absence of products and accumulation of metabolites from the alternate pathways.

• The trait which we inherit from our parents is controlled by a pair of genes and there about 50, 000 gene pairs on a xme in man. • The-one-gene-one ezyme hypothesis whereby each gene is responsible for the synthesis of a single enzyme (or protein) implies that absence or abnormality or deficiency of a gene- automatically result in the corresponding absence, abnormality or defy of corresponding protein or enzyme.

• Recent advances in molecular biology has made it clear that IMD results from mutation in the DNA base sequence that codes for specific amino acid sequence of a particular enzyme protein. • A look at this schematic representation is needed to explain the metabolic basis of IMD.

• • Z x =Substrate E X Y y= product of major pathway z =product of minor (alternate) pathway E=enzyme catalyzing conversion of x – y • Note: absence, or deficiency of the response E, can lead to the following: •

• Deficient product of y (major product) as seen in deficient products of cortisol in congenital adrenal hyperplasia (CAH). • Accumulation of substrate x as seen in (PKU) Phyenylketonuria. • Xs production of product z of minor pathway (alternate) – as in xs production of androgen in CAH and phenyl ketones in PKU.

INTRODUCTION • In born errors of metabolism affect conversion of nutrients into one another or energy and are due to impaired activity of enzymes, transporters or cofactors-resulting in the accumulation of abnormal metabolites (substrates) proximal to the metabolic block.

• Abnormal products can also be produced when alternative pathways are used to dispose of the excess metabolites.

MODE OF INHERITANCE • Metabolic disorders are caused by mutations in the genes that code of specific enzymes involved in metabolic pathways. The bulk of these disorders have autosomal recessive inheritance and d/4 affect both boys and girls equally.

• Affected individuals have a mutation in both alleles encoding for specific enzyme/transporter. Parents or such affected offsprings are carriers of the condition and they carry one normal allele and one mutant allele.

• They have a 25% chance/risk of having an affected child in each pregnancy, a 50% chance of having children who are carriers like them and 25% chance of having a child with two normal allele.

• Diagnosis of IMD/IEM can be achieved at 3 levels • Identification of defective enzyme • Identification or detection of abnormal DNA • Identification of abnormal metabolites

• More than 1400 human dxs are now known to be caused by alteration of a single gene out of the 50, 000 gene pairs in each human cell. Sickle Cell Anemia (SCA) is an important example.

SIGNIFICANCE OF IMD • The bulk of sufferers of IMD are infants and children. In view of this any bizarre clinical and biochemical manifestations noted in infancy and childhood should cause a suspicion of an IMD and should be investigated further.

• The following features are suggestive, especially if causes are not obvious. • Failure to thrive (FTT) • Vomiting • Jaundice • Hepatomepaly

• Splenomegaly • Staining of napkins • Mental retardation, seizures, spasticity • Peculiar smell • Hypoglycemia • Rickets (is unresponsive to treatment)

• Its pertinent to note that while some IMD have deleterious consequences others have no clinical effects. • (a) Irreversible damage or death may be prevented if defects are • identified early e. g.

• Phenylketonuria (PKU) • Galactosemia • Neonatal Hypothyroidism • (b) Precipitating factors of ill-effect can be avoided in patients with • G 6 PD deficiency • Acute porphyria • Wilson dx (Hepatolenticular degeneration) • Cystinuria • Hemochromatosis • Cholinesterase deficiency

• Harmless disorders can be treated symptomatically but are clinically input because their effects may cause diagnostic problems e. g. • Renal glycosuria • Alkaptonuria • Gilberts dx

• Major classes of MDs = CHO, PAS and Lipids • IMD of Gas: These usually result in acidemias and aminoacidurias e. g.
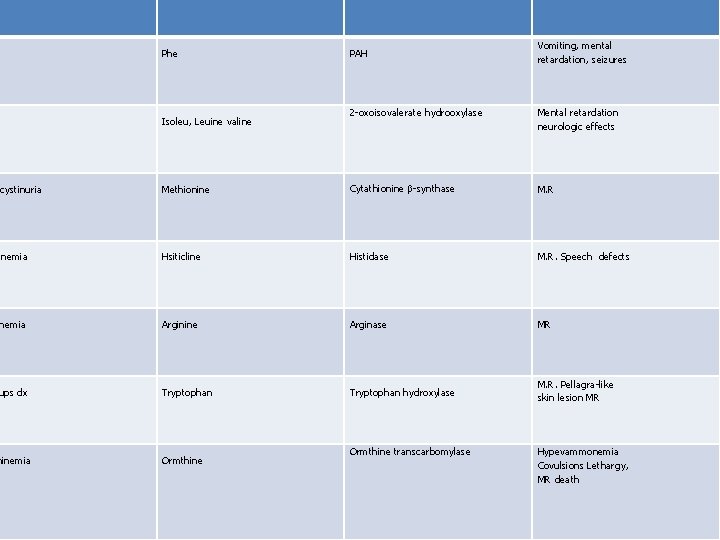
Phe PAH Vomiting, mental retardation, seizures 2 -oxoisovalerate hydrooxylase Mental retardation neurologic effects Methionine Cytathionine β-synthase M. R inemia Hsiticline Histidase M. R. Speech defects nemia Arginine Arginase MR ups dx Tryptophan hydroxylase M. R. Pellagra-like skin lesion MR hinemia Ormthine D Isoleu, Leuine valine ocystinuria Ormthine transcarbomylase Hypevammonemia Covulsions Lethargy, MR death

• Aminoacidurias are due to defects • metabolic defects • - tipt

Phenylketonuria • Phe is an essential amino acid and as such must be supplied in diet.

• PAH is a complex enzyme system involving 4 enzymes and 4 cofactors. • PKU first reported by following (1932) and later by Bickel (1953) in Birminghamautosomal recessive disorder.

Clinical features • Vomiting • Mental retardation and in irreversible after 4 -6 months without treatment • Fits and irritability • Feeding difficulties

C/F cont’d • Mousy odour due to phenylketones • Pale skin • Generalized eczema • Blue eyes • Patchy hair

• The cause of MR due to brain damage is not known for certain but is believed to be due to: • Inhibition of synthesis of serotonin (5 HT). Nor adrenaline and adrenaline by high levels of Phe.

• Phenylpyruvate is a potent inhibitor of mitoenzyme-pyouvate translocase. This reduces the level of pyruvate supply to the brain mito, thus reduced ATP production. The reduction in mito ATP interferes with aminoacid and protein synthesis – deficient myelination and brain function.

Chemical Diagnosis • Screening – Urine Fecl 3 test. Phepyruvate reacts. This text is not tvely with Fecl 3 (phenistix – Ames teststrip). This test is not positive until bady is about 6 weeks old by which time permanent brain damage might have occurred.

• Guthrie & Susi bacterial inhibition method. Blood Phe can be measured in heel-prick blood samples collected 6 -9 days after birth and before baby is discharged from hospital. Many developed countries e. g. US, France, UK, Sweden and Japan now undertakes mass screening for PKU using this method.

• Direct enzyme away from biopsy samples • Prenatal - aminotic fluid cells in early pregnancy using DNA technique.

Management of PKU • Dietary Phe restriction but this is difficult, expensive • Lower Phe plasma levels • Gene therapy (still under trial)

ALKAPTONURIA • Due to deficiency of homogentistic acid oxidase (HGA). The accumulated homogentistic acid polymerizes to form alkapton and in deposited in cartilages causing ochronosis – sites- ears & joints – arthritis in later life

Clinical features • Urine in black or blue • Urine darkens on standing and is enhanced by alkaline conditions • Napkins turn black

• Alkaptonuria is compatible with life. It is autosomal recessive. • * Note homogentisic acid is a reducing agent and so is a cause of false +ve result with Benedicts or clinitest tablets.

ALBINISM • Due to deficiency of tyrosinase in melanocytes, it is AR in inheritance. Pigment of skin, iris, hair is • Eyes may appear pink • Photosentivity due to – pigmentation of the iris • Tendency to development of certain skin cancers due to low–pigmentation of the skin.

MAPLE SYRUP URINE DISEASE (MSUD) • AR inheritance due to defy of decarboxylase • Enzyme that handles three (3) branched-chain amino acids namely leu, ile, val. As a result, these amino acids accumulate in plasma and are excreted in urine together with their corresponding oxoacids.

• Because of the sweet smell of the urine and is akin to that of maple symptoms, the condition has been given the name MSUD. • Presents in the 1 st week of life and if not treated – severe neurological defects and later death within weeks or months.

• - demonstration of high levels of branched chain amino acids in plasma and urine confirmation is by showing defy in leucocytes.

TREATMENT • Diet low in branched chain amino acids, normal development is possible.

HOMOCYSTINURIA • AR disorder due to defy of the enzyme: • Cystathionine synthase in metal of sulfur containing amino acids. • N 5 N 10 methylene tetrahydrofolate reductase • N 5 methyltetrahydroflate – homocystein methylase is low

CLINICAL FEATURES • cataracts • Progressive CNS dysfunction • Thromobsis • Cardiovascular problems

• - presence of plasma and urine homocystine & plasma methionine concentration. • Definitive is by enzyme assay in cultured skin fibroblasts

TPT DISORDERS • Usually due to defective tubular reabsorption. • Cytinoria - AR inheritance due to abnormality in tabular reabsorption of dibasic amino acids with resultant excretion of excessive levels in urine.

• C = Cystine • O = • A = • L = Ornithine Arginine Lysine O-L: They are however synthesized in the body and so deficiency does not occur. Because cystine is insoluble it can precipitate to form calculi in renal tract

• - establishment of high urinary level of the amino acids. • Management – Aims to prevent calculi from - fluid intake • Alkalinizing the urine and so solubility of cystine • -D- penicillamine – this forms chelates and is more soluble than cystine.

• Cystinosis – rare, but dangerous. Characterized intracellular deposition of cystine in many tissues. Affected individual may die young. • Hartnupdx – rare AR. This is xterized by both renal and intestinal transport defects involving neutral amino absorption and tryptophan. acids. There is intestinal increased urinary loss of

CLINICAL FEATURES • pellagra like- scaly rashes on exposed areas of the skin. • Cerebellar ataxia which is reversible • Mental confusion • Demonstration of the characteristics amino acid in urine.

LYSOSOMAL DISORDERS • Mucopolysaccharidoses (MPS) – rare conditions due to defects in any of the enzymes that hydrolyse mucopolysacchs and hence accumulation occurs in such tissues as liver, spleen, eyes, CNS, cartilages and bones e. g.

• Hurler’s syndrome (MPS 1 H) AR inheritance. Affected individuals present in infancy or early childhood and features. • Short stature • Coarse features • M. R. • Clouding of cornea

• Products usually die young due to cardiopulmonary dx Scheie’s syndrome (MPSIS) – similar to Hurler’s but has better prognosis and there is little mental retardation. • Hunter’s syndrome (MPS II) – inherited as xlinked recessive

LIPID STORAGE DISORDERS • There is defy of lysosomal hydrolase – accumulation of sphingolipids e. g. • Tay-sachs dx (GM 2 gangliosidosis) due to defy of hexoraminidase. • GM I gangliosidosis – defy of β-galactosidase

• Niemann-Pick dx – due to defy of sphingomylinase • Gaucher’s ox – due to defy of β-glucosidase (glucocerebrosidase) • Fabry’s dx – resulting from -galactosidase A defy • Metachromic leucodystrophy – due to defy of arylsulfatase.

CLINICAL FEATURES • Organomegaly • - Skeletal abnormalities • - Cherry-red macular spots • - Pulmonary infiltration

Galactosemia • AR disorder due to Gal – I-Puridlyl transferase deficiency. Galacose is utitlised in the synthesis of cerebrosides, some glycoproteins, Xs amount is converted to glucose. Symptoms appear affected infants are normal at birth but symptoms appear only when the baby is fed on milk or milk products. Plasma galactose concentration then.

CLINICAL FEATURES • Vomiting and diarrhea FTT • Cataract formation • FTT • Hepato splenomegaly with jaundice and cirrhosis • M. R.

• Renal tubular damage due to deposition of Gal-1 P in the tubular cells – This damage may lead to generalized aminoaciduria. • - identification of galactose by thin layer chromatography (TLC) and arraying for the Gal-1 PUT) activity in rbc.

TREATMENT • – Eliminating galactose in milk and milk products from the diet.

LACTOSE/GALACTOSE CYCLE • In the absence of G-I PUT, gal-1 -(P) which is insoluble and toxic accumulates in body cell and damages the liver, kidneys, brain. The defect that causes galactosemia is inherited as autosomal recessive gene and results from mutation of the three different genes, GALT (9 p 13) GALK(17 q 24. 1) and GALE (1 p 36 -p 35) that code of the enzymes that metabolise galactose.

• These defects are due to point mutations in the amino acid polypetpic chain. • The mutation in the GALT gene that causes the classic galactosemia (which is severe) is due to replacement of leu for serine (ser 135 leu) at postn 135. A mild form of this x, the Duarte variant is due to replacement of Asp. for Asparagine (Asn 314 Asp) at position 314.

• Note: In classic galactosemia, there is hardly any enzyme activity and both galactose and Gal-1 -p accumulate in the cells – injury to tissue and organs.

• Other types – Ga. ALk 1 gene – produces enzyme galactosidase which Converts galactose to gal-1 -(P). Mutation in this gene affects the activity and stability of the enzyleading to accumulation of galactose and galactitol, a byproduct in the cells – resulting in signs and symptoms of galactosemia e. g. cataract.

• GALE gene – This gene produces UDP – galactose 4 -epimerase required for interconversion between UDP – galactose and UDP glucose. • Mutation of the gene - 2 subtypes Generalised sever form • Mild benign form

CASE STUDY • A 4 - year old boy was seen in the pediatric OPD because of hepatomegaly metabolic acidosis and growth retardation. Investigation: Results turned out like so • Plasma (fasting) • Glucose – 2. 0 mmol/l (3. 0 -5. 5)

• Urate – 0. 61 mmol/l (0. 20 -0. . 43) • Lactate – 3. 7 mmol/L (0. 5 -1. 5) • Cholesterol – 5. 4 mmol/l (3. 0 -5. 0) • Triglycerides – 6. 7 mmol/l (0. 5 -1. 5) • Q. Comment of the result.

GALACTOSEMIA • A disorder of CHO metabolism resulting in the accumulation of galactose. It was first decribed by Von. Reuss- a German physician (1908). The most common form is caused by deficiency of galactose-I- phosphate puridyl transferase, (GALT), the enzyme that converts gal-1 -(P) to glucose-1 -(P).

• Typical traditional screening methods include measurement of galactose –gal-1 -(P) or assay of GALT enzyme activity from a dried blood sport (Beutler test). •

• Affected infants present with - FTT (Failure To Thrive) - Jaundice - Liver failure - Death if left untreated

GLYCOGEN STORAGE DISEASE • The metabolite affected is glycogen.

Types • GSD type I - known as von Gierke’s disease and is due to deficiency of the enzyme glucose – 6 phosphatase Patient presents with a hypoglycemia, hyperuricemia, lactic acidosis and hypertriglyceridemia.

GSD TYPE II (POMPE’S DISEASE) • Due to maltase deficiency ( , 1, 4 glucocidase)- & is a lysosomal defect. • c/f - Hypotonia • - Cardiomyopathy • - Skeletal myopathy
GSD TYPE III (FORBES-CORI DX) • characterized by accumulation of glycogen in skeletal muscle, heart and liver • C/f - Growth retardation • - Muscular weakness • - Cardiomyopathy

GSD TYPE IV (ANDERSEN’S DX) • defect of glycogen branching enzyme • C/f - Hepatomegaly • - Cardiac defects • - Skeletal muscle defects • - Splenomegaly

GSD TYPE V (Mc. ARDLES DX) • deficiency of muscle phosphorylase • C/f - Muscle cramps • - Fatique on heavy exertion • - Myoglobinuria – due to muscle breakdown • - Urine is burgundy red in colour due to myoglobin

GSD TYPE VI (HER’S DISEASE) • due to deficiency of hepatic phosphorylase growth retardation may occur.

MSD TYPE VII (TARUI’S DISEASE) • due to deficiency of phosphofructokinase (PFK) symptoms similar to Mc. Ardles dx.
- Slides: 76